Abstract
Loss of TGF-β tumour suppressive response is a hallmark of human cancers. As a central player in TGF-β signal transduction, SMAD4 (also known as DPC4) is frequently mutated or deleted in gastrointestinal and pancreatic cancer. However, such genetic alterations are rare in most cancer types and the underlying mechanism for TGF-β resistance is not understood. Here we describe a mechanism of TGF-β resistance in ALK-positive tumours, including lymphoma, lung cancer and neuroblastoma. We demonstrate that, in ALK-positive tumours, ALK directly phosphorylates SMAD4 at Tyr 95. Phosphorylated SMAD4 is unable to bind to DNA and fails to elicit TGF-β gene responses and tumour suppressing responses. Chemical or genetic interference of the oncogenic ALK restores TGF-β responses in ALK-positive tumour cells. These findings reveal that SMAD4 is tyrosine-phosphorylated by an oncogenic tyrosine kinase during tumorigenesis. This suggests a mechanism by which SMAD4 is inactivated in cancers and provides guidance for targeted therapies in ALK-positive cancers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
RNA-Seq data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) under the accession code GSE121188. Statistics source data for Figs. 3a–m,o, 4c–f, 6a–i and 7c,e–h and Supplementary Figs. 3a–c, 4b–d and 6b,c,e are provided in Supplementary Table 2. Unprocessed original scans of western blots are shown in Supplementary Fig. 8. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Change history
02 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41556-021-00638-5
References
Iwahara, T. et al. Molecular characterization of ALK, a receptor tyrosine kinase expressed specifically in the nervous system. Oncogene 14, 439–449 (1997).
Hallberg, B. & Palmer, R. H. Mechanistic insight into ALK receptor tyrosine kinase in human cancer biology. Nat. Rev. Cancer 13, 685–700 (2013).
Holla, V. R. et al. ALK: a tyrosine kinase target for cancer therapy. Cold Spring Harb. Mol. Case Stud. 3, a001115 (2017).
Katayama, R., Lovly, C. M. & Shaw, A. T. Therapeutic targeting of anaplastic lymphoma kinase in lung cancer: a paradigm for precision cancer medicine. Clin. Cancer Res. 21, 2227–2235 (2015).
Werner, M. T., Zhao, C., Zhang, Q. & Wasik, M. A. Nucleophosmin-anaplastic lymphoma kinase: the ultimate oncogene and therapeutic target. Blood 129, 823–831 (2017).
Morris, S. W. et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science 263, 1281–1284 (1994).
Soda, M. et al. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007).
Gridelli, C. et al. ALK inhibitors in the treatment of advanced NSCLC. Cancer Treat. Rev. 40, 300–306 (2014).
Mologni, L. Inhibitors of the anaplastic lymphoma kinase. Expert Opin. Investig. Drugs 21, 985–994 (2012).
Hida, T. et al. Alectinib versus crizotinib in patients with ALK-positive non-small-cell lung cancer (J-ALEX): an open-label, randomised phase 3 trial. Lancet 390, 29–39 (2017).
Peters, S. et al. Alectinib versus crizotinib in untreated ALK-positive non-small-cell lung cancer. N. Engl. J. Med. 377, 829–838 (2017).
Recondo, G., Facchinetti, F., Olaussen, K. A., Besse, B. & Friboulet, L. Making the first move in EGFR-driven or ALK-driven NSCLC: first-generation or next-generation TKI? Nat. Rev. Clin. Oncol. 15, 694–708 (2018).
Rotow, J. & Bivona, T. G. Understanding and targeting resistance mechanisms in NSCLC. Nat. Rev. Cancer 17, 637–658 (2017).
Shirinian, M., Varshney, G., Loren, C. E., Grabbe, C. & Palmer, R. H. Drosophila Anaplastic Lymphoma Kinase regulates Dpp signalling in the developing embryonic gut. Differentiation 75, 418–426 (2007).
Loren, C. E. et al. A crucial role for the Anaplastic lymphoma kinase receptor tyrosine kinase in gut development in Drosophila melanogaster. EMBO Rep. 4, 781–786 (2003).
Reiner, D. J., Ailion, M., Thomas, J. H. & Meyer, B. J. C. elegans anaplastic lymphoma kinase ortholog SCD-2 controls dauer formation by modulating TGF-beta signaling. Curr. Biol. 18, 1101–1109 (2008).
