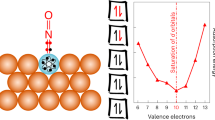
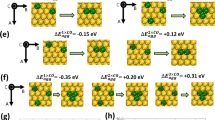
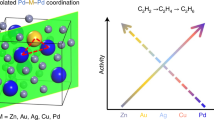
Abstract
Alloying provides a means by which to tune a metal catalyst’s electronic structure and thus tailor its performance; however, mean-field behaviour in metals imposes limits. To access unprecedented catalytic behaviour, materials must exhibit emergent properties that are not simply interpolations of the constituent components’ properties. Here we show an emergent electronic structure in single-atom alloys, whereby weak wavefunction mixing between minority and majority elements results in a free-atom-like electronic structure on the minority element. This unusual electronic structure alters the minority element’s adsorption properties such that the bonding with adsorbates resembles the bonding in molecular metal complexes. We demonstrate this phenomenon with AgCu alloys, dilute in Cu, where the Cu d states are nearly unperturbed from their free-atom state. In situ electron spectroscopy demonstrates that this unusual electronic structure persists in reaction conditions and exhibits a 0.1 eV smaller activation barrier than bulk Cu in methanol reforming. Theory predicts that several other dilute alloys exhibit this phenomenon, which offers a design approach that may lead to alloys with unprecedented catalytic properties.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Sabatier, P. Hydrogénations et déshydrogénations par catalyse. Ber. Deutsch. Chem. Gesellschaft 44, 1984–2001 (1911).
Medford, A. J. et al. From the Sabatier principle to a predictive theory of transition-metal heterogeneous catalysis. J. Catal. 328, 36–42 (2015).
Bligaard, T. & Nørskov, J. K. in Heterogeneous Catalysis Chemical Bonding at Surfaces and Interfaces (eds Nilsson, A., Pettersson, L. & Nørskov, J. K.) 255–321 (Elsevier, Amsterdam, 2008).
Hammer, B. & Nørskov, J. K. Electronic factors determining the reactivity of metal surfaces. Surf. Sci. 343, 211–220 (1995).
Abild-Pedersen, F. et al. Scaling properties of adsorption energies for hydrogen-containing molecules on transition-metal surfaces. Phys. Rev. Lett. 99, 016105 (2007).
Jacobsen, C. J. H. et al. Catalyst design by interpolation in the periodic table: bimetallic ammonia synthesis catalysts. J. Am. Chem. Soc. 123, 8404–8405 (2001).
Hansen, H. A., Varley, J. B., Peterson, A. A. & Nørskov, J. K. Understanding trends in the electrocatalytic activity of metals and enzymes for CO2 reduction to CO. J. Phys. Chem. Lett. 4, 388–392 (2013).
Vojvodic, A. & Nørskov, J. K. New design paradigm for heterogeneous catalysts. Natl Sci. Rev. 2, 140–143 (2015).
Man, I. C. et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. ChemCatChem 3, 1159–1165 (2011).
Studt, F. et al. Identification of non-precious metal alloy catalysts for selective hydrogenation of acetylene. Science 320, 1320–1322 (2008).
Kyriakou, G. et al. Isolated metal atom geometries as a strategy for selective heterogeneous hydrogenations. Science 335, 1209–1212 (2012).
Tierney, H. L., Baber, A. E., Kitchin, J. R. & Sykes, E. C. H. Hydrogen dissociation and spillover on individual isolated palladium atoms. Phys. Rev. Lett. 103, 246102 (2009).
Baber, A. E., Tierney, H. L., Lawton, T. J. & Sykes, E. C. H. An atomic-scale view of palladium alloys and their ability to dissociate molecular hydrogen. ChemCatChem 3, 607–614 (2011).
Rupprechter, G. Popping up to the surface. Nat. Chem. 9, 833–834 (2017).
Taccardi, N. et al. Gallium-rich Pd–Ga phases as supported liquid metal catalysts. Nat. Chem. 9, 862–867 (2017).
