Abstract
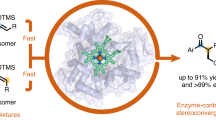
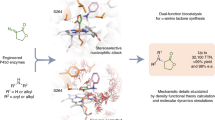
The ability to selectively functionalize ubiquitous C–H bonds streamlines the construction of complex molecular architectures from easily available precursors. Here we report enzyme catalysts derived from a cytochrome P450 that use a nitrene transfer mechanism for the enantioselective amination of primary, secondary and tertiary C(sp3)–H bonds. These fully genetically encoded enzymes are produced and function in bacteria, where they can be optimized by directed evolution for a broad spectrum of enantioselective C(sp3)–H amination reactions. These catalysts can aminate a variety of benzylic, allylic and aliphatic C–H bonds in excellent enantioselectivity with access to either antipode of product. Enantioselective amination of primary C(sp3)–H bonds in substrates that bear geminal dimethyl substituents furnished chiral amines that feature a quaternary stereocentre. Moreover, these enzymes enabled the enantioconvergent transformation of racemic substrates that possess a tertiary C(sp3)–H bond to afford products that bear a tetrasubstituted stereocentre, a process that has eluded small-molecule catalysts. Further engineering allowed for the enantioselective construction of methyl–ethyl stereocentres, which is notoriously challenging in asymmetric catalysis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data necessary to support the paper’s conclusions are available in the main text and the Supplementary Information. Solid-state structures of 2a, 4a, 5a and 5f are available free of charge from the Cambridge Crystallographic Data Centre under reference nos CCDC 1905551, 1905553, 1905552 and 1905554. Plasmids encoding the enzymes reported in this study are available for research purposes from F.H.A. under a material transfer agreement with the California Institute of Technology.
References
Saint-Denis, T. G., Zhu, R.-Y., Chen, G., Wu, Q.-F. & Yu, J.-Q. Enantioselective C(sp 3)‒H bond activation by chiral transition metal catalysts. Science 359, eaao4798 (2018).
Gutekunst, W. R. & Baran, P. S. C–H functionalization logic in total synthesis. Chem. Soc. Rev. 40, 1976–1991 (2011).
Hartwig, J. F. & Larsen, M. A. Undirected, homogeneous C–H bond functionalization: challenges and opportunities. ACS Cent. Sci. 2, 281–292 (2016).
Lu, H. & Zhang, X. P. Catalytic C–H functionalization by metalloporphyrins: recent developments and future directions. Chem. Soc. Rev. 40, 1899–1909 (2011).
Davies, H. M. L. & Manning, J. R. Catalytic C–H functionalization by metal carbenoid and nitrenoid insertion. Nature 451, 417–424 (2008).
Smalley, A. P., Cuthbertson, J. D. & Gaunt, M. J. Palladium-catalyzed enantioselective C–H activation of aliphatic amines using chiral anionic binol-phosphoric acid ligands. J. Am. Chem. Soc. 139, 1412–1415 (2017).
Quasdorf, K. W. & Overman, L. E. Catalytic enantioselective synthesis of quaternary carbon stereocentres. Nature 516, 181–191 (2014).
Bhat, V., Welin, E. R., Guo, X. & Stoltz, B. M. Advances in stereoconvergent catalysis from 2005 to 2015: transition-metal-mediated stereoablative reactions, dynamic kinetic resolutions, and dynamic kinetic asymmetric transformations. Chem. Rev. 117, 4528–4561 (2017).
Lewis, J. C., Coelho, P. S. & Arnold, F. H. Enzymatic functionalization of carbon–hydrogen bonds. Chem. Soc. Rev. 40, 2003–2021 (2011).
Ortiz de Montellano, P. R. Hydrocarbon hydroxylation by cytochrome P450 enzymes. Chem. Rev. 110, 932–948 (2010).
Fasan, R. Tuning P450 enzymes as oxidation catalysts. ACS Catal. 2, 647–666 (2012).
Coelho, P. S., Brustad, E. M., Kannan, A. & Arnold, F. H. Olefin cyclopropanation via carbene transfer catalyzed by engineered cytochrome P450 enzymes. Science 339, 307–310 (2013).
Kan, S. B. J., Lewis, R. D., Chen, K. & Arnold, F. H. Directed evolution of cytochrome c for carbon–silicon bond formation: bringing silicon to life. Science 354, 1048–1051 (2016).