Derynck, R. & Miyazono, K. The TGF-β Family (Cold Spring Harbor Lab Press, New York, 2008).
Morikawa, M., Derynck, R. & Miyazono, K. TGF-beta and the TGF-beta family: context-dependent roles in cell and tissue physiology. Cold Spring Harb. Perspect. Biol. 8, a021873 (2016).
David, C. J. & Massague, J. Contextual determinants of TGFbeta action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 19, 419–435 (2018).
Moses, H. L., Roberts, A. B. & Derynck, R. The discovery and early days of TGF-beta: a historical perspective. Cold Spring Harb. Perspect. Biol. 8, a021865 (2016).
Massague, J. TGFbeta signalling in context. Nat. Rev. Mol. Cell Biol. 13, 616–630 (2012).
Massague, J. TGFbeta signalling in context. Cancer Cell. 134, 215–230 (2008).
Katsuno, Y., Lamouille, S. & Derynck, R. TGF-beta signaling and epithelial–mesenchymal transition in cancer progression. Curr. Opin. Oncol. 25, 76–84 (2013).
Nickel, J., Ten Dijke, P. & Mueller, T. D. TGF-beta family co-receptor function and signaling. Acta Biochim. Biophys. Sin. (Shanghai) 50, 12–36 (2018).
Feng, X. H. & Derynck, R. Specificity and versatility in TGF-beta signaling through Smads. Annu. Rev. Cell Dev. Bio. 21, 659–693 (2005).
Zhao, M., Mishra, L. & Deng, C. X. The role of TGF-beta/SMAD4 signaling in cancer. Int. J. Biol. Sci. 14, 111–123 (2018).
Wrighton, K. H. & Feng, X. H. To (TGF)beta or not to (TGF)beta: Fine-tuning of Smad signaling via post-translational modifications. Cell. Signal. 20, 1579–1591 (2008).
Xu, P., Lin, X. & Feng, X. H. Posttranslational Regulation of Smads. Cold Spring Harb. Perspect. Biol. 8, a022087 (2016).
Liang, M. et al. Ubiquitination and proteolysis of cancer-derived Smad4 mutants by SCFSkp2. Mol. Cell. Biol. 24, 7524–7537 (2004).
George, R. E. et al. Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature 455, 975–978 (2008).
Ambrogio, C. et al. p130Cas mediates the transforming properties of the anaplastic lymphoma kinase. Blood 106, 3907–3916 (2005).
Zawel, L. et al. Human Smad3 and Smad4 are sequence-specific transcription activators. Mol. Cell 1, 611–617 (1998).
Dennler, S. et al. Direct binding of Smad3 and Smad4 to critical TGF beta-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Keeton, M. R., Curriden, S. A., van Zonneveld, A. J. & Loskutoff, D. J. Identification of regulatory sequences in the type 1 plasminogen activator inhibitor gene responsive to transforming growth factor beta. J. Biol. Chem. 266, 23048–23052 (1991).
Zhou, S., Zawel, L., Lengauer, C., Kinzler, K. W. & Vogelstein, B. Characterization of human FAST-1, a TGF beta and activin signal transducer. Mol. Cell 2, 121–127 (1998).
Kaneki, H. et al. Tumor necrosis factor promotes Runx2 degradation through up-regulation of Smurf1 and Smurf2 in osteoblasts. J. Biol. Chem. 281, 4326–4333 (2006).
Feng, X. H., Zhang, Y., Wu, R. Y. & Derynck, R. The tumor suppressor Smad4/DPC4 and transcriptional adaptor CBP/p300 are coactivators for smad3 in TGF-beta-induced transcriptional activation. Genes Dev. 12, 2153–2163 (1998).
Moustakas, A. & Kardassis, D. Regulation of the human p21/WAF1/Cip1 promoter in hepatic cells by functional interactions between Sp1 and Smad family members. Proc. Natl Acad. Sci. USA 95, 6733–6738 (1998).
Scandura, J. M., Boccuni, P., Massague, J. & Nimer, S. D. Transforming growth factor beta-induced cell cycle arrest of human hematopoietic cells requires p57KIP2 up-regulation. Proc. Natl Acad. Sci. USA 101, 15231–15236 (2004).
