Abstract
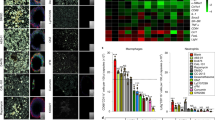
Silicone is widely used in chronic implants and is generally perceived to be safe. However, textured breast implants have been associated with immune-related complications, including malignancies. Here, by examining for up to one year the foreign body response and capsular fibrosis triggered by miniaturized or full-scale clinically approved breast implants with different surface topography (average roughness, 0–90 μm) placed in the mammary fat pads of mice or rabbits, respectively, we show that surface topography mediates immune responses to the implants. We also show that the surface surrounding human breast implants collected during revision surgeries also differentially alters the individual’s immune responses to the implant. Moreover, miniaturized implants with an average roughness of 4 μm can largely suppress the foreign body response and fibrosis (but not in T-cell-deficient mice), and that tissue surrounding these implants displayed higher levels of immunosuppressive FOXP3+ regulatory T cells. Our findings suggest that, amongst the topographies investigated, implants with an average roughness of 4 μm provoke the least amount of inflammation and foreign body response.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the findings of this study are available within the paper and its Supplementary Information. The raw and analysed datasets generated during the study are available for research purposes from the corresponding authors on reasonable request. High-throughput sequencing data have been deposited in the Gene Expression Omnibus (GEO) database, with series accession number GSE164645.
References
Teo, A. J. T. et al. Polymeric biomaterials for medical implants and devices. ACS Biomater. Sci. Eng. 2, 454–472 (2016).
Lloyd, A. W., Faragher, R. G. & Denyer, S. P. Ocular biomaterials and implants. Biomaterials 22, 769–785 (2001).
McLaughlin, K., Jones, B., Mactier, R. & Porteus, C. Long-term vascular access for hemodialysis using silicon dual-lumen catheters with guidewire replacement of catheters for technique salvage. Am. J. Kidney Dis. 29, 553–559 (1997).
Khoo, C. T. Silicone synovitis. The current role of silicone elastomer implants in joint reconstruction. J. Hand Surg. Br. 18, 679–686 (1993).
VandeVord, P. J. et al. Immune reactions associated with silicone-based ventriculo-peritoneal shunt malfunctions in children. Biomaterials 25, 3853–3860 (2004).
Gabriel, A. & Maxwell, G. P. The evolution of breast implants. Clin. Plast. Surg. 42, 399–404 (2015).
Yoda, R. Elastomers for biomedical applications. J. Biomater. Sci. Polym. Ed. 9, 561–626 (1998).
Hanak, B. W., Bonow, R. H., Harris, C. A. & Browd, S. R. Cerebrospinal fluid shunting complications in children. Pediatr. Neurosurg. 52, 381–400 (2017).
O’Malley, J. T., Burgess, B. J., Galler, D. & Nadol, J. B. Jr. Foreign body response to silicone in cochlear implant electrodes in the human. Otol. Neurotol. 38, 970–977 (2017).
Coroneos, C. J. et al. US FDA breast implant postapproval studies: long-term outcomes in 99,993 patients. Ann. Surg. 269, 30–36 (2019).
Headon, H., Kasem, A. & Mokbel, K. Capsular contracture after breast augmentation: an update for clinical practice. Arch. Plast. Surg. 42, 532–543 (2015).
Castel, N., Soon-Sutton, T., Deptula, P., Flaherty, A. & Parsa, F. D. Polyurethane-coated breast implants revisited: a 30-year follow-up. Arch. Plast. Surg. 42, 186–193 (2015).
Xu, H. et al. Hydrogel-coated ventricular catheters for high-risk patients receiving ventricular peritoneum shunt. Medicine 95, e4252 (2016).
Barnea, Y., Hammond, D. C., Geffen, Y., Navon-Venezia, S. & Goldberg, K. Plasma activation of a breast implant shell in conjunction with antibacterial irrigants enhances antibacterial activity. Aesthet. Surg. J. 38, 1188–1196 (2018).
