Abstract
Epithelial integrity is maintained by the cytoskeleton and through cell adhesion. However, it is not yet known how a deregulated cytoskeleton is associated with cancer. We identified cancer-related regulator of actin dynamics (CRAD) as frequently mutated or transcriptionally downregulated in colorectal cancer. We found that CRAD stabilizes the cadherin–catenin–actin complex via capping protein inhibition. The loss of CRAD inhibits F-actin polymerization and subsequently disrupts the cadherin–catenin–actin complex, which leads to β-catenin release and Wnt signalling hyperactivation. In mice, CRAD knockout induces epithelial cell integrity loss and Wnt signalling activation, resulting in the development of intestinal mucinous adenoma. With APC mutation, CRAD knockout initiates and accelerates mucinous and invasive adenoma development in the colorectum. These results define CRAD as a tumour suppressor, the inactivation of which deregulates the cytoskeleton and hyperactivates Wnt signalling thus initiating mucinous colorectal cancer. Our study reveals the unexpected roles of an actin cytoskeletal regulator in maintaining epithelial cell integrity and suppressing tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Microarray data that support the findings of this study have been deposited in GEO (accession code GDS2947). Mass spectrometry data used in this study is available from Supplementary Table 2. The CRAD expression data in CRC cells were derived from the cBioportal using the TCGA Research Network (http://cancergenome.nih.gov/) and Genetech datesets. The dataset derived from this resource that supports the findings of this study is available in Oncomine (https://www.oncomine.org/resource). CRAD expression data were also derived from cBioportal (http://www.cbioportal.org/) and COSMIC database (https://cancer.sanger.ac.uk/cosmic). Source data for Figs. 1–8 and Supplementary Figs. 1–8 have been provided as Supplementary Table 4. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
References
Mege, R. M., Gavard, J. & Lambert, M. Regulation of cell-cell junctions by the cytoskeleton. Curr. Opin. Cell Biol. 18, 541–548 (2006).
Gumbiner, B. M. & Kim, N. G. The Hippo-YAP signaling pathway and contact inhibition of growth. J. Cell Sci. 127, 709–717 (2014).
Oda, H. & Takeichi, M. Evolution: structural and functional diversity of cadherin at the adherens junction. J. Cell Biol. 193, 1137–1146 (2011).
Maiden, S. L. & Hardin, J. The secret life of alpha-catenin: moonlighting in morphogenesis. J. Cell Biol. 195, 543–552 (2011).
Bianchini, J. M. et al. Reevaluating αE-catenin monomer and homodimer functions by characterizing E-cadherin/αE-catenin chimeras. J. Cell Biol. 210, 1065–1074 (2015).
Edwards, M. et al. Capping protein regulators fine-tune actin assembly dynamics. Nat. Rev. Mol. Cell Biol. 15, 677–689 (2014).
Bear, J. E. et al. Antagonism between Ena/VASP proteins and actin filament capping regulates fibroblast motility. Cell 109, 509–521 (2002).
Xue, B. & Robinson, R. C. Guardians of the actin monomer. Eur. J. Cell Biol. 92, 316–332 (2013).
Schafer, D. A., Jennings, P. B. & Cooper, J. A. Dynamics of capping protein and actin assembly in vitro: uncapping barbed ends by polyphosphoinositides. J. Cell Biol. 135, 169–179 (1996).
Taoka, M. et al. V-1, a protein expressed transiently during murine cerebellar development, regulates actin polymerization via interaction with capping protein. J. Biol. Chem. 278, 5864–5870 (2003).
George, J. et al. Comprehensive genomic profiles of small cell lung cancer. Nature 524, 47–53 (2015).
Ambros, V. R., Chen, L. B. & Buchanan, J. M. Surface ruffles as markers for studies of cell transformation by Rous sarcoma virus. Proc. Natl Acad. Sci. USA 72, 3144–3148 (1975).
de Curtis, I. & Meldolesi, J. Cell surface dynamics - how Rho GTPases orchestrate the interplay between the plasma membrane and the cortical cytoskeleton. J. Cell Sci. 125, 4435–4444 (2012).
