Abstract
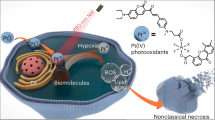
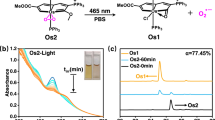
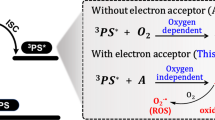
Hypoxic tumours are a major problem for cancer photodynamic therapy. Here, we show that photoredox catalysis can provide an oxygen-independent mechanism of action to combat this problem. We have designed a highly oxidative Ir(iii) photocatalyst, [Ir(ttpy)(pq)Cl]PF6 ([1]PF6, where ‘ttpy’ represents 4′-(p-tolyl)-2,2′:6′,2′′-terpyridine and ‘pq’ represents 3-phenylisoquinoline), which is phototoxic towards both normoxic and hypoxic cancer cells. Complex 1 photocatalytically oxidizes 1,4-dihydronicotinamide adenine dinucleotide (NADH)—an important coenzyme in living cells—generating NAD• radicals with a high turnover frequency in biological media. Moreover, complex 1 and NADH synergistically photoreduce cytochrome c under hypoxia. Density functional theory calculations reveal π stacking in adducts of complex 1 and NADH, facilitating photoinduced single-electron transfer. In cancer cells, complex 1 localizes in mitochondria and disrupts electron transport via NADH photocatalysis. On light irradiation, complex 1 induces NADH depletion, intracellular redox imbalance and immunogenic apoptotic cancer cell death. This photocatalytic redox imbalance strategy offers a new approach for efficient cancer phototherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information files, or from the corresponding authors on reasonable request. Crystallographic data for the complex [1]PF6·(1.5 toluene) reported in this Article have been deposited at the Cambridge Crystallographic Data Centre (under deposition number CCDC 1840366). After the Open Access agreement has been established, underpinning datasets will be deposited in Warwick’s Institutional Repository—Warwick Research Archive Portal, according to the Open Access Agreement.
References
Riddell, I. A. & Lippard, S. J. Cisplatin and oxaliplatin: our current understanding of their actions. Met Ions Life Sci. 18, 1–42 (2018).
Meier-Menches, S. M., Gerner, C., Berger, W., Hartinger, C. G. & Keppler, B. K. Structure–activity relationships for ruthenium and osmium anticancer agents—towards clinical development. Chem. Soc. Rev. 47, 909–928 (2018).
Bergamo, A., Dyson, P. J. & Sava, G. The mechanism of tumour cell death by metal-based anticancer drugs is not only a matter of DNA interactions. Coord. Chem. Rev. 360, 17–33 (2018).
Farrer, N. J., Salassa, L. & Sadler, P. J. Photoactivated chemotherapy (PACT): the potential of excited-state d-block metals in medicine. Dalton Trans. 10690–10701 (2009).
Banerjee, S. & Chakravarty, A. R. Metal complexes of curcumin for cellular imaging, targeting, and photoinduced anticancer activity. Acc. Chem. Res. 48, 2075–2083 (2015).
Lo, K. K. W. Luminescent rhenium(i) and iridium(iii) polypyridine complexes as biological probes, imaging reagents, and photocytotoxic agents. Acc. Chem. Res. 48, 2985–2995 (2015).
Knoll, J. D. & Turro, C. Control and utilization of ruthenium and rhodium metal complex excited states for photoactivated cancer therapy. Coord. Chem. Rev. 282–283, 110–126 (2015).
Liu, J. et al. Harnessing ruthenium(ii) as photodynamic agents: encouraging advances in cancer therapy. Coord. Chem. Rev. 363, 17–28 (2018).
Heinemann, F., Karges, J. & Gasser, G. Critical overview of the use of Ru (ii) polypyridyl complexes as photosensitizers in one-photon and two-photon photodynamic therapy. Acc. Chem. Res. 50, 2727–2736 (2017).
Shi, G. et al. Ru(ii) dyads derived from α-oligothiophenes: a new class of potent and versatile photosensitizers for PDT. Coord. Chem. Rev. 282–283, 127–138 (2015).
