Abstract
Many cancers have an unusual dependence on glutamine. However, most previous studies have focused on the contribution of glutamine to metabolic building blocks and the energy supply. Here, we report that cancer cells with aberrant expression of glutamate decarboxylase 1 (GAD1) rewire glutamine metabolism for the synthesis of γ-aminobutyric acid (GABA)—a prominent neurotransmitter—in non-nervous tissues. An analysis of clinical samples reveals that increased GABA levels predict poor prognosis. Mechanistically, we identify a cancer-intrinsic pathway through which GABA activates the GABAB receptor to inhibit GSK-3β activity, leading to enhanced β-catenin signalling. This GABA-mediated β-catenin activation both stimulates tumour cell proliferation and suppresses CD8+ T cell intratumoural infiltration, such that targeting GAD1 or GABABR in mouse models overcomes resistance to anti-PD-1 immune checkpoint blockade therapy. Our findings uncover a signalling role for tumour-derived GABA beyond its classic function as a neurotransmitter that can be targeted pharmacologically to reverse immunosuppression.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
RNA-seq data supporting the findings of this study (Figs. 3d, 5e and 6e,f and Extended Data Fig. 5a) have been deposited at the Gene Expression Omnibus (GEO) under accession code GSE165889. The human tumour and adjacent data (Figs. 1e and 7g,h and Extended Data Fig. 1a) were derived from the TCGA Research Network (http://cancergenome.nih.gov/). The dataset derived from this resource supporting the findings of this study is available in Tumor IMmune Estimation Resource (TIMER) (https://cistrome.shinyapps.io/timer/). All other data supporting the findings of this study are available from the corresponding author on reasonable request. Source data are provided with this paper.
Code availability
Gene-expression heat maps, t-SNE analysis, enrichment pathway analysis and volcano plots were generated using R package. Accurate mass extraction from CE–MS for each precursor was performed using Skyline v.19.1.9.349 (https://www.skyline.ms).
References
Cluntun, A. A., Lukey, M. J., Cerione, R. A. & Locasale, J. W. Glutamine metabolism in cancer: understanding the heterogeneity. Trends Cancer 3, 169–180 (2017).
Hensley, C. T., Wasti, A. T. & DeBerardinis, R. J. Glutamine and cancer: cell biology, physiology, and clinical opportunities. J. Clin. Invest. 123, 3678–3684 (2013).
Altman, B. J., Stine, Z. E. & Dang, C. V. From Krebs to clinic: glutamine metabolism to cancer therapy. Nat. Rev. Cancer 16, 773–773 (2016).
Wise, D. R. & Thompson, C. B. Glutamine addiction: a new therapeutic target in cancer. Trends Biochem. Sci. 35, 427–433 (2010).
Erlander, M. G. & Tobin, A. J. The structural and functional heterogeneity of glutamic acid decarboxylase: a review. Neurochem. Res. 16, 215–226 (1991).
Watanabe, M., Maemura, K., Kanbara, K., Tamayama, T. & Hayasaki, H. GABA and GABA receptors in the central nervous system and other organs. Int. Rev. Cytol. 213, 1–47 (2002).
Wong, C. G., Bottiglieri, T. & Snead, O. C. III GABA, gamma-hydroxybutyric acid, and neurological disease. Ann. Neurol. 54, S3–S12 (2003).
Tillakaratne, N. J., Medina-Kauwe, L. & Gibson, K. M. γ-Aminobutyric acid (GABA) metabolism in mammalian neural and nonneural tissues. Comp. Biochem. Physiol. A Physiol. 112, 247–263 (1995).
Matuszek, M., Jesipowicz, M. & Kleinrok, Z. GABA content and GAD activity in gastric cancer. Med. Sci. Monit. 7, 377–381 (2001).
Mazurkiewicz, M., Opolski, A., Wietrzyk, J., Radzikowski, C. & Kleinrok, Z. GABA level and GAD activity in human and mouse normal and neoplastic mammary gland. J. Exp. Clin. Cancer Res. 18, 247–253 (1999).
Kleinrok, Z. et al. GABA content and GAD activity in colon tumors taken from patients with colon cancer or from xenografted human colon cancer cells growing as s.c. tumors in athymic nu/nu mice. J. Physiol. Pharm. 49, 303–310 (1998).