Stevens, W. G. et al. Risk factor analysis for capsular contracture: a 5-year Sientra study analysis using round, smooth, and textured implants for breast augmentation. Plast. Reconstr. Surg. 132, 1115–1123 (2013).
Mempin, M., Hu, H., Chowdhury, D., Deva, A. & Vickery, K. The A, B and C’s of silicone breast implants: anaplastic large cell lymphoma, biofilm and capsular contracture. Materials 11, 2393 (2018).
Mendonça, A. M., Santanelli di Pompeo, F. & De Mezerville, R. Nanotechnology, nanosurfaces and silicone gel breast implants: current aspects. Case Rep. Plast. Surg. Hand Surg. 4, 99–113 (2017).
Munhoz, A. M., Clemens, M. W. & Nahabedian, M. Y. Breast implant surfaces and their impact on current practices: where we are now and where are we going. Plast. Reconstr. Surg. Global Open 7, e2466 (2019).
Technical Committee ISO/TC 150. ISO 14607 Non-Active Surgical Implants—Mammary Implants—Particular Requirements 3rd edn (ISO copyright office, 2018).
Barnsley, G. P., Sigurdson, L. J. & Barnsley, S. E. Textured surface breast implants in the prevention of capsular contracture among breast augmentation patients: a meta-analysis of randomized controlled trials. Plast. Reconstr. Surg. 117, 2182–2190 (2006).
Derby, B. M. & Codner, M. A. Textured silicone breast implant use in primary augmentation: core data update and review. Plast. Reconstr. Surg. 135, 113–124 (2015).
Tevis, S. E. et al. Breast implant-associated anaplastic large cell lymphoma: a prospective series of 52 patients. Ann. Surg. https://doi.org/10.1097/SLA.0000000000004035 (2020).
Clemens, M. W. et al. Complete surgical excision is essential for the management of patients with breast implant-associated anaplastic large-cell lymphoma. J. Clin. Oncol. 34, 160–168 (2016).
Kadin, M. E. et al. Biomarkers provide clues to early events in the pathogenesis of breast implant-associated anaplastic large cell lymphoma. Aesthet. Surg. J. 36, 773–781 (2016).
Chung, L. et al. Interleukin-17 and senescent cells regulate the foreign body response to synthetic material implants in mice and humans. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aax3799 (2020).
Loch-Wilkinson, A. et al. Breast implant-associated anaplastic large cell lymphoma in Australia and New Zealand: high-surface-area textured implants are associated with increased risk. Plast. Reconstr. Surg. 140, 645–654 (2017).
Hallab, N. J., Samelko, L. & Hammond, D. The inflammatory effects of breast implant particulate shedding: comparison with orthopedic implants. Aesthet. Surg. J. 39, S36–S48 (2019).
Parham, C. S. et al. Advising patients about breast implant associated anaplastic large cell lymphoma. Gland Surg. 10, 417–442 (2020).
Hall-Findlay, E. J. Breast implant complication review: double capsules and late seromas. Plast. Reconstr. Surg. 127, 56–66 (2011).
Wolfram, D. et al. T regulatory cells and TH17 cells in peri-silicone implant capsular fibrosis. Plast. Reconstr. Surg. 129, 327–337 (2012).
Chung, L. et al. Interleukin 17 and senescent cells regulate the foreign body response to synthetic material implants in mice and humans. Sci. Transl. Med. 12, eaax3799 (2020).
Flemming, R. G., Murphy, C. J., Abrams, G. A., Goodman, S. L. & Nealey, P. F. Effects of synthetic micro- and nano-structured surfaces on cell behavior. Biomaterials 20, 573–588 (1999).
Jain, N. & Vogel, V. Spatial confinement downsizes the inflammatory response of macrophages. Nat. Mater. 17, 1134–1144 (2018).
Madden, L. R. et al. Proangiogenic scaffolds as functional templates for cardiac tissue engineering. Proc. Natl Acad. Sci. USA 107, 15211–15216 (2010).
