Abstract
A key step in nutrient sensing is activation of the master growth regulator, mTORC1 kinase, on the lysosomal membrane. Nutrients enable mTORC1 scaffolding by a complex composed of the Rag GTPases (Rags) and Ragulator, but the underlying mechanism of mTORC1 capture is poorly understood. Combining dynamic imaging in cells and reconstituted systems, we uncover an affinity switch that controls mTORC1 lifetime and activation at the lysosome. Nutrients destabilize the Rag–Ragulator interface, causing cycling of the Rags between lysosome-bound Ragulator and the cytoplasm, and rendering mTORC1 capture contingent on simultaneous engagement of two Rag-binding interfaces. Rag GTPase domains trigger cycling by coordinately weakening binding of the C-terminal domains to Ragulator in a nucleotide-controlled manner. Cancer-specific Rag mutants override release from Ragulator and enhance mTORC1 recruitment and signalling output. Cycling in the active state sets the Rags apart from most signalling GTPases, and provides a mechanism to attenuate mTORC1 signalling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Perera, R. M. & Zoncu, R. The lysosome as a regulatory hub. Annu. Rev. Cell Dev. Biol. 32, 223–253 (2016).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell 169, 361–371 (2017).
Binda, M. et al. The Vam6 GEF controls TORC1 by activating the EGO complex. Mol. Cell 35, 563–573 (2009).
Castellano, B. M. et al. Lysosomal cholesterol activates mTORC1 via an SLC38A9-Niemann-Pick C1 signaling complex. Science 355, 1306–1311 (2017).
Efeyan, A. et al. Regulation of mTORC1 by the Rag GTPases is necessary for neonatal autophagy and survival. Nature 493, 679–683 (2013).
Kim, E., Goraksha-Hicks, P., Li, L., Neufeld, T. P. & Guan, K. L. Regulation of TORC1 by Rag GTPases in nutrient response. Nat. Cell Biol. 10, 935–945 (2008).
Sancak, Y. et al. Ragulator–Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141, 290–303 (2010).
Sancak, Y. et al. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science 320, 1496–1501 (2008).
Demetriades, C., Doumpas, N. & Teleman, A. A. Regulation of TORC1 in response to amino acid starvation via lysosomal recruitment of TSC2. Cell 156, 786–799 (2014).
Menon, S. et al. Spatial control of the TSC complex integrates insulin and nutrient regulation of mTORC1 at the lysosome. Cell 156, 771–785 (2014).
Pacold, M. E. et al. Crystal structure and functional analysis of Ras binding to its effector phosphoinositide 3-kinase gamma. Cell 103, 931–943 (2000).
Yang, H. et al. Mechanisms of mTORC1 activation by RHEB and inhibition by PRAS40. Nature 552, 368–373 (2017).
Bar-Peled, L. et al. A Tumor suppressor complex with GAP activity for the Rag GTPases that signal amino acid sufficiency to mTORC1. Science 340, 1100–1106 (2013).
Okosun, J. et al. Recurrent mTORC1-activating RRAGC mutations in follicular lymphoma. Nat. Genet. 48, 183–188 (2016).
Ying, Z. X. et al. Recurrent Mutations in the MTOR Regulator RRAGC in Follicular Lymphoma. Clin. Cancer Res 22, 5383–5393 (2016).
Bar-Peled, L., Schweitzer, L. D., Zoncu, R. & Sabatini, D. M. Ragulator is a GEF for the rag GTPases that signal amino acid levels to mTORC1. Cell 150, 1196–1208 (2012).
Panchaud, N., Peli-Gulli, M. P. & De Virgilio, C. Amino acid deprivation inhibits TORC1 through a GTPase-activating protein complex for the Rag family GTPase Gtr1. Sci. Signal. 6, ra42 (2013).
Peli-Gulli, M. P., Sardu, A., Panchaud, N., Raucci, S. & De Virgilio, C. Amino acids stimulate TORC1 through Lst4-Lst7, a GTPase-activating protein complex for the Rag family GTPase Gtr2. Cell Rep. 13, 1–7 (2015).
