Abstract
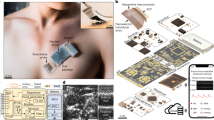
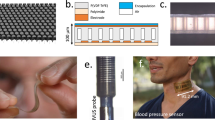
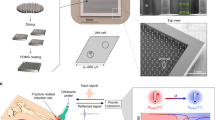
Stretchable wearable devices for the continuous monitoring of physiological signals from deep tissues are constrained by the depth of signal penetration and by difficulties in resolving signals from specific tissues. Here, we report the development and testing of a prototype skin-conformal ultrasonic phased array for the monitoring of haemodynamic signals from tissues up to 14 cm beneath the skin. The device allows for active focusing and steering of ultrasound beams over a range of incident angles so as to target regions of interest. In healthy volunteers, we show that the phased array can be used to monitor Doppler spectra from cardiac tissues, record central blood flow waveforms and estimate cerebral blood supply in real time. Stretchable and conformal skin-worn ultrasonic phased arrays may open up opportunities for wearable diagnostics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Data generated in this study, including source data and the data used to produce the figures, are available from Harvard Dataverse at https://dataverse.harvard.edu/dataset.xhtml?persistentId=doi:10.7910/DVN/EPITIY.
References
Tran, D. D., Groeneveld, A., Nauta, J., van Schijndel Strack, R. & Thijs, L. Age, chronic disease, sepsis, organ system failure, and mortality in a medical intensive care unit. Crit. Care Med. 18, 474–479 (1990).
Knaus, W. A., Draper, E. A., Wagner, D. P. & Zimmerman, J. E.Prognosis in acute organ-system failure. Ann. Surg. 202, 685–693 (1985).
Tsai, M.-H. et al. Organ system failure scoring system can predict hospital mortality in critically ill cirrhotic patients. J. Clin. Gastroenterol. 37, 251–257 (2003).
Agrawal, D. R. et al. Conformal phased surfaces for wireless powering of bioelectronic microdevices. Nat. Biomed. Eng. 1, 0043 (2017).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Someya, T., Bao, Z. & Malliaras, G. G. The rise of plastic bioelectronics. Nature 540, 379–385 (2016).
Rogers, J. A., Someya, T. & Huang, Y. Materials and mechanics for stretchable electronics. Science 327, 1603–1607 (2010).
Kim, J. et al. Battery-free, stretchable optoelectronic systems for wireless optical characterization of the skin. Sci. Adv. 2, e1600418 (2016).
Chung, H. U. et al. Binodal, wireless epidermal electronic systems with in-sensor analytics for neonatal intensive care. Science 363, eaau0780 (2019).
Khan, Y. et al. A flexible organic reflectance oximeter array. Proc. Natl Acad. Sci. USA 115, E11015–E11024 (2018).
Webb, R. C. et al. Epidermal devices for noninvasive, precise, and continuous mapping of macrovascular and microvascular blood flow. Sci. Adv. 1, e1500701 (2015).
Tian, L. et al. Flexible and stretchable 3ω sensors for thermal characterization of human skin. Adv. Funct. Mater. 27, 1701282 (2017).
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotech. 37, 389–406 (2019).
Suzuki, D., Oda, S. & Kawano, Y. A flexible and wearable terahertz scanner. Nat. Photon. 10, 809–813 (2016).
Garbacz, P. Terahertz imaging—principles, techniques, benefits, and limitations. J. Mach. Constr. Maint. 1, 81–92 (2016).
You, B., Chen, C.-Y., Yu, C.-P., Wang, P.-H. & Lu, J.-Y. Frequency-dependent skin penetration depth of terahertz radiation determined by water sorption–desorption. Opt. Express 26, 22709–22721 (2018).
Hu, H. et al. Stretchable ultrasonic transducer arrays for three-dimensional imaging on complex surfaces. Sci. Adv. 4, eaar3979 (2018).
Wang, C. et al. Monitoring of the central blood pressure waveform via a conformal ultrasonic device. Nat. Biomed. Eng. 2, 687–695 (2018).
Schulze, R. K., Ćurić, D. & d’Hoedt, B. B-mode versus A-mode ultrasonographic measurements of mucosal thickness in vivo. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 93, 110–117 (2002).
