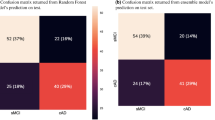
Abstract
The prevalence of concomitant proteinopathies and heterogeneous clinical symptoms in neurodegenerative diseases hinders the identification of individuals who might be candidates for a particular intervention. Here, by applying an unsupervised clustering algorithm to post-mortem histopathological data from 895 patients with degeneration in the central nervous system, we show that six non-overlapping disease clusters can simultaneously account for tau neurofibrillary tangles, α-synuclein inclusions, neuritic plaques, inclusions of the transcriptional repressor TDP-43, angiopathy, neuron loss and gliosis. We also show that membership to the six transdiagnostic disease clusters, which explains more variance in cognitive phenotypes than can be explained by individual diagnoses, can be accurately predicted from scores of the Mini-Mental Status Exam, protein levels in cerebrospinal fluid, and genotype at the APOE and MAPT loci, via cross-validated multiple logistic regression. This combination of unsupervised and supervised data-driven tools provides a framework that could be used to identify latent disease subtypes in other areas of medicine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Source data for all figures and pathology scores for the 895 patients analysed here are available from figshare at https://doi.org/10.6084/m9.figshare.12519488.v1. The raw patient data are available from the authors, subject to approval from the Institutional Review Board of the University of Pennsylvania. For data requests, please visit https://www.med.upenn.edu/cndr/biosamples-brainbank.html and complete a Biosample Request Form. Source data are provided with this paper.
Code availability
All analysis code is available at https://github.com/ejcorn/neuropathcluster.
References
Hebert, L. E., Scherr, P. A., Bienias, J. L., Bennett, D. A. & Evans, D. A. Alzheimer disease in the US population. Arch. Neurol. 60, 1119 (2003).
Alzheimer’s Association 2019 Alzheimer’s disease facts and figures. Alzheimers Dement. 15, 321–387 (2019).
Dorsey, E. R. et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 68, 384–386 (2007).
Brookmeyer, R., Gray, S. & Kawas, C. Projections of Alzheimer’s disease in the United States and the public health impact of delaying disease onset. Am. J. Public Health 88, 1337–1342 (1998).
Hyman, B. T. et al. National Institute on Aging–Alzheimer’s association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimers Dement. 8, 1–13 (2012).
Jankovic, J. Parkinson’s disease: clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 79, 368–376 (2008).
Irwin, D. J. et al. Frontotemporal lobar degeneration: defining phenotypic diversity through personalized medicine. Acta Neuropathol. 129, 469–491 (2015).
Gilman, S. et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 71, 670–676 (2008).
Selkoe, D. J. & Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 8, 595–608 (2016).
Thal, D. R. & Fändrich, M. Protein aggregation in Alzheimer’s disease: Aβ and τ and their potential roles in the pathogenesis of AD. Acta Neuropathologica 129, 163–165 (2015).
Raj, A., Kuceyeski, A. & Weiner, M. A network diffusion model of disease progression in dementia. Neuron 73, 1204–1215 (2012).
Pandya, S., Mezias, C. & Raj, A. Predictive model of spread of progressive supranuclear palsy using directional network diffusion. Front. Neurol. 8, 692 (2017).
Wyss-Coray, T. Inflammation in Alzheimer disease: driving force, bystander or beneficial response? Nat. Med. 12, 1005–1015 (2006).
Akiyama, H. et al. Inflammation and Alzheimer’s disease. Neurobiol. Aging 21, 383–421 (2000).
Rademakers, R., Cruts, M. & van Broeckhoven, C. The role of tau in frontotemporal dementia and related tauopathies (MAPT). Hum. Mutat. 24, 277–295 (2004).
Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 (2006).
Robinson, J. L. et al. Neurodegenerative disease concomitant proteinopathies are prevalent, age-related and APOE4-associated. Brain 141, 2181–2193 (2018).
Higashi, S. et al. Concurrence of TDP-43, tau and α-synuclein pathology in brains of Alzheimer’s disease and dementia with Lewy bodies. Brain Res. 1184, 284–294 (2007).
Nakashima-Yasuda, H. et al. Co-morbidity of TDP-43 proteinopathy in Lewy body related diseases. Acta Neuropathol. 114, 221–229 (2007).