Lawler, S. et al. The type II transforming growth factor-beta receptor autophosphorylates not only on serine and threonine but also on tyrosine residues. J. Biol. Chem. 272, 14850–14859 (1997).
Ma, L. & Chung, W. K. The genetic basis of pulmonary arterial hypertension. Hum. Genet. 133, 471–479 (2014).
Le Goff, C. & Cormier-Daire, V. From tall to short: the role of TGFbeta signaling in growth and its disorders. Am. J. Med. Genet. C 160C, 145–153 (2012).
Dupuis-Girod, S., Bailly, S. & Plauchu, H. Hereditary hemorrhagic telangiectasia: from molecular biology to patient care. J. Thromb. Haemost. 8, 1447–1456 (2010).
Moog-Lutz, C. et al. Activation and inhibition of anaplastic lymphoma kinase receptor tyrosine kinase by monoclonal antibodies and absence of agonist activity of pleiotrophin. J. Biol. Chem. 280, 26039–26048 (2005).
Feng, X. H. & Derynck, R. Ligand-independent activation of transforming growth factor (TGF) beta signaling pathways by heteromeric cytoplasmic domains of TGF-beta receptors. J. Biol. Chem. 271, 13123–13129 (1996).
Lin, X. et al. Activation of transforming growth factor-beta signaling by SUMO-1 modification of tumor suppressor Smad4/DPC4. J. Biol. Chem. 278, 18714–18719 (2003).
Piva, R. et al. Ablation of oncogenic ALK is a viable therapeutic approach for anaplastic large-cell lymphomas. Blood 107, 689–697 (2006).
Lin, X. et al. PPM1A functions as a Smad phosphatase to terminate TGFbeta signaling. Cell 125, 915–928 (2006).
Feng, Y. et al. Zinc finger protein 451 is a novel Smad corepressor in transforming growth factor-beta signaling. J. Biol. Chem. 289, 2072–2083 (2014).
Liu, T. et al. Tumor suppressor bromodomain-containing protein 7 cooperates with Smads to promote transforming growth factor-beta responses. Oncogene 36, 362–372 (2017).
Wang, G. et al. STAT3 selectively interacts with Smad3 to antagonize TGF-beta signalling. Oncogene 35, 4388–4398 (2016).
Maddalo, D. et al. In vivo engineering of oncogenic chromosomal rearrangements with the CRISPR/Cas9 system. Nature 516, 423–427 (2014).
Boukamp, P. et al. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell Biol. 106, 761–771 (1988).
Yu, Y. et al. Smad7 enables STAT3 activation and promotes pluripotency independent of TGF-beta signaling. Proc. Natl Acad. Sci. USA 114, 10113–10118 (2017).
Zhao, Y., Liu, J., Chen, F. & Feng, X. H. C-terminal domain small phosphatase-like 2 promotes epithelial-to-mesenchymal transition via Snail dephosphorylation and stabilization. Open Biol. 8, 170274 (2018).
Acknowledgements
We thank R. Chiarle for NPM–ALK and kinase-dead NPM–ALK(K210R), N. Fusenig for the HaCaT cell line, D. Luskutoff for p800(PAI-1)–Luc, Y. P. Mossé for the NB-1643 cell line, M. Vigny for the full-length ALK complementary DNA, B. Vogelstein for WWP1(p21)–Luc and SBE–Luc and X.-F. Wang for p15–Luc. We are indebted to L. Ding and Y. Ma at Betta Pharmaceutical for sharing the ALK inhibitor X396. We thank colleagues at Beijing Proteome Research Center and National Center for Protein Sciences (The PHOENIX Center, Beijing) for assistance with mass spectrometry analysis. We are grateful to K. Yu for sharing reagents, and J. Peng, Y. Zhu, N. Xu, H. Xia and members of the Feng laboratory for helpful discussion and technical assistance. We also thank K. Wrighton for editing the manuscript. We are grateful to L. Su for her contribution. This research was partly supported by grants from the NSFC (31730057, 91540205, 31090360, 31571447), the NIH (R21CA209007) and the DoD (DAMD W81XWH-15–1–0650/0651), and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
Q.Z. contributed most of the experimental data, data analysis and participated manuscript writing. X.L. performed the initial antibody array experiment. M.X., S.G., Y.X., L.Q., Y.Y., H.L. and X.L. performed some biochemical/cellular experiments and analysed the respective data. F.C., Y.W., H.J., Z.C., S.M., P.X., B.Z., Y. Zu, Y.L. and T. Liang provided essential experimental materials. H.W., C.D. and J.Q. performed the mass spectrometry experiment and data analyses. J.J. performed the statistical analyses of ALK–SMAD4 correlation in human lymphoma. M.X., Y. Zhu and L.S. conducted the RNA-Seq data analysis. T. Liu and X.L. participated in experimental design and data analyses. X.-H.F. designed and directed the project, analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 ALK interacts with SMAD4 in vitro and in vivo.