Liu, J. et al. Tackling CO poisoning with single-atom alloy catalysts. J. Am. Chem. Soc. 138, 6396–6399 (2016).
Thirumalai, H. & Kitchin, J. R. Investigating the reactivity of single atom alloys using density functional theory. Top. Catal. 61, 462–474 (2018).
Inderwildi, O. R., Jenkins, S. J. & King, D. A. When adding an unreactive metal enhances catalytic activity: NOx decomposition over silver–rhodium bimetallic surfaces. Surf. Sci. 601, L103–L108 (2007).
Greiner, M. T. et al. Phase coexistence of multiple copper oxides on AgCu catalysts during ethylene epoxidation. ACS Catal. 8, 2286–2295 (2018).
Abbati, I. et al. Solid-state effects on the valence-band 4d-photoionization cross sections at the Cooper minimum. Phys. Rev. Lett. 50, 1799–1802 (1983).
Subramanian, P. R. & Perepezko, J. H. The Ag–Cu (silver–copper) system. J. Phase Equilibria 14, 62–75 (1993).
Dweydari, A. W. & Mee, C. H. B. Work function measurements on (100) and (110) surfaces of silver. Phys. Status Solidi 27, 223–230 (1975).
Greiner, M. T., Chai, L., Helander, M. G., Tang, W. M. & Lu, Z. H. Transition metal oxide work functions: the influence of cation oxidation state and oxygen vacancies. Adv. Funct. Mater. 22, 4557–4568 (2012).
Pearson, R. G. Absolute electronegativity and hardness: application to inorganic chemistry. Inorg. Chem. 27, 734–740 (1988).
Weinert, M. & Watson, R. E. Core-level shifts in bulk alloys and surface adlayers. Phys. Rev. B 51, 17168–17180 (1995).
Olovsson, W., Göransson, C., Pourovskii, L. V., Johansson, B. & Abrikosov, I. A. Core-level shifts in fcc random alloys: a first-principles approach. Phys. Rev. B 72, 064203 (2005).
Bagus, P. S., Ilton, E. S. & Nelin, C. J. The interpretation of XPS spectra: insights into materials properties. Surf. Sci. Rep. 68, 273–304 (2013).
Ebert, H., Stöhr, J., Parkin, S. S. P., Samant, M. & Nilsson, A. L-edge X-ray absorption in fcc and bcc Cu metal: comparison of experimental and first-principles theoretical results. Phys. Rev. B 53, 16067–16073 (1996).
Anderson, P. W. Localized magnetic states in metals. Phys. Rev. 124, 41–53 (1961).
Newns, D. M. Self-consistent model of hydrogen chemisorption. Phys. Rev. 178, 1123–1135 (1969).
Vojvodic, A., Nørskov, J. K. & Abild-Pedersen, F. Electronic structure effects in transition metal surface chemistry. Top. Catal. 57, 25–32 (2013).
Hammer, B. & Nørskov, J. K. in Advances in Catalysis Vol. 45 (eds Gates, B. & Knoezinger, H.) 71–129 (Academic, Cambridge MA, 2000).
Grimley, T. B. The indirect interaction between atoms or molecules adsorbed on metals. Proc. Phys. Soc. 90, 751–764 (1967).
Haynes, W. M. CRC Handbook of Chemistry and Physics: a Ready-Reference Book of Chemical and Physical Data (CRC, Boca Raton, 2009).
Yong, S. T., Ooi, C. W., Chai, S. P. & Wu, X. S. Review of methanol reforming-Cu-based catalysts, surface reaction mechanisms, and reaction schemes. Int. J. Hydrogen Energy 38, 9541–9552 (2013).
Matthias, B. T. et al. Magnetic moment of transition metal atoms in dilute solution and their effect on superconducting transition temperature. Phys. Rev. Lett. 5, 542–544 (1960).
Giannozzi, P. et al. QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple [Phys. Rev. Lett. 77, 3865 (1996)]. Phys. Rev. Lett. 77, 3865–3868 (1996); erratum 78, 1396 (19987).