Petit, C. S., Roczniak-Ferguson, A. & Ferguson, S. M. Recruitment of folliculin to lysosomes supports the amino acid-dependent activation of Rag GTPases. J. Cell Biol. 202, 1107–1122 (2013).
Tsun, Z. Y. et al. The folliculin tumor suppressor is a GAP for the RagC/D GTPases that signal amino acid levels to mTORC1. Mol. Cell 52, 495–505 (2013).
Chantranupong, L. et al. The CASTOR proteins are arginine sensors for the mTORC1 pathway. Cell 165, 153–164 (2016).
Rebsamen, M. et al. SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1. Nature 519, 477–481 (2015).
Saxton, R. A., Chantranupong, L., Knockenhauer, K. E., Schwartz, T. U. & Sabatini, D. M. Mechanism of arginine sensing by CASTOR1 upstream of mTORC1. Nature 536, 229–233 (2016).
Wang, S. et al. Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1. Science 347, 188–194 (2015).
Wolfson, R. L. et al. Sestrin2 is a leucine sensor for the mTORC1 pathway. Science 351, 43–48 (2016).
Teis, D., Wunderlich, W. & Huber, L. A. Localization of the MP1-MAPK scaffold complex to endosomes is mediated by p14 and required for signal transduction. Dev. Cell 3, 803–814 (2002).
Nada, S. et al. The novel lipid raft adaptor p18 controls endosome dynamics by anchoring the MEK-ERK pathway to late endosomes. EMBO J. 28, 477–489 (2009).
Manifava, M. et al. Dynamics of mTORC1 activation in response to amino acids. eLife 5, e19960 (2016).
Kulak, N. A., Pichler, G., Paron, I., Nagaraj, N. & Mann, M. Minimal, encapsulated proteomic-sample processing applied to copy-number estimation in eukaryotic cells. Nat. Methods 11, 319–324 (2014).
Sprague, B. L., Pego, R. L., Stavreva, D. A. & McNally, J. G. Analysis of binding reactions by fluorescence recovery after photobleaching. Biophys. J. 86, 3473–3495 (2004).
Inoue, T., Heo, W. D., Grimley, J. S., Wandless, T. J. & Meyer, T. An inducible translocation strategy to rapidly activate and inhibit small GTPase signaling pathways. Nat. Methods 2, 415–418 (2005).
Zoncu, R. et al. A phosphoinositide switch controls the maturation and signaling properties of APPL endosomes. Cell 136, 1110–1121 (2009).
Abu-Remaileh, M. et al. Lysosomal metabolomics reveals V-ATPase- and mTOR-dependent regulation of amino acid efflux from lysosomes. Science 358, 807–813 (2017).
Schroder, B., Wrocklage, C., Hasilik, A. & Saftig, P. Molecular characterisation of ‘transmembrane protein 192’ (TMEM192), a novel protein of the lysosomal membrane. Biol. Chem. 391, 695–704 (2010).
Liberles, S. D., Diver, S. T., Austin, D. J. & Schreiber, S. L. Inducible gene expression and protein translocation using nontoxic ligands identified by a mammalian three-hybrid screen. Proc. Natl Acad. Sci. USA 94, 7825–7830 (1997).
Fracchiolla, D. et al. Mechanism of cargo-directed Atg8 conjugation during selective autophagy. eLife 5, e18544 (2016).
Vink, M. et al. In vitro FRAP identifies the minimal requirements for Mad2 kinetochore dynamics. Curr. Biol. 16, 755–766 (2006).
Young, L. N., Cho, K., Lawrence, R., Zoncu, R. & Hurley, J. H. Dynamics and architecture of the NRBF2-containing phosphatidylinositol 3-kinase complex I of autophagy. Proc. Natl Acad. Sci. USA 113, 8224–8229 (2016).
Schmidt, G. et al. Biochemical and biological consequences of changing the specificity of p21ras from guanosine to xanthosine nucleotides. Oncogene 12, 87–96 (1996).