(a) ALK interacts with SMAD4 under physiological conditions. Exponentially growing SUDHL-1 cells were treated with TGF-β and/or TAE684 for 3 h before harvest. Cell lysates were subject to IP-Western analysis using appropriate antibodies. The experiment was repeated three times independently with similar results. (b) SMAD4 directly binds to the kinase domain of ALK in vitro. GST-SMAD4 proteins were purified from E. coli BL21 (DE3) strain and mixed with in vitro synthesized FLAG-tagged NPM-ALK, NPM or ALK deletion mutants (as depicted in Figure 1e), respectively. GST-bound proteins and input were resolved by SDS-PAGE and detected by Western blotting. GST protein is used as a negative control. The experiment was repeated three times independently with similar results. (c) ALK directly binds to the MH1 domain of SMAD4. Purified GST-ALK-Δ2 proteins were mixed with in vitro synthesized HA-tagged SMAD4 deletion mutants respectively. GST-bound proteins and input were resolved by SDS-PAGE and detected by Western blotting. The experiment was repeated three times independently with similar results. Unprocessed blots are shown in Supplementary Fig. 8.
Supplementary Figure 2 ALK phosphorylates SMAD4 on tyrosine-95.
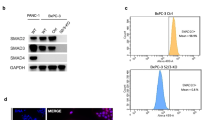
(a) Oncogenic ALK variants phosphorylate SMAD4. HEK293T cells were transfected with expression plasmids encoding FLAG-SMAD4 and ALK(WT), ALK(F1174L), NPM-ALK or EML4-ALK. Anti-FLAG IP-Western blotting analysis was carried out as described in Figure 2a. PY95 antibody was used to detect Y95 phosphorylation of SMAD4. The experiment was repeated three times independently with similar results. (b) Phosphorylation of GST-SMAD4 by immunopurified NPM-ALK. Recombinant NPM-ALK proteins immunopurified from transfected HEK293T cells were incubated with recombinant GST-SMAD4 purified from E. coli. In an in vitro kinase assay, the level of SMAD4 tyrosine phosphorylation was examined by Western blotting with PY100 antibody. The experiment was repeated three times independently with similar results. (c) NPM-ALK phosphorylates SMAD4 in the MH1 domain. NPM-ALK and FLAG-SMAD4 mutants were transfected into HEK293T cells. IP-Western blotting was carried out as described in Figure 2a. Domains of SMAD4 are described in Figure 1g. The experiment was repeated three times independently with similar results. (d) NPM-ALK phosphorylates SMAD4 in the MH1 domain. NPM-ALK and HA-SMAD4 truncations were transfected into HEK293T cells. The experiment was repeated three times independently with similar results. (e) NPM-ALK fails to phosphorylate the MH1 domain with Y95F substitution. HEK293T cells were transfected with NPM-ALK and FLAG-SMAD4MH1 (WT or Y95F). The experiment was repeated three times independently with similar results. (f) Depletion of NPM-ALK abolishes tyrosine phosphorylation of SMAD4 in Karpas 299 cells. Western blotting was done using appropriate antibodies. The experiment was repeated three times independently with similar results. Unprocessed blots are shown in Supplementary Fig. 8.
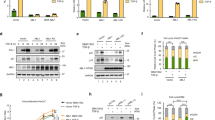
Supplementary Figure 3 ALK functionally inactivates SMAD4-dependent TGF-β signalling.
