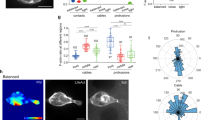
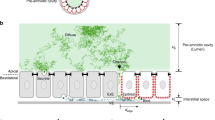
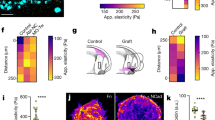
Abstract
Chemoattractant gradients frequently guide migrating cells. To achieve the most directional signal, such gradients should be maintained with concentrations around the dissociation constant (Kd)1,2,3,4,5,6 of the chemoreceptor. Whether this actually occurs in animals is unknown. Here we investigate whether a moving tissue, the zebrafish posterior lateral line primordium, buffers its attractant in this concentration range to achieve robust migration. We find that the Cxcl12 (also known as Sdf1) attractant gradient ranges from 0 to 12 nM, values similar to the 3.4 nM Kd of its receptor Cxcr4. When we increase the Kd of Cxcl12 for Cxcr4, primordium migration is less directional. Furthermore, a negative-feedback loop between Cxcl12 and its clearance receptor Ackr3 (also known as Cxcr7) regulates the Cxcl12 concentrations. Breaking this negative feedback by blocking the phosphorylation of the cytoplasmic tail of Ackr3 also results in less directional primordium migration. Thus, directed migration of the primordium is dependent on a close match between the Cxcl12 concentration and the Kd of Cxcl12 for Cxcr4, which is maintained by buffering of the chemokine levels. Quantitative modelling confirms the plausibility of this mechanism. We anticipate that buffering of attractant concentration is a general mechanism for ensuring robust cell migration.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ueda, M. & Shibata, T. Stochastic signal processing and transduction in chemotactic response of eukaryotic cells. Biophys. J. 93, 11–20 (2007).
Schwarz, J. et al. Dendritic cells interpret haptotactic chemokine gradients in a manner governed by signal-to-noise ratio and dependent on GRK6. Curr. Biol. 27, 1314–1325 (2017).
Rappel, W.-J. & Levine, H. Receptor noise limitations on chemotactic sensing. Proc. Natl Acad. Sci. USA 105, 19270–19275 (2008).
Fuller, D. et al. External and internal constraints on eukaryotic chemotaxis. Proc. Natl Acad. Sci. USA 107, 9656–9659 (2010).
Herzmark, P. et al. Bound attractant at the leading vs. the trailing edge determines chemotactic prowess. Proc. Natl Acad. Sci. USA 104, 13349–13354 (2007).
Van Haastert, P. J. M. & Postma, M. Biased random walk by stochastic fluctuations of chemoattractant–receptor interactions at the lower limit of detection. Biophys. J. 93, 1787–1796 (2007).
David, N. B. et al. Molecular basis of cell migration in the fish lateral line: role of the chemokine receptor CXCR4 and of its ligand, SDF1. Proc. Natl Acad. Sci. USA 99, 16297–16302 (2002).
Donà, E. et al. Directional tissue migration through a self-generated chemokine gradient. Nature 503, 285–289 (2013).
Venkiteswaran, G. et al. Generation and dynamics of an endogenous, self-generated signaling gradient across a migrating. Tissue Cell 155, 674–687 (2013).
Hanes, M. S. et al. Dual targeting of the chemokine receptors CXCR4 and ACKR3 with novel engineered chemokines. J. Biol. Chem. 290, 22385–22397 (2015).
Burns, J. M. et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J Exp. Med. 203, 2201–2213 (2006).
Ziarek, J. J. et al. Structural basis for chemokine recognition by a G protein-coupled receptor and implications for receptor activation. Sci. Signal. 10, eaah5756 (2017).
Boldajipour, B. et al. Control of chemokine-guided cell migration by ligand sequestration. Cell 132, 463–473 (2008).
Sánchez-Alcañiz, J. A. et al. Cxcr7 controls neuronal migration by regulating chemokine responsiveness. Neuron 69, 77–90 (2011).
Kennedy, J. E. & Marchese, A. et al. Regulation of GPCR Trafficking by Ubiquitin. Prog. Mol. Biol. Transl. Sci. 132, 15–38 (2015).
