Abstract
Heavy metals are both integral parts of cells and environmental toxicants, and their deregulation is associated with severe cellular dysfunction and various diseases. Here we show that the Hippo pathway plays a critical role in regulating heavy metal homeostasis. Hippo signalling deficiency promotes the transcription of heavy metal response genes and protects cells from heavy metal-induced toxicity, a process independent of its classic downstream effectors YAP and TAZ. Mechanistically, the Hippo pathway kinase LATS phosphorylates and inhibits MTF1, an essential transcription factor in the heavy metal response, resulting in the loss of heavy metal response gene transcription and cellular protection. Moreover, LATS activity is inhibited following heavy metal treatment, where accumulated zinc directly binds and inhibits LATS. Together, our study reveals an interplay between the Hippo pathway and heavy metals, providing insights into this growth-related pathway in tissue homeostasis and stress response.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The RNAseq dataset is available in the Gene Expression Omnibus under the accession number GSE163156. The lung squamous cell carcinoma RNAseq datasets were downloaded from The Cancer Genome Atlas data portal (https://portal.gdc.cancer.gov/). The protein structure information (PDB: 1ZW8) was downloaded from the Protein Data Bank (https://www.rcsb.org/structure/1zw8). All other data supporting the findings of this study are available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Gunther, V., Lindert, U. & Schaffner, W. The taste of heavy metals: gene regulation by MTF-1. Biochim. Biophys. Acta 1823, 1416–1425 (2012).
Baltaci, A. K., Yuce, K. & Mogulkoc, R. Zinc metabolism and metallothioneins. Biol. Trace Elem. Res. 183, 22–31 (2017).
Coyle, P., Philcox, J. C., Carey, L. C. & Rofe, A. M. Metallothionein: the multipurpose protein. Cell. Mol. Life Sci. 59, 627–647 (2002).
Nath, R., Kambadur, R., Gulati, S., Paliwal, V. K. & Sharma, M. Molecular aspects, physiological function, and clinical significance of metallothioneins. Crit. Rev. Food Sci. Nutr. 27, 41–85 (1988).
Hamer, D. H. Metallothionein. Annu. Rev. Biochem. 55, 913–951 (1986).
Robbins, A. H. et al. Refined crystal structure of Cd, Zn metallothionein at 2.0 Å resolution. J. Mol. Biol. 221, 1269–1293 (1991).
Dong, G., Chen, H., Qi, M., Dou, Y. & Wang, Q. Balance between metallothionein and metal response element binding transcription factor 1 is mediated by zinc ions (review). Mol. Med. Rep. 11, 1582–1586 (2015).
Davis, S. R. & Cousins, R. J. Metallothionein expression in animals: a physiological perspective on function. J. Nutr. 130, 1085–1088 (2000).
Stuart, G. W., Searle, P. F. & Palmiter, R. D. Identification of multiple metal regulatory elements in mouse metallothionein-I promoter by assaying synthetic sequences. Nature 317, 828–831 (1985).
Westin, G. & Schaffner, W. A zinc-responsive factor interacts with a metal-regulated enhancer element (MRE) of the mouse metallothionein-I gene. EMBO J. 7, 3763–3770 (1988).
Radtke, F. et al. Cloned transcription factor MTF-1 activates the mouse metallothionein I promoter. EMBO J. 12, 1355–1362 (1993).
Brugnera, E. et al. Cloning, chromosomal mapping and characterization of the human metal-regulatory transcription factor MTF-1. Nucleic Acids Res. 22, 3167–3173 (1994).
Lindert, U., Cramer, M., Meuli, M., Georgiev, O. & Schaffner, W. Metal-responsive transcription factor 1 (MTF-1) activity is regulated by a nonconventional nuclear localization signal and a metal-responsive transactivation domain. Mol. Cell. Biol. 29, 6283–6293 (2009).
Bittel, D., Dalton, T., Samson, S. L., Gedamu, L. & Andrews, G. K. The DNA binding activity of metal response element-binding transcription factor-1 is activated in vivo and in vitro by zinc, but not by other transition metals. J. Biol. Chem. 273, 7127–7133 (1998).
Egli, D. et al. Knockout of ‘metal-responsive transcription factor’ MTF-1 in Drosophila by homologous recombination reveals its central role in heavy metal homeostasis. EMBO J. 22, 100–108 (2003).