Takahashi, R. H., Capetillo-Zarate, E., Lin, M. T., Milner, T. A. & Gouras, G. K. Co-occurrence of Alzheimer’s disease β-amyloid and tau pathologies at synapses. Neurobiol. Aging 31, 1145–1152 (2010).
Fang, Y. S. et al. Full-length TDP-43 forms toxic amyloid oligomers that are present in frontotemporal lobar dementia-TDP patients. Nat. Commun. 5, 4824 (2014).
He, Z. et al. Amyloid-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation. Nat. Med. 24, 29–38 (2017).
Giasson, B. I. et al. Initiation and synergistic fibrillization of tau and alpha-synuclein. Science 300, 636–640 (2003).
Clinton, L. K., Blurton-Jones, M., Myczek, K., Trojanowski, J. Q. & LaFerla, F. M. Synergistic interactions between Aβ, tau, and α-synuclein: acceleration of neuropathology and cognitive decline. J. Neurosci. 30, 7281–7289 (2010).
Styr, B. & Slutsky, I. Imbalance between firing homeostasis and synaptic plasticity drives early-phase Alzheimer’s disease. Nat. Neurosci. 21, 463–473 (2018).
Jeong, S. Molecular and cellular basis of neurodegeneration in Alzheimer’s disease. Mol. Cells 40, 613–620 (2017).
Yankner, B. A. & Lu, T. Amyloid β-protein toxicity and the pathogenesis of Alzheimer disease. J. Biol. Chem. 284, 4755–4759 (2009).
Li, A. et al. Unsupervised analysis of transcriptomic profiles reveals six glioma subtypes. Cancer Res. 69, 2091–2099 (2009).
Liu, J. J. et al. Multiclass cancer classification and biomarker discovery using GA-based algorithms. Bioinformatics 21, 2691–2697 (2005).
Bartolomei, F. et al. Seizures of temporal lobe epilepsy: identification of subtypes by coherence analysis using stereo-electro-encephalography. Clin. Neurophysiol. 110, 1741–1754 (1999).
Cragar, D. E., Berry, D. T., Schmitt, F. A. & Fakhoury, T. A. Cluster analysis of normal personality traits in patients with psychogenic nonepileptic seizures. Epilepsy Behav. 6, 593–600 (2005).
Xia, C. H. et al. Linked dimensions of psychopathology and connectivity in functional brain networks. Nat. Commun. 9, 3003 (2018).
Drysdale, A. T. et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat. Med. 23, 28–38 (2017).
Grisanzio, K. A. et al. Transdiagnostic symptom clusters and associations with brain, behavior, and daily function in mood, anxiety, and trauma disorders. JAMA Psychiatry 75, 201–209 (2018).
König, A. et al. Automatic speech analysis for the assessment of patients with predementia and Alzheimer’s disease. Alzheimers Dement. 1, 112–124 (2015).
Avants, B. B., Cook, P. A., Ungar, L., Gee, J. C. & Grossman, M. Dementia induces correlated reductions in white matter integrity and cortical thickness: a multivariate neuroimaging study with sparse canonical correlation analysis. NeuroImage 50, 1004–1016 (2010).
Brier, M. R. et al. Tau and Aβ imaging, CSF measures, and cognition in Alzheimer’s disease. Sci. Transl. Med. 8, 338ra66 (2016).
Khanna, S. et al. Using multi-scale genetic, neuroimaging and clinical data for predicting Alzheimer’s disease and reconstruction of relevant biological mechanisms. Sci. Rep. 8, 11173 (2018).
Toledo, J. B. et al. CSF biomarkers cutoffs: the importance of coincident neuropathological diseases. Acta Neuropathol. 124, 23–35 (2012).
Kansal, K. & Irwin, D. J. The use of cerebrospinal fluid and neuropathologic studies in neuropsychiatry practice and research. Psychiatr. Clin. North Am. 38, 309–22 (2015).
Olsson, U. Maximum likelihood estimation of the polychoric correlation coefficient. Psychometrika 44, 443–460 (1979).
Revelle, W. psych: Procedures for Psychological, Psychometric, and Personality Research (CRAN, 2019); https://cran.r-project.org/web/packages/psych/index.html
Montine, T. J. et al. National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol. 123, 1–11 (2012).
Jenkinson, M., Beckmann, C. F., Behrens, T. E., Woolrich, M. W. & Smith, S. M. FSL. NeuroImage 62, 782–790 (2012).