Moore, C. A. C., Milano, S. K. & Benovic, J. L. Regulation of receptor trafficking by GRKs and arrestins. Annu. Rev. Physiol. 69, 451–482 (2007).
Hoffmann, F. et al. Rapid uptake and degradation of CXCL12 depend on CXCR7 carboxyl-terminal serine/threonine residues. J. Biol. Chem. 287, 28362–28377 (2012).
Canals, M. et al. Ubiquitination of CXCR7 controls receptor trafficking. PLoS ONE 7, e34192 (2012).
Saaber, F. et al. ACKR3 regulation of neuronal migration requires ACKR3 phosphorylation, but not β-arrestin. Cell Rep. 26, 1473–1488 (2019).
Swaney, K. F., Huang, C.-H. & Devreotes, P. N. Eukaryotic chemotaxis: a network of signaling pathways controls motility, directional sensing, and polarity. Annu. Rev. Biophys. 39, 265–289 (2010).
Mantovani, A., Bonecchi, R. & Locati, M. Tuning inflammation and immunity by chemokine sequestration: decoys and more. Nat. Rev. Immunol 6, 907–918 (2006).
Korsensky, L. & Ron, D. Regulation of FGF signaling: recent insights from studying positive and negative modulators. Semin. Cell Dev. Biol. 53, 1–14 (2016).
Chung, A. S. & Ferrara, N. Developmental and pathological angiogenesis. Annu. Rev. Cell Dev. Biol. 27, 563–584 (2011).
Pocha, S. M. & Montell, D. J. Cellular and molecular mechanisms of single and collective cell migrations in Drosophila: themes and variations. Annu. Rev. Genet. 48, 295–318 (2014).
Friedl, P. & Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Biol. 10, 445–457 (2009).
Knaut, H., et al. A zebrafish homologue of the chemokine receptor Cxcr4 is a germ-cell guidance receptor. Nature 421, 279–282 (2003).
Bussmann, J., Wolfe, S. A. & Siekmann, A. F. Arterial–venous network formation during brain vascularization involves hemodynamic regulation of chemokine signaling. Development 138, 1717–1726 (2011).
Valentin, G., Haas, P. & Gilmour, D. The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr. Biol. 17, 1026–1031 (2007).
Kettleborough, R. N. W. et al. A systematic genome-wide analysis of zebrafish protein-coding gene function. Nature 496, 494–497 (2013).
Knaut, H., Blader, P., Strähle, U. & Schier, A. F. Assembly of trigeminal sensory ganglia by chemokine signaling. Neuron 47, 653–666 (2005).
Haas, P. & Gilmour, D. Chemokine signaling mediates self-organizing tissue migration in the zebrafish lateral line. Dev. Cell 10, 673–680 (2006).
Wang, J. et al. Anosmin1 shuttles Fgf to facilitate its diffusion, increase its local concentration, and induce sensory organs. Dev. Cell 46, 751–766 (2018).
Kozlovskaja-Gumbrienė, A. et al. Proliferation-independent regulation of organ size by Fgf/Notch signaling. eLife 6, e21049 (2017).
Fuentes, F., Reynolds, E., Lewellis, S. W., Venkiteswaran, G. & Knaut, H. A plasmid set for efficient bacterial artificial chromosome (BAC) transgenesis in zebrafish. G3 6, 829–834 (2016).
Yamaguchi, N., colak-Champollion, T. & Knaut, H. zGrad is a nanobody-based degron system that inactivates proteins in zebrafish. eLife 8, 4640 (2019).
colak-Champollion, T. et al. Cadherin-mediated cell coupling coordinates chemokine sensing across collectively migrating cells. Curr. Biol. 29, 2570–2579.e7 (2019).
Tsai, W.-H. Moment-preserving thresolding: a new approach. Comput. Gr. Image Process. 29, 377–393 (1985).
Lauffenburger, D. A. & Linderman, J. J. Receptors: Models for Binding, Trafficking, and Signaling (Oxford Univ. Press, 1993).
Thisse, C. & Thisse, B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat. Protoc. 3, 59–69 (2008).
Ho, J., Tumkaya, T., Aryal, S., Choi, H. & Claridge-Chang, A. Moving beyond P values: data analysis with estimation graphics. Nat. Methods 16, 1–4 (2019).