Gunes, C. et al. Embryonic lethality and liver degeneration in mice lacking the metal-responsive transcriptional activator MTF-1. EMBO J. 17, 2846–2854 (1998).
Wang, Y. et al. Metal-responsive transcription factor-1 (MTF-1) is essential for embryonic liver development and heavy metal detoxification in the adult liver. FASEB J. 18, 1071–1079 (2004).
Hua, H. et al. Toxicity of Alzheimer’s disease-associated Aβ peptide is ameliorated in a Drosophila model by tight control of zinc and copper availability. Biol. Chem. 392, 919–926 (2011).
Saini, N., Georgiev, O. & Schaffner, W. The parkin mutant phenotype in the fly is largely rescued by metal-responsive transcription factor (MTF-1). Mol. Cell. Biol. 31, 2151–2161 (2011).
Shi, Y. et al. The metal-responsive transcription factor-1 protein is elevated in human tumors. Cancer Biol. Ther. 9, 469–476 (2010).
Yu, F. X., Zhao, B. & Guan, K. L. Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell 163, 811–828 (2015).
Halder, G. & Johnson, R. L. Hippo signaling: growth control and beyond. Development 138, 9–22 (2011).
Pan, D. The Hippo signaling pathway in development and cancer. Dev. Cell 19, 491–505 (2010).
Yu, F. X. & Guan, K. L. The Hippo pathway: regulators and regulations. Genes Dev. 27, 355–371 (2013).
Piccolo, S., Dupont, S. & Cordenonsi, M. The biology of YAP/TAZ: Hippo signaling and beyond. Physiol. Rev. 94, 1287–1312 (2014).
Zanconato, F., Cordenonsi, M. & Piccolo, S. YAP/TAZ at the roots of Cancer. Cancer Cell 29, 783–803 (2016).
Zheng, Y. et al. Identification of Happyhour/MAP4K as alternative Hpo/Mst-like kinases in the Hippo kinase cascade. Dev. Cell 34, 642–655 (2015).
Li, S., Cho, Y. S., Yue, T., Ip, Y. T. & Jiang, J. Overlapping functions of the MAP4K family kinases Hppy and Msn in Hippo signaling. Cell Discov. 1, 15038 (2015).
Meng, Z. et al. MAP4K family kinases act in parallel to MST1/2 to activate LATS1/2 in the Hippo pathway. Nat. Commun. 6, 8357 (2015).
Li, Q. et al. The conserved Misshapen–Warts–Yorkie pathway acts in enteroblasts to regulate intestinal stem cells in Drosophila. Dev. Cell 31, 291–304 (2014).
Li, Q. et al. Ingestion of food particles regulates the mechanosensing Misshapen–Yorkie pathway in Drosophila intestinal growth. Dev. Cell 45, 433–449 (2018).
Boggiano, J. C., Vanderzalm, P. J. & Fehon, R. G. Tao-1 phosphorylates Hippo/MST kinases to regulate the Hippo–Salvador–Warts tumor suppressor pathway. Dev. Cell 21, 888–895 (2011).
Poon, C. L., Lin, J. I., Zhang, X. & Harvey, K. F. The sterile 20-like kinase Tao-1 controls tissue growth by regulating the Salvador–Warts–Hippo pathway. Dev. Cell 21, 896–906 (2011).
Plouffe, S. W. et al. Characterization of Hippo pathway components by gene inactivation. Mol. Cell 64, 993–1008 (2016).
Hamaratoglu, F. et al. The tumour-suppressor genes NF2/Merlin and Expanded act through Hippo signalling to regulate cell proliferation and apoptosis. Nat. Cell Biol. 8, 27–36 (2006).
Yin, F. et al. Spatial organization of Hippo signaling at the plasma membrane mediated by the tumor suppressor Merlin/NF2. Cell 154, 1342–1355 (2013).
Yu, F. X., Meng, Z., Plouffe, S. W. & Guan, K. L. Hippo pathway regulation of gastrointestinal tissues. Annu Rev. Physiol. 77, 201–227 (2015).
Wu, S., Liu, Y., Zheng, Y., Dong, J. & Pan, D. The TEAD/TEF family protein Scalloped mediates transcriptional output of the Hippo growth-regulatory pathway. Dev. Cell 14, 388–398 (2008).
Goulev, Y. et al. SCALLOPED interacts with YORKIE, the nuclear effector of the hippo tumor-suppressor pathway in Drosophila. Curr. Biol. 18, 435–441 (2008).