Meggers, E. Asymmetric catalysis activated by visible light. Chem. Commun. 51, 3290–3301 (2015).
Spring, B. Q., Rizvi, I., Xu, N. & Hasan, T. The role of photodynamic therapy in overcoming cancer drug resistance. Photochem. Photobiol. Sci. 14, 1476–1491 (2015).
Teicher, B. A. Hypoxia and drug resistance. Cancer Metast. Rev. 13, 139–168 (1994).
Wang, H. P. et al. Phospholipid hydroperoxide glutathione peroxidase protects against singlet oxygen-induced cell damage of photodynamic therapy. Free Radical Biol. Med. 30, 825–835 (2001).
Henderson, B. W. & Miller, A. C. Effects of scavengers of reactive oxygen and radical species on cell survival following photodynamic treatment in vitro: comparison to ionizing radiation. Radiat. Res. 108, 196–205 (1986).
Gomer, C. J. et al. Photodynamic therapy-mediated oxidative stress can induce expression of heat shock proteins. Cancer Res. 56, 2355–2360 (1996).
Ruhdorfer, S., Sanovic, R., Sander, V., Krammer, B. & Verwanger, T. Gene expression profiling of the human carcinoma cell line A-431 after 5-aminolevulinic acid-based photodynamic treatment. Int. J. Oncol. 30, 1253–1262 (2007).
Tong, Z., Singh, G. & Rainbow, A. J. Sustained activation of the extracellular signal-regulated kinase pathway protects cells from photofrin-mediated photodynamic therapy. Cancer Res. 62, 5528–5535 (2002).
Edmonds, C., Hagan, S., Gallagher-Colombo, S. M., Busch, T. M. & Cengel, K. A. Photodynamic therapy activated signaling from epidermal growth factor receptor and STAT3: targeting survival pathways to increase PDT efficacy in ovarian and lung cancer. Cancer Biol. Ther. 13, 1463–1470 (2012).
Assefa, Z. et al. The activation of the c-Jun N-terminal kinase and p38 mitogen-activated protein kinase signaling pathways protects HeLa cells from apoptosis following photodynamic therapy with hypericin. J. Biol. Chem. 274, 8788–8796 (1999).
Kessel, D. & Erickson, C. Porphyrin photosensitization of multi-drug resistant cell types. Photochem. Photobiol. 55, 397–399 (1992).
Höckel, M. & Vaupel, P. Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J. Natl Cancer Inst. 93, 266–276 (2001).
Klimova, T. & Chandel, N. S. Mitochondrial complex III regulates hypoxic activation of HIF. Cell Death Differ. 15, 660–666 (2008).
Chiarugi, A., Dölle, C., Felici, R. & Ziegler, M. The NAD metabolome—a key determinant of cancer cell biology. Nat. Rev. Cancer 12, 741–752 (2012).
Prier, C. K. et al. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Huo, H. et al. Asymmetric photoredox transition-metal catalysis activated by visible light. Nature 515, 100–103 (2014).
Demas, J. N. et al. Oxygen sensors based on luminescence quenching. Anal. Chem. 71, 793A–800A (1999).
Liu, Z. et al. The potent oxidant anticancer activity of organoiridium catalysts. Angew. Chem. Int. Ed. 53, 3941–3946 (2014).
Ma, J. et al. Preparation of chiral-at-metal catalysts and their use in asymmetric photoredox chemistry. Nat. Protoc. 13, 605–632 (2018).
Peterson, J. R., Smith, T. A. & Thordarson, P. Synthesis and room temperature photo-induced electron transfer in biologically active bis(terpyridine) ruthenium(ii)–cytochrome c bioconjugates and the effect of solvents on the bioconjugation of cytochrome c. Org. Biomol. Chem. 8, 151–162 (2010).
Boaz, H. & Rollefson, G. K. The quenching of fluorescence. Deviations from the Stern–Volmer law. J. Am. Chem. Soc. 72, 3435–3443 (1950).