Acknowledgements
We thank T. Colak-Champollion, N. Yamaguchi, D. Nagelberg, J. Torres-Vazquez and his laboratory, R. Lehmann, C. Desplan, A. Mogilner and T. Prüstel for critical discussions, L. Lan for help with Matlab scripts, T. Gerson and J. Proietti for excellent fish care, and F. Fuentes, J. Ryou and A. Epifano for help with cloning. This work was supported by an American Heart Association fellowship (18PRE33960083 to S.L.) and the National Institutes of Health (R01NS102322 to H.K., F30HD079229 to J.W. and R01 AI058072 to B.F.V.) and, in part, by the intramural programme of NIH/NIAID (M.M.-S.)
Author information
Authors and Affiliations
Contributions
S.L., M.M.-S. and H.K. conceived the project. S.L. and H.K. designed the experiments and analysed the data. S.L. performed the experimental work with the following exceptions: J.W. and A.F. performed experiments involving the quantification of the Cxcl12a concentrations. S.W.L. and G.V. generated and characterized the Ackr3b–GFP expression in zebrafish. C.A.K., F.C.P. and B.F.V. purified the chemokine proteins. M.M.-S. performed the computational modelling. H.K. helped with the generation of the transgenic lines. S.L., M.M.-S. and H.K. wrote, reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
B.F.V., C.A.K. and F.C.P. have ownership interests in Protein Foundry. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Measurements of the dissociation constants of Cxcl12 binding to Cxcr4.
a, One-dimensional 1 H NMR of purified zebrafish Cxcl12a protein indicates protein folding since the amide region (6–10 ppm) of the spectrum contains peak dispersion, and upfield methyl peaks (peaks present at < 1 ppm) are present. b, Mass spectrometry analysis of purified zebrafish Cxcl12a protein yielded an experimental weight of 8,974.8 daltons. This agrees with the expected weight of 8,975.6 daltons. c, Ratio of membrane red-to-green fluorescence of the signaling sensor in T-REx 293 cells upon induction over time indicates stability of membrane red-to-green fluorescence ratios over time. Mean (dots) and SD (grey bars) are shown. n = 1240678, 1098868, 1076961, 3307144, 4260821 voxels analyzed for 12, 21, 24, 27, 36 h post induction, respectively. d, Human CXCL12α-human CXCR4 binding curve fitted to a one-site specific binding model. The mean (dots), SD (grey bars) and extracted Kd and Bmax (dotted cyan lines) are indicated. n = 406108, 385060, 416200, 369700, 296400, 338460 voxels analyzed for 0, 1.5, 5, 10, 50, 100 nM concentrations, respectively. e, Zebrafish Cxcl12a-human CXCR4 binding curve fitted to a one-site specific binding model. The mean (dots), SD (grey bars) and extracted Kd and Bmax (dotted cyan lines) are indicated. n = 248544, 308098, 350872, 324908, 282572, 259696 voxels analyzed for 0, 50, 100, 500, 1000, 2000 nM concentations, respectively. f, Human CXCL12α -zebrafish Cxcr4b binding curve fitted to a one-site specific binding model. The mean (dots), SD (grey bars) and extracted Kd and Bmax (dotted cyan lines) are indicated. n = 399632, 529120, 453188, 292848, 325436 voxels analyzed for 0, 20, 50, 100, 500 nM concentrations, respectively.
Extended Data Fig. 2 Low human Cxcr4 affinity to fish ligand affects cell migration.