Chen, J. et al. Single-molecule dynamics of enhanceosome assembly in embryonic stem cells. Cell 156, 1274–1285 (2014).
Hansen, A. S., Pustova, I., Cattaglio, C., Tijan, R. & Darzacq, X. CTCF and cohesin regulate chromatin loop stability with distinct dynamics. eLife 6, e25776 (2017).
Knight, S. C. et al. Dynamics of CRISPR–Cas9 genome interrogation in living cells. Science 350, 823–826 (2015).
Caussinus, E., Kanca, O. & Affolter, M. Fluorescent fusion protein knockout mediated by anti-GFP nanobody. Nat. Struct. Mol. Biol. 19, 117–121 (2011).
Gong, R. et al. Crystal structure of the Gtr1p–Gtr2p complex reveals new insights into the amino acid-induced TORC1 activation. Genes Dev. 25, 1668–1673 (2011).
Kurzbauer, R. et al. Crystal structure of the p14/MP1 scaffolding complex: how a twin couple attaches mitogen-activated protein kinase signaling to late endosomes. Proc. Natl Acad. Sci. USA 101, 10984–10989 (2004).
Powis, K. et al. Crystal structure of the Ego1–Ego2–Ego3 complex and its role in promoting Rag GTPase-dependent TORC1 signaling. Cell Res. 25, 1043–1059 (2015).
Zhang, T., Peli-Gulli, M. P., Yang, H., De Virgilio, C. & Ding, J. Ego3 functions as a homodimer to mediate the interaction between Gtr1–Gtr2 and Ego1 in the ego complex to activate TORC1. Structure 20, 2151–2160 (2012).
de Araujo, M. E. G. et al. Crystal structure of the human lysosomal mTORC1 scaffold complex and its impact on signaling. Science 358, 377–381 (2017).
Su, M. Y. et al. Hybrid structure of the RagA/C-Ragulator mTORC1 activation complex. Mol. Cell 68, 835–846 (2017).
Yonehara, R. et al. Structural basis for the assembly of the Ragulator–Rag GTPase complex. Nat. Commun. 8, 1625 (2017).
Zhang, T. et al. Structural basis for Ragulator functioning as a scaffold in membrane-anchoring of Rag GTPases and mTORC1. Nat. Commun. 8, 1394 (2017).
Jeong, J. H. et al. Crystal structure of the Gtr1p(GTP)–Gtr2p(GDP) protein complex reveals large structural rearrangements triggered by GTP-to-GDP conversion. J. Biol. Chem. 287, 29648–29653 (2012).
Cherfils, J. & Zeghouf, M. Regulation of small GTPases by GEFs, GAPs, and GDIs. Physiol. Rev. 93, 269–309 (2013).
Horiuchi, H. et al. A novel Rab5 GDP/GTP exchange factor complexed to Rabaptin-5 links nucleotide exchange to effector recruitment and function. Cell 90, 1149–1159 (1997).
Lippe, R., Miaczynska, M., Rybin, V., Runge, A. & Zerial, M. Functional synergy between Rab5 effector Rabaptin-5 and exchange factor Rabex-5 when physically associated in a complex. Mol. Biol. Cell 12, 2219–2228 (2001).
Groves, J. T. & Kuriyan, J. Molecular mechanisms in signal transduction at the membrane. Nat. Struct. Mol. Biol. 17, 659–665 (2010).
Gureasko, J. et al. Membrane-dependent signal integration by the Ras activator Son of sevenless. Nat. Struct. Mol. Biol. 15, 452–461 (2008).
Shen, K., Choe, A. & Sabatini, D. M. Intersubunit crosstalk in the Rag GTPase heterodimer enables mTORC1 to respond rapidly to amino acid availability. Mol. Cell 68, 552–565 (2017).
Martina, J. A. & Puertollano, R. Rag GTPases mediate amino acid-dependent recruitment of TFEB and MITF to lysosomes. J. Cell Biol. 200, 475–491 (2013).