Young, S. Z. & Bordey, A. GABA’s control of stem and cancer cell proliferation in adult neural and peripheral niches. Physiology 24, 171–185 (2009).
Jiang, S. H. et al. GABRP regulates chemokine signalling, macrophage recruitment and tumour progression in pancreatic cancer through tuning KCNN4-mediated Ca2+ signalling in a GABA-independent manner. Gut 68, 1994–2006 (2019).
Gumireddy, K. et al. The mRNA-edited form of GABRA3 suppresses GABRA3-mediated Akt activation and breast cancer metastasis. Nat. Commun. 7, 10715 (2016).
Xia, S. et al. GABABR-Induced EGFR transactivation promotes migration of human prostate cancer cells. Mol. Pharmacol. 92, 265–277 (2017).
Li, B. et al. Comprehensive analyses of tumor immunity: implications for cancer immunotherapy. Genome Biol. 17, 174 (2016).
Li, T. et al. TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Cancer Res. 77, e108–e110 (2017).
Wilhelm, M. et al. Mass-spectrometry-based draft of the human proteome. Nature 509, 582–587 (2014).
Samaras, P. et al. ProteomicsDB: a multi-omics and multi-organism resource for life science research. Nucleic Acids Res. 48, D1153–D1163 (2020).
Kaupmann, K. et al. Expression cloning of GABA(B) receptors uncovers similarity to metabotropic glutamate receptors. Nature 386, 239–246 (1997).
Roth, F. C. & Draguhn, A. GABA metabolism and transport: effects on synaptic efficacy. Neural Plast. 2012, 805830 (2012).
Zhan, T., Rindtorff, N. & Boutros, M. Wnt signaling in cancer. Oncogene 36, 1461–1473 (2017).
Kolligs, F. T., Hu, G., Dang, C. V. & Fearon, E. R. Neoplastic transformation of RK3E by mutant beta-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression. Mol. Cell. Biol. 19, 5696–5706 (1999).
Lu, F. F., Su, P., Liu, F. & Daskalakis, Z. J. Activation of GABA(B) receptors inhibits protein kinase B/glycogen synthase kinase 3 signaling. Mol. Brain 5, 41 (2012).
Bjurstom, H. et al. GABA, a natural immunomodulator of T lymphocytes. J. Neuroimmunol. 205, 44–50 (2008).
Tian, J., Chau, C., Hales, T. G. & Kaufman, D. L. GABA(A) receptors mediate inhibition of T cell responses. J. Neuroimmunol. 96, 21–28 (1999).
Bhat, R. et al. Inhibitory role for GABA in autoimmune inflammation. Proc. Natl Acad. Sci. USA 107, 2580–2585 (2010).
Tian, J. et al. Gamma-aminobutyric acid inhibits T cell autoimmunity and the development of inflammatory responses in a mouse type 1 diabetes model. J. Immunol. 173, 5298–5304 (2004).
Roberts, E. W. et al. Critical Role for CD103+/CD141+ dendritic cells bearing CCR7 for tumor antigen trafficking and priming of T cell immunity in melanoma. Cancer Cell 30, 324–336 (2016).
Salmon, H. et al. Expansion and activation of CD103+ dendritic cell progenitors at the tumor site enhances tumor responses to therapeutic PD-L1 and BRAF Inhibition. Immunity 44, 924–938 (2016).
Hildner, K. et al. Batf3 deficiency reveals a critical role for CD8α+ dendritic cells in cytotoxic T cell immunity. Science 322, 1097–1100 (2008).
Dangaj, D. et al. Cooperation between constitutive and inducible chemokines enables T cell engraftment and immune attack in solid tumors. Cancer Cell 35, 885–900 (2019).
Williford, J. M. et al. Recruitment of CD103+ dendritic cells via tumor-targeted chemokine delivery enhances efficacy of checkpoint inhibitor immunotherapy. Sci. Adv. 5, eaay1357 (2019).
Ruiz de Galarreta, M. et al. β-Catenin activation promotes immune escape and resistance to anti-PD-1 therapy in hepatocellular carcinoma. Cancer Discov. 9, 1124–1141 (2019).
Spranger, S., Bao, R. & Gajewski, T. F. Melanoma-intrinsic beta-catenin signalling prevents anti-tumour immunity. Nature 523, 231–235 (2015).