Gauthier, N. C., Masters, T. A. & Sheetz, M. P. Mechanical feedback between membrane tension and dynamics. Trends Cell Biol. 22, 527–535 (2012).
Pollard, T. D. & Cooper, J. A. Actin, a central player in cell shape and movement. Science 326, 1208–1212 (2009).
Bryce, N. S. et al. Cortactin promotes cell motility by enhancing lamellipodial persistence. Curr. Biol. 15, 1276–1285 (2005).
Montross, W. T., Ji, H. & McCrea, P. D. A beta-catenin/engrailed chimera selectively suppresses Wnt signaling. J. Cell Sci. 113, 1759–1770 (2000).
Kwiatkowski, A. V. et al. In vitro and in vivo reconstitution of the cadherin–catenin–actin complex from Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 107, 14591–14596 (2010).
Maiden, S. L. et al. Specific conserved C-terminal amino acids of Caenorhabditis elegans HMP-1/α-catenin modulate F-actin binding independently of vinculin. J. Biol. Chem. 288, 5694–5706 (2013).
Hong, S., Troyanovsky, R. B. & Troyanovsky, S. M. Binding to F-actin guides cadherin cluster assembly, stability, and movement. J. Cell Biol. 201, 131–143 (2013).
Braga, V. M. & Yap, A. S. The challenges of abundance: epithelial junctions and small GTPase signalling. Curr. Opin. Cell Biol. 17, 466–474 (2005).
Tanaka, Y. et al. Frequent β-catenin mutation and cytoplasmic/nuclear accumulation in pancreatic solid-pseudopapillary neoplasm. Cancer Res. 61, 8401–8404 (2001).
Peterson, L. W. & Artis, D. Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat. Rev. Immunol. 14, 141–153 (2014).
van Es, J. H. et al. Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nat. Cell Biol. 7, 381–386 (2005).
Moser, A. R., Pitot, H. C. & Dove, W. F. A dominant mutation that predisposes to multiple intestinal neoplasia in the mouse. Science 247, 322–324 (1990).
Bara, J., Forgue-Lafitte, M. E., Maurin, N., Flejou, J. F. & Zimber, A. Abnormal expression of gastric mucin in human and rat aberrant crypt foci during colon carcinogenesis. Tumour Biol. 24, 109–115 (2003).
Sachs, N. & Clevers, H. Organoid cultures for the analysis of cancer phenotypes. Curr. Opin. Genet. Dev. 24, 68–73 (2014).
Podolsky, D. K., Fournier, D. A. & Lynch, K. E. Human colonic goblet cells. Demonstration of distinct subpopulations defined by mucin-specific monoclonal antibodies. J. Clin. Invest. 77, 1263–1271 (1986).
Yamachika, T. et al. Establishment and characterization of a human colonic mucinous carcinoma cell line with predominant goblet-cell differentiation from liver metastasis. Pathol. Int. 55, 550–557 (2005).
van Es, J. H. et al. Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature 435, 959–963 (2005).
Hugen, N., Brown, G., Glynne-Jones, R., de Wilt, J. H. & Nagtegaal, I. D. Advances in the care of patients with mucinous colorectal cancer. Nat. Rev. Clin. Oncol. 13, 361–369 (2016).
Brabletz, T. et al. Nuclear overexpression of the oncoprotein beta-catenin in colorectal cancer is localized predominantly at the invasion front. Pathol. Res. Pract. 194, 701–704 (1998).
Vermeulen, L. et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 12, 468–476 (2010).
Goentoro, L. & Kirschner, M. W. Evidence that fold-change, and not absolute level, of beta-catenin dictates Wnt signaling. Mol. Cell 36, 872–884 (2009).