Van Keuk, G. & Blackman, S. S. On phased-array radar tracking and parameter control. IEEE Trans. Aerosp. Electron. Syst. 29, 186–194 (1993).
Saucan, A.-A., Chonavel, T., Sintes, C. & Le Caillec, J.-M. CPHD-DOA tracking of multiple extended sonar targets in impulsive environments. IEEE Trans. Signal Process. 64, 1147–1160 (2015).
Nissen, S. E. et al. Application of a new phased-array ultrasound imaging catheter in the assessment of vascular dimensions. In vivo comparison to cineangiography. Circulation 81, 660–666 (1990).
Baribeau, Y. et al. Hand-held point of care ultrasound probes—the new generation of POCUS. J. Cardiothorac. Vasc. Anesth. 34, 3139–3145 (2020).
Cheng, J., Potter, J. N., Croxford, A. J. & Drinkwater, B. W. Monitoring fatigue crack growth using nonlinear ultrasonic phased array imaging. Smart Mater. Struct. 26, 055006 (2017).
J. Lee, H.-S. & Sodini, C. G. in Intracranial Pressure & Neuromonitoring XVI 111–114 (Springer, 2018).
Daum, D. R., Smith, N. B., King, R. & Hynynen, K. In vivo demonstration of noninvasive thermal surgery of the liver and kidney using an ultrasonic phased array. Ultrasound Med. Biol. 25, 1087–1098 (1999).
Shi, K., Que, K. & Guo, D. Flexible ultrasonic phased-array probe. Tsinghua Sci. Technol. 9, 574–577 (2004).
Bhuyan, A. et al. Miniaturized, wearable, ultrasound probe for on-demand ultrasound screening. In Proc. IEEE International Ultrasonics Symposium 1060–1063 (IEEE, 2011).
Pashaei, V. et al. Flexible body-conformal ultrasound patches for image-guided neuromodulation. IEEE Trans. Biomed. Circuits Syst. 14, 305–318 (2019).
Abdelfattah, R. et al. Outbreak of Burkholderia cepacia bacteraemia in a tertiary care centre due to contaminated ultrasound probe gel. J. Hosp. Infect. 98, 289–294 (2018).
Thomenius, K. E. Evolution of ultrasound beamformers. In Proc. IEEE International Ultrasonics Symposium 1615–1622 (IEEE, 1996).
Smith, S. W., Pavy, H. G. & von Ramm, O. T. High-speed ultrasound volumetric imaging system. I. Transducer design and beam steering. IEEE T. Ultrason. Ferr. 38, 100–108 (1991).
Gill, R. W. Measurement of blood flow by ultrasound: accuracy and sources of error. Ultrasound Med. Biol. 11, 625–641 (1985).
Wooh, S.-C. & Shi, Y. A simulation study of the beam steering characteristics for linear phased arrays. J. Nondestruct. Eval. 18, 39–57 (1999).
Cloostermans, M. & Thijssen, J. A beam corrected estimation of the frequency dependent attenuation of biological tissues from backscattered ultrasound. Ultrason. Imaging 5, 136–147 (1983).
Ng, A. & Swanevelder, J. Resolution in ultrasound imaging. Continuing Educ. Anaesthesia Crit. Care Pain 11, 186–192 (2011).
Gururaja, T. et al. Piezoelectric composite materials for ultrasonic transducer applications. Part I: resonant modes of vibration of PZT rod-polymer composites. IEEE Trans. Sonics Ultrason. 32, 481–498 (1985).
Food & Drug Administration Marketing Clearance of Diagnostic Ultrasound Systems and Transducers (Center for Devices and Radiological Health, 2019).
Gazit, S., Szameit, A., Eldar, Y. C. & Segev, M. Super-resolution and reconstruction of sparse sub-wavelength images. Opt. Express 17, 23920–23946 (2009).
Mills, D. M. Medical imaging with capacitive micromachined ultrasound transducer (CMUT) arrays. In Proc. IEEE Ultrasonics Symposium 384–390 (IEEE, 2004).
Ophir, J., McWhirt, R., Maklad, N. & Jaeger, P. A narrowband pulse-echo technique for in vivo ultrasonic attenuation estimation. In Proc. IEEE Transactions on Biomedical Engineering 205–212 (IEEE, 1985).