Roczniak-Ferguson, A. et al. The transcription factor TFEB links mTORC1 signaling to transcriptional control of lysosome homeostasis. Sci. Signal. 5, ra42 (2012).
Settembre, C. et al. A lysosome-to-nucleus signalling mechanism senses and regulates the lysosome via mTOR and TFEB. EMBO J. 31, 1095–1108 (2012).
Zhou, X. et al. Dynamic visualization of mTORC1 activity in living cells. Cell Rep. http://doi.org/crv5 (2015).
Filipek, P. A. et al. LAMTOR/Ragulator is a negative regulator of Arl8b- and BORC-dependent late endosomal positioning. J. Cell Biol. 216, 4199–4215 (2017).
Pu, J., Keren-Kaplan, T. & Bonifacino, J. S. A Ragulator–BORC interaction controls lysosome positioning in response to amino acid availability. J. Cell Biol. 216, 4183–4197 (2017).
Wolfson, R. L. et al. KICSTOR recruits GATOR1 to the lysosome and is necessary for nutrients to regulate mTORC1. Nature 543, 438–442 (2017).
Zoncu, R. et al. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H+-ATPase. Science 334, 678–683 (2011).
Grabiner, B. C. et al. A diverse array of cancer-associated MTOR mutations are hyperactivating and can predict rapamycin sensitivity. Cancer Discov. 4, 554–563 (2014).
Rodrik-Outmezguine, V. S. et al. Overcoming mTOR resistance mutations with a new-generation mTOR inhibitor. Nature 534, 272–276 (2016).
Acknowledgements
The authors thank all members of the Zoncu Lab for helpful insights, M. Rape and R. Perera for critical reading of the manuscript, A. Hansen and X. Darzacq for assistance with analysis of single molecule data sets, S. Knight and L. Bosanac for assistance with FRAP analysis software, and H. Garcia for insights on co-localization analysis. This work was supported by the NIH Director’s New Innovator Award (1DP2CA195761-01), the Pew–Stewart Scholarship for Cancer Research, the Damon Runyon-Rachleff Innovation Award, the Edward Mallinckrodt, Jr Foundation Grant and the Packer Wentz Endowment to R.Z., and a National Science Foundation Graduate Research Fellowship (DGE 1106400) to R.E.L.
Author information
Authors and Affiliations
Contributions
R.E.L. and R.Z. conceived of the study. R.E.L., K.F.C., J.H.H. and R.Z. designed experiments. R.E.L., K.F.C., R.R., A.T., M.T., O.M. and R.Z. performed the experiments. R.E.L. and K.F.C. performed quantitative analysis of the results. D.J.K. generated reagents. R.E.L. and R.Z. wrote the manuscript. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Control experiments for FRAP analysis of mTORC1 pathway components.
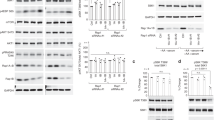
(a) Representative 3D images of endogenous LAMP2 staining from U2OS cells that were starved for amino acids and glucose (−AA/G) or starved and restimulated (+AA/G) (top), and HAP-1 Raptor:GFP cells that were starved for amino acids (−AA) or starved and restimulated (+AA) (bottom), followed by 3-D volumetric analysis of z-stacks. Scale bar 10 μm. Experiment repeated 2 times. (b) Subcellular fractionation. HEK-293T cells were starved of amino acids for 1 hour, or starved for 50 minutes then restimulated with amino acids for 10 minutes, followed by fractionation and collection of light membrane and cytoplasm. Fractions were immunoblotted for the indicated proteins. Experiment repeated 2 times. Unprocessed scans are shown in Supplementary Fig. 8. (c) mTOR localization in U2OS cells is not affected by incubation in nocodazole. U2OS cells were starved for amino acids and glucose, or starved and restimulated, and 2.5 μg/mL nocodazole was added for the last 20 minutes where indicated. Cells were fixed and subjected to immunofluorescence for mTOR and LAMP2. Scale bar 10 μm. Experiment repeated 2 times. (d) mTOR signalling in U2OS cells is not affected by nocodazole treatment. U2OS cells were starved for amino acids and glucose, or starved and restimulated, and 2.5 μg/mL nocodazole was added for the last 20 minutes where indicated. Cells were lysed, followed by immunoblotting for the indicated proteins and phosphor-proteins. Experiment repeated 2 times. Unprocessed scans are shown in Supplementary Fig. 8. (e) Fluorescence recovery over time curves from FRAP experiments in Raptor:GFP edited HAP-1 cells. Curve is the average ± S.E.M. of N = 15 lysosomes. (f) Fluorescence recovery over time curves from FRAP experiments in (g). Each curve is the average ± S.E.M. of [GFP-MP1 N = 23, p18-GFP +AA N = 28, GFP-RagB N = 24, GFP-RagC N = 21] lysosomes. (g) Time-lapse montage of single lysosome fluorescence recovery after photobleaching (FRAP) in U2OS cells expressing the indicated GFP-tagged Ragulator and Rag GTPase constructs (along with non-fluorescent Rag heterodimer partner). Scale bar 1 μm. See Supplementary Table 1 for statistical source data.