Zhang, L. et al. The TEAD/TEF family of transcription factor Scalloped mediates Hippo signaling in organ size control. Dev. Cell 14, 377–387 (2008).
Yu, F. X. et al. Regulation of the Hippo–YAP pathway by G-protein-coupled receptor signaling. Cell 150, 780–791 (2012).
Zhao, B. et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 21, 2747–2761 (2007).
Hao, Y., Chun, A., Cheung, K., Rashidi, B. & Yang, X. Tumor suppressor LATS1 is a negative regulator of oncogene YAP. J. Biol. Chem. 283, 5496–5509 (2008).
Wang, Z. et al. Solution structure of a Zap1 zinc-responsive domain provides insights into metalloregulatory transcriptional repression in Saccharomyces cerevisiae. J. Mol. Biol. 357, 1167–1183 (2006).
Li, Y., Kimura, T., Huyck, R. W., Laity, J. H. & Andrews, G. K. Zinc-induced formation of a coactivator complex containing the zinc-sensing transcription factor MTF-1, p300/CBP, and Sp1. Mol. Cell. Biol. 28, 4275–4284 (2008).
Datta, J. et al. Metallothionein expression is suppressed in primary human hepatocellular carcinomas and is mediated through inactivation of CCAAT/enhancer binding protein α by phosphatidylinositol 3-kinase signaling cascade. Cancer Res. 67, 2736–2746 (2007).
Fan, F. et al. Pharmacological targeting of kinases MST1 and MST2 augments tissue repair and regeneration. Sci. Transl. Med. 8, 352ra108 (2016).
Chen, X. et al. Copper sensing function of Drosophila metal-responsive transcription factor-1 is mediated by a tetranuclear Cu(I) cluster. Nucleic Acids Res. 36, 3128–3138 (2008).
Selvaraj, A. et al. Metal-responsive transcription factor (MTF-1) handles both extremes, copper load and copper starvation, by activating different genes. Genes Dev. 19, 891–896 (2005).
Han, H. et al. Regulation of the Hippo pathway by phosphatidic acid-mediated lipid–protein interaction. Mol. Cell 72, 328–340 e328 (2018).
Lv, X. B. et al. PARD3 induces TAZ activation and cell growth by promoting LATS1 and PP1 interaction. EMBO Rep. 16, 975–985 (2015).
Rahmat, M. B., Zhang, S. & Koh, C. G. POPX2 is a novel LATS phosphatase that regulates the Hippo pathway. Oncotarget 10, 1525–1538 (2019).
Rottenberg, S., Disler, C. & Perego, P. The rediscovery of platinum-based cancer therapy. Nat. Rev. Cancer 21, 37–50 (2021).
Chen, P. H. et al. Kinome screen of ferroptosis reveals a novel role of ATM in regulating iron metabolism. Cell Death Differ. 27, 1008–1022 (2020).
LaRochelle, O., Gagne, V., Charron, J., Soh, J. W. & Seguin, C. Phosphorylation is involved in the activation of metal-regulatory transcription factor 1 in response to metal ions. J. Biol. Chem. 276, 41879–41888 (2001).
Murphy, J. T. et al. Histidine protects against zinc and nickel toxicity in Caenorhabditis elegans. PLoS Genet. 7, e1002013 (2011).
Bafaro, E., Liu, Y., Xu, Y. & Dempski, R. E. The emerging role of zinc transporters in cellular homeostasis and cancer. Signal Transduct. Target. Ther. 2, 17029 (2017).
Yang, H. & Shu, Y. Cadmium transporters in the kidney and cadmium-induced nephrotoxicity. Int. J. Mol. Sci. 16, 1484–1494 (2015).
Gupta, A. & Lutsenko, S. Human copper transporters: mechanism, role in human diseases and therapeutic potential. Future Med. Chem. 1, 1125–1142 (2009).
Garrick, M. D. Human iron transporters. Genes Nutr. 6, 45–54 (2011).
Tsang, T., Davis, C. I. & Brady, D. C. Copper biology. Curr. Biol. 31, R421–R427 (2021).
Zhang, B. et al. Activity of metal-responsive transcription factor 1 by toxic heavy metals and H2O2 in vitro is modulated by metallothionein. Mol. Cell. Biol. 23, 8471–8485 (2003).
Pace, N. J. & Weerapana, E. Zinc-binding cysteines: diverse functions and structural motifs. Biomolecules 4, 419–434 (2014).