(a) NPM-ALK inhibits the TGF-β-induced transcriptional responses using reporter 3TP-Luc. HaCaT cells were transfected with expression plasmids for NPM-ALK or its kinase-dead K210R mutant, lucifersae reporter plasmid and Renilla luciferase. Relative luciferase activity was measured 18 h after TGF-β treatment (2 ng/ml). Values are normalized for transfection efficiency against Renilla luciferase activities. n = 3 independent experiments; Mean ± s.e.m. (b) NPM-ALK inhibits TGF-β-induced CAGA-Luc reporter response in HeLa cells. Assays were carried out as in panel a. n = 3 independent experiments; Mean ± s.e.m. (c) NPM-ALK inhibits TGF-β-induced SBE-Luc response in B lymphocyte BL41 cells. Assays were carried out as in panel a. n = 3 independent experiments; Mean ± s.e.m. (d) ALK(F1174L) blocks TGF-β-induced Caspase 3 activation. NMuMG cells (4x105) stably expressing ALK(F1174L) are treated with or without 10 ng/ml TGF-β for 24 h and then subjected to Western blotting. β-actin is a loading control. The experiment was repeated three times independently with similar results. (e) siRNA-mediated knockdown efficacy on NPM-ALK expression was measured in Karpas 299 cells. GAPDH is a loading control. The experiment was repeated three times independently with similar results. Unprocessed blots are shown in Supplementary Fig. 8. Statistical source data for panels a-c are provided in Supplementary Table 2.
Supplementary Figure 4 ALK inhibitor TAE-684 sensitizes ALK-positive tumour cells to TGF-β-induced target gene expression.
(a) SMAD4 knockout in Karpas 299 cells was demonstrated by anti-SMAD4 Western blotting. Clone 5 and 20 are two individual clones that were also confirmed by DNA sequencing. The experiment was repeated three times independently with similar results. (b) TAE-684 restores TGF-β-dependent CDKN1A transcription in H2228 cells. n = 3 independent experiments; Mean ± s.e.m. (c) TAE-684 restores TGF-β-dependent SERPINE1 transcription in Karpas 299 cells. n = 3 independent experiments; Mean ± s.e.m. (d) TAE-684 restores TGF-β-dependent SERPINE1 transcription in NB-1643 cells. n = 3 independent experiments; Mean ± s.e.m. Unprocessed blots are shown in Supplementary Fig. 8. Statistical source data for panels b-d are provided in Supplementary Table 2.
Supplementary Figure 5 ALK blocks the DNA-binding activity of SMAD4, but not its nuclear accumulation or SMAD complex assembly.
(a) Depletion of NPM-ALK has no effect on TGF-β-induced SMAD2/3 phosphorylation. H2228 cells were transfected with siRNA against ALK, treated with TGF-β (2 ng/ml, 1 h). Cell lysates were harvested for Western blotting analysis. The experiment was repeated three times independently with similar results. (b, c) Mutation at Y95 of SMAD4 has no effect on its interaction with SMAD2 (b) or SMAD3 (c). HEK293T cells were transfected with HA-SMAD2/SMAD3, FLAG-SMAD4 or a Y95 mutant (Y95E or Y95F), together with NPM-ALK (WT or K210R) and treated with TGF-β (2 ng/ml, 1 h). IP-Western blotting analysis was carried out. WT, wild-type; KR, K210R substitution; YE, Y95E substitution; YF, Y95F substitution. The experiments were repeated three times independently with similar results. (d, e) NPM-ALK represses the DNA binding of endogenous SMAD4. HaCaT-NPM-ALK(WT or K210R mutant) cells were treated with Dox and/or TGF-β (2 ng/ml, 1 h). DNA pull-down was done as described in Figure 5g. (d) Protein levels of SMAD4 and Dox-induced NPM-ALK (WT or K210R mutant) in whole cell lysates. (e) Protein levels of SMAD4 in DNA pull-down fractions. Biotin-SBEm, a biotin-labeled mutated SBE. The experiment was repeated three times independently with similar results. (f) SMAD4(Y95E) exerts a stronger interaction with p300 than wild-type SMAD4. The experiment was repeated three times independently with similar results. Unprocessed blots are shown in Supplementary Fig. 8.
Supplementary Figure 6 Y95 phosphorylation of SMAD4 inhibits its tumour suppressor activity.