Voloshanenko, O. et al. Wnt secretion is required to maintain high levels of Wnt activity in colon cancer cells. Nat. Commun. 4, 2610 (2013).
Hulsken, J., Birchmeier, W. & Behrens, J. E-cadherin and APC compete for the interaction with beta-catenin and the cytoskeleton. J. Cell Biol. 127, 2061–2069 (1994).
Fagotto, F., Funayama, N., Gluck, U. & Gumbiner, B. M. Binding to cadherins antagonizes the signaling activity of beta-catenin during axis formation in Xenopus. J. Cell Biol. 132, 1105–1114 (1996).
Brannon, A. R. et al. Comparative sequencing analysis reveals high genomic concordance between matched primary and metastatic colorectal cancer lesions. Genome Biol. 15, 454 (2014).
Okada, K. et al. Adenomatous polyposis coli protein nucleates actin assembly and synergizes with the formin mDia1. J. Cell Biol. 189, 1087–1096 (2010).
Kufe, D. W. Mucins in cancer: function, prognosis and therapy. Nat. Rev. Cancer 9, 874–885 (2009).
Edgar, R., Domrachev, M. & Lash, A. E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 30, 207–210 (2002).
Forbes, S. A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2011).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 6, pl1 (2013).
Rhodes, D. R. et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6, 1–6 (2004).
Cerami, E. et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2, 401–404 (2012).
Jung, H. Y. et al. PAF and EZH2 induce Wnt/β-catenin signaling hyperactivation. Mol. Cell 52, 193–205 (2013).
Jun, S. et al. PAF-mediated MAPK signaling hyperactivation via LAMTOR3 induces pancreatic tumorigenesis. Cell Rep. 5, 314–322 (2013).
Elias, J. E. & Gygi, S. P. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat. Methods 4, 207–214 (2007).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Symons, M. H. & Mitchison, T. J. Control of actin polymerization in live and permeabilized fibroblasts. J. Cell Biol. 114, 503–513 (1991).
Acknowledgements
We thank J.A. Cooper for his insightful comments and recommendations regarding the experiment and result analysis. We thank S.-H. Lee, H.N. Suh, S.H. Lee and K.-S. Park for helpful comments on the manuscript. This work was supported by the Cancer Prevention Research Institute of Texas (grant no. RP140563 to J-.I.P.), the National Institutes of Health (grant nos R01 CA193297-01 (to J-.I.P.), 5R01 GM107079 (to P.D.M.) and R01 GM126048 (to W.W.)), the Department of Defense Peer Reviewed Cancer Research Program (grant no. CA140572 to J-.I.P.), a Duncan Family Institute for Cancer Prevention and Risk Assessment Grant (IRG-08-061-01; to J-.I.P.), a Center for Stem Cell and Developmental Biology Transformative Grant (MD Anderson Cancer Center to J-.I.P.), an Institutional Research Grant (MD Anderson Cancer Center; to J-.I.P.), a New Faculty Award (MD Anderson Cancer Center Support Grant; to J-.I.P.), a Metastasis Research Center Grant (MD Anderson; to J-.I.P.) and a Uterine SPORE Career Enhancement Program (MD Anderson; to J-.I.P.). The core facility (DNA sequencing and Genetically Engineered Mouse Facility) was supported by the MD Anderson Cancer Center Support Grant (CA016672).
Author information
Authors and Affiliations
Contributions
Y.-S.J. and J.-I.P. conceived the experiments. Y.-S.J., W.W., S.J., J.Z., M.S., M.J.K., E.M.L., J.S. and J.-I.P. performed the experiments. Y.-S.J., J.C., P.D.M., S.Z. and J.-I.P. analysed the data. Y.-S.J. and J.-I.P. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Genetic inactivation of CRAD in cancer.