a, Schematics of the cxcr4b BAC transgenes. b, Ratio of normalized mean Cxcr4b-Kate2 fluorescence intensity to normalized mean CXCR4-Kate2 fluorescence intensity shown in Fig. 2d fitted to the expected difference in receptor internalization for two receptors with Kds of 3.4 nM and 782 nM (black line). c, Membrane Cxcr4b-Kate2 and CXCR4-Kate2 fluorescence intensities with increasing levels of Cxcl12a normalized to heat-shocked control embryos over time. Individual means (circles), averaged means (horizontal lines), and fit of the data (t > 60 min) to a one-exponential decay model (lines) are indicated for Cxcr4b-Kate2 (black, n = 3 embryos) and CXCR4-Kate2 (magenta, n = 4 embryos). Scale bar is 20 μm. Note, this is a biologically independent experiment of Fig. 2d. d, Ratio of normalized mean Cxcr4b-Kate2 fluorescence intensity to normalized mean CXCR4-Kate2 fluorescence intensity shown in c fitted to the expected difference in receptor internalization for two receptors with Kds of 3.4 nM and 782 nM (black line). e, Semi-circular histogram plots of the directional angle frequencies of leader cells in cxcr4b:cxcr4b-Citrine; cxcr4b-/- or cxcr4b-/+ (left, n = 4244) and cxcr4b:CXCR4-Kate2; cxcr4b-/- primordia (right, n = 4085) from H2A-GFP labeled nuclei (Video 4). Radial axes are log10-scale. The difference in directional angle distributions is significantly different (p = 0.0001, two-sided Kolmogorov-Smirnov test). f, Directional indices of leader cells in cxcr4b:cxcr4b-Citrine; cxcr4b-/- or cxcr4b:cxcr4b-Citrine; cxcr4b-/+ (n = 205) and cxcr4b:CXCR4-Kate2; cxcr4b-/- primordia (n = 212) (Video 4). p = 1.8 × 10−10, two-sided Mann-Whitney test. g, Neighbor-neighbor distances for leader cells in cxcr4b:cxcr4b-Citrine; cxcr4b-/- or cxcr4b-/+ (n = 305) and cxcr4b:CXCR4-Kate2; cxcr4b-/- primordia (n = 324) (Video 4). p = 6.3 × 10−5, two-sided Mann-Whitney test. In f and g, the mean difference is shown as a Gardner-Altman estimation plot. Both groups are plotted on the left axes; the mean difference is plotted on the right axes as a bootstrap sampling distribution. Mean (horizontal line), mean difference (dot), the 95% confidence interval (vertical bars) are indicated. h, Speed of leader cells in cxcr4b:cxcr4b-Citrine; cxcr4b-/- or cxcr4b-/+ (n = 4159) and cxcr4b:CXCR4-Kate2; cxcr4b-/- primordia (n = 4317, Video 4). Mean (horizontal line) and SD (vertical bars) are indicated. The difference in speed is significantly different, p = 0.0001, two-sided Kolmogorov-Smirnov test. In c, e–h, n represents the number of tracked cells extracted from seven embryos for each genetic scenario.
Extended Data Fig. 3 Determination of the binding affinity of Cxcl12a for Cxcr4bI7E,I8E.
a, Comparison of the N-terminal extracellular sequence of human and zebrafish Cxcr4. Mutations known to lower the affinity to Cxcl12 are highlighted in red for the human CXCR4 and, based on sequence conservation, for the zebrafish Cxcr4b receptors. b, Top. Construct of the Cxcl12a-signaling sensor for the Cxcr4bI7E,I8E receptor expressed in T-REx 293 cells. Bottom. Zebrafish Cxcl12a- zebrafish Cxcr4bI7E,I8E binding curve fitted to a one-site specific binding model. The mean (dots), SD (grey bars) and extracted Kd and Bmax (dotted cyan lines) are indicated. n = 410452, 581584, 386060, 266532, 300640, 203284 voxels analyzed for 0, 5, 20, 50, 100, 200 nM concentrations, respectively.
Extended Data Fig. 4 Ackr3b BAC transgenes recapitulate Acrk3b function.
