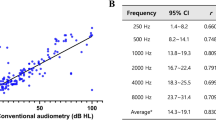
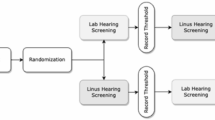
Abstract
Otoacoustic emissions (OAEs) provide information about the function of the outer hair cells of the cochlea. In high-income countries, infants undergo OAE tests as part of the screening protocols for hearing. However, the cost of the necessary equipment hinders early screening for hearing in low- and middle-income countries, which disproportionately bear the brunt of disabling hearing loss. Here we report the design and clinical testing of a low-cost probe for OAEs. The device, which has a material cost of approximately US$10, uses an off-the-shelf microphone and off-the-shelf earphones connected to a smartphone through a headphone jack. It sends two pure tones through each of the headphone’s earbuds and algorithmically detects the distortion-product OAEs generated by the cochlea and recorded via the microphone. In a clinical study involving 201 paediatric ears across three healthcare sites, the device detected hearing loss with 100% sensitivity and 88.9% specificity, comparable to the performance of a commercial device. Low-cost devices for OAE testing may aid the early detection of hearing loss in resource-constrained settings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data supporting the findings from this study are available within the article and its Supplementary Information. The dataset used to generate the results for this study is available at https://github.com/uw-x/oae and https://zenodo.org/record/7032657 (ref. 48). Source data are provided with this paper.
Code availability
The custom code used in this study is available at https://github.com/uw-x/oae and https://zenodo.org/record/7032657 (ref. 48).
References
Millions of People in the World Have Hearing Loss That Can Be Treated or Prevented (WHO, 2013); https://www.who.int/news/item/27-02-2013-millions-have-hearing-loss-that-can-be-improved-or-prevented
Childhood Hearing Loss—Strategies for Prevention and Care (WHO, 2016); https://apps.who.int/iris/bitstream/handle/10665/204632/9789241510325_eng.pdf
Patel, H. & Feldman, M. Universal newborn hearing screening. Paediatr. Child Health 16, 301–305 (2011).
McPherson, B. Newborn hearing screening in developing countries: needs & new directions. Indian J. Med. Res. 135, 152 (2012).
Swanepoel, D. W. et al. Telehealth in audiology: the need and potential to reach underserved communities. Int. J. Audiol. 49, 195–202 (2010).
Chan, J. & Gollakota, S. Inner-ear cochlea testing with earphones. In Proc. 20th Annual International Conference on Mobile Systems, Applications and Services 609–610 (ACM, 2022).
Abdala, C. & Visser-Dumont, L. Distortion product otoacoustic emissions: a tool for hearing assessment and scientific study. Volta. Rev. 103, 281 (2001).
Walker, J. J., Cleveland, L. M., Davis, J. L. & Seales, J. S. Audiometry screening and interpretation. Am. Fam. Physician 87, 41–47 (2013).
Mimi Hearing Test (Mimi Hearing Technologies GmbH, 2022); https://apps.apple.com/us/app/mimi-hearing-test/id932496645
Hearing Test—Audiometry, Tone (IT4YOU Corp., 2022); https://apps.apple.com/us/app/hearing-test-audiometry-tone/id1368396053
Hearing Test & Ear Age Test (PortTownSoft, 2022); https://apps.apple.com/us/app/hearing-test-ear-age-test/id1067630100
Thompson, D. C. et al. Universal newborn hearing screening: summary of evidence. JAMA 286, 2000–2010 (2001).
Torre, P. III, Cruickshanks, K. J., Nondahl, D. M. & Wiley, T. L. Distortion product otoacoustic emission response characteristics in older adults. Ear Hear. 24, 20–29 (2003).
Heitmann, N., Rosner, T. & Chakraborty, S. Designing a single speaker-based ultra low-cost otoacoustic emission hearing screening probe. In 2020 IEEE Global Humanitarian Technology Conference (GHTC) 1–8 (IEEE, 2020).