Srivastava, A. & Kumar, M. Prediction of zinc binding sites in proteins using sequence derived information. J. Biomol. Struct. Dyn. 36, 4413–4423 (2018).
Shu, N., Zhou, T. & Hovmoller, S. Prediction of zinc-binding sites in proteins from sequence. Bioinformatics 24, 775–782 (2008).
Beyersmann, D. & Haase, H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 14, 331–341 (2001).
Maret, W. Zinc in cellular regulation: the nature and significance of “Zinc Signals”. Int. J. Mol. Sci. 18, 2285 (2017).
Brady, D. C. et al. Copper is required for oncogenic BRAF signalling and tumorigenesis. Nature 509, 492–496 (2014).
Tsang, T. et al. Copper is an essential regulator of the autophagic kinases ULK1/2 to drive lung adenocarcinoma. Nat. Cell Biol. 22, 412–424 (2020).
Turski, M. L. et al. A novel role for copper in Ras/mitogen-activated protein kinase signaling. Mol. Cell. Biol. 32, 1284–1295 (2012).
Weinman, S. Calcium-binding proteins: an overview. J. Biol. Buccale 19, 90–98 (1991).
Bialasek, M. et al. Exploiting iron-binding proteins for drug delivery. J. Physiol. Pharmacol. 70, 675–685 (2019).
Karlsson, M. & Kurz, T. Attenuation of iron-binding proteins in ARPE-19 cells reduces their resistance to oxidative stress. Acta Ophthalmol. 94, 556–564 (2016).
Tristao, G. B. et al. Predicting copper-, iron-, and zinc-binding proteins in pathogenic species of the Paracoccidioides genus. Front. Microbiol. 5, 761 (2014).
Sanchez, M. et al. Iron regulatory protein-1 and -2: transcriptome-wide definition of binding mRNAs and shaping of the cellular proteome by iron regulatory proteins. Blood 118, e168-79 (2011).
Piovesan, D., Profiti, G., Martelli, P. L. & Casadio, R. The human “magnesome”: detecting magnesium binding sites on human proteins. BMC Bioinform. 13, S10 (2012).
Wang, W., Huang, J. & Chen, J. Angiomotin-like proteins associate with and negatively regulate YAP1. J. Biol. Chem. 286, 4364–4370 (2011).
Wang, W., Chen, L., Ding, Y., Jin, J. & Liao, K. Centrosome separation driven by actin-microfilaments during mitosis is mediated by centrosome-associated tyrosine-phosphorylated cortactin. J. Cell Sci. 121, 1334–1343 (2008).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Lee, T. I., Johnstone, S. E. & Young, R. A. Chromatin immunoprecipitation and microarray-based analysis of protein location. Nat. Protoc. 1, 729–748 (2006).
Wang, X. et al. The proteasome-interacting Ecm29 protein disassembles the 26S proteasome in response to oxidative stress. J. Biol. Chem. 292, 16310–16320 (2017).
Maier, J. A. et al. ff14SB: improving the accuracy of protein side chain and backbone parameters from ff99SB. J. Chem. Theory Comput. 11, 3696–3713 (2015).
Peters, M. B. et al. Structural survey of zinc containing proteins and the development of the zinc AMBER Force Field (ZAFF). J. Chem. Theory Comput. 6, 2935–2947 (2010).
Roe, D. R. & Cheatham, T. E.III PTRAJ and CPPTRAJ: software for processing and analysis of molecular dynamics trajectory data. J. Chem. Theory Comput. 9, 3084–3095 (2013).
Johnson, M. S., Sutcliffe, M. J. & Blundell, T. L. Molecular anatomy: phyletic relationships derived from three-dimensional structures of proteins. J. Mol. Evol. 30, 43–59 (1990).
Krissinel, E. & Henrick, K. Secondary-structure matching (SSM), a new tool for fast protein structure alignment in three dimensions. Acta Crystallogr. D 60, 2256–2268 (2004).