Jones, T. E. et al. Thermodynamic and spectroscopic properties of oxygen on silver under an oxygen atmosphere. Phys. Chem. Chem. Phys. 17, 9288–9312 (2015).
Greiner, M. T. et al. The oxidation of copper catalysts during ethylene epoxidation. Phys. Chem. Chem. Phys. 17, 25073–25089 (2015).
Marzari, N., Vanderbilt, D., De Vita, A. & Payne, M. C. Thermal contraction and disordering of the Al(110) surface. Phys. Rev. Lett. 82, 3296–3299 (1999).
Pehlke, E. & Scheffler, M. Evidence for site-sensitive screening of core holes at the Si and Ge (001) surface. Phys. Rev. Lett. 71, 2338–2341 (1993).
Shirley, E. L. Ti 1s pre-edge features in rutile: a Bethe–Salpeter calculation. J. Electron Spectrosc. 136, 77–83 (2004).
Soininen, J. A. & Shirley, E. L. Scheme to calculate core hole–electron interactions in solids. Phys. Rev. B 64, 165112 (2001).
Vinson, J. & Rehr, J. J. Ab initio Bethe–Salpeter calculations of the X-ray absorption spectra of transition metals at the L-shell edges. Phys. Rev. B 86, 195135 (2012).
Vinson, J., Rehr, J. J., Kas, J. J. & Shirley, E. L. Bethe–Salpeter equation calculations of core excitation spectra. Phys. Rev. B 83, 115106 (2011).
Fuchs, M. & Scheffler, M. Ab initio pseudopotentials for electronic structure calculations of poly-atomic systems using density-functional theory. Comput. Phys. Commun. 119, 67–98 (1999).
Perdew, J. P. & Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B 45, 13244–13249 (1992).
Methfessel, M. & Paxton, A. T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40, 3616–3621 (1989).
Krause, M. O. & Oliver, J. H. Natural widths of atomic K and L levels, Kα X‐ray lines and several KLL Auger lines. J. Phys. Chem. Ref. Data 8, 329–338 (1979).
Fonseca, G. C., Handgraaf, J.-W., Baerends, E. J. & Bickelhaupt, F. M. Voronoi deformation density (VDD) charges: assessment of the Mulliken, Bader, Hirshfeld, Weinhold, and VDD methods for charge analysis. J. Comput. Chem. 25, 189–210 (2004).
Löwdin, P. O. On the non‐orthogonality problem connected with the use of atomic wave functions in the theory of molecules and crystals. J. Chem. Phys. 18, 365–375 (1950).
Acknowledgements
Funding for this project was provided by the Alexander von Humboldt Foundation and the Max-Planck Society. We also thank the Helmholtz-Zentrum Berlin for their infrastructure support and Höchstleistungsrechenzentrum Stuttgart for access to the supercomputer HazelHen.
Author information
Authors and Affiliations
Contributions
M.T.G. and T.E.J. conceived, designed and planned the project. M.T.G. measured and analysed the photoemission spectra as well as activation energies. T.E.J. performed the theoretical calculations. R.S., S.P. and A.K.-G. provided direction, guidance and assistance in the data interpretation. S.B. assisted in photoemission measurements. L.Z. synthesized the samples used for the activity measurements. M.A. synthesized the bulk metal samples. M.S. and F.G. measured and analysed the X-ray diffraction data. M.T.G. and T.E.J. wrote the manuscript. All the authors provided input and advice while writing manuscript drafts. M.T.G. and T.E.J. contributed to this work equally.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–12, Supplementary Tables 1 and 2, Supplementary Data and Discussion
Rights and permissions
About this article
Cite this article
Greiner, M.T., Jones, T.E., Beeg, S. et al. Free-atom-like d states in single-atom alloy catalysts. Nature Chem 10, 1008–1015 (2018). https://doi.org/10.1038/s41557-018-0125-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0125-5