Hunter, A. J., Drinkwater, B. W., Wilcox, P. & Russell, J. Automatic image correction for flexible ultrasonic phased array inspection. In Proc. 8th International Conference on NDE in Relation to Structural Integrity for Nuclear and Pressurised Components (JRC-NDE, 2010).
Shung, K. K., Sigelmann, R. A. & Reid, J. M. Scattering of ultrasound by blood. In Proc. IEEE Transactions on Biomedical Engineering 460–467 (IEEE, 1976).
Von Ramm, O. T. & Smith, S. W. Beam steering with linear arrays. In Proc. IEEE Transactions on Biomedical Engineering 438–452 (IEEE, 1983).
Ketterling, J. A., Aristizabal, O., Turnbull, D. H. & Lizzi, F. L. Design and fabrication of a 40-MHz annular array transducer. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 52, 672–681 (2005).
Abbasi, A. S., Chahine, R. A., Mac Alpin, R. N. & Kattus, A. A. Ultrasound in the diagnosis of primary congestive cardiomyopathy. Chest 63, 937–942 (1973).
Mohan, J. C. A Practical Approach to Clinical Echocardiography (JP Medical, 2014).
Ommen, S. R. et al. Clinical utility of Doppler echocardiography and tissue Doppler imaging in the estimation of left ventricular filling pressures: a comparative simultaneous Doppler-catheterization study. Circulation 102, 1788–1794 (2000).
Galiuto, L., Ignone, G. & DeMaria, A. N. Contraction and relaxation velocities of the normal left ventricle using pulsed-wave tissue Doppler echocardiography. J. Am. Coll. Cardiol. 81, 609–614 (1998).
Søgaard, P. et al. Tissue Doppler imaging predicts improved systolic performance and reversed left ventricular remodeling during long-term cardiac resynchronization therapy. Am. J. Cardiol. 40, 723–730 (2002).
Auricchio, A. et al. Long-term clinical effect of hemodynamically optimized cardiac resynchronization therapy in patients with heart failure and ventricular conduction delay. Am. J. Cardiol. 39, 2026–2033 (2002).
Auricchio, A. & Abraham, W. T. Cardiac resynchronization therapy: current state of the art: cost versus benefit. Circulation 109, 300–307 (2004).
Thimot, J. & Shepard, K. L. Bioelectronic devices: wirelessly powered implants. Nat. Biomed. Eng. 1, 0051 (2017).
Joyner, C. R. Jr, Hey, E. B. Jr, Johnson, J. & Reid, J. M. Reflected ultrasound in the diagnosis of tricuspid stenosis. Am. J. Cardiol. 19, 66–73 (1967).
Flammer, A. J. et al. The assessment of endothelial function: from research into clinical practice. Circulation 126, 753–767 (2012).
Petrofsky, J. S. Resting blood flow in the skin: does it exist, and what is the influence of temperature, aging, and diabetes? J. Diabetes Sci. Technol. 6, 674–685 (2012).
Herholz, K. et al. Regional cerebral blood flow in patients with leuko-araiosis and atherosclerotic carotid artery disease. Arch. Neurol. 47, 392–396 (1990).
Boutry, C. M. et al. Biodegradable and flexible arterial-pulse sensor for the wireless monitoring of blood flow. Nat. Biomed. Eng. 3, 47–57 (2019).
Dymling, S. O., Persson, H. W. & Hertz, C. H. Measurement of blood perfusion in tissue using Doppler ultrasound. Ultrasound Med. Bio. 17, 433–444 (1991).
Chung, C. P. et al. More severe white matter changes in the elderly with jugular venous reflux. Ann. Neurol. 69, 553–559 (2011).
Merritt, C. R. Doppler colour flow imaging. J. Clin. Ultrasound 15, 591–597 (1987).
Bland, J. M. & Altman, D. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 327, 307–310 (1986).
Bondar, R. L. et al. Cerebral blood flow velocities by transcranial Doppler during parabolic flight. J. Clin. Pharmacol. 31, 915–919 (1991).
Jawad, K., Miller, J. D., Wyper, D. J. & Rowan, J. O. Measurement of CBF and carotid artery pressure compared with cerebral angiography in assessing collateral blood supply after carotid ligation. J. Neurosurg. 46, 185–196 (1977).