Khuu, C. H., Barrozo, R. M., Hai, T. & Weinstein, S. L. Activating transcription factor 3 (ATF3) represses the expression of CCL4 in murine macrophages. Mol. Immunol. 44, 1598–1605 (2007).
Inoue, M. et al. The stress response gene ATF3 is a direct target of the Wnt/beta-catenin pathway and inhibits the invasion and migration of HCT116 human colorectal cancer cells. PLoS ONE13, e0194160 (2018).
Evenseth, L. S. M., Gabrielsen, M. & Sylte, I. The GABAB receptor-structure, ligand binding and drug development. Molecules 25, 3093 (2020).
George, K. & Sadiq, N. M. StatPearls (StatPearls, 2020).
Jiang, S. H., Hu, L. P., Wang, X., Li, J. & Zhang, Z. G. Neurotransmitters: emerging targets in cancer. Oncogene 39, 503–515 (2020).
Mendu, S. K., Bhandage, A., Jin, Z. & Birnir, B. Different subtypes of GABA-A receptors are expressed in human, mouse and rat T lymphocytes. PLoS ONE 7, e42959 (2012).
Jones, K. A. et al. Signal transduction by GABA(B) rehceptor heterodimers. Neuropsychopharmacology 23, S41–S49 (2000).
Akk, G., Shin, D. J., Germann, A. L. & Steinbach, J. H. GABA type A receptor activation in the allosteric coagonist model framework: relationship between EC50 and basal activity. Mol. Pharmacol. 93, 90–100 (2018).
Spranger, S., Dai, D., Horton, B. & Gajewski, T. F. Tumor-residing Batf3 dendritic cells are required for effector T cell trafficking and adoptive T cell therapy. Cancer Cell 31, 711–723 (2017).
Riaz, N. et al. Tumor and microenvironment evolution during immunotherapy with nivolumab. Cell 171, 934–949 (2017).
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Zhang, B. et al. B cell-derived GABA elicits IL-10+ macrophages to limit anti-tumour immunity. Nature 599, 471–476 (2021).
Lin, R. et al. Targeting miR-23a in CD8+ cytotoxic T lymphocytes prevents tumor-dependent immunosuppression. J. Clin. Invest. 124, 5352–5367 (2014).
Martz, C. A. et al. Systematic identification of signaling pathways with potential to confer anticancer drug resistance. Sci. Signal. 7, ra121 (2014).
Fuerer, C. & Nusse, R. Lentiviral vectors to probe and manipulate the Wnt signaling pathway. PLoS ONE 5, e9370 (2010).
Tan, J. et al. Capillary morphogenesis protein 2 is a novel prognostic biomarker and plays oncogenic roles in glioma. J. Pathol. 245, 160–171 (2018).
Acknowledgements
We thank the staff at the Duke University Flow Cytometry Shared Resource and the Duke University Proteomics and Metabolomics Shared Resource for assistance with flow cytometry and metabolic profiling, respectively; Z. Zhang (Duke University) and Y. Zhang (Whitehead Institute) for their comments on the preparation of this manuscript. This work was supported by R33‐CA‐225325 (to Q.-J.L.) and R01‐CA233205 (to Q.-J.L. and X.-F.W.) from the National Cancer Institute.
Author information
Authors and Affiliations
Contributions
D.H., Q.-J.L. and X.-F.W. conceived the study. D.H., P.B.A., Y. Wan., Q.-J.L. and X.-F.W. designed the experiments. Y. Wang and D.H. performed and analysed IHC staining. J.W.T. analysed CE–MS metabolic profiling. P.M. and H.W. analysed the RNA-seq data. D.H., T.Y. and D.Q. performed and analysed flow cytometry experiments. D.H., Y.L., L.T., C.P. and L.Y. performed the other experiments. D.H. wrote the original draft. P.B.A., Q.-J.L. and X.-F.W. reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Q.-J.L. is a scientific co-founder and shareholder of TCRCure Biopharma. The remaining authors declare no competing interests.
