Abstract
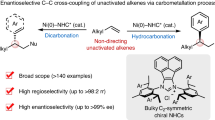
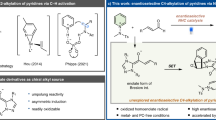
The enantioselective construction of carbon–heteroatom and carbon–carbon bonds that are alpha to ketones leads to the formation of substructures that are ubiquitous in natural products, pharmaceuticals and agrochemicals. Traditional methods to form such bonds have relied on combining ketone enolates with electrophiles. Reactions with heteroatom-based electrophiles require special reagents in which the heteroatom, which is typically nucleophilic, has been rendered electrophilic by changes to the oxidation state. The resulting products usually require post-synthetic transformations to unveil the functional group in the final desired products. Moreover, different catalytic systems are typically required for the reaction of different electrophiles. Here, we report a strategy for the formal enantioselective α-functionalization of ketones to form products containing a diverse array of substituents at the alpha position with a single catalyst. This strategy involves an unusual reversal of the role of the nucleophile and electrophile to form C–N, C–O, C–S and C–C bonds from a series of masked ketone electrophiles and a wide range of conventional heteroatom and carbon nucleophiles catalysed by a metallacyclic iridium catalyst.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All relevant data are available with the manuscript, in the Supplementary Information files.
References
Bellina, F. & Rossi, R. Transition metal-catalyzed direct arylation of substrates with activated sp 3 -hybridized C–H bonds and some of their synthetic equivalents with aryl halides and pseudohalides. Chem. Rev. 110, 1082–1146 (2010).
Cano, R., Zakarian, A. & McGlacken, G. P. Direct asymmetric alkylation of ketones: still unconquered. Angew. Chem. Int. Ed. 56, 9278–9290 (2017).
Janey, J. M. Recent advances in catalytic, enantioselective α aminations and α oxygenations of carbonyl compounds. Angew. Chem. Int. Ed. 44, 4292–4300 (2005).
Smith, A. M. R. & Hii, K. K. Transition metal catalyzed enantioselective α heterofunctionalization of carbonyl compounds. Chem. Rev. 111, 1637–1656 (2011).
Zhou, F., Liao, F.-M., Yu, J.-S. & Zhou, J. Catalytic asymmetric electrophilic amination reactions to form nitrogen-bearing tetrasubstituted carbon stereocenters. Synthesis 46, 2983–3003 (2014).
Maji, B. & Yamamoto, H. Use of in situ generated nitrosocarbonyl compounds in catalytic asymmetric α-hydroxylation and α-amination reactions. Bull. Chem. Soc. Jpn 88, 753–762 (2015).
Bøgevig, A., Sundén, H. & Córdova, A. Direct catalytic enantioselective α-aminoxylation of ketones: a stereoselective synthesis of α-hydroxy and α,α-dihydroxy ketones. Angew. Chem. Int. Ed. 43, 1109–1112 (2004).
Zhou, L. et al. Development of tartaric acid derived chiral guanidines and their application to catalytic enantioselective α-hydroxylation of β-dicarbonyl compounds. Org. Lett. 15, 3106–3109 (2013).
Yao, H., Lian, M., Li, Z., Wang, Y. & Meng, Q. Asymmetric direct α-hydroxylation of β-oxo esters catalyzed by chiral quaternary ammonium salts derived from cinchona alkaloids. J. Org. Chem. 77, 9601–9608 (2012).
Wang, Y. et al. A series of cinchona-derived N-oxide phase-transfer catalysts: application to the photo-organocatalytic enantioselective α-hydroxylation of β-dicarbonyl compounds. J. Org. Chem. 81, 7042–7050 (2016).
Yu, J.-S. et al. Catalytic enantioselective construction of sulfur-containing tetrasubstituted carbon stereocenters. ACS Catal. 6, 5319–5344 (2016).
Kumaragurubaran, N., Juhl, K., Zhuang, W., Bøgevig, A. & Jørgensen, K. A. Direct l-proline-catalyzed asymmetric α-amination of ketones. J. Am. Chem. Soc. 124, 6254–6255 (2002).
Yang, X. & Toste, F. D. Direct asymmetric amination of α-branched cyclic ketones catalyzed by a chiral phosphoric acid. J. Am. Chem. Soc. 137, 3205–3208 (2015).
Ohmatsu, K., Ando, Y., Nakashima, T. & Ooi, T. A modular strategy for the direct catalytic asymmetric α-amination of carbonyl compounds. Chemistry 1, 802–810 (2016).
Huang, X., Webster, R. D., Harms, K. & Meggers, E. Asymmetric catalysis with organic azides and diazo compounds initiated by photoinduced electron transfer. J. Am. Chem. Soc. 138, 12636–12642 (2016).
Shang, M. et al. Frustrated Lewis acid/Brønsted base catalysts for direct enantioselective α-amination of carbonyl compounds. J. Am. Chem. Soc. 139, 95–98 (2017).
Guo, F., Clift, M. D. & Thomson, R. J. Oxidative coupling of enolates, enol silanes, and enamines: methods and natural product synthesis. Eur. J. Org. Chem. 2012, 4881–4896 (2012).