Cope, M. & Delpy, D. T. System for long-term measurement of cerebral blood and tissue oxygenation on newborn infants by near infra-red transillumination. Med. Biol. Eng. Comput. 26, 289–294 (1988).
Soustiel, J. et al. Assessment of cerebral blood flow by means of blood-flow-volume measurement in the internal carotid artery: comparative study with a 133xenon clearance technique. Stroke 34, 1876–1880 (2003).
Gunst, M. et al. Accuracy of cardiac function and volume status estimates using the bedside echocardiographic assessment in trauma/critical care. J. Trauma Acute Care Surg. 65, 509–516 (2008).
Podstawski, R., Kasietczuk, B., Boraczyński, T., Boraczyński, M. & Choszcz, D. Relationship between BMI and endurance-strength abilities assessed by the 3 minute burpee test. Int. J. Sports Sci. 3, 28–35 (2013).
Ma, Y.-C., Peng, C.-T., Huang, Y.-C., Lin, H.-Y. & Lin, J.-G.The depths from skin to the major organs at chest acupoints of pediatric patients. Evid. Based Complement. Altern. Med. 2015, 126028 (2015).
Huang, Z. et al. Three-dimensional integrated stretchable electronics. Nat. Electron. 1, 473–480 (2018).
Burckhardt, C. B. Speckle in ultrasound B-mode scans. IEEE Trans. Sonics Ultrason. 25, 1–6 (1978).
Liu, Y. et al. Epidermal mechano-acoustic sensing electronics for cardiovascular diagnostics and human-machine interfaces. Sci. Adv. 2, e1601185 (2016).
Delgado-Trejos, E., Quiceno-Manrique, A., Godino-Llorente, J., Blanco-Velasco, M. & Castellanos Dominguez, G. Digital auscultation analysis for heart murmur detection. Ann. Biomed. Eng. 37, 337–353 (2009).
Lee, K. et al. Mechano-acoustic sensing of physiological processes and body motions via a soft wireless device placed at the suprasternal notch. Nat. Biomed. Eng. 4, 148–158 (2019).
Acknowledgements
We thank Z. Wu and L. Chen for guidance on the ultrasonic imaging algorithm and data processing; S. Xiang for feedback on manuscript preparation; and Y. Hu and Z. Liu for advice on the cardiac tissue Doppler experiments. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health (NIH). All biological experiments were conducted in accordance with the ethical guidelines of the NIH and with the approval of the IRB of the University of California, San Diego (IRB number 170812). We acknowledge support from the NIH (grant 1R21EB027303-01A1) and the Center for Wearable Sensors at the University of California, San Diego.
Author information
Authors and Affiliations
Contributions
C.W., B.Q., M.L., Z.Z. and S.X. designed the research. C.W., B.Q., M.L., B.L. and Z.Z. performed the experiments. C.W., B.Q. and Z.Z. performed the simulations. C.W., Z.Z., B.Q. and M.L. analysed the data. C.W. and S.X. wrote the paper. All authors provided active and valuable feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Biomedical Engineering thanks John Ho and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Comparison of existing flexible or stretchable electronic devices for deep-tissue monitoring.
a, Comparison of the detection range (shaded areas underneath each sensing mode) of the stretchable phased array with all other flexible/stretchable biosensors, including optoelectronics, thermal electronics, iontophoresis based electrochemical sensors, terahertz wave-based sensors, a single ultrasonic transducer, and electronic stethoscopes. The phased array has distinct advantages compared to the other sensing modalities in both penetration depth and spatial resolution. With phased array beamforming, the stretchable ultrasonic probe can focus the beam to achieve longer penetration (up to 14 cm in human tissues) and steer the beam to scan over a 40° range with a high signal-to-noise ratio (SNR). The insonation area in 2D can reach ~68.41 cm2, 380 times larger than the insonation area of a single element, ~0.18 cm2, calculated by Field II, MATLAB, MathWorks, Natick, MA. b, Comparison of the penetration depth and detectable object size of representative flexible/stretchable biosensors in the literature. Stretchable optoelectronics illuminates the tissue to analyze changes in blood oxygen saturation, but the depth of penetration is generally limited to ~8 mm8,9,10. Stretchable thermal electronics suffer from similar limitations11,12. Although flexible terahertz devices show high penetration in dielectric materials14, terahertz electromagnetic waves attenuate quickly in water15, leading to a maximum penetration depth of only 0.3 mm in the human tissue16. Flexible electromagnetic devices in the frequency range of ~GHz can penetrate more than 5 cm of human tissue, but their spatial resolution ranges from 5 to 50 cm, which is inadequate for most biomedical applications4. Stretchable stethoscopes based on kinematics can sense vibrations far beneath the skin72; but due to the omnidirectional propagation of vibrations, these devices can receive signals from all organs in their sensing range, and thus lack spatial resolution73,74. Additionally, they can only detect low-frequency pulsations, missing critical information from static objects (for example, their location, dimension, and modulus) or high-frequency pulsations (for example, hemodynamics). The result shows the stretchable phased array has a long penetration depth with sufficient resolving capabilities for deep tissue monitoring.
