Abstract
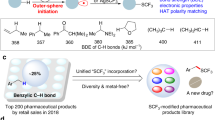
The development of new methodologies that enable chemo- and stereoselective construction of fluorinated substituents, such as the trifluoromethyl (CF3) group, plays an essential role in the synthesis of new pharmaceutical agents. The exceptional ability of the CF3 moiety to prevent in vivo metabolism as well as improve other pharmacological properties has led to numerous innovative strategies for installing this unique functional group. One potential yet underdeveloped approach to access these trifluoromethylated products is direct substitution of α-trifluoromethyl carbanions. Although the electron-withdrawing nature of the CF3 group should facilitate deprotonation of adjacent hydrogens, the propensity of the resulting carbanions to undergo α-elimination of fluoride renders this process highly challenging. Herein, we describe a new strategy for stabilizing and utilizing transient α-trifluoromethyl carbanions that relies on a neighbouring cationic π-allyl palladium complex. These palladium-stabilized zwitterions participate in asymmetric [3 + 2] cycloadditions with a broad range of acceptors, generating valuable di- and trifluoromethylated cyclopentanes, pyrrolidines and tetrahydrofurans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All of the characterization data and experimental protocols are provided in this Article and its Supplementary Information. Data are also available from the corresponding author upon request. Crystallographic data for compound 7k have been deposited at the Cambridge Crystallographic Data Centre under deposition number 1909105 and can be obtained free of charge (http://www.ccdc.cam.ac.uk/data_request/cif).
References
Muller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Meanwell, N. A. Synopsis of some recent tactical application of bioisosteres in drug design. J. Med. Chem. 54, 2529–2591 (2011).
Purser, S., Moore, P. R., Swallow, S. & Gouverneur, V. Fluorine in medicinal chemistry. Chem. Soc. Rev. 37, 320–330 (2008).
Le, C., Chen, T. Q., Liang, T., Zhang, P. & MacMillan, D. W. C. A radical approach to the copper oxidative addition problem: trifluoromethylation of bromoarenes. Science 360, 1010–1014 (2018).
Yang, X., Wu, T., Phipps, R. J. & Toste, F. D. Advances in catalytic enantioselective fluorination, mono-, di-, and trifluoromethylation, and trifluoromethylthiolation reactions. Chem. Rev. 115, 826–870 (2015).
Ma, J.-A. & Cahard, D. Asymmetric fluorination, trifluoromethylation, and perfluoroalkylation reactions. Chem. Rev. 104, 6119–6146 (2004).
Deng, Q.-H., Wadepohl, H. & Gade, L. H. Highly enantioselective copper-catalyzed electrophilic trifluoromethylation of β-ketoesters. J. Am. Chem. Soc. 134, 10769–10772 (2012).
Iseki, K., Nagai, T. & Kobayashi, Y. Asymmetric trifluoromethylation of aldehydes and ketones with trifluoromethyltrimethylsilane catalyzed by chiral quaternary ammonium fluorides. Tetrahedron Lett. 35, 3137–3138 (1994).
Nagib, D. A., Scott, M. E. & MacMillan, D. W. C. Enantioselective α-trifluoromethylation of aldehydes via photoredox organocatalysis. J. Am. Chem. Soc. 131, 10875–10877 (2009).
Kornfilt, D. J. P. & MacMillan, D. W. C. Copper-catalyzed trifluoromethylation of alkyl bromides. J. Am. Chem. Soc. 141, 6853–6858 (2019).
Kautzky, J. A., Wang, T., Evans, R. W. & MacMillan, D. W. C. Decarboxylative trifluoromethylation of aliphatic carboxylic acids. J. Am. Chem. Soc. 140, 6522–6526 (2018).
Nie, J., Guo, H.-C., Cahard, D. & Ma, J.-A. Asymmetric construction of stereogenic carbon centers featuring a trifluoromethyl group from prochiral trifluoromethylated substrates. Chem. Rev. 111, 455–529 (2011).
Morandi, B., Mariampillai, B. & Carreira, E. M. Enantioselective cobalt-catalyzed preparation of trifluoromethyl-substituted cyclopropanes. Angew. Chem. Int. Ed. 50, 1101–1104 (2011).
He, X.-H., Ji, Y.-L., Peng, C. & Han, B. Organocatalytic asymmetric synthesis of cyclic compounds bearing a trifluoromethylated stereogenic center: recent developments. Adv. Synth. Catal. 361, 1923–1957 (2019).