Acknowledgements
We thank K.-L. Guan (University of California, San Diego), D. Pan (The University of Texas Southwestern Medical Center), D. Fruman (University of California, Irvine) and A. Edinger (University of California, Irvine) for comments on this study. We thank J. W. C. Leung (University of Arkansas for Medical Sciences), R. Edwards (University of California, Irvine), G. MacGregor (University of California, Irvine) and R. Geertsema (University of California, Irvine) for their technical help. This work was supported by NIH grants (grant nos R01GM126048 and R21ES031642), an American Cancer Society Research Scholar grant (grant no. RSG-18-009-01-CCG), a University of California Cancer Research Coordinating Committee Research Award (grant no. CRN-19-585568), and a Tower Cancer Research Foundation Cancer Free Generation Career Development Grant to W.W.; and was also supported in part by NIH grants to R.L. (grant no. R35GM130367), K.W.Y.C. (grant no. R35GM139617) and L.H. (grant no. R01GM130144). W.W. is a member of the Chao Family Comprehensive Cancer Center (award no. P30CA062203) at the University of California, Irvine.
Author information
Authors and Affiliations
Contributions
H.H. and W.W. conceived and designed the study. W.W. supervised the study. H.H. and H.J.N. performed all of the experiments with the assistance from G.S., R.E.V. and B.Y. L.H. and R.W. performed the Drosophila survival study. C.Y. and L.H. performed the mass spectrometry analysis. L.B. helped with the in vitro kinase assay. J.N., L.Z., R.Q. and R.L. performed the simulation analysis. J.J.Z. and K.W.Y.C. helped with the RNAseq analysis. D.A.F. helped with the circular dichroism analysis. W.W. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review information
Nature Cell Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Inhibition of the Hippo pathway activates heavy metal response.
(a) Validation of the YAP-5SA overexpressed HEK239A cells. Western blot was performed using the indicated antibodies. (b) Validation of the YAP/TAZ shRNAs-transduced LATS1/2 DKO HEK293A cells. Western blot was performed using the indicated antibodies. (c,d) Western blot was performed using the indicated antibodies (c). MT1A gene transcription was examined in the indicated cells treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours by qPCR (mean ± s.d., n = 3 biological replicates) (d). *** p < 0.001 (two-tailed Student’s t-test). Relative expression fold change numbers are shown. (e,f) Transcription of MT1F (e) and CTGF (f) was examined in the HEK293A cells cultured under serum starvation and serum stimulation by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). (g,h) Transcription of MT1F (g) and CTGF (h) was examined in the HEK293A cells cultured under high density and low density by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). (i,j) Serum-starved wild-type and the LATS1/2 DKO HEK293A cells were treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. Transcription of MT1A (i) and MT1F (j) was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). Relative expression fold change numbers are shown. (k-l) Wild-type and the LATS1/2 DKO HEK293A cells were treated with 250 μM Zn with the indicated anions for 4 hours. The transcription of MT1A (i) and MT1F (j) was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). (m) Validation of the indicated HEK293A cells stably expressing SFB-MTF1. Western blot was performed using the indicated antibodies. Data shown represent 2 independent experiments in a-c, m.
Extended Data Fig. 2 LATS1 binds and phosphorylates MTF1.
(a) HEK293A and Hep3B cells stably expressing SFB-tagged MTF1 were subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. (b) HEK293A cells were transfected with the construct encoding SFB-tagged MTF1, treated with CdCl2 (50 µM) and ZnCl2 (250 µM) for 1 hour, and subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. (c) SFB-tagged LATS1 and its kinase dead (K734R) mutant were expressed in HEK293T cells, purified using S protein beads, and subjected to the ATP-γ-S-based in vitro kinase assay. Bacterially purified MBP-MTF1-R2, MBP-R2-5A mutant (S152A/T153A/T241A/S301A/T302A) and GST-YAP proteins were used as substrates. Western blot was performed using the indicated antibodies. (d) HEK293A cells stably expressing SFB-tagged MTF1 were transfected with the constructs encoding HA-tagged LATS1 and its kinase dead (K734R) mutant, and subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. Arrow indicates the exogenously expressed SFB-MTF1. Pound sign indicates endogenous MTF1. (e) SFB-tagged LATS1, STK38 and STK38L were expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay. Bacterially purified MBP-tagged MTF1-R2 protein was used as substrate. Western blot was performed using the indicated antibodies. Data shown represent 2 independent experiments in a-e.
Extended Data Fig. 3 MTF1 S152 phosphorylation inhibits heavy metal response.