Peer review information
Nature Cell Biology thanks Lei Cao, Stefani Spranger and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 GABA content is not associated with ABAT expression in cancer patient samples.
a, GAD2 expression in tumors and adjacent normal tissues for indicated cancer types were analyzed by Tumor IMmune Estimation Resource (TIMER). Number of patients for each cancer type for the analysis is marked as ‘n’. b, Quantification of ABAT staining (classified as high, medium, or low ABAT expression by histoscores) of different clinical stages (I–III) specimens from NSCLC and COAD patients. c, Pearson correlation of ABAT expression with GABA levels in NSCLC and COAD patient samples. d, Kaplan-Meier curves with univariate analysis of 113 NSCLC and 89 COAD patient survival based on high versus low expression of ABAT. Number of patients for each cancer type is marked as ‘n’. Box plots represent a range from the 25th to 75th percentile, the median value and the minima to maxima (whiskers). P-values were calculated by two-sided Wilcoxon test (a), Chi-square test (b), and Log-rank test (d). The correlation coefficient (r) and P-values in (c) were obtained from two-tailed Pearson correlation analysis. *P < 0.05, **P < 0.01, ***P < 0.001; NS, not significant. Exact P values can be found in the Source Data.
Extended Data Fig. 2 Aberrant GAD1 expression in cancer cells rewires glutamine metabolism for GABA synthesis.
a, Protein expression analysis of ABAT, GAD1, and GAD2 in the indicated human tissues. Data were obtained from the human proteomic database ProteomicsDB. Sample number of each tissue is marked as ‘n’. b, GABA levels in cell lysates (bottom, left) and culture medium (bottom, right) of LLC-ova, LG1233, and MC38 cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC were measured by ELISA (n = 3 per group, biological replicates). Knockdown efficiencies of GAD1 in LLC-ova, LG1233, and MC38 cells were determined by western blot (top), which is representative of two independent replicates. c, Model depicting GABA synthesis in normal lung or colon and transformed NSCLC or COAD cells. β-actin served as loading control for western blot analysis. Data are presented as mean ± SD. P-values were calculated by two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001. Exact P values can be found in the Source Data.
Extended Data Fig. 3 Tumor cell-derived GABA induces β-catenin signaling to support proliferation.
a, Growth curves of H1650, HCC827, HCT116, SW480 and LLC-ova cells stably expressing GAD1/Gad1 shRNAs (sh1 and sh2) or NTC (n = 3 per group). b, Cell numbers were examined in LLC-ova (left) and MC38 (right) cells stably expressing Gad1 shRNAs or NTC in the absence or presence of 10 μM or 100 μM GABA for 3 days (n = 3 per group). c, HT29 cells stably expressing GAD1 shRNAs or NTC were subcutaneously injected into nude mice (n = 5 per group). Intratumoral injection of saline or saline containing GABA was initiated on day 13. d, Cellular ATP levels in H520, HT29, LLC-ova and MC38 cells stably expressing GAD1/Gad1 shRNAs (sh1 and sh2) or NTC were determined by ATP assay kit (n = 3 per group). e, Western blot analysis of β-catenin expression in NHBE cells obtained from three independent donors and the indicated NSCLC cell lines. f, Western blot analysis of β-catenin expression in the indicated normal human colon cell lines and COAD cell lines. g, Quantitative real-time PCR analysis of CTNNB1/Ctnnb1 expression in the indicated cancer cell lines stably expressing GAD1/Gad1 shRNAs (sh1 and sh2) or NTC (n = 3 per group). h, Western blot analysis of β-catenin and cyclin D1 expression in LLC-ova, LG1233, and MC38 cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC. i, Quantitative real-time PCR analysis of CCND1, VEGFA, CMYC, and LEF1 expression in H1650 cells stably expressing GAD1 shRNAs (sh1 and sh2) or NTC (n = 3 per group). j, Quantitative real-time PCR analysis of Atf3, Vegf, Lef1, and Ccnd1 expression in LLC-ova, LG1233, and MC38 cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC (n = 3 per group). k, Western blot analysis of GAD1, β-catenin, and cyclin D1 expression in LLC-ova (left) and MC38 (right) cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC, with subsequent GABA (50 μM) treatment for 48 hours. l, LLC-ova and MC38 cells stably expressing Gad1 shRNAs or NTC were further infected with virus expressing mutant β-catenin (∆GSK) or empty vector (n = 3 per group). Cell numbers were examined on day 3 after cells were seeded. β-actin served as loading control in western blot analysis. Western blot data shown in (e, f, h, k) are repeated independently at least two times with similar results. n indicates the number of biological (a-d, l) or technical (g, i, j) replicates. Data are presented as the mean ± SD. P-values were calculated by two-tailed Student’s t test (a, b, d, g, i, j, l) and two-way ANOVA (c). *P < 0.05, **P < 0.01, ***P < 0.001; NS, not significant. Exact P values can be found in the Source Data.