Cammoun, L. et al. Mapping the human connectome at multiple scales with diffusion spectrum MRI. J. Neurosci. Methods 203, 386–397 (2012).
Beach, T. G., Monsell, S. E., Phillips, L. E. & Kukull, W. Accuracy of the clinical diagnosis of Alzheimer disease at National Institute on Aging Alzheimer disease centers, 2005–2010. J. Neuropathol. Exp. Neurol. 71, 266–273 (2012).
Irwin, D. J. et al. Neuropathological and genetic correlates of survival and dementia onset in synucleinopathies: a retrospective analysis. Lancet Neurol. 16, 55 (2017).
Holgado-Tello, F. P., Chacón-Moscoso, S., Barbero-García, I. & Vila-Abad, E. Polychoric versus Pearson correlations in exploratory and confirmatory factor analysis of ordinal variables. Qual. Quant. 44, 153–166 (2010).
Toledo, J. B. et al. Pathological α-synuclein distribution in subjects with coincident Alzheimer’s and Lewy body pathology. Acta Neuropathol. 131, 393–409 (2016).
Nasreddine, Z. S. et al. The Montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J. Am. Geriatrics Soc. 53, 695–699 (2005).
Irwin, J. D. & Hurtig, I. H. The contribution of tau, amyloid-beta and alpha-synuclein pathology to dementia in Lewy body disorders. J. Alzheimer’s Dis. Parkinsonism 08, 444 (2018).
McKeith, I. G. et al. Diagnosis and management of dementia with Lewy bodies: Fourth Consensus Report of the DLB Consortium. Neurology 89, 88–100 (2017).
Tapiola, T. et al. Cerebrospinal fluid β-amyloid 42 and tau proteins as biomarkers of Alzheimer-type pathologic changes in the brain. Arch. Neurol. 66, 382–389 (2009).
Kang, J. H., Korecka, M., Toledo, J. B., Trojanowski, J. Q. & Shaw, L. M. Clinical utility and analytical challenges in measurement of cerebrospinal fluid amyloid-1–42 and proteins as Alzheimer disease biomarkers. Clin. Chem. 59, 903–916 (2013).
Perneczky, R. et al. CSF soluble amyloid precursor proteins in the diagnosis of incipient Alzheimer disease. Neurology 77, 35–38 (2011).
Irwin, D. J. et al. CSF tau and amyloid-β predict cerebral synucleinopathy in autopsied Lewy body disorders Alzheimer disease biomarkers and synucleinopathy. Neurology 90, 1038–1046 (2018).
Liu, C. C., Kanekiyo, T., Xu, H. & Bu, G. Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat. Rev. Neurol. 9, 106–118 (2013).
Baker, M. et al. Association of an extended haplotype in the Tau gene with progressive supranuclear palsy. Hum. Mol. Genet. 8, 711–715 (1999).
Tobin, J. E. et al. Haplotypes and gene expression implicate the MAPT region for Parkinson disease: the GenePD study. Neurology 71, 28–34 (2008).
Myers, A. J. et al. The H1c haplotype at the MAPT locus is associated with Alzheimer’s disease. Hum. Mol. Genet. 14, 2399–2404 (2005).
Tsuang, D. et al. APOE ϵ4 increases risk for dementia in pure synucleinopathies. JAMA Neurol. 70, 223–228 (2013).
Liaw, A. et al. Classification and regression by RandomForest. R News 2, 18–22 (2002).
Uchikado, H., Lin, W. L., Delucia, M. W. & Dickson, D. W. Alzheimer disease with amygdala Lewy bodies: a distinct form of α-synucleinopathy. J. Neuropathol. Exp. Neurol. 65, 685–697 (2006).
Covell, D. J. et al. Novel conformation-selective α-synuclein antibodies raised against different in vitro fibril forms show distinct patterns of Lewy pathology in Parkinson’s disease. Neuropathol. Appl. Neurobiol. 43, 604–620 (2017).
Guo, J. L. & Lee, V. M. Y. Cell-to-cell transmission of pathogenic proteins in neurodegenerative diseases. Nat. Med. 20, 130–138 (2014).
Guo, J. L. et al. Distinct α-synuclein strains differentially promote tau inclusions in neurons. Cell 154, 103–117 (2013).