a, Schematics of the ackr3b BAC transgenes. Exons 1 and 2 of ackr3b and upstream and downstream genomic regions are indicated. The unmodified genomic locus is shown for reference (top). For the ackr3b:ackr3b-GFP transgene, GFP is inserted before the stop codon in exon 2 (middle). For the ackr3b transcriptional reporter, the coding sequence of ackr3b in exon 2 was replaced with the coding sequence for sfGFP (bottom). b, Analysis of completed primordium migration in ackr3b-/- (n = 9 embryos) and ackr3b-/-; ackr3b:ackr3b-GFP embryos (n = 29 embryos) using in situ hybridization against the primordium marker epcam at 48 hpf. The arrowhead indicates the position of the primordium. Scale bar corresponds to 200 μm. c, Total Cxcr4b-GFP (n = 2 embryos) and Ackr3b-sfGFP (n = 5 embryos) fluorescence intensities within the primordia with increasing levels of Cxcl12a over time. Note that decreasing Cxcr4b-GFP intensity reflects increasing Cxcl12a levels outside the primordium. Fluorescence intensities in primordia of individual embryos (circles) and averaged total fluorescence intensities (horizontal lines) are indicated. The initial rate of Cxcr4b-GFP intensity decrease is 2.8 × 10−2 AU/s and initial rate of Ackr3b-GFP intensity increase is 1.8 × 10−3 AU/s. The experiment was independently repeated once with similar results. d, Increasing Ackr3b-sfGFP expression plotted against decreasing Cxcr4b-GFP expression from 50 min to 200 min after induction of increasing Cxcl12a expression as shown in c. The initial rate of Ackr3b-sfGFP intensity increase per Cxcr4b-GFP intensity decrease is 16. Mean (dot) and SD (grey bars) are indicated. n as indicated in c represents the number of embryos.
Extended Data Fig. 5 Characterization of Ackr3b ST/A and K/A cytoplasmic tail mutants.
a, Amino acid sequence of the cytoplasmic tail of wild-type, ST/A and K/A mutant Ackr3b (in red). b, Design of ackr3b-sfGFP control and cytoplasmic tail mutant BAC transgenic lines. c, Quantification of migration distance of primordia in 48 hpf embryos of indicated genotypes. The mean difference for five comparisons against the shared ackr3b-/+ control embryos are shown as a Cumming estimation plot. The raw data is plotted on the left axis. On the right axis, the mean differences are plotted as bootstrap sampling distributions. Mean differences (dots) and 95% confidence interval (vertical bars) are indicated for ackr3b-/+ ( < x > = 1.00, n = 32), ackr3b-/- ( < x > = 0.35, n = 102, p = 1.7 × 10−17), ackr3b-/-; ackr3b:ackr3bwt tail-sfGFP ( < x > = 1.02, n = 15, p = 0.41), ackr3b-/-; ackr3b:ackr3bST/A tail-sfGFP ( < x > = 0.59, n = 19, p = 3.4 × 10−9), ackr3b-/-; ackr3b:ackr3bwt tail-sfGFP ( < x > = 1.03, n = 24, p = 0.03), and ackr3b-/-; ackr3b:ackr3bK/A tail-sfGFP ( < x > = 1.01, n = 34, p = 0.27), where < x > represents the mean, n represents the number of embryos, and p represents p-values (two-sided Mann-Whitney test). d,e, Left. Ackr3bwt tail-sfGFP and Ackr3bST/A tail-sfGFP (in d), and Ackr3bwt tail-sfGFP and Ackr3bK/A tail-sfGFP (in e) fluorescence intensities in the primordia are shown together with the primordium marker prim:lyn2mCherry (top panels) and separately as a heat map (bottom panels). The fluorescence intensities in all images are scaled identically. Right. Mean (dots) and SD (vertical bars) of Ackr3b-sfGFP fluorescence intensities along the front-back axis of primordia in wild-type and ST/A tail embryos (in d) and wild-type and K/A tail embryos (in e). n = 12 and 11 embryos for wt and ST/A tail, respectively and n = 5 embryos for wt and K/A tail. f, Response of Ackr3bwt tail-sfGFP and Ackr3bK/A tail-sfGFP fusion proteins in the primordium of embryos with increasing Cxcl12a levels. The control ackr3b:loxP-ackr3bwt tail-sfGFP-stop-loxP-ackr3bK/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry and the experimental ackr3b:ackr3bK/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry; hsp70:cxcl12a embryos were imaged at indicated times past a 30 min heat-shock that induced Cxcl12a expression from the heat shock promoter. The Ackr3b-sfGFP fluorescence intensities in the primordia are shown together with the primordium marker prim:lyn2mCherry (top panels) and separately as a heat-map (bottom panels). The fluorescence intensities in all images are scaled identically and quantified in g. g, Mean Ackr3bwt tail-sfGFP and Ackr3bK/A tail-sfGFP fluorescence intensities (dots) and SD (vertical bars) of heat-shocked control embryos (black, n = 4 and 3 embryos, respectively) and Cxcl12a-overexpressing embryos (blue, n = 3 and 5 embryos, respectively) along the front-back axis of primordia. In d–f, the scale bar corresponds to 20 μm. Anterior is to the left and the front of the primordium is to the right.