Key, H. M., Dydio, P., Clark, D. S. & Hartwig, J. F. Abiological catalysis by artificial haem proteins containing noble metals in place of iron. Nature 534, 534–537 (2016).
Dydio, P., Key, H. M., Hayashi, H., Clark, D. S. & Hartwig, J. F. Chemoselective, enzymatic C–H bond amination catalyzed by a cytochrome P450 containing an Ir(Me)-PIX cofactor. J. Am. Chem. Soc. 139, 1750–1753 (2017).
Brandenberg, O. F., Fasan, R. & Arnold, F. H. Exploiting and engineering hemoproteins for abiological carbene and nitrene transfer reactions. Curr. Opin. Biotechnol. 47, 102–111 (2017).
Singh, R., Bordeaux, M. & Fasan, R. P450-catalyzed intramolecular sp 3 C–H amination with arylsulfonyl azide substrates. ACS Catal. 4, 546–552 (2014).
Zhang, R. K. et al. Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp 3 C–H functionalization. Nature 565, 67–72 (2019).
McIntosh, J. A. et al. Enantioselective intramolecular C–H amination catalyzed by engineered cytochrome P450 enzymes in vitro and in vivo. Angew. Chem. Int. Ed. 52, 9309–9312 (2013).
Hyster, T. K., Farwell, C. C., Buller, A. R., McIntosh, J. A. & Arnold, F. H. Enzyme-controlled nitrogen-atom transfer enables regiodivergent C–H amination. J. Am. Chem. Soc. 136, 15505–15508 (2014).
Prier, C. K., Zhang, R. K., Buller, A. R., Brinkmann-Chen, S. & Arnold, F. H. Enantioselective, intermolecular benzylic C–H amination catalysed by an engineered iron–haem enzyme. Nat. Chem. 9, 629–634 (2017).
Kurokawa, T., Kim, M. & Du Bois, J. Synthesis of 1,3-diamines through rhodium-catalyzed C–H insertion. Angew. Chem. Int. Ed. 48, 2777–2779 (2009).
Lu, H., Jiang, H., Wojtas, L. & Zhang, X. P. Selective intramolecular C–H amination through the metalloradical activation of azides: synthesis of 1,3-diamines under neutral and nonoxidative conditions. Angew. Chem. Int. Ed. 49, 10192–10196 (2010).
Zalatan, D. N. & Du Bois, J. A chiral rhodium carboxamidate catalyst for enantioselective C–H amination. J. Am. Chem. Soc. 130, 9220–9221 (2008).
Ichinose, M. et al. Enantioselective intramolecular benzylic C–H bond amination: efficient synthesis of optically active benzosultams. Angew. Chem. Int. Ed. 50, 9884–9887 (2011).
Roizen, J. L., Harvey, M. E. & Du Bois, J. Metal-catalyzed nitrogen-atom transfer methods for the oxidation of aliphatic C–H bonds. Acc. Chem. Res. 45, 911–922 (2012).
Paradine, S. M. & White, M. C. Iron-catalyzed intramolecular allylic C–H amination. J. Am. Chem. Soc. 134, 2036–2039 (2012).
Paradine, S. M. et al. A manganese catalyst for highly reactive yet chemoselective intramolecular C(sp 3)–H amination. Nat. Chem. 7, 987–994 (2015).
Clark, J. R., Feng, K., Sookezian, A. & White, M. C. Manganese-catalysed benzylic C(sp 3)–H amination for late-stage functionalization. Nat. Chem. 10, 583–591 (2018).
Li, C. et al. Catalytic radical process for enantioselective amination of C(sp 3)−H bonds. Angew. Chem. Int. Ed. 57, 16837–16841 (2018).
Lu, H., Lang, K., Jiang, H., Wojtas, L. & Zhang, X. P. Intramolecular 1,5-C(sp 3)–H radical amination via Co(ii)-based metalloradical catalysis for five-membered cyclic sulfamides. Chem. Sci. 7, 6934–6939 (2016).
Capdevila, J. H. et al. The highly stereoselective oxidation of polyunsaturated fatty acids by cytochrome P450BM-3. J. Biol. Chem. 271, 22663–22671 (1996).
Chen, K., Huang, X., Kan, S. B. J., Zhang, R. K. & Arnold, F. H. Enzymatic construction of highly strained carbocycles. Science 360, 71–75 (2018).
Nagib, D. A. Catalytic desymmetrization by C–H functionalization as a solution to the chiral methyl problem. Angew. Chem. Int. Ed. 56, 7354–7356 (2017).