Extended Data Fig. 4 GABABR activation dampens GSK-3β activity to stabilize β-catenin.
a, Antagonists and agonists for GABAAR or GABABR used in this study. b, Western blot analysis of β-catenin expression in LLC-ova cells treated with bicuculline or 2-OH-saclofen at concentrations of 0, 10, 50, and 100 μM. c, d, Western blot analysis of β-catenin, p-GSK-3β (S9), and GSK-3β levels in HT29 (c) and LLC-ova (d) cells stably expressing GAD1/Gad1 shRNAs (sh1 and sh2) or NTC, followed by treatment with muscimol (50 μM) or baclofen (50 μM) for 48 hours. e, f, MC38 (e) or H520 (f) cells were subcutaneously injected into nude mice. Intratumoral injection of saline, 3-MPA or 2-OH-saclofen was initiated on day 15 (n = 5 per group, biological replicates). Tumor masses were weighed at the end of the experiment. g, Western blot analysis of p-GSK-3β (S9), GSK-3β, and β-catenin levels in HT29 cells stably expressing GAD1 shRNAs (sh1 and sh2) or NTC, followed by treatment with or without LiCl (20 mM) for 48 hours. β-actin served as loading control in western blot analysis. Western blot data shown in (b-d, g) are repeated independently at least two times with similar results. Data are presented as the mean ± SD (e, f left) and mean ± SEM (e, f right). P-values were calculated by two-way ANOVA (e, f left) and two-tailed Student’s t test (e, f right). *P < 0.05, **P < 0.01, ***P < 0.001; NS, not significant. Exact P values can be found in the Source Data.
Extended Data Fig. 5 Tumor cell-derived GABA excludes T cells from the tumor microenvironment.
a, Two-dimensional T-distributed stochastic neighbor embedding (t-SNE) analysis of RNA-seq data (n = 3 per group). b, Flow cytometry analysis of percentages of CD4+ T cells, CD8+ T cells, Treg, DC, CD103+ DC, and MDSC in subcutaneous LG1233 tumors as indicated conditions from C57BL/6 mice (n = 6 per group). c, Flow cytometry analysis of representative examples of CD4 and CD8 staining (gated live, TCRβ) in LLC-ova (top) and LG1233 (bottom) tumors stably expressing Gad1 shRNAs (sh1 and sh2) or NTC. d, Flow cytometry analysis of percentages of neutrophils, eosinophils, and macrophages in subcutaneous MC38 tumors as indicated conditions from C57BL/6 mice (n = 5 per group). e, MC38 cells stably expressing Gad1 shRNAs or NTC were subcutaneously injected into C57BL/6 mice (n = 5 per group). Intratumoral injection of saline or saline containing GABA was initiated on day 15. Average food consumption per mouse per day and mouse body weight on the indicated days were shown. n indicates the number of biological replicates. Data are presented as the mean ± SEM (b, d) and mean ± SD (e). P-values were calculated by two-tailed Student’s t test (b, d, e, left) and two-way ANOVA (e, right). *P < 0.05, **P < 0.01, ***P < 0.001; NS, not significant. Exact P values can be found in the Source Data.
Extended Data Fig. 6 GABA suppresses CCL4/5 production in tumor cells to create a non-T cell-inflamed microenvironment.