(a-c) SMAD4(Y95E) fails to suppress tumour growth, while SMAD4(Y95F) exhibits the most potent tumour suppressor activity. SUDHL cells stably expressing GFP, SMAD4, SMAD4(Y95E) or SMAD4(Y95F) were injected subcutaneously into ancillary lymph nodes of nude mice to determine their tumorigenic activity. SUDHL-1 cells expressing SMAD4(Y95F) generate significantly smaller tumours than other groups as evaluated by tumour size (a), and weight (b). Panel c shows the time (days) of tumour appearance post injection. (b,c) n = 5 mice; Mean ± s.e.m.***, P < 0.001, **, P < 0.01, N.S., P > 0.05, by two-sided Student’s t test. The experiments were repeated three times independently with similar results. The data shown was obtained from one single experiment. (d, e) ALK inhibitor suppresses EAp9 lung tumour growth. EAp9 cells derived from EML4-ALK-induced mouse lung tumours were used to induce tumours in nude mice as described in panel a and treated with ALK inhibitor X396 at a dosage of 50 mg/kg every 2 days. Tumours were evaluated by tumour size (d) and weight (e). (e) n = 10 mice; Mean ± s.e.m. ***, P < 0.001, by two-sided Student’s t test. The experiment was repeated three times independently with similar results. The data shown was obtained from one single experiment. (f) ALK inhibitor blocks SMAD4 phosphorylation and enhances SMAD4 responses in EAp9-induced tumours. Tumour tissues from Panel d were subjected to Western blotting analysis. Phosphorylation levels of ALK and SMAD4 are indicated at top blots. The expression levels of PAI-1, p15Ink4B, p21Cip1 and MYC (SMAD4 target proteins), H3pS10 (proliferation index) and cleaved Caspase 3 (apoptosis index) are shown. The experiment was repeated three times independently with similar results. Unprocessed blots are shown in Supplementary Fig. 8. Statistical source data for panels b, c and e are provided in Supplementary Table 2.
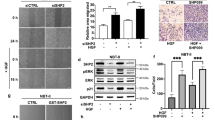
Supplementary Figure 7 SMAD4 Y95 phosphorylation is correlated with ALK positivity in lymphoma.
(a) SMAD4 tyrosine phosphorylation at Y95 is correlated with the presence of NPM-ALK in human lymphoma. IHC staining of SMAD4, p-SMAD4(PY95) and ALK, as well as H&E staining are shown in one ALK-positive representative and one ALK-negative representative ALCL tumour sections. Pictures were taken under microscope in 10x, 20x, 40x, and 100x magnification, respectively. Scale Bars, 25 μm. Sections from 41 human patients were analyzed. One section for each patient was assessed and analyzed under microscope for five fields. (b) Y95 Phosphorylation is absent in Karpas 299(SMAD4-KO) tumours. To test the specificity of the PY95 antibody, IHC staining of SMAD4, PY95 and ALK was carried out in tumours derived from nude mice injected with Karpas 299 cells (2 samples) and Karpas 299(SMAD4-KO) (2 samples). Pictures were taken under microscope in 4x, 10x, 20x, and 40x magnification, respectively. Scale Bars, 50 μm. The experiment was repeated three times independently with similar results.
Supplementary Figure 8 Unprocessed original scans of blots.
Unprocessed images of immunoblots shown in Figures and Supplementary Figures are provided.
Supplementary information
Supplementary Information
Supplementary Figures 1–8, and legends for Supplementary Tables 1 and 2.
Rights and permissions
About this article
Cite this article
Zhang, Q., Xiao, M., Gu, S. et al. ALK phosphorylates SMAD4 on tyrosine to disable TGF-β tumour suppressor functions. Nat Cell Biol 21, 179–189 (2019). https://doi.org/10.1038/s41556-018-0264-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0264-3
This article is cited by
-
SNHG17 alters anaerobic glycolysis by resetting phosphorylation modification of PGK1 to foster pro-tumor macrophage formation in pancreatic ductal adenocarcinoma
Journal of Experimental & Clinical Cancer Research (2023)
-
Imatinib blocks tyrosine phosphorylation of Smad4 and restores TGF-β growth-suppressive signaling in BCR-ABL1-positive leukemia
Signal Transduction and Targeted Therapy (2023)
-
Integrated circulating tumour DNA and cytokine analysis for therapy monitoring of ALK-rearranged lung adenocarcinoma
British Journal of Cancer (2023)
-
Targeting TGF-β signal transduction for fibrosis and cancer therapy
Molecular Cancer (2022)
-
Inhibiting ALK-TOPK signaling pathway promotes cell apoptosis of ALK-positive NSCLC
Cell Death & Disease (2022)