Pitterna, T., Böger, M. & Maienfisch, P. gem-Difluorovinyl derivatives as insecticides and acaricides. Chimia 58, 108–116 (2004).
Shimizu, R., Egami, H., Hamashima, Y. & Sodeoka, M. Copper-catalyzed trifluoromethylation of allylsilanes. Angew. Chem. Int. Ed. 51, 4577–4580 (2012).
Seebach, D., Beck, A. K. & Renaud, P. Di- and trifluoro-substituted dilithium compounds for organic syntheses. Angew. Chem. Int. Ed. Engl. 25, 98–99 (1986).
Komatsu, Y., Sakamoto, T. & Kitazume, T. Synthetic applications of the carbanion with a fluoroalkyl group generated by palladium(0) catalyst under neutral conditions. J. Org. Chem. 64, 8369–8374 (1999).
Itoh, Y., Yamanaka, M. & Mikami, K. Direct generation of Ti-enolate of α-CF3 ketone: theoretical study and high-yielding and diastereoselective aldol reaction. J. Am. Chem. Soc. 126, 13174–13175 (2004).
Yin, L., Brewitz, L., Kumagai, N. & Shibasaki, M. Catalytic generation of α-CF3 enolate: direct catalytic asymmetric Mannich-type reaction of α-CF3 amide. J. Am. Chem. Soc. 136, 17958–17961 (2014).
Wu, Y., Hu, L., Li, Z. & Deng, L. Catalytic asymmetric umpolung reactions of imines. Nature 523, 445–450 (2015).
Sun, Q. et al. The squaramide-catalyzed 1,3-dipolar cycloaddition of nitroalkenes with N-2,2,2-trifluoroethylisatin ketimines: an approach for the synthesis of 5′-trifluoromethyl-spiro[pyrrolidin-3,2′-oxindoles]. Adv. Synth. Catal. 357, 3187–3196 (2015).
Trost, B. M. [3+2] Cycloaddition approaches to five-membered rings via trimethylenemethane and its equivalents. Angew. Chem. Int. Ed. 25, 1–20 (1986).
Nakamura, E. [3+2] Cycloaddition of trimethylenemethane and its synthetic equivalents. Org. React. 61, 1–217 (2003).
Trost, B. M., Stambuli, J. P., Silverman, S. M. & Schwörer, U. Palladium-catalyzed asymmetric [3+2] trimethylenemethane cycloaddition reactions. J. Am. Chem. Soc. 128, 13328–13329 (2006).
Trost, B. M., Silverman, S. M. & Stambuli, J. P. Palladium-catalyzed asymmetric [3+2] cycloaddition of trimethylenemethane with imines. J. Am. Chem. Soc. 129, 12398–12399 (2007).
Trost, B. M., Bringley, D. A. & Silverman, S. M. Asymmetric synthesis of methylenetetrahydrofurans by palladium-catalyzed [3+2] cycloaddition of trimethylenemethane with aldehydes—a novel ligand design. J. Am. Chem. Soc. 133, 7664–7667 (2011).
Trost, B. M. & Bringley, D. A. Enantioselective synthesis of 2,2-disubstituted tetrahydrofurans: palladium-catalyzed [3+2] cycloadditions of trimethylenemethane with ketones. Angew. Chem. Int. Ed. 52, 4466–4469 (2013).
Allen, B. D. W., Lakeland, C. P. & Harrity, J. P. A. Utilizing palladium-stabilized zwitterions for the construction of N-heterocycles. Chem. Eur. J. 23, 13830–13857 (2017).
Rivinoja, D. J., Gee, Y. S., Gardiner, M. G., Ryan, J. H. & Hyland, C. J. T. The diastereoselective synthesis of pyrroloindolines by Pd-catalyzed dearomative cycloaddition of 1-tosyl-2-vinylaziridine to 3-nitroindoles. ACS Catal. 7, 1053–1056 (2017).
Laugeois, M. et al. Palladium(0)-catalyzed dearomative [3+2] cycloaddition of 3-nitroindoles with vinylcyclopropanes: an entry to stereodefined 2,3-fused cyclopentannulated indoline derivatives. Org. Lett. 19, 2266–2269 (2017).