Supplementary information
41556_2020_465_MOESM2_ESM.avi
Supplementary Video 1 Time lapses of zebrafish and human Cxcr4 internalization upon Cxcl12a overexpression. Time lapses of cxcr4b:cxcr4b-Kate2; cldnB:lyn2-GFP and cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP control (left) and cxcr4b:cxcr4b-Kate2; cldnB:lyn2-GFP; hsp70:cxcl12a and cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP; hsp70:cxcl12a embryos (right) after a 10 min heat shock to induce Cxcl12a expression. Time stamp indicates minutes past the end of the heat shock. Scale bar = 50 μm. Time lapses start at 35 hpf. Each time frame is a sum projection of an individual Z-stack. Heatshock control cxcr4b:cxcr4b-Kate2; cldnB:lyn2-GFP embryos, n = 4. Heatshock control cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP embryos, n = 3. cxcr4b:cxcr4b-Kate2; cldnB:lyn2-GFP; hsp70:cxcl12a embryos, n = 2. cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP; hsp70:cxcl12a embryos, n = 3. n represents the number of embryos.
41556_2020_465_MOESM3_ESM.avi
Supplementary Video 2 Time lapses of migrating primordia expressing zebrafish Cxcr4b-Citrine and human CXCR4-Kate2. Time lapses of cxcr4b-/+; cldnB:lyn2-GFP (first and second from top), cxcr4b-/-; cldnB:lyn2-GFP (third from top), cxcr4b-/-; cxcr4b:cxcr4b-Citrine; cldnB:lyn2-GFP (fourth and fifth from top), and cxcr4b-/-; cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP embryos (bottom two). Scale bar = 50 μm, time stamp in min. Time lapses start at 36 hpf. Each time frame is a sum projection of an individual Z-stack. Note, Kate2 and Citrine channels are not shown. cxcr4b-/+; cldnB:lyn2-GFP embryos, n = 4. cxcr4b-/-; cldnB:lyn2-GFP embryos, n = 2. cxcr4b-/-; cxcr4b:cxcr4b-Citrine; cldnB:lyn2-GFP embryos, n = 2. cxcr4b-/-; cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP embryos, n = 4. n represents the number of embryos.
41556_2020_465_MOESM4_ESM.avi
Supplementary Video 3 Time lapses of front cells of migrating primordia in control embryos and cxcr4b mutant embryos expressing the human CXCR4 receptor. Time lapses of primordia in cxcr4b-/+; cldnB:lyn2-GFP (top) and cxcr4b-/-; cxcr4b:CXCR4-Kate2; cldnB:lyn2-GFP embryos (middle and bottom). Scale bar = 50 μm, time stamp in min. Time lapses start at 36 hpf. Each time frame is a maximum projection of an individual Z-stack. cxcr4b-/+ embryos, n = 1. cxcr4b:CXCR4-Kate2; cxcr4b-/- embryos, n = 2. n represents the number of embryos.
41556_2020_465_MOESM5_ESM.avi
Supplementary Video 4 Time lapses of nuclei-labeled cells in migrating primordia guided by the zebrafish Cxcr4b receptor and the human CXCR4 receptor. Time lapses of cells in primordia whose nuclei are labeled with H2A-GFP in cxcr4b:cxcr4b-Citrine; cxcr4b-/- or cxcr4b-/+ (top) and cxcr4b: CXCR4-Kate2; cxcr4b-/- embryos (bottom). Scale bar = 20μm, time stamp in min. Time lapses start at 33hpf. Each time frame is a maximum projection of an individual Z-stack. cxcr4b:cxcr4b-Citrine; cxcr4b:H2A-GFP; cxcr4b-/- or cxcr4b-/+, n = 7. cxcr4b:CXCR4-Kate2; cxcr4b:H2A-GFP; cxcr4b-/- embryos, n = 7. n represents the number of embryos.