Matsuzaki, S., Kotake, Y. & Humphries, K. M. Identification of mitochondrial electron transport chain-mediated NADH radical formation by EPR spin-trapping techniques. Biochemistry 50, 10792–10803 (2011).
Lapuente-Brun, E. et al. Supercomplex assembly determines electron flux in the mitochondrial electron transport chain. Science 340, 1567–1570 (2013).
Li, H. et al. Cytochrome c release and apoptosis induced by mitochondrial targeting of nuclear orphan receptor TR3. Science 289, 1159–1164 (2000).
Margoliash, E. & Frohwirt, N. Spectrum of horse-heart cytochrome c. Biochem. J. 71, 570–572 (1959).
Huang, H. et al. Highly charged ruthenium(ii) polypyridyl complexes as lysosome-localized photosensitizers for two-photon photodynamic therapy. Angew. Chem. Int. Ed. 54, 14049–14052 (2015).
Peng, Q. et al. 5-Aminolevulinic acid-based photodynamic therapy. Clinical research and future challenges. Cancer 79, 2282–2308 (1997).
Huang, H. et al. Real-time tracking mitochondrial dynamic remodeling with two-photon phosphorescent iridium(iii) complexes. Biomaterials 83, 321–331 (2016).
Berezin, M. Y. & Achilefu, S. Fluorescence lifetime measurements and biological imaging. Chem. Rev. 110, 2641–2684 (2010).
Berridge, M. V., Herst, P. M. & Tan, A. S. Tetrazolium dyes as tools in cell biology: new insights into their cellular reduction. Biotechnol. Annu. Rev. 11, 127–152 (2005).
Waghray, M. et al. Hydrogen peroxide is a diffusible paracrine signal for the induction of epithelial cell death by activated myofibroblasts. FASEB J. 19, 854–856 (2005).
He, Y. Y. & Häder, D. P. UV-B-induced formation of reactive oxygen species and oxidative damage of the cyanobacterium Anabaena sp.: protective effects of ascorbic acid and N-acetyl-l-cysteine. J. Photochem. Photobiol. B 66, 115–124 (2002).
Ricci, J. E. et al. Disruption of mitochondrial function during apoptosis is mediated by caspase cleavage of the p75 subunit of complex I of the electron transport chain. Cell 117, 773–786 (2004).
Tesniere, A. et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene 29, 482–491 (2010).
Coverdale, J. P. C. et al. Asymmetric transfer hydrogenation by synthetic catalysts in cancer cells. Nat. Chem. 10, 347–354 (2018).
Moan, J. Effect of bleaching of porphyrin sensitizers during photodynamic therapy. Cancer Lett. 33, 45–53 (1986).
Gorrini, C., Harris, I. S. & Mak, T. W. Modulation of oxidative stress as an anticancer strategy. Nat. Rev. Drug Discov. 12, 931–947 (2013).
Porras, J. A., Mills, I. N., Transue, W. J. & Bernhard, S. Highly fluorinated Ir(iii)-2,2′:6′,2″-terpyridine–phenylpyridine–X complexes via selective C–F activation: robust photocatalysts for solar fuel generation and photoredox catalysis. J. Am. Chem. Soc. 138, 9460–9472 (2016).
Tinker, L. L. et al. Visible light induced catalytic water reduction without an electron relay. Chem. Eur. J. 13, 8726–8732 (2007).
Gȩbicki, J., Marcinek, A. & Zielonka, J. Transient species in the stepwise interconversion of NADH and NAD+. Acc. Chem. Res. 37, 379–386 (2004).
Fukuzumi, S., Inada, O. & Suenobu, T. Mechanisms of electron-transfer oxidation of NADH analogues and chemiluminescence. Detection of the keto and enol radical cations. J. Am. Chem. Soc. 125, 4808–4816 (2003).
Tanaka, M., Ohkubo, K. & Fukuzumi, S. DNA cleavage by UVA irradiation of NADH with dioxygen via radical chain processes. J. Phys. Chem. A 110, 11214–11218 (2006).