(a,b) The indicated HEK293A cells were treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. The transcription of MT1A (a) and MT1F (b) was examined by qPCR (mean ± s.d., n = 3 biological replicates). * p < 0.05, ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). ns, no significance. Relative expression fold change numbers are shown. (c) Validation of the MTF1 KO HEK293A cells reconstituted with the indicated SFB-tagged MTF1 and its mutants. Western blot was performed using the indicated antibodies. Arrow indicates the exogenously expressed SFB-tagged MTF1 and its mutants. (d,e) The MTF1KO HEK293A cells were reconstituted with the indicated SFB-tagged MTF1 and its mutants, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 12 hours, visualized by crystal violet staining (d), and quantified for relative viability (mean ± s.d., n = 4 biological replicates) (e). *** p < 0.001 (two-tailed Student’s t-test). (f,g) The transcription of MT1A (f) and MT1F (g) was examined in wild-type and the MTF1 KO HEK293A cells reconstituted with the indicated SFB-tagged MTF1 and its mutants by qPCR (mean ± s.d., n = 3 biological replicates). Cells were treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. * p < 0.05, ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). ns, no significance. (h) The structure comparison of the ZAP1-ZNF1 in khaki with the phosphorylated ZAP1-ZNF1 (p-S591) in pink (left) and with ZAP1-ZNF1 S591D mutant in blue (right). The overall RMSD values of wild-type ZAP1-ZNF1 with its phosphorylated form (p-S591) and S591D mutant are shown. Arrows indicate the S591 site. (i) The MTF1 KO HEK293A cells were reconstituted with the indicated SFB-MTF1 and its mutants, and treated with ZnCl2 (250 μM) for 1 hour. Immunofluorescent staining was performed. Scale bar, 40 μm. Data shown represent 3 independent experiments. (j) The MTF1 KO HEK293A cells were reconstituted with the indicated SFB-MTF1 and its mutants, treated with ZnCl2 (250 μM) for 1 hour, and subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. Data shown represent 2 independent experiments in c, j.
Extended Data Fig. 4 Characterization of XMU-MP-1 in regulating the Hippo pathway and heavy metal response.
(a) Immunohistochemical analyses of Mtf1 and its S151 phosphorylation were performed in the liver and kidney tissues of the mice treated with DMSO, XMU-MP-1 alone or combined with VP post CdCl2 intoxication. The indicated regions in the box are shown 3 times enlarged. Scale bar, 150 μm. Data shown represent 3 independent experiments. (b) Wild-type and the MST1/2 DKO Hep3B and HEK293A cells were treated with DMSO and XMU-MP-1 (10 μM) for 4 hours, and subjected to Western blot analysis using the indicated antibodies. (c,d) The indicated Hep3B cells (c) and HEK293A cells (d) were pre-incubated with XMU-MP-1 (10 μM) for 2 hours, and treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. The transcription of MT1F was examined by qPCR (mean ± s.d., n = 3 biological replicates). * p < 0.05, ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). ns, no significance. (e-h) Hep3B and HEK293A cells stably expressing SFB-tagged TAOK1 (e), MAP4K2 (f), MAP4K4 (g) and MAP4K6 (h) were serum starved for 24 hours, and treated with XMU-MP-1 (10 μM) for 4 hours. The indicated SFB-tagged kinases were purified using S protein beads and subjected to in vitro kinase assay using the bacterially purified MBP-tagged LATS1-C3 protein as substrate. Western blot was performed using the indicated antibodies Data shown represent 2 independent experiments in b, e-h.
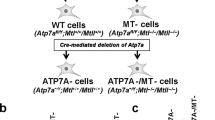
Extended Data Fig. 5 The Hippo pathway-mediated regulation of MTF1 and heavy metal response is conserved in Drosophila.
(a) Heterozygous mats Drosophila mutant and homozygous mtf1 Drosophila mutant were used for adult viability assay. Adult viability was assayed by feeding adult Drosophila (~7-day old) with normal food supplemented with 5 mM CuSO4 and determining the number of survivors daily. **, p < 0.01; ***, p < 0.001 (Log-rank test). n = 19 for wild-type Drosophila, n = 14 for mtf-/- Drosophila, n = 24 for mats−/+ Drosophila. (b) The MTF1 KO HEK293A cells were transfected with the constructs encoding the SFB-tagged human MTF1 and fly-mtf1. Western blot was performed using the indicated antibodies. (c) The MTF1 KO HEK293A cells were transfected with the constructs encoding the SFB-tagged human MTF1 and fly-mtf1, and treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. MT1A gene transcription was examined by qPCR (mean ± s.d., n = 3 biological replicates). *** p < 0.001 (two-tailed Student’s t-test). (d) HEK293A cells were transfected with the constructs encoding the SFB-tagged fly-mtf1 and its S126D mutant, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 1 hour, and subjected to immunofluorescent staining. Scale bar, 40 μm (e) The LATS/MTF1 TKO HEK293A cells were transduced with the constructs encoding the SFB-tagged fly-mtf1 and its S126D mutant. Western blot was performed using the indicated antibodies. (f) The LATS/MTF1 TKO HEK293A cells stably expressing the SFB-tagged fly-mtf1 and its S126D mutant were treated with CdCl2 (50 μM), ZnCl2 (250 μM) and CuSO4 (250 μM) for 4 hours. MT1F gene transcription was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). Data shown represent 2 independent experiments in b, d, e.