a, Lymphocytes were isolated from lymph nodes of OTI mice, and then treated for 3 days with peptide antigen and the indicated concentrations of GABA. Cells were then collected and analyzed for IFNγ and TNFα expression by flow cytometry (left, n = 3 per group). Representative examples of IFNγ and TNFα staining with or without 0.1 μM GABA (gated live, TCRβ, CD8; right). b, MC38 cells stably expressing Gad1 shRNAs or NTC were subcutaneously injected into C57BL/6 mice. Tumor-bearing mice were given anti-mouse CD8β antibody or IgG1 isotype control by intraperitoneal injection on days -1, 0, 7, 14 and 21 (n = 5 per group). c, Lymphocytes were isolated from lymph nodes of OTII or OTI mice, and then treated for 3 days with peptide antigen and the indicated concentrations of GABA. CFSE staining was used for analyzing proliferation of OTII CD4+ T cells and OTI CD8+ T cells. d, Quantitative real-time PCR analysis of CCL4 and CCL5 expression in H520 (left) and HT29 (right) cells stably expressing GAD1 shRNAs (sh1 and sh2) or NTC, as indicated (n = 3 per group). e, Western blot analysis of GAD1 and β-catenin expression in tumor samples of LLC-ova, LG1233, and MC38 tumors stably expressing Gad1 shRNAs (sh1 and sh2) or NTC, as indicated. f, LLC-ova cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC were further infected with virus expressing β-catenin (∆GSK) mutant or empty vector (pRRLSIN). Ccl4 and Ccl5 mRNA levels were analyzed by quantitative real-time PCR (n = 3 per group). g, Quantitative real-time PCR analysis of CCL4 and CCL5 expression in H520 cells stably expressing GAD1 shRNAs or NTC were further infected with virus expressing mutant β-catenin (S33Y) or EV (n = 3 per group). h, LLC-ova cells stably expressing Gad1 shRNAs (sh1 and sh2) or NTC were further infected with virus expressing β-catenin (∆GSK) mutant or empty vector (pRRLSIN). GAD1, β-catenin, cyclin D1, and ATF3 protein levels were analyzed by western blot. i, Quantitative real-time PCR analysis of Ccl4 and Ccl5 expression in MC38 cells expressing Ccl4 and Ccl5 shRNAs or NTC (n = 3 per group). β-actin served as loading control in western blot analysis. Western blot data shown in (e, h) are repeated independently at least two times with similar results. n indicates the number of biological (a, b) or technical (d, f, g, i) replicates. Data are presented as the mean ± SEM (a) or mean ± SD (b, d, f, g, i). P-values were calculated by two-tailed Student’s t test (a, d, f, g, i) and two-way ANOVA (b). *P < 0.05, **P < 0.01, ***P < 0.001. Exact P values can be found in the Source Data.
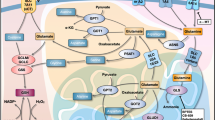
Extended Data Fig. 7 Graphical summary.
Unlike their corresponding normal tissues, NSCLC and COAD cancer cells with aberrant GAD1 expression hijack glutamine metabolism for GABA synthesis. Consequently, GABA accumulates within tumors. Rather than providing metabolic fuel or building blocks, GABA activates GABABR to enhance β-catenin signaling by repressing GSK-3β activity. On one hand, GABA-mediated β-catenin activation directly contributes to autonomous tumor growth; on the other hand, enhanced β-catenin signaling by cancer cells suppresses production of CCL4 and CCL5 production in tumor cells to create a non-T cell-inflamed microenvironment. Targeting GAD1 or GABABR in mouse models overcomes resistance to anti-PD-1 immune checkpoint blockade therapy.
Supplementary information
Supplementary Information
Supplementary Fig. 1.
Supplementary Table 1
RT–qPCR primer sequences.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 2
Unprocessed western blot.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed western blot.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 4
Unprocessed western blot.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 6
Unprocessed western blot.
Source Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed western blot.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 3
Unprocessed western blot.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed western blot.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed western blot.
Rights and permissions
About this article
Cite this article
Huang, D., Wang, Y., Thompson, J.W. et al. Cancer-cell-derived GABA promotes β-catenin-mediated tumour growth and immunosuppression. Nat Cell Biol 24, 230–241 (2022). https://doi.org/10.1038/s41556-021-00820-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-021-00820-9
This article is cited by
-
Modulation of alveolar macrophage and mitochondrial fitness by medicinal plant-derived nanovesicles to mitigate acute lung injury and viral pneumonia
Journal of Nanobiotechnology (2024)
-
The cancer-immune dialogue in the context of stress
Nature Reviews Immunology (2024)
-
Effects of dietary intervention on human diseases: molecular mechanisms and therapeutic potential
Signal Transduction and Targeted Therapy (2024)
-
WNT/β-catenin regulatory roles on PD-(L)1 and immunotherapy responses
Clinical and Experimental Medicine (2024)
-
Perineural invasion in colorectal cancer: mechanisms of action and clinical relevance
Cellular Oncology (2024)