41556_2020_465_MOESM6_ESM.avi
Supplementary Video 5 Time lapses of Ackr3b-GFP expression in primordia of control and Cxcl12a-overexpressing embryos. Time lapses of migrating primordia in control (left) and Cxcl12a-overexpressing ackr3b:ackr3b-GFP; prim:lyn2mCherry embryos (right). The second set of movies are identical to the first set but only show the GFP intensities of Ackr3b-sfGFP (false-coloured as a heat map). Scale bar = 20 μm, time stamp in min after the end of a 30-minute heat shock at 36 hpf. Each time frame is a sum projection of an individual Z-stack. Heat-shocked ackr3b:ackr3b-GFP; prim:lyn2mCherry embryos, n = 4. ackr3b:ackr3b-GFP; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 13. n represents the number of embryos.
41556_2020_465_MOESM7_ESM.avi
Supplementary Video 6 Time lapses of the ackr3b transcriptional reporter in control and Cxcl12a-overexpressing primordial. Time lapses of control (left) and Cxcl12a-overexpressing primordia (right) in ackr3b:sfGFP; prim:lyn2mCherry embryos. The second set of movies are identical to the first set but only show the sfGFP intensities from the ackr3b:sfGFP transcriptional reporter (false-colored as a heat map). Scale bar = 20 μm, time stamp in min. Each time frame is a sum projection of an individual Z-stack. The movies start 90 min after a 30 min heat shock. Heat-shocked ackr3b:sfGFP; prim:lyn2mCherry embryos, n = 3. ackr3b:sfGFP; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 3. n represents the number of embryos.
41556_2020_465_MOESM8_ESM.avi
Supplementary Video 7 Time lapses of Ackr3b-GFP expression in primordia of cxcr4 mutant control and cxcr4 mutant Cxcl12a-overexpressing embryos. Time lapses of control (left) and Cxcl12a-overexpressing primordium (right) in ackr3b:ackr3b-GFP; prim:lyn2mCherry; cxcr4a-/-; cxcr4b-/- embryos. The embryo shown on the left do not carry the hsp70:cxcl12a. The embryo shown on the right carry the hsp70:cxcl12a transgene. The second set of movies are identical to the first set but only show the GFP intensities of Ackr3b-GFP (false-colored as a heat map). Scale bar = 20 μm, time stamp in min. Each time frame is a sum projection of an individual Z-stack. The movies start at 90 min after a 30 min heat shock. Heat-shocked ackr3b:ackr3b-GFP; prim:lyn2mCherry; cxcr4a-/-; cxcr4b-/- embryos, n = 5. ackr3b:ackr3b-GFP; prim:lyn2mCherry; cxcr4a-/-; cxcr4b-/-; hsp70:cxcl12a embryos, n = 3. n represents the number of embryos.
41556_2020_465_MOESM9_ESM.avi
Supplementary Video 8 Time lapses of Ackr3bwt tail-sfGFP and Ackr3bK/A tail-sfGFP expression in primordia of control and Cxcl12a overexpressing embryos. Time lapses of control (top) and Cxcl12a-overexpressing embryos (bottom) in ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry (left) and ackr3b:ackr3bK/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos (right). The second set of movies is identical to the first set but only shows the sfGFP intensities of Ackr3bwt tail-sfGFP and Ackr3bK/A tail-sfGFP (false-colored as a heat map). Scale bar = 50 μm, time stamp in min after the end of a 30-minute heat shock at 36 hpf. Each time frame is a sum projection of an individual Z-stack. Heat-shocked control ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos, n = 4. Heat-shocked control ackr3b:ackr3bK/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos, n = 3. ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 3. ackr3b:ackr3bK/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 5. n represents the number of embryos.
41556_2020_465_MOESM10_ESM.avi
Supplementary Video 9 Time lapses of Ackr3bwt tail-sfGFP and Ackr3bST/A tail-sfGFP expression in primordia of control and Cxcl12a overexpressing embryos. Time lapses of control (top) and Cxcl12a-overexpressing embryos (bottom) in ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry (left) and ackr3b:ackr3bST/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos (right). The second set of movies are identical to the first set but only show the sfGFP intensities of Ackr3bwt tail-sfGFP and Ackr3bST/A tail-sfGFP (false-colored as a heat map). Scale bar = 50 μm, time stamp in min after the end of a 30-minute heat shock. Each time frame is a sum projection of an individual Z-stack. Heat-shocked control ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos, n = 4. Heat-shocked control ackr3b:ackr3bST/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry embryos, n = 4. ackr3b:ackr3bwt tail-sfGFP; ackr3b-/-; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 1. ackr3b:ackr3bST/A tail-sfGFP; ackr3b-/-; prim:lyn2mCherry; hsp70:cxcl12a embryos, n = 3. n represents the number of embryos.