Masliah, E. et al. β-Amyloid peptides enhance α-synuclein accumulation and neuronal deficits in a transgenic mouse model linking Alzheimer’s disease and Parkinson’s disease. Proc. Natl Acad. Sci. USA 98, 12245–12250 (2001).
Williams-Gray, C. H. et al. The distinct cognitive syndromes of Parkinson’s disease: 5 year follow-up of the CamPaIGN cohort. Brain 132, 2958–2969 (2009).
Perry, D. et al. Building a roadmap for developing combination therapies for Alzheimer’s disease. Expert Rev. Neurother. 15, 327–333.
Neltner, J. H. et al. Digital pathology and image analysis for robust high-throughput quantitative assessment of Alzheimer disease neuropathologic changes. J. Neuropathol. Exp. Neurol. 71, 1075–1085 (2012).
Irwin, D. J. et al. Semi-automated digital image analysis of Pick’s disease and TDP-43 proteinopathy. J. Histochem. Cytochem. 64, 54–66 (2016).
Eickhoff, S. B., Yeo, B. T. & Genon, S. Imaging-based parcellations of the human brain. Nat. Rev. Neurosci. 19, 672–686 (2018).
Alegro, M. et al. Deep learning based large-scale histological tau protein mapping for neuroimaging biomarker validation in Alzheimer’s disease. Preprint at https://www.biorxiv.org/content/10.1101/698902v1 (2019).
Demirovic, J. et al. Prevalence of dementia in three ethnic groups: the South Florida program on aging and health. Ann. Epidemiol. 13, 472–478 (2003).
Van Cauwenberghe, C., Van Broeckhoven, C. & Sleegers, K. The genetic landscape of Alzheimer disease: clinical implications and perspectives. Genet. Med. 18, 421–430 (2016).
Kalinderi, K., Bostantjopoulou, S. & Fidani, L. The genetic background of Parkinson’s disease: current progress and future prospects. Acta Neurol. Scand. 134, 314–326 (2016).
Jack, C. R. et al. NIA–AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 14, 535–562 (2018).
Landau, S. M. et al. Comparing positron emission tomography imaging and cerebrospinal fluid measurements of β-amyloid. Ann. Neurol. 74, 826–836 (2013).
Toledo, J. B. et al. A platform for discovery: the University of Pennsylvania Integrated Neurodegenerative Disease Biobank. Alzheimers Dement. 10, 477–484 (2014).
Xie, S. X. et al. Building an integrated neurodegenerative disease database at an academic health center. Alzheimers Dement. 7, e84–e93 (2011).
Crary, J. F. et al. Primary age-related tauopathy: a common pathology associated with human aging (PART). Acta Neuropathol. 128, 755–66 (2014).
Shaw, L. M. et al. Cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann. Neurol. 65, 403–413 (2009).
Kaufman, L. & Rousseeuw, P. J. Finding Groups in Data: An Introduction to Cluster Analysis Vol. 344 (John Wiley & Sons, 1990).
Hastie, T., Tibshirani, R. & Friedman, J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction (Springer Science & Business Media, 2009).
Kerr, M. K. & Churchill, G. A. Bootstrapping cluster analysis: assessing the reliability of conclusions from microarray experiments. Proc. Natl Acad. Sci. USA 98, 8961–8965 (2001).
VonLuxburg, U. Clustering stability: an overview. Found. Trends Mach. Learn. 2, 235–274 (2010).
Ben-Hur, A., Elisseeff, A. & Guyon, I. in Biocomputing 2002 (eds Altman, R. B. et al.) 6–17 (World Scientific, 2001).
Clarke, G. M. et al. Basic statistical analysis in genetic case-control studies. Nat. Protoc. 6, 121–33 (2011).
Cook, J. P., Mahajan, A. & Morris, A. P. Guidance for the utility of linear models in meta-analysis of genetic association studies of binary phenotypes. Eur. J. Hum. Genet. 25, 240–245 (2016).
Kohavi, R. A study of cross-validation and bootstrap for accuracy estimation and model selection. IJCAI 14, 1137–1145 (1995).
Drummond, C. & Holte, R. C. C4.5, Class Imbalance, and Cost Sensitivity: Why Under-Sampling Beats Over-Sampling (ICML, 2003).
Mani, I. & Zhang, J. kNN Approach to Unbalanced Data Dstributions: A Case Study Involving Information Extraction (ICML, 2003).