Kainz, Q. M. et al. Asymmetric copper-catalyzed C–N cross-couplings induced by visible light. Science 351, 681–684 (2016).
Wendlandt, A. E., Vangal, P. & Jacobsen, E. N. Quaternary stereocentres via an enantioconvergent catalytic SN1 reaction. Nature 556, 447–451 (2018).
Zhang, X. et al. An enantioconvergent halogenophilic nucleophilic substitution (SN2X) reaction. Science 363, 400–404 (2019).
Hennessy, E. T., Liu, R. Y., Iovan, D. A., Duncan, R. A. & Betley, T. A. Iron-mediated intermolecular N-group transfer chemistry with olefinic substrates. Chem. Sci. 5, 1526–1532 (2014).
Singh, R., Kolev, J. N., Sutera, P. A. & Fasan, R. Enzymatic C(sp 3)–H amination: P450-catalyzed conversion of carbonazidates into oxazolidinones. ACS Catal. 5, 1685–1691 (2015).
Fandrick, K. R. et al. A general copper–BINAP-catalyzed asymmetric propargylation of ketones with propargyl boronates. J. Am. Chem. Soc. 133, 10332–10335 (2011).
Yang, Y., Shi, S.-L., Niu, D., Liu, P. & Buchwald, S. L. Catalytic asymmetric hydroamination of unactivated internal olefins to aliphatic amines. Science 349, 62–66 (2015).
Jacobs, B. P., Wolczanski, P. T., Jiang, Q., Cundari, T. R. & MacMillan, S. N. Rare examples of Fe(iv) alkyl-imide migratory insertions: impact of Fe–C covalency in (Me2IPr)Fe(═NAd)R2 (R = neoPe, 1-nor). J. Am. Chem. Soc. 139, 12145–12148 (2017).
Acknowledgements
This work was supported by the NSF (grant nos MCB-1513007 for F.H.A. and CHE-1654122 for P.L.). Y.Y. thanks the National Institutes of Health for a postdoctoral fellowship (grant no. 1F32GM133126-01). Calculations were performed at the Center for Research Computing at the University of Pittsburgh. We thank R. K. Zhang, K. Chen, D. C. Miller, D. K. Romney (Caltech) and Y. Wang (University of Pittsburgh) for helpful discussions and comments on the manuscript, L. Henling for X-ray diffraction analysis and S. Virgil for assistance with chiral supercritical fluid chromatography analysis.
Author information
Authors and Affiliations
Contributions
Y.Y. designed the overall research with F.H.A. providing guidance. Y.Y. and I.C. designed and performed the initial screening of haem proteins and directed evolution experiments. Y.Y. designed and performed the substrate scope study and mechanistic study. X.Q. carried out the computational studies with P.L. providing guidance. Y.Y. and F.H.A. wrote the manuscript with the input of all other authors.
Corresponding authors
Ethics declarations
Competing interests
A provisional patent application (inventors Y.Y. and I.C.) has been filed through the California Institute of Technology. The provisional patent covers the development and application of engineered cytochromes P450 for the synthesis of chiral diamine derivatives by C–H amination.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
NMR spectra
1H, 13C and 19F NMR spectra.
X-ray crystal structure of 2a
Crystallographic data for compound 2a.
X-ray crystal structure of 4a
Crystallographic data for compound 4a.
X-ray crystal structure of 5a
Crystallographic data for compound 5a.
X-ray crystal structure of 5f
Crystallographic data for compound 5f.
Rights and permissions
About this article
Cite this article
Yang, Y., Cho, I., Qi, X. et al. An enzymatic platform for the asymmetric amination of primary, secondary and tertiary C(sp3)–H bonds. Nat. Chem. 11, 987–993 (2019). https://doi.org/10.1038/s41557-019-0343-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0343-5
This article is cited by
-
Biocatalytic, enantioenriched primary amination of tertiary C–H bonds
Nature Catalysis (2024)
-
Recent Progress on Peroxidase Modification and Application
Applied Biochemistry and Biotechnology (2024)
-
Enzyme-controlled stereoselective radical cyclization to arenes enabled by metalloredox biocatalysis
Nature Catalysis (2023)
-
Stereoselective construction of β-, γ- and δ-lactam rings via enzymatic C–H amidation
Nature Catalysis (2023)
-
Biocatalytic, stereoconvergent alkylation of (Z/E)-trisubstituted silyl enol ethers
Nature Synthesis (2023)