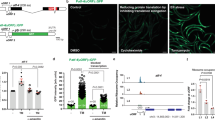
Extended Data Fig. 6 Heavy metals inhibit the Hippo pathway in different cells and tissues.
(a) Hep3B cells were serum starved for 24 hours, and treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for the indicated time points. Western blot was performed using the indicated antibodies. (b) MCF10A and HeLa cells were serum starved for 24 hours, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 1 hour, and subjected to immunofluorescent staining. Scale bar, 40 μm (c) MCF10A and HeLa cells were serum starved for 24 hours, and treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 4 hours. The transcription of YAP downstream genes CTGF and CYR61 was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). (d) Hep3B cells stably expressing SFB-tagged MTF1 were serum starved for 24 hours, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for the indicated time points, and subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. (e) Hep3B and HEK293A cells were serum starved for 24 hours, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 1 hour, and subjected to immunoprecipitation using LATS1 antibody. Western blot was performed using the indicated antibodies. Arrow indicates the correct p-LATS1 S909 signal. Asterisk indicates the non-specific signal. (f) The transcription of heavy metal response gene Mt1 was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). Relative expression fold change numbers are shown. (g) The transcription of Yap downstream gene Ctgf was examined by qPCR (mean ± s.d., n = 3 biological replicates). * p < 0.05, ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). Relative expression fold change numbers are shown. (h-j) The Human Protein Atlas (https://www.proteinatlas.org/) was used to examine the expression of MTF1 and LATS1 in different human tissues (h), human cells (i) and commonly used human cell lines (j). Data shown represent 2 independent experiments in a, b, d, e.
Extended Data Fig. 7 Zn binds and inhibits LATS.
(a) Illustration of the metal beads. (b) HEK293T cells were transfected with the constructs encoding the indicated Hippo pathway components and subjected to pulldown assay using Cu beads. The input blot is shared with the one in Fig. 8c. (c,d) HEK293T cells were transfected with the constructs encoding SFB-tagged LATS1 (c) and LATS2 (d), and subjected to pulldown assay using Zn beads in the presence of TPEN at the indicated concentrations. (e-f) SFB-tagged MST1 was expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay in the presence of CaCl2 (e) and ZnCl2 (f) at the indicated concentrations. MBP-tagged LATS1-C3 protein was used as substrate. (g) SFB-tagged MAP4K2 was expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay in the presence of ZnCl2 at the indicated concentrations. MBP-tagged LATS1-C3 protein was used as substrate. (h) SFB-tagged MST1 was expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay in the presence of ZnCl2 at the indicated concentrations. GST-tagged MOB1 protein was used as substrate. (i-j) HEK293A cells were transfected with the constructs encoding the SFB-tagged LATS1 with HA-tagged PPP1CA (i) and POPX2 (j), treated with ZnCl2 (250 μM) and CaCl2 (250 μM) for 1 hour, and subjected to pulldown assay using S protein beads. (k-l) SFB-tagged LATS1 was expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay in the presence of CaCl2 (k) or ZnCl2 (l) at the indicated concentrations. GST-tagged YAP protein was used as substrate. (m) SFB-tagged LATS2 was expressed in HEK293T cells, purified using S protein beads, and subjected to in vitro kinase assay in the presence of ZnCl2 at the indicated concentrations. GST-tagged YAP protein was used as substrate. (n) The potential Zn-binding Cys, His, Glu and Asp residues in the LATS1-C3 region are shown. The conserved Cys and His residues are highlighted in yellow. The conserved Glu and Asp residues are highlighted in blue. LATS1 T1079 site is indicated by arrow. Data shown represent 2 independent experiments in b-m.