Campbell, L. J. & Slater, K. D. Personalization of auditory stimulus. US patent 10154333B2 (2018).
Heitmann, N., Rosner, T. & Chakraborty, S. Mass-deployable smartphone-based objective hearing screening with otoacoustic emissions. In Proc. 2021 International Conference on Multimodal Interaction 653–661 (ACM, 2021).
Heitmann, N. et al. Sound4all: towards affordable large-scale hearing screening. In 2017 12th International Conference on Design & Technology of Integrated Systems in Nanoscale Era (DTIS) 1–6 (IEEE, 2017).
Novel pediatric hearing screening device. Johns Hopkins Technological Ventures http://jhu.technologypublisher.com/technology/40355 (2018).
Generic eartips from Grason Associates fitment guide eartips. Electro-Medical Instruments https://www.emi-canada.com/new-page-5 (2021).
Newborn hearing screening protocol. Iowa Department of Public Health https://idph.iowa.gov/Portals/1/userfiles/77/Newborn%20Hearing%20Screening%20Protocol-11-08.pdf (2008).
Parthasarathy, T. K. & Klostermann, B. Similarities and differences in distortion-product otoacoustic emissions among four FDA-approved devices. J. Am. Acad. Audiol. 12, 397–405 (2001).
Frequently asked questions: EroScan OAE Test System. Maico https://www.schoolhealth.com/media/pdf/51075_FAQ%20EroScan.pdf (2022).
Chan, J., Michaelsen, K., Estergreen, J. K., Sabath, D. E. & Gollakota, S. Micro-mechanical blood clot testing using smartphones. Nat. Commun. 13, 1–12 (2022).
Funk, J. Technology change, economic feasibility, and creative destruction: the case of new electronic products and services. Ind. Corp. Change 27, 65–82 (2018).
Device compatibility overview. Android Developers https://developer.android.com/guide/practices/compatibility (2022).
For iPhone headphone adapter 3.5 mm jack aux cord dongle audio cable connector US. eBay https://www.ebay.com/itm/313802818623 (2022).
Device software functions including mobile medical applications. US FDA https://www.fda.gov/medical-devices/digital-health-center-excellence/device-software-functions-including-mobile-medical-applications (2019).
Policy for device software functions and mobile medical applications. US FDA https://www.fda.gov/regulatory-information/search-fda-guidance-documents/policy-device-software-functions-and-mobile-medical-applications (2019).
Software as a medical device (SaMD). US FDA https://www.fda.gov/medical-devices/digital-health-center-excellence/software-medical-device-samd (2018).
Software as a Medical Device: Possible Framework for Risk Categorization and Corresponding Considerations (2014); http://www.imdrf.org/docs/imdrf/final/technical/imdrf-tech-140918-samd-framework-risk-categorization-141013.pdf
What are examples of software as a medical device? US FDA https://www.fda.gov/medical-devices/software-medical-device-samd/what-are-examples-software-medical-device (2017).
Examples of premarket submissions that include MMAs cleared or approved by the FDA. US FDA https://www.fda.gov/medical-devices/device-software-functions-including-mobile-medical-applications/examples-premarket-submissions-include-mmas-cleared-or-approved-fda (2019).
Chan, J., Raju, S., Nandakumar, R., Bly, R. & Gollakota, S. Detecting middle ear fluid using smartphones. Sci. Transl. Med. 11, eaav1102 (2019).
Chan, J. et al. Performing tympanometry using smartphones. Commun. Med. 2, 57 (2022).
Siegel, J. & Hirohata, E. Sound calibration and distortion product otoacoustic emissions at high frequencies. Hear. Res. 80, 146–152 (1994).
Siegel, J. Ear-canal standing waves and high-frequency sound calibration using otoacoustic emission probes. J. Acoust. Soc. Am. 95, 2589–2597 (1994).
Charaziak, K. K. & Shera, C. A. Compensating for ear-canal acoustics when measuring otoacoustic emissions. J. Acoust. Soc. Am. 141, 515–531 (2017).