Supplementary Figure 2 The Rag-mTORC1 complex is transient and can be stabilized by anchoring either component to the lysosomal membrane.
(a) Fluorescence recovery over time curves from FRAP experiments in U2OS cells stably expressing the indicated TMEM192 complex along with FRB-myc RagC and GFP-RagB. Cells were treated with the indicated concentration of rapalogue (rapa) for 30 minutes prior to imaging. Each curve is the average ± S.E.M. of [TMEM192-FLAG ctrl N = 27, TMEM192-FLAG +rapa N = 23, TMEM192-FKBP ctrl N = 31, TMEM192-FKBP +rapa N = 23] lysosomes. (b) Immunofluorescence of mTOR and Lamp2 in U2OS cells stably expressing the indicated constructs. Cells were either kept in complete media or treated for two hours in amino acid- and glucose-depleted media. Where indicated, cells were treated with 50 nM rapalogue (rapa) prior to fixation. Scale bar 10 μm. Experiment repeated 3 times. (c) HEK-293T cells stably expressing FLAG-Raptor or FLAG-Raptor-Rheb15 were starved of amino acids for 1 hour, or starved for 50 minutes then restimulated with amino acids for 10 minutes, followed by fractionation and collection of light membrane and cytoplasm fractions. Fractions were immunoblotted for the indicated proteins. Experiment repeated 3 times. Unprocessed scans are shown in Supplementary Fig. 8. (d) HEK- 293T cells expressing FLAG-Raptor or FLAG-Raptor-Rheb15 were starved of amino acids for 1 hour, or starved for 50 minutes then restimulated with amino acids for 10 minutes, followed by immunostaining for the indicated proteins. Scale bar 10 μm. (e) Quantitation of RagA Lysosomal Enrichment Score for IF images in (d) (mean ± S.D., [grey N = 30 black N = 28, blue N = 30, red N = 29] cells/condition respectively, p = 0.0094; ****p < 0.0001 two-sided unpaired t-tests). See Supplementary Table 1 for statistical source data.
Supplementary Figure 3 Rag GTPase cycling is independent of the RagC/D GAP, FLCN.
(a) Fluorescence recovery over time curves from FRAP experiments in UOK257-1 FLCN-null or UOK257-2 FLCN rescue cells. Each curve is the average ± S.E.M. of [UOK257-1 N = 25, UOK257-2 N = 27] lysosomes. (b) MP1 and Rab7 FRAP is independent of nutrient conditions. Fluorescence recovery over time curves from FRAP experiments in U2OS cells expressing GFP-tagged MP1 or Rab7. Cells were either starved or restimulated for amino acids and glucose. Each curve is the average ± S.E.M. of [Mp1 –AA/G N = 27, Mp1 +AA/G N = 27, Rab7 –AA/G N = 30, Rab7 +AA/G N = 22] lysosomes.
Supplementary Figure 4 Control experiments for in vitro FRAP of Rag GTPases.