Zhu, Y., Zhang, L. & Luo, S. Asymmetric α-photoalkylation of β-ketocarbonyls by primary amine catalysis: facile access to acyclic all-carbon quaternary stereocenters. J. Am. Chem. Soc. 136, 14642–14645 (2014).
Jang, H.-Y., Hong, J.-B. & MacMillan, D. W. C. Enantioselective organocatalytic singly occupied molecular orbital activation: the enantioselective α-enolation of aldehydes. J. Am. Chem. Soc. 129, 7004–7005 (2007).
Hartwig, J. F. & Stanley, L. M. Mechanistically driven development of iridium catalysts for asymmetric allylic substitution. Acc. Chem. Res. 43, 1461–1475 (2010).
Liu, W.-B., Xia, J.-B. & You, S.-L. Iridium-catalyzed asymmetric allylic substitutions. Top. Organomet. Chem. 38, 155–208 (2012).
Hethcox, J. C., Shockley, S. E. & Stoltz, B. M. Iridium-catalyzed diastereo-, enantio-, and regioselective allylic alkylation with prochiral enolates. ACS Catal. 6, 6207–6213 (2016).
Qu, J. & Helmchen, G. Applications of iridium-catalyzed asymmetric allylic substitution reactions in target-oriented synthesis. Acc. Chem. Res. 50, 2539–2555 (2017).
Murphy, K. E. & Hoveyda, A. H. Enantioselective synthesis of α-alkyl-β,γ-unsaturated esters through efficient Cu-catalyzed allylic alkylations. J. Am. Chem. Soc. 125, 4690–4691 (2003).
Lee, Y. & Hoveyda, A. H. Lewis base activation of Grignard reagents with N-heterocyclic carbenes, Cu-free catalytic enantioselective additions to γ-chloro-α,β-unsaturated esters. J. Am. Chem. Soc. 128, 15604–15605 (2006).
Gao, F., Lee, Y., Mandai, K. & Hoveyda, A. H. Quaternary carbon stereogenic centers through copper-catalyzed enantioselective allylic substitutions with readily accessible aryl- or heteroaryllithium reagents and aluminum chlorides. Angew. Chem., Int. Ed. 49, 8370–8374 (2010).
Den Hartog, T., Maciá, B., Minaard, A. & Feringa, B. L. Copper-catalyzed asymmetric allylic alkylation of halocrotonates: efficient synthesis of versatile chiral multifunctional building blocks. Adv. Synth. Catal. 352, 999–1013 (2010).
Ashfeld, B. L., Miller, K. A. & Martin, S. F. Direct, stereoselective substitution in [Rh(CO)2 Cl]2 -catalyzed allylic alkylations of unsymmetrical substrates. Org. Lett. 6, 1321–1324 (2004).
Ohmatsu, K., Ito, M., Kunieda, T. & Ooi, T. Exploiting the modularity of ion-paired chiral ligands for palladium-catalyzed enantioselective allylation of benzofuran-2(3H)-ones. J. Am. Chem. Soc. 135, 590–593 (2013).
Leitner, A., Shu, C. & Hartwig, J. F. Effects of catalyst activation and ligand steric properties on the enantioselective allylation of amines and phenoxides. Org. Lett. 7, 1093–1096 (2005).
Shu, C., Leitner, A. & Hartwig, J. F. Enantioselective allylation of aromatic amines after in situ generation of an activated cyclometalated iridium catalyst. Angew. Chem., Int. Ed. 43, 4797–4800 (2004).
Tosatti, P. et al. Iridium-catalyzed asymmetric allylic amination with polar amines: access to building blocks with lead-like molecular properties. Adv. Synth. Catal. 352, 3153–3157 (2010).
Trost, B. M. α-Sulfenylated carbonyl compounds in organic synthesis. Chem. Rev. 78, 363–382 (1978).
Hegedus, L. L. & McCabe, R. W. Catalyst Poisoning (Marcel Dekker, New York, 1984).
Manabe, Y., Kanematsu, M., Hiromasa, Y., Yoshida, M. & Shishido, K. Concise total syntheses of heliannuols B and D. Tetrahedron 70, 742–748 (2014).
Acknowledgements
Financial support for this work was provided by the National Institutes of Health (GM-55382). Z.-T.H. thanks a joint postdoc fellowship from Pharmaron and Shanghai Institute of Organic Chemistry (SIOC). We thank Sophie I. Arlow for assistance in the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Z.-T.H. and J.F.H. conceived and designed the project. Z.-T.H. performed the experiments and collected and analysed the data. J.F.H. directed the project. Z.-T.H. and J.F.H. co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Details of experiments for reaction development, experimental procedures, characterization data of all new molecules, spectra and references.
Rights and permissions
About this article
Cite this article
He, ZT., Hartwig, J.F. Enantioselective α-functionalizations of ketones via allylic substitution of silyl enol ethers. Nature Chem 11, 177–183 (2019). https://doi.org/10.1038/s41557-018-0165-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0165-x