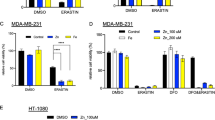
Extended Data Fig. 8 The Hippo pathway mediates cellular response to different heavy metals.
(a) HEK293A cells were transfected with the construct encoding SFB-tagged MOB1, treated with CdCl2 (50 μM) and ZnCl2 (250 μM) for 1 hour, and subjected to pulldown assay using S protein beads. Western blot was performed using the indicated antibodies. short, short exposure; long, long exposure. (b) HEK293T cells were transfected with the construct encoding SFB-tagged MOB1 and subjected to pulldown assay using the indicated metal beads. (c) Hep3B and HEK293A cells were serum starved for 24 hours and treated with CuSO4 (250 μM) and ZnCl2 (250 μM) for 4 hours. The transcription of MT1A, MT1F and CYR61 was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.01, *** p < 0.001 (two-tailed Student’s t-test). (d) The indicated cells were treated with CuSO4 (250 μM) and ZnCl2 (250 μM) for 4 hours. The transcription of MT1A was examined by qPCR (mean ± s.d., n = 3 biological replicates). *** p < 0.001 (two-tailed Student’s t-test). (e) SFB-tagged LATS1 was expressed in HEK293T cells, purified using S protein beads, washed thoroughly with high-salt buffer containing 250 mM NaCl, incubated with Zn chelator TPEN (25 μM) or Cu chelator TTM (25 μM), and subjected to in vitro kinase assay. GST-YAP protein used as substrate. (f) Illustration of cisplatin and carboplatin compound structures, where platinum is labelled in red. (g) The indicated cells were treated with cisplatin (10 μM) and carboplatin (100 μM) for 6 hours. The transcription of MT1A was examined by qPCR (mean ± s.d., n = 3 biological replicates). ** p < 0.05, *** p < 0.001 (two-tailed Student’s t-test). ns, no significance. Relative expression fold change numbers are shown. (h-k) The lung squamous cell carcinoma (LUSC) RNAseq datasets with clinic data were downloaded from the Cancer Genome Atlas (TCGA) data portal. The indicated gene expression from a total of 119 patients treated with cisplatin or carboplatin were subjected to Spearman correction analysis. Correlation coefficient R value and p value were calculated by GraphPad prism software. (l) A proposed model for the Hippo pathway-mediated heavy metal response through MTF1. Data shown represent 2 independent experiments in a, b, e.
Supplementary information
Supplementary Table 1
Supplementary Table 1. A total of 181 LATS1/2-regulated heavy metal response genes are listed based on clustered groups as shown in Fig. 2c. Supplementary Table 2. Sequence information of the qPCR primers used in this study.
Supplementary Video 1
The H2O control-treated mice are shown (upper left). Mice were pretreated with CdCl2 for 1 h and subjected to treatment with vehicle (upper right), XMU-MP-1 (lower left) or XMU-MP-1 + verteporfin (lower right). This video was taken 4 h post the CdCl2 intoxication.
Supplementary Video 2
The H2O control-treated mice are shown (upper left). Mice were pretreated with CdCl2 for 1 h and subjected to treatment with vehicle (upper right), XMU-MP-1 (lower left) and XMU-MP-1 + verteporfin (lower right). This video was taken 100 h post the CdCl2 intoxication.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Unprocessed western blots and gel.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 5
Unprocessed western blots.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 6
Unprocessed western blots.
Source Data Fig. 7
Statistical source data.
Source Data Fig. 7
Unprocessed western blots.
Source Data Fig. 8
Statistical source data.
Source Data Fig. 8
Unprocessed western blots and gel.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed western blots.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 3
Unprocessed western blots.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 5
Unprocessed western blots.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 7
Unprocessed western blots.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Han, H., Nakaoka, H.J., Hofmann, L. et al. The Hippo pathway kinases LATS1 and LATS2 attenuate cellular responses to heavy metals through phosphorylating MTF1. Nat Cell Biol 24, 74–87 (2022). https://doi.org/10.1038/s41556-021-00813-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-021-00813-8
This article is cited by
-
Angiotensin II type-2 receptor signaling facilitates liver injury repair and regeneration via inactivation of Hippo pathway
Acta Pharmacologica Sinica (2024)
-
LncRNA modulates Hippo-YAP signaling to reprogram iron metabolism
Nature Communications (2023)
-
Cuproptosis in lung cancer: mechanisms and therapeutic potential
Molecular and Cellular Biochemistry (2023)