Extended Data Fig. 2 Performance of the stretchable phased array at all incidence angles in the xz plane on the phantom replicated from the human neck.
a, A schematic setup of the characterization. Ultrasonic field mapping is carried out with the stretchable phased array on a flat (top left) or a curvilinear (top right) surface. The characterization system is composed of a water tank and a 3D motor system with a hydrophone at its tip. b, Mapped ultrasonic fields with the stretchable phased array on a flat (left) or a curvilinear (right) surface with an incidence angle range of 0° - 25°. Scale bars are 2 cm. c, Normalized beam intensity of the stretchable phased array at a typical carotid artery depth (~3 cm) with different incident angles. The inset illustrates the depth at which the intensity measurements are taken. d, Beam directivity of the stretchable phased array at a typical carotid artery depth (~3 cm) with different incident angles. The beam directivity of the stretchable phased array is comparable on the two surfaces at all incident angles in the range tested.
Extended Data Fig. 3 Raw radiofrequency signals of the tissue Doppler in the time domain, and corresponding human anatomy.
a, Raw radiofrequency signals of the tissue Doppler in one cardiac cycle. Note that the y-axis is time. The x-axis is converted to depth by multiplying the ultrasonic speed by the time-of-flight of ultrasonic pulses in the human tissue. As a proof of concept, we neglect the non-uniformity of the ultrasonic speed in the human body. From the pattern, we can clearly observe the reflection peaks from the anterior wall of the right ventricle (RV) in the blue dashed box and the posterior wall of the left ventricle (LV) in the orange dashed box. The positions of the reflection peaks shift during the cardiac cycle. The RV anterior wall peak shifts to the left from 0 to 0.6 s, corresponding to RV diastole, and shifts to the right from 0.6 to 0.8 s, corresponding to RV systole. Meanwhile, the LV posterior wall peak shifts to the right from 0 to 0.6 s, corresponding to LV diastole, and shifts to the left from 0.6 to 0.8 s, corresponding to LV systole. The peak shifting clearly indicates the relaxation and contraction of the cardiac chambers. b, The cross-sectional structure of the parasternal short-axis view corresponding to the signal peaks in a.
Supplementary information
Supplementary Information
Supplementary Discussions 1–10, Figs. 1–38 and captions for Supplementary Videos 1–3.
Supplementary Video 1
Phased-array steerability.
Supplementary Video 2
Comparison of wave propagation of the stretchable phased array on planar and curved surfaces.
Supplementary Video 3
Real-time imaging of the conformal probe on a deep-tissue phantom.
Rights and permissions
About this article
Cite this article
Wang, C., Qi, B., Lin, M. et al. Continuous monitoring of deep-tissue haemodynamics with stretchable ultrasonic phased arrays. Nat Biomed Eng 5, 749–758 (2021). https://doi.org/10.1038/s41551-021-00763-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-021-00763-4
This article is cited by
-
Injectable and retrievable soft electronics
Nature Materials (2024)
-
Permanent fluidic magnets for liquid bioelectronics
Nature Materials (2024)
-
A three-dimensional liquid diode for soft, integrated permeable electronics
Nature (2024)
-
Wearable ultrasound for continuous deep-tissue monitoring
Nature Biotechnology (2024)
-
A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects
Nature Biotechnology (2024)