Dworkin, J. D. et al. The extent and drivers of gender imbalance in neuroscience reference lists. Nat. Neurosci. https://doi.org/10.1038/s41593-020-0658-y (2020).
Maliniak, D., Powers, R. & Walter, B. F. The gender citation gap in international relations. Int. Organ. 67, 889–922 (2013).
Caplar, N., Tacchella, S. & Birrer, S. Quantitative evaluation of gender bias in astronomical publications from citation counts. Nat. Astron. 1, 0141 (2017).
Chakravartty, P., Kuo, R., Grubbs, V. & McIlwain, C. #CommunicationSoWhite. J. Commun. 68, 254–266 (2018).
Thiem, Y., Sealey, K. F., Ferrer, A. E., Trott, A. M. & Kennison, R. Just Ideas? The Status and Future of Publication Ethics in Philosophy: A White Paper (Publication Ethics, 2018); https://publication-ethics.org/white-paper/
Dion, M. L., Sumner, J. L. & Mitchell, S. M. L. Gendered citation patterns across political science and social science methodology fields. Political Anal. 26, 312–327 (2018).
Zhou, D. et al. Gender Diversity Statement and Code Notebook v1.0; (2020); https://zenodo.org/record/3672110
Bejamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Sci. B 57, 289–300 (1995).
Kassambara, A. ggpubr: ‘ggplot2’ Based Publication Ready Plots v.0.2.4 (CRAN, 2019).
Acknowledgements
D.S.B. and E.J.C. acknowledge support from the John D. and Catherine T. MacArthur Foundation, the Alfred P. Sloan Foundation, the ISI Foundation, the Paul Allen Foundation, the Army Research Laboratory (W911NF-10-2-0022), the Army Research Office (Bassett-W911NF-14-1-0679, Grafton-W911NF-16-1-0474, DCIST-W911NF-17-2-0181), the Office of Naval Research, the National Institute of Mental Health (2-R01-DC-009209-11, R01-MH112847, R01-MH107235, R21-M MH-106799), the National Institute of Child Health and Human Development (1R01HD086888-01), National Institute of Neurological Disorders and Stroke (R01 NS099348) and the National Science Foundation (BCS-1441502, BCS-1430087, NSF PHY-1554488 and BCS-1631550). E.J.C. also acknowledges support from the National Institute of Mental Health (F30 MH118871-01). D.J.I. acknowledges the National Institute of Neurological Disorders and Stroke (R01-NS109260). J.Q.T., V.M.-Y.L. and E.B.L. thank members of the Center for Neurodegenerative Disease Research who contributed to the studies reviewed here. J.Q.T., V.M.-Y.L. and E.B.L. also thank the patients and their families for brain donation. J.Q.T., V.M.-Y.L. and E.B.L. acknowledge funding support from AG10124, AG17586, AG62418 and the Woods Foundation. The authors thank D. Wolk for helpful comments on the manuscript during the review process. The content is solely the responsibility of the authors and does not necessarily represent the official views of any of the funding agencies.
Author information
Authors and Affiliations
Contributions
E.J.C. performed all analyses. E.J.C. and D.S.B. designed all analyses and wrote the manuscript. J.Q.T., E.B.L., J.L.R., and V.M.-Y.L. oversaw data collection. All authors edited the manuscript and interpreted data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, figures, tables and references.
Rights and permissions
About this article
Cite this article
Cornblath, E.J., Robinson, J.L., Irwin, D.J. et al. Defining and predicting transdiagnostic categories of neurodegenerative disease. Nat Biomed Eng 4, 787–800 (2020). https://doi.org/10.1038/s41551-020-0593-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-0593-y
This article is cited by
-
The neural substrates of transdiagnostic cognitive-linguistic heterogeneity in primary progressive aphasia
Alzheimer's Research & Therapy (2023)
-
Shared genetic risk loci between Alzheimer’s disease and related dementias, Parkinson’s disease, and amyotrophic lateral sclerosis
Alzheimer's Research & Therapy (2023)
-
A scoping review of neurodegenerative manifestations in explainable digital phenotyping
npj Parkinson's Disease (2023)
-
CRISPR and iPSCs: Recent Developments and Future Perspectives in Neurodegenerative Disease Modelling, Research, and Therapeutics
Neurotoxicity Research (2022)
-
Deep learning reveals disease-specific signatures of white matter pathology in tauopathies
Acta Neuropathologica Communications (2021)