41556_2020_465_MOESM11_ESM.avi
Supplementary Video 10 Time lapses of migrating primordia expressing Ackr3bwt tail-sfGFP and Ackr3bST/A tail sfGFP. Time lapses of primordia in ackr3b-/-; prim:lyn2mCherry (top two movies), ackr3b-/-; ackr3b:loxP-ackr3bwt tail-sfGFP-stop-loxP-ackr3bST/A tail-sfGFP; prim:lyn2mCherry (middle two movies) and ackr3b-/-; ackr3b:ackr3bST/A tail-sfGFP; prim:lyn2mCherry embryos (bottom two movies). Scale bar = 50 μm, time stamp in min. Each time frame is a max projection of an individual Z-stack. The movies start at 36 hpf for 15 hours. Note the GFP channel is not shown. ackr3b-/-; ackr3b:loxP-ackr3bwt tail-sfGFP-stop-loxP-ackr3bST/A tail-sfGFP; prim:lyn2mCherry embryos, n = 6. ackr3b-/-; ackr3b:loxP-ackr3bwt tail-sfGFP-stop-loxP-ackr3bST/A tail-sfGFP; prim:lyn2mCherry embryos, n = 4. ackr3b-/-; prim:lyn2mCherry embryos, n = 3. n represents the number of embryos.
41556_2020_465_MOESM13_ESM.py
Supplementary Data 2 Python script to calculate pooled mean, standard deviation, and number of values from text images from cultured cells
41556_2020_465_MOESM15_ESM.py
Supplementary Data 4 Python script to calculate pooled mean, standard deviation, and number of values from text images from primordium
41556_2020_465_MOESM18_ESM.zip
Supplementary Data 7 Matlab scripts to calculate the angles, directionality indices, and neighbor-neighbor distance from nuclei-tracked cell data exported from Imaris
41556_2020_465_MOESM22_ESM.xlsx
Supplementary Table 1 Table of the dissociation constants for the indicated Cxcl12-Cxcr4 pairs with 95% confidence intervals. Supplementary Table 2 Table of the dissociation constants for Cxcl12a-Cxcr4bI7E,I8E with 95% confidence interval. Supplementary Table 3 Table listing the measured Cxcl12a concentrations and Cxcr4b internalization along the front-back axis of the primordium and the calculated values from the model
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Source Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 1
Statistical Source Data
Source Data Extended Data Fig. 2
Statistical Source Data
Source Data Extended Data Fig. 3
Statistical Source Data
Source Data Extended Data Fig. 4
Statistical Source Data
Source Data Extended Data Fig. 5
Statistical Source Data
Rights and permissions
About this article
Cite this article
Lau, S., Feitzinger, A., Venkiteswaran, G. et al. A negative-feedback loop maintains optimal chemokine concentrations for directional cell migration. Nat Cell Biol 22, 266–273 (2020). https://doi.org/10.1038/s41556-020-0465-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-020-0465-4
This article is cited by
-
Atypical chemokine receptors in the immune system
Nature Reviews Immunology (2024)
-
Type 2 and Non-type 2 Inflammation in the Upper Airways: Cellular and Molecular Alterations in Olfactory Neuroepithelium Cell Populations
Current Allergy and Asthma Reports (2024)
-
Neuronal chemokine concentration gradients mediate effects of embryonic ethanol exposure on ectopic hypocretin/orexin neurons and behavior in zebrafish
Scientific Reports (2023)
-
The chemokines CXCL8 and CXCL12: molecular and functional properties, role in disease and efforts towards pharmacological intervention
Cellular & Molecular Immunology (2023)
-
Proteolytic inactivation of CXCL12 in the lungs and circulation of COVID-19 patients
Cellular and Molecular Life Sciences (2023)