(a) In vitro FRAP experiment in which Ragulator-coated beads were incubated with increasing amounts of GFP-RagB + Flag RagC. Each curve is the average ± S.E.M. of [0.3 uL N = 11, 1 uL N = 8, 3 uL N = 11, 9 uL N = 12] bead regions. Experiment repeated 2 times. (b) Halftime (t1/2) calculations on single exponential fits of FRAP recovery curves in (a). Shown are best fit values with 95% confidence intervals. (c) Recovery fraction calculations on single exponential fits of FRAP recovery curves in (a) the presence of increasing concentrations of soluble GFP-tagged Rag GTPases. Shown are best fit values with 95% confidence intervals. (d) Montages showing fluorescence recovery over time for in vitro FRAP experiment of GFP-labeled Rag GTPases bound to GST-tagged Ragulator on beads, either in the presence or absence of excess GFP-labeled Rag GTPase heterodimers. Scale bar 10 μm. (e) Fluorescence recovery over time curves for (d). Fluorescence recovery occurs only when excess GFP-Rag heterodimers are present, indicating absence of lateral diffusion of the bead-bound Ragulator-Rag complexes into the bleached area. Each curve is the average ± S.E.M. of [+excess GFP-Rags N = 12, w/o excess GFP-Rags N = 9] bead regions. (f) Montages showing fluorescence recovery over time for in vitro FRAP experiment of bead-bound GST-RagB + GFP-RagC in the presence of excess FLAG-RagB + GFP-RagC. Scale bar 10 μm. (g) Fluorescence recovery over time curves for (f). No fluorescence recovery occurs, indicating that Rag heterodimers are stable and that no GFP-RagC exchanges between bead-bound GST-RagB and soluble FLAG-RagB. Curve is the average ± S.E.M. of N = 12 bead regions. See Supplementary Table 1 for statistical source data.
Supplementary Figure 5 Single molecule studies of mTORC1 scaffolding interactions.
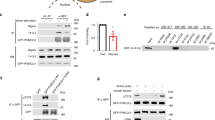
(a) Confocal images of glutathione beads coated with GST-Ragulator (left) or GST-inactive-loaded Rags (middle) or GST-active-loaded Rags (right) and incubated with an excess of GFP-Raptor (co-expressed along with mTOR and mLST8). Notice binding of GFP-Raptor to the surface of beads bearing active Rags, but not inactive Rags or Ragulator. Scale bar 100 μm. Experiment repeated 3 times. (b) Two-component exponential fits of survival probability curves determined from single molecule imaging of the indicated nucleotide loading combinations of GFP-tagged Rag GTPase heterodimers binding to GST-Ragulator on GST affinity beads. Graphs were randomly generated from ten percent of all single-molecule detections per condition. Datasets were each modeled by a two-component exponential fit and slow and fast time constants are reported as average +/- SEM. (c) Fluorescence recovery over time curves from FRAP experiments in U2OS cells expressing p18-VhH (p18 fused with a GFP nanobody) or p18 alone along with GFP-tagged Rag GTPases. Each curve is the average ± S.E.M. [blue N = 15, grey, N = 17] lysosomes. Experiment repeated 2 times. See Supplementary Table 1 for statistical source data.
Supplementary Figure 6 Investigation of Rag truncation interactions and the effects of RagC cancer mutant expression on mTORC1 accumulation on lysosomes.