Li, W.-K., Liu, Z.-S., He, L., Kang, T.-R. & Liu, Q.-Z. Enantioselective cycloadditions of vinyl cyclopropanes and nitroolefins for functionally and optically enriched nitrocyclopentanes. Asian J. Org. Chem. 4, 28–32 (2015).
Kobayashi, S. & Jørgensen, K. A. Cycloaddition Reactions in Organic Synthesis (Wiley, 2001).
Gordon, D. J., Fenske, R. F., Nanninga, T. N. & Trost, B. M. Molecular orbital study of substituted trimethylenemethane-bis(phosphine)palladium complexes. J. Am. Chem. Soc. 103, 5974–5976 (1981).
Trost, B. M. & Chan, D. M. T. New conjunctive reagents. 2-acetoxymethyl-3-allyltrimethylsilane for methylenecyclopentane annulations catalyzed by palladium(0). J. Am. Chem. Soc. 101, 6429–6432 (1979).
Shimuzu, I., Ohashi, Y. & Tsuji, J. Palladium-catalyzed [3+2] cycloaddition reaction using 2-(sulfonylmethyl)- or 2-(cyanomethyl)allyl carbonate. Tetrahedron Lett. 25, 5183–5186 (1984).
Trost, B. M. & Wang, Y. A deprotonation approach to the unprecedented amino-trimethylenemethane chemistry: regio, diastereo, and enantioselective synthesis of complex amino cycles. Angew. Chem. Int. Ed. 57, 11025–11029 (2018).
Bunten, K. A., Chen, L., Fernandez, A. L. & Poë, A. J. Cone angles: Tolman’s and Plato’s. Coord. Chem. Rev. 233–234, 41–51 (2002).
Trost, B. M. & Lam, T. M. Development of diamidophosphite ligands and their application to the palladium-catalyzed vinyl-substituted trimethylenemethane asymmetric [3+2] cycloaddition. J. Am. Chem. Soc. 134, 11319–11321 (2012).
Adams, G. L. et al. Il-8 receptor antagonists. International patent WO2008070707A1 (2008).
Butora, G. et al. Heterocyclic cyclopentyl tetrahydroisoquinoline and tetrahydropyridopyridine modulators of chemokine receptor activity. International patent WO2004094371A2 (2004).
Swann, S. L. & Vasudevan, A. Compounds useful as inhibitors of ROCK kinases. US patent 20090258907A1 (2009).
Zafrani, Y. et al. Difluoromethyl bioisostere: examining the ‘lipophilic hydrogen bond donor’ concept. J. Med. Chem. 60, 797–804 (2017).
Kim, K. H., Lin, N.-H. & Anderson, D. J. Quantitative structure–activity relationships of nicotine analogues as neuronal nicotinic acetylcholine receptor ligands. Bioorg. Med. Chem. 4, 2211–2217 (1996).
Trost, B. M. & Silverman, S. M. Enantioselective construction of pyrrolidines by palladium-catalyzed asymmetric [3+2] cycloaddition of trimethylenemethane with imines. J. Am. Chem. Soc. 134, 4941–4954 (2012).
Trost, B. M., Bringley, D. A. & O’Keefe, B. M. Highly substituted enantioenriched cyclopentane derivatives by palladium-catalyzed [3+2] trimethylenemethane cycloadditions with disubstituted nitroalkenes. Org. Lett. 15, 5630–5633 (2013).
Acknowledgements
We are grateful to the Tamaki Foundation and Chugai Pharmaceutical for financial support. C. Kalnmals is acknowledged for proofreading the manuscript. We also thank Z. Jiao for technical assistance and S. Lynch for conducting some nuclear Overhauser effect and 19F-decoupled experiments. Finally, we acknowledge J. Maclaren (Stanford University) for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
B.M.T. conceived and designed the project. Y.W. and C.-I.H. carried out the experiments. B.M.T., Y.W. and C.-I.H. wrote the manuscript. All authors analysed the data and discussed the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Experimental protocols and characterization data (NMR, infrared, optical rotation, high-performance liquid chromatography and so on) for every new product mentioned in this work.
Crystallographic data
Crystallographic data for compound 7k (CCDC reference 1909105).
Rights and permissions
About this article
Cite this article
Trost, B.M., Wang, Y. & Hung, CI.(. Use of α-trifluoromethyl carbanions for palladium-catalysed asymmetric cycloadditions. Nat. Chem. 12, 294–301 (2020). https://doi.org/10.1038/s41557-019-0412-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0412-9