Kyle, D. J., Oikonomou, A., Hill, E. & Bayat, A. Development and functional evaluation of biomimetic silicone surfaces with hierarchical micro/nano-topographical features demonstrates favourable in vitro foreign body response of breast-derived fibroblasts. Biomaterials 52, 88–102 (2015).
Sforza, M. et al. Preliminary 3-year evaluation of experience with SilkSurface and VelvetSurface Motiva silicone breast implants: a single-center experience with 5813 consecutive breast augmentation cases. Aesthet. Surg. J. 38, S62–S73 (2018).
Doloff, J. C. et al. Colony stimulating factor-1 receptor is a central component of the foreign body response to biomaterial implants in rodents and non-human primates. Nat. Mater. 16, 671–680 (2017).
Vegas, A. J. et al. Combinatorial hydrogel library enables identification of materials that mitigate the foreign body response in primates. Nat. Biotechnol. 34, 345–352 (2016).
Veiseh, O. et al. Size- and shape-dependent foreign body immune response to materials implanted in rodents and non-human primates. Nat. Mater. 14, 643–651 (2015).
Anderson, J. M., Rodriguez, A. & Chang, D. T. Foreign body reaction to biomaterials. Semin. Immunol. 20, 86–100 (2008).
Kenneth Ward, W. A review of the foreign-body response to subcutaneously-implanted devices: the role of macrophages and cytokines in biofouling and fibrosis. J. Diabetes Sci. Technol. Online 2, 768–777 (2008).
Bain, C. C. et al. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat. Immunol. 15, 929–937 (2014).
Abbas, A., Lichtman, A. & Pillai, S. in Cellular and Molecular Immunology 8th edn (Elsevier Saunders, 2014).
Efanov, J. I., Giot, J. P., Fernandez, J. & Danino, M. A. Breast-implant texturing associated with delamination of capsular layers: a histological analysis of the double capsule phenomenon. Ann. Chir. Plast. Esthet. 62, 196–201 (2017).
Glicksman, C. A., Danino, M. A., Efanov, J. I., El Khatib, A. & Nelea, M. A step forward toward the understanding of the long-term pathogenesis of double capsule formation in macrotextured implants: a prospective histological analysis. Aesthet. Surg. J. 39, 1191–1199 (2018).
Maxwell, G. P., Scheflan, M., Spear, S., Nava, M. B. & Heden, P. Benefits and limitations of macrotextured breast implants and consensus recommendations for optimizing their effectiveness. Aesthet. Surg. J. 34, 876–881 (2014).
Farah, S. et al. Long-term implant fibrosis prevention in rodents and non-human primates using crystallized drug formulations. Nat. Mater. 18, 892–904 (2019).
Cappellano, G. et al. Immunophenotypic characterization of human T cells after in vitro exposure to different silicone breast implant surfaces. PLoS ONE 13, e0192108 (2018).
Katzin, W. E., Feng, L. J., Abbuhl, M. & Klein, M. A. Phenotype of lymphocytes associated with the inflammatory reaction to silicone gel breast implants. Clin. Diagn. Lab Immunol. 3, 156–161 (1996).
Wynn, T. A. & Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat. Med. 18, 1028–1040 (2012).
Sharabi, A. et al. Regulatory T cells in the treatment of disease. Nat. Rev. Drug Discov. 17, 823–844 (2018).
Watad, A. et al. Silicone breast implants and the risk of autoimmune/rheumatic disorders: a real-world analysis. Int J. Epidemiol. 47, 1846–1854 (2018).
Sadighi Akha, A. A. Aging and the immune system: an overview. J. Immunol. Methods 463, 21–26 (2018).
Durbin, J. E., Hackenmiller, R., Simon, M. C. & Levy, D. E. Targeted disruption of the mouse Stat1 gene results in compromised innate immunity to viral disease. Cell 84, 443–450 (1996).
O’Shea, J. J., Holland, S. M. & Staudt, L. M. JAKs and STATs in immunity, immunodeficiency, and cancer. N. Engl. J. Med. 368, 161–170 (2013).
Liu, M. et al. CXCL10/IP-10 in infectious diseases pathogenesis and potential therapeutic implications. Cytokine Growth Factor Rev. 22, 121–130 (2011).