(a) HEK-293T cells were transiently transfected with FLAG-tagged full-length or truncated RagB, along with GFP-tagged, full-length or truncated RagC. Cells were subjected to lysis and FLAG immunoprecipitation, followed by western blotting for FLAG and GFP. Experiment performed 1 time, Unprocessed scans are shown in Supplementary Fig. 8. (b) Subcellular localization of the indicated truncated GFP-RagB + FLAG-RagC heterodimers in U2OS cells. Scale bar 10 μm. Experiment repeated 2 times. (c) Images of Raptor:GFP localization in U2OS cells stably coexpressing RaptorGFP and Flag-tagged RagC containing the indicated mutations. Cells were maintained in the indicated nutrient conditions for 2 hours. Scale bar 10 μm. Experiment repeated 2 times. (d) Quantitation of the ratio between lysosomal and cytosolic fluorescence intensities (lyso:cyto ratios) from the images in (c) (mean ± S.D., N = 19, 24, 24 cells/condition respectively, left to right, **** p<.0001, two-sided unpaired t-tests). (e,f) 293T cells stably expressing the indicated FLAG-RagC constructs were starved for amino acids for 90 min, or starved and restimulated for the indicated times (5 min, 10 min, 20 min, or 30 min), followed by cell lysis and immunoblotting for the indicated proteins and phosphor-proteins. Experiment repeated 3 times. Unprocessed scans are shown in Supplementary Fig. 8. See Supplementary Table 1 for statistical source data.
Supplementary Figure 7 mTORC1 efficiently phosphorylates substrates when localized to mitochondrial membranes that contain Rheb.
(a) Immunofluorescence images of HEK-293T cells expressing FLAG-Raptor-Omp25 stained for FLAG and for the TOM20 mitochondrial marker. Scale bar 10 μm. Experiment repeated 2 times. (b) Immunofluorescence images of HEK-293T cells expressing Flag-Raptor-Omp25 (Raptor-OMP25) stained for endogenous mTOR and for FLAG (Raptor-OMP25). Experiment repeated 2 times. (c) U2OS cells transiently overexpressing FLAG-Raptor-Omp25 (Raptor-OMP25) and/or MYC-Rheb-Omp25 (Rheb-OMP25) were starved for amino acids for 50 min, or starved and restimulated for 10 min, followed by cell lysis and immunoblotting for the indicated proteins and phosphor-proteins. Experiment repeated 4 times. Unprocessed scans are shown in Supplementary Fig. 8. (d) HEK-293T cells stably expressing Flag-RagC containing the indicated mutations were treated with BafA for 2h, fixed and immunostained for LC3 and LAMP2. Images correspond to quantitative Lysosomal Enrichment Scores reported in Fig. 7c. Scale bar 10 μm. Experiment repeated 1 time.
Supplementary Figure 8 Unprocessed scans for all films presented in figures.
Included here are uncropped blots corresponding to the indicated figures.
Supplementary information
Supplementary Information
Supplementary Figures 1–8, Supplementary Table and Supplementary Video legends.
Supplementary Table 1
Statistical source data.
Supplementary Table 2
Antibody information.
Supplementary Table 3
Reagents and resources.
Supplementary Video 1
Rag GTPases cycle between cytoplasmic and lysosomal pools more rapidly in nutrient restimulated cells than in nutrient starved cells.
Supplementary Video 2
In vitro single molecule measurement of Rag residence times.
Supplementary Video 3
Rag heterodimers lacking a G domain have impaired cycling in vitro.
Rights and permissions
About this article
Cite this article
Lawrence, R.E., Cho, K.F., Rappold, R. et al. A nutrient-induced affinity switch controls mTORC1 activation by its Rag GTPase–Ragulator lysosomal scaffold. Nat Cell Biol 20, 1052–1063 (2018). https://doi.org/10.1038/s41556-018-0148-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0148-6
This article is cited by
-
The molecular basis of nutrient sensing and signalling by mTORC1 in metabolism regulation and disease
Nature Reviews Molecular Cell Biology (2023)
-
EGOC inhibits TOROID polymerization by structurally activating TORC1
Nature Structural & Molecular Biology (2023)
-
Molecular basis and design principles of switchable front-rear polarity and directional migration in Myxococcus xanthus
Nature Communications (2023)
-
Crosstalk of organelles in Parkinson’s disease – MiT family transcription factors as central players in signaling pathways connecting mitochondria and lysosomes
Molecular Neurodegeneration (2022)
-
Brain-enriched RagB isoforms regulate the dynamics of mTORC1 activity through GATOR1 inhibition
Nature Cell Biology (2022)