(a), Genetic inactivation of CRAD in CRC. cBioportal database analysis of CRAD in CRC. CRAD shows 10.18 % incidence rate of truncated mutants (Nonsense + Frameshift) in CRC. APC, DCC, CTNNB1, and AXIN2 were analyzed as the positive control. HPRP1 served as negative control. Total 502 CRC cases were analyzed. (b), Genetic alteration of CRAD in human cancer. cBioportal analysis. (c), Potent epigenetic suppression of CRAD promoter. Upregulated CRAD expression by the inhibition of methylation. IECs and CRC cells were treated with 5-Azacytidine (5-AZA; 20 μM) for 24 hr. n = 3 independent experiments; Error bars: average +/− S.D.; NS: not significant (P > 0.05); Two-sided unpaired t-test. (d), COSMIC analysis of CRAD mutations in CRC cell lines.
Supplementary Figure 2 Positive regulation of the actin polymerization by CRAD-induced capping protein inhibition.
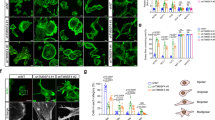
(a and b), Morphological comparison of IECs and CRC cells. IF staining (a). Quantification of the cell area of IECs and CRC cells by AxioVision software (b; n = 5 independent experiments). (c and d), Depletion of endogenous CRAD by shRNAs. FHC cells were transfected with shRNAs (c). shCRAD#1 stably expressing IECs were analyzed for IB (d). IB was performed once. (e-g), Cell shrinkage by CRAD depletion. 48 hr after transfection, cells were analyzed for IF staining (e) and cell area quantification (f; n = 5 (independent experiments). Cells were infected with lenti-shCRAD for 48 hr. After infection, cells were selected by puromycin (2 μg/ml) treatment for 72 hr. The cell morphology was analyzed by IF staining for Phalloidin (g). (h-k), Extended cell morphology by CRAD in CRC cells. 48 h after transfection (Vec [empty vector] or FLAG-CRAD), cells were analysed for IF staining (actin, h; Phalloidin, k) and cell area quantification (i; n = 5 independent experiments). The cell morphology was monitored using blight field microscope (j). (l), Physical interaction of CRAD with CPs. Purified FLAG-Actin, FLAG-CPs, and HA-CRAD proteins were used for co-IP. These results recapitulate in vitro interaction between CRAD and CPs performed using GST recombinant proteins. Experiment was performed three times with similar results. (m-o), Decreased F-actin formation by CRAD depletion. Quantification of the F-actin in the CRAD depleted (m), or the CRAD ectopic expressed condition (n and o). The level of F-actin and G-actin was examined by immunoblots (n) and quantified by the subsequent ImageJ analysis (m and o; n = 3 independent experiments). After measurement, F-actin was normalized by G-actin. (p), Coomassie Brilliant Blue (CBB) staining of the purified recombinant proteins. Each recombinant protein was expressed in E. coli and purified, followed by GST cleavage. Experiment was performed once. (q), Inhibition of interaction between CAPZs and actin by CRAD. Co-IP assay using purified recombinant proteins (see Fig. 2m) was quantified by ImageJ. n = 3 independent experiments). (r and s). No effect of A511V-CRAD on Wnt/β-catenin signaling. AXIN2 qRT-PCR (r; n = independent experiments) and IB (s) using WT-CRAD or A511V-CRAD transfected CRC cells. IB was performed three times with similar results. Representative images are shown; Scale bars indicate 20 μm; Error bars: mean +/− S.D.; NS: not significant (P > 0.05); Two-sided unpaired t-test.
Supplementary Figure 3 Illustration of the working model of CRAD-induced F-actin polymerization.
CRAD inhibits CPs’ binding to the barbed (+) end, resulting in the increased actin polymerization.
Supplementary Figure 4 CRC cell growth inhibition by CRAD.