Lee, H. Y., Chen, S., Zhang, M. H. & Shen, T. Studies on the synthesis of two hydrophilic hypocrellin derivatives with enhanced absorption in the red spectral region and on their photogeneration of O2 • − and O2 (1Δg). J. Photochem. Photobiol. B 71, 43–50 (2003).
Hvasanov, D., Mason, A. F., Goldstein, D. C., Bhadbhade, M. & Thordarson, P. Optimising the synthesis, polymer membrane encapsulation and photoreduction performance of Ru(ii)-and Ir(iii)-bis(terpyridine) cytochrome c bioconjugates. Org. Biomol. Chem. 11, 4602–4612 (2013).
Sabharwal, S. S. & Schumacker, P. T. Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles’ heel? Nat. Rev. Cancer 14, 709–721 (2014).
Wenzel, C. et al. 3D high-content screening for the identification of compounds that target cells in dormant tumor spheroid regions. Exp. Cell Res. 323, 131–143 (2014).
Ji, J., Zhang, Y., Chen, W. R. & Wang, X. DC vaccine generated by ALA-PDT-induced immunogenic apoptotic cells for skin squamous cell carcinoma. Oncoimmunology 5, e1072674 (2016).
Galluzzi, L., Buqué, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 17, 97–111 (2017).
Acknowledgements
We thank the EPSRC (grants EP/G006792, EP/F034210/1 and EP/P030572/1 to P.J.S.; platform grant EP/P001459/1 to M.J.P.; EPSRC DTP studentship to T.M.; EP/N010825/1 to M.S.; and EP/N010825 to V.G.S.), MRC (grant G0701062 to P.J.S.), The Royal Society (Newton International Fellowship NF160307 to H.H.; and Newton-Bhahba International Fellowship NF151429 to S.B.), Leverhulme Trust (Senior Research Fellowship to V.G.S.), National Science Foundation of China (NSFC grant 21701113 to P.Z.; and 21525105, 21471164 and 21778079 to H.C.), 973 Program (2015CB856301 to H.C.), The Fundamental Research Funds for the Central Universities (to H.C.), ERC (Consolidator Grant GA 681679 PhotoMedMet to G.G.), French Government (Investissements d’Avenir grant ANR-10-IDEX-0001-02 PSL to G.G.) and the Sun Yat-sen University Startup fund (75110-18841213 to H.H.). We also thank W. Zhang, L. Song and P. Aston for assistance with mass spectrometry, J. P. C. Coverdale for assistance with ICP-MS experiments, and I. Prokes for assistance with NMR spectroscopy.
Author information
Authors and Affiliations
Contributions
All authors were involved with the design and interpretation of the experiments, and with the writing of the manuscript. Chemical and biological experiments were carried out by H.H., S.B., K.Q. and P.Z. X-ray crystallography was carried out by S.B. and G.J.C. DFT calculations were carried by O.B., T.M. and M.J.P. M.S. and V.G.S. carried out the excited-state photochemistry experiments and analysed the data. H.H., S.B., H.C., G.G. and P.J.S analysed the data and co-wrote the paper. All authors discussed the results and commented on the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–9, Figs. 1–36 and references.
Complex-1.cif
Crystallographic data for compound 1 (CCDC reference 1840366).
Rights and permissions
About this article
Cite this article
Huang, H., Banerjee, S., Qiu, K. et al. Targeted photoredox catalysis in cancer cells. Nat. Chem. 11, 1041–1048 (2019). https://doi.org/10.1038/s41557-019-0328-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0328-4
This article is cited by
-
Metallopolymer strategy to explore hypoxic active narrow-bandgap photosensitizers for effective cancer photodynamic therapy
Nature Communications (2024)
-
Oxygen-independent organic photosensitizer with ultralow-power NIR photoexcitation for tumor-specific photodynamic therapy
Nature Communications (2024)
-
Metal complex catalysts broaden bioorthogonal reactions
Science China Chemistry (2024)
-
Supramolecular systems for bioapplications: recent research progress in China
Science China Chemistry (2024)
-
Targeted activation in localized protein environments via deep red photoredox catalysis
Nature Chemistry (2023)