Le Page, C., Genin, P., Baines, M. G. & Hiscott, J. Interferon activation and innate immunity. Rev. Immunogenet. 2, 374–386 (2000).
Murray, P. J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Acknowledgements
This work was supported by Establishment Labs. We thank members of the Establishment team for help with sourcing full-scale, commercial implants and manufacturing the miniaturized implants used in this study; M. C. Quiros for his clinical contributions; and D. Wolfram for critique on the presented data that went into this manuscript. The laboratory of H.C.H. is supported by Gabrielle’s Angel Foundation for Cancer Research. We acknowledge the use of resources at Core Facilities (Swanson Biotechnology Center, David H. Koch Institute for Integrative Cancer Research at MIT), W. M. Keck Biological Imaging Facility for Flow Cytometry and Histology, as well as the Division of Comparative Medicine for animal work, and the Sidney Kimmel Comprehensive Cancer Center (SKCCC) Immune Monitoring Core for additional NanoString analysis (at Johns Hopkins).
Author information
Authors and Affiliations
Contributions
J.C.D., O.V. and R.L. designed the studies, analysed data and wrote the paper. J.C.D., O.V., M.S., J. Haupt, M.J., C.C., A.N., S.A.-F., J.L.S., S.J.B., S.Y.N., N.A.R., Y.E.H., I.M.L., H.C.H., R.N.M. and M.W.C. conducted the experiments. J.C.D., H.C.H. and O.V. carried out the statistical analyses and prepared displays communicating datasets. R.d.M., M.S., T.A.P., J. Hancock, A.M.M., A.B., B.M.K. and R.L. provided advice and technical support throughout, and J.C.D., O.V. and R.L. contributed with supervision of the study. All authors discussed the results and the preparation of the paper.
Corresponding authors
Ethics declarations
Competing interests
J.H., A.B., B.K., T.A.P and R.L. are members of the Scientific Advisory Board of Establishment Labs Holdings and each hold equity in the company. M.S. and A.M.M. are members of the Medical Advisory Board and each hold equity in Establishment Labs Holdings. M.W.C. and B.M.K. are Investigators on the US IDE Clinical Trial for the Study of Safety and Effectiveness of Motiva Implants. R.D.M., N.A.R., Y.E.H. and I.M.L. are employees of Establishment Labs S.A., and hold equity in Establishment Labs Holdings. J.C.D. and O.V. are paid consultants for Establishment Labs S.A. For a list of entities with which R.L. is involved, compensated or uncompensated, see the listing in the Supplementary Information.
Additional information
Peer review information Nature Biomedical Engineering thanks Pamela Moalli and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures, tables, video captions, references and additional detailed competing interests.
Supplementary Video 1
Double capsules for Mentor Siltex.
Supplementary Video 2
Double capsule for Allergan Microcell, with Velcro effect.
Supplementary Video 3
Double capsule for Allergan Biocell, with Velcro effect.
Rights and permissions
About this article
Cite this article
Doloff, J.C., Veiseh, O., de Mezerville, R. et al. The surface topography of silicone breast implants mediates the foreign body response in mice, rabbits and humans. Nat Biomed Eng 5, 1115–1130 (2021). https://doi.org/10.1038/s41551-021-00739-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-021-00739-4
This article is cited by
-
Miniaturized implantable temperature sensors for the long-term monitoring of chronic intestinal inflammation
Nature Biomedical Engineering (2024)
-
Reply: Comment on BIA-ALCL: Comparing the Risk Profiles of Smooth and Textured Breast Implants
Aesthetic Plastic Surgery (2024)
-
Single-cell transcriptomics in tissue engineering and regenerative medicine
Nature Reviews Bioengineering (2023)
-
Screening hydrogels for antifibrotic properties by implanting cellularly barcoded alginates in mice and a non-human primate
Nature Biomedical Engineering (2023)
-
Controlled swelling of biomaterial devices for improved antifouling polymer coatings
Scientific Reports (2023)