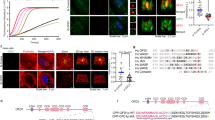
(a), The mutually exclusive expression between CRAD and AXIN2 in IECs and CRC. Oncomine analysis of CRAD and AXIN2 expression in CRC. (b), Inversed correlation between CRAD and β-catenin. IECs and CRC cells were fractionated into the nucleus and cytosol fractions using fractionation kit. WCL: whole cell lysates. (c and d), Decreased nuclear β-catenin by CRAD. IECs (c) and CRC cells (d) were transfected with shCtrl or shCRAD and Vec or CRAD, respectively. After 48 hr, cells were fractionated into the cytosolic and nucleus fractions, followed by IB. (e and f), Increased interaction between E-Cadherin and β-catenin by CRAD. Cell extracts from IEC (e) and CRC cell (f) were used for IP analysis. Binding between E-Cadherin and β-catenin was quantified by ImageJ. n = 3. (g), Increased interaction between E-cadherin and catenins by CRAD. HCT116 cells were transiently transfected with FLAG-CRAD plasmid and analyzed for IF staining using Super Resolution microscope (see Fig. 3p). E-cadherin intensity was quantified by ZEN software (Zeiss). n = 10. (h and i), Blockade of the actin cytoskeleton increases Wnt/β-catenin reporter activity. CCD-841CoN cells were transfected with β-catenin reporter plasmids (pMEGA-TOP/FOP-FLASH). 24 hr after, cells were treated with Cytochalasin D (Cyto D; 2 μM; barbed [+] end inhibition)51, Latrunculin B (Lat B; 1 μM; monomeric G-actin inhibition)52, and Jasplakinolide (2 μM; a stabilizer of actin cytoskeleton) for 72 hr. β-catenin transcription activity was measured by TOP/FOP luciferase activity (h). CCD-841CoN cells were treated with indicated reagents as same concentration of (h). 72 h after treatment, cells were collected for qRT-PCR (i). n = 3 independent experiments. j, Illustration of the working model: the molecular mechanism of CRAD loss-induced hyperactivation of Wnt/β-catenin signaling. In normal epithelial cells, CRAD inhibits CPs, which results in actin polymerization. Subsequently, increased F-actin stabilizes complex formation composed of E-cadherin-catenins-F-actin. In the absence of CRAD, F-actin is disrupted by CPs, which leads to destabilization of E-cadherin-catenin complex. Disturbance of E-cadherin-catenin complex releases β-catenin into the cytosols and the nucleus, which transactivates Wnt/β-catenin target genes. Error bars: average +/− S.D.; Two-sided unpaired t-test; Centre: Average.
Supplementary Figure 5 CRC cell growth inhibition by CRAD.
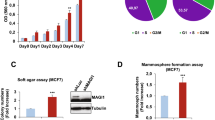
(a and b), Suppression of shCRAD-induced cell hyperproliferation by β-catenin inhibition in IECs. FHC (shCtrl and shCRAD) cells were treated with iCRT14 for 14 days and stained with crystal violet (a). Cell proliferation was quantified by measurement of absorbance at 590 nm (b; n = 3 independent experiments). (c-e), Engrailed-LEF1 inhibits CRAD depletion-induced IEC hyperproliferation. 1,000 cells of CCD-841CoN were seeded onto tissue culture plate. Cells were transfected with indicated plasmids. Eng-LEF1 was used as a dominant-negative mutant blocker of β-catenin-mediated gene transactivation53,54. 48 h after transfection, cells were incubated in 37 °C tissue culture CO2 chamber for 14 days. Colony forming was visualized by crystal violet staining (c) and quantified (d and e; n = 3 independent experiments). (f-h), CRC cell growth inhibition by CRAD expression. CRC cells (Vec [control] and CRAD expressing) were analyzed for cell counting (f; n = 3 independent experiments), crystal violet staining (g), and quantification (h). (i-o), β-catenin rescues CRAD-induced CRC cell growth inhibition. CRC cells were transfected with CRAD or-catenin plasmids and analyzed for crystal violet staining (i,k,m), quantification (j, l, n), and cell counting (o). (p and q), CRC cell growth inhibition by CPI motif-containing CRAD mutants. CRAD (full length, ΔCPI, and M1-M4)-transfected CRC cells were analysed for quantification of cell proliferation. HCT116 (p); SW620 cells (q). Representative images are shown; Error bars: mean +/− S.D.; NS: not significant (P > 0.05); Two-sided unpaired t-test.