Maico Diagnostics. School Nurses Guide to Otoacoustic Emissions (OAEs) (2009); https://www.schoolhealth.com/media/pdf/School%20Nurse%20Guide%20to%20OAEs_web%202016.pdf
Hall, J. W. III The Important Role of Otoacoustic Emissions (OAEs) in Preschool Hearing Screening (2018); https://www.audiologyonline.com/articles/important-role-otoacoustic-emissions-oaes-22091
Technical specifications—Lyra. Interacoustics https://www.interacoustics.com/download/lyra/3111-datasheet-for-lyra (2019).
OtoRead™ – instructions for use. Interacoustics https://www.interacoustics.com/download/otoread/manuals-otoread/1236-instructions-for-use-otoread (2021).
Welch Allyn® OAE hearing screener—directions for use. Welch Allyn https://www.henryschein.com/assets/Medical/80022349LITPDF.pdf (2019).
r140 Portable OAE hearing screener. Resonance http://www.resonance-audiology.com/wp-content/uploads/2018/05/R14O-TECNICAL-SPECIFICATION.pdf (2018).
AVLinearPCMBitDepthKey. Apple Developer Documentation https://developer.apple.com/documentation/avfaudio/avlinearpcmbitdepthkey (2022).
USB Digital Audio. Android Open Source Project https://source.android.com/devices/audio/usb (2022).
Reavis, K. M. et al. Distortion-product otoacoustic emission test performance for ototoxicity monitoring. Ear Hear. 32, 61 (2011).
Goodman, S. S., Fitzpatrick, D. F., Ellison, J. C., Jesteadt, W. & Keefe, D. H. High-frequency click-evoked otoacoustic emissions and behavioral thresholds in humans. J. Acoust. Soc. Am. 125, 1014–1032 (2009).
Chan, J. et al. A low-cost smartphone-based otoacoustic emission device for hearing loss. Zenodo https://doi.org/10.5281/zenodo.7032657 (2022).
Acknowledgements
We thank our participants and their families at Seattle Children’s Hospital. We thank K. Sie, D. Horn and H. Ou at the Seattle Children’s Hospital and Research Institute for permitting us to recruit patients from their clinics. We thank S. Norton for her critical and important feedback on the manuscript and methods. N.A. discloses support for the research described in this study from the National Institute on Deafness and Other Communication Disorders award T32DC000018. R.B. and S.G. disclose support for the research described in this study from the Washington Research Foundation, Seattle Children’s Research Institute, Research Integration Hub. S.G. discloses support for the research described in this study from the Moore Inventor Fellow award #10617.
Author information
Authors and Affiliations
Contributions
All authors designed the experiments and interpreted the results. J.C. and S.G. wrote the manuscript, and N.A., A.M., L.R.M., E.G. and R.B. edited the manuscript. J.C., N.A. and A.M. conducted the experiments and performed the analysis, under technical supervision by S.G. and J.C.; A.N. and S.G. designed the algorithms. J.C. and S.G. conceptualized the study.
Corresponding authors
Ethics declarations
Competing interests
S.G., J.C. and R.B. are co-founders of Wavely Diagnostics, Inc. S.G. is a co-founder of Jeeva Wireless, Inc. and Sound Life Sciences. R.B. is a co-founder of EigenHealth, Inc. R.B. is a consultant and stockholder of Spiway, LLC. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary note, figures and tables.
Supplementary data
Source data for Supplementary Figs. 2, 3 and 8.
Source data
Source data for Figs. 2–7.
Source data for all main figures.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chan, J., Ali, N., Najafi, A. et al. An off-the-shelf otoacoustic-emission probe for hearing screening via a smartphone. Nat. Biomed. Eng 6, 1203–1213 (2022). https://doi.org/10.1038/s41551-022-00947-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00947-6
This article is cited by
-
Affordable hearing screening
Nature Biomedical Engineering (2022)