Supplementary Figure 6 Intestinal adenoma development by CRAD KO.
(a), CRISPR/Cas9-mediated targeting of CRAD alleles. Exon2 of CRAD was targeted using gRNAs. gRNA and Cas9 mRNA were injected into the pronuclei of C57BL/B6 mouse embryos. (b), CRAD expression in the small intestine. IHC of mouse intestine. (c), Validation of CRAD KO. The cell lysates were extracted from the small intestine of six independent CRAD KO mice for IB. (d), Sequencing analysis of potential off-target genes of CRAD gRNAs. No mutations in 14 off-target genes were detected. (e), Increased Wnt signaling target genes in CRAD KO mouse. CRAD KO-induced tumors were analyzed for qRT-PCR of Hippo, Notch, Shh, BMP, and Wnt signaling pathway target genes. n = 3 independent mice. f,g, Tumor development in the lung and pancreas of CRAD KO mice. Development of the early lesion of SCLC. The multiple sites displaying the early lesion of SCLC (black arrows; f) and the pancreatic tumors (g; i and ii) were observed. Scale bars indicate 100 μm (f) or 20 μm (g). (h), Disruption of epithelial cell integrity. Cytokeratin 19 (CK19). Arrows: Villi not expressing CK19. (i and j), Analysis of inflammation in CRAD KO mouse. Small intestine samples of WT and CRAD KO mouse were stained with H&E (i; n = 8 mice). Intestinal inflammation (foci size > 200 μm) was assessed by counting (j). Scale bars indicate 200 μm. (k-n), Cell hyperproliferation in CRAD KO small intestine. Phospho-histone H3 (k) and Ki67 (n) staining and quantification (l and m; n = 10 crypts). Scale bars indicate 20 μm. o, Analysis of apoptosis in CRAD KO-induced tumor lesion. WT and CRAD KO mice were analyzed for cleaved Caspase-3 (Casp3). Scale bars indicate 20 μm. (p), Abnormal differentiation of IECs by CRAD KO. WT and CRAD KO small intestine were immunostained with ChgA (arrows). Scale bars indicate 20 μm. (q), Disorganized cell adhesion in CRAD KO mice. Cells were stained with E-cadherin. Scale bars indicate 20 μm. (r), Increased β-catenin target genes in the intestinal adenoma of CRAD KO mice. IHC for CD44. Scale bars indicate 20 μm. Representative images of three independent experiments; Error bars: mean +/− S.D.; NS: not significant (P > 0.05); Two-sided unpaired t-test.
Supplementary Figure 7 Accelerated intestinal tumorigenesis by CRAD heterogeneous KO.
(a), Heterozygous mutation of CRAD gene in CRC. Zygosity analysis of CRAD mutation in CRC patient samples using COSMIC and cBioportal databases. Of note, the frequency of heterozygous mutation is higher than that of homozygous mutation. (b-e), IHC of the non-tumor region of the colorectum of APCMIN and APCMIN:CRAD+/− mice (4mo of age). Cyclin D1 (b); Ki67 (d). Quantification (c; n = 9 crypts, (e); n = 10 crypts). (f), Micro-invasion by CRAD KO. Micro-invasion was observed in the tumor region of APCMIN:CRAD+/− small intestine. The small intestine samples were stained with αSMA (α-Smooth Muscle Actin: green) to visualize the basement membrane. The basement membrane of APCMIN:CRAD+/− is disrupted/discontinued tumor cell infiltration. Arrow indicates invasive tumor cell and yellow dashed lines mark the border of the basement membrane. (g), No EMT in tumour region of APCMIN:CRAD+/− mice. EMT marker analysis in APCMIN and APCMIN:CRAD+/− small intestine samples. Markers of mesenchymal cell (Vimentin: green; N-cadherin: red) were not detected in tumors of both strains. Representative images of three independent experiments; Scale bars indicate 20 μm; Error bars: mean +/− S.D.; Two-sided unpaired t-test.
Supplementary Figure 8 Accelerated intestinal tumorigenesis by CRAD KO.
(a and b), IHC analysis of the organoids derived from CRAD WT and KO mouse intestine. Compared to WT, CRAD KO-derived cystic spheroids showed the increased cell proliferation (Ki67; a) and the increase of β-catenin (b). Quantification was performed using 10 cystic spheroids. n = 10 organoids from three different experiments. (c), Increased Mucin expression in CRAD KO. The small intestine samples from CRAD WT (2mo) and CRAD KO mice (from different ages as indicated) were examined by qRT-PCR for mMUC1, mMUC2, mMUC4, and mMUC5AC. n = 3 independent experiments. (d), Increased goblet cell in CRAD KO mice. After fixation and paraffin embedding, each sample was stained with PAS and quantified. n = 10 villi. (e), Increased mucin deposition in CRAD KO-induced tumors. qRT-PCR of MUC1 and MUC2 from the small intestine samples of each mice. n = 3 independent experiments. (f), The increase of TOP-1 expression in CRAD KO mouse. Tumor of APCMIN (4mo) and CRAD KO (3, 4, 6, 8, and 12mo) were immunostained with a TOP-1 antibody. (g), Upregulation of TOP-1 in CRAD KO tumors. CRAD WT (2mo) intestine and CRAD KO tumors (from different age were examined by qRT-PCR of mTOP-1. n = 3 independent experiments. (h), Mutual exclusive expression of CRAD and MUCs. Oncomine analysis of TCGA datasets; 10% gene rank; P < 0.0001; fold change >2; compared with normal cells. Representative images of three independent experiments; Scale bars indicate 20 μm; Error bars: mean +/− S.D.; NS: not significant (P > 0.05); Two-sided unpaired t-test.
Supplementary information
Supplementary Information
Supplementary Figures 1–9, Supplementary Table legends and Supplementary References.
Supplementary Table 1
Analysis of CRC tumour microarray for CRAD expression.
Supplementary Table 2
CRAD-interacting proteins identified by tandem affinity purification and mass spectrometry.
Supplementary Table 3
Information of MC microarray for CRAD expression.
Supplementary Table 4
Primer information.
Supplementary Table 5
Antibody information.
Supplementary Table 6
Statistics Source Data.
Rights and permissions
About this article
Cite this article
Jung, YS., Wang, W., Jun, S. et al. Deregulation of CRAD-controlled cytoskeleton initiates mucinous colorectal cancer via β-catenin. Nat Cell Biol 20, 1303–1314 (2018). https://doi.org/10.1038/s41556-018-0215-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0215-z
This article is cited by
-
Exploiting synergistic effect of CO/NO gases for soft tissue transplantation using a hydrogel patch
Nature Communications (2023)
-
Wnt signaling in cancer: therapeutic targeting of Wnt signaling beyond β-catenin and the destruction complex
Experimental & Molecular Medicine (2020)
-
Clinical impact of first-line bevacizumab plus chemotherapy in metastatic colorectal cancer of mucinous histology: a multicenter, retrospective analysis on 685 patients
Journal of Cancer Research and Clinical Oncology (2020)
-
Prognostic impact of tumor mutation burden and the mutation in KIAA1211 in small cell lung cancer
Respiratory Research (2019)
-
Recent progress in mapping the emerging landscape of the small-cell lung cancer genome
Experimental & Molecular Medicine (2019)