Abstract
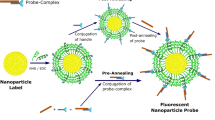
The detection and quantification of low-abundance molecular biomarkers in biological samples is challenging. Here, we show that a plasmonic nanoscale construct serving as an ‘add-on’ label for a broad range of bioassays improves their signal-to-noise ratio and dynamic range without altering their workflow and readout devices. The plasmonic construct consists of a bovine serum albumin scaffold with approximately 210 IRDye 800CW fluorophores (with a fluorescence intensity approximately 6,700-fold that of a single 800CW fluorophore), a polymer-coated gold nanorod acting as a plasmonic antenna and biotin as a high-affinity biorecognition element. Its emission wavelength can be tuned over the visible and near-infrared spectral regions by modifying its size, shape and composition. It improves the limit of detection in fluorescence-linked immunosorbent assays by up to 4,750-fold and is compatible with multiplexed bead-based immunoassays, immunomicroarrays, flow cytometry and immunocytochemistry methods, and it shortens overall assay times (to 20 min) and lowers sample volumes, as shown for the detection of a pro-inflammatory cytokine in mouse interstitial fluid and of urinary biomarkers in patient samples.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Raw imaging data (collected and analysed via the software indicated in the Reporting Summary) are available from figshare with the identifier https://doi.org/10.6084/m9.figshare.11888055.
References
Cohen, L. & Walt, D. R. Highly sensitive and multiplexed protein measurements. Chem. Rev. 119, 293–321 (2018).
Hanash, S. M., Pitteri, S. J. & Faca, V. M. Mining the plasma proteome for cancer biomarkers. Nature 452, 571–579 (2008).
Shaw, L. M., Korecka, M., Clark, C. M., Lee, V. M.-Y. & Trojanowski, J. Q. Biomarkers of neurodegeneration for diagnosis and monitoring therapeutics. Nat. Rev. Drug Discov. 6, 295–303 (2007).
Blennow, K. & Zetterberg, H. Understanding biomarkers of neurodegeneration: ultrasensitive detection techniques pave the way for mechanistic understanding. Nat. Med. 21, 217–219 (2015).
Savage, M. J. et al. A sensitive aβ oligomer assay discriminates Alzheimer’s and aged control cerebrospinal fluid. J. Neurosci. 34, 2884–2897 (2014).
Westermann, D., Neumann, J. T., Sörensen, N. A. & Blankenberg, S. High-sensitivity assays for troponin in patients with cardiac disease. Nat. Rev. Cardiol. 14, 472–483 (2017).
Rissin, D. M. et al. Single-molecule enzyme-linked immunosorbent assay detects serum proteins at subfemtomolar concentrations. Nat. Biotechnol. 28, 595–599 (2010).
Tabakman, S. M. et al. Plasmonic substrates for multiplexed protein microarrays with femtomolar sensitivity and broad dynamic range. Nat. Commun. 2, 466 (2011).
Zhang, B. et al. Diagnosis of Zika virus infection on a nanotechnology platform. Nat. Med. 23, 548–550 (2017).
Zhang, B., Kumar, R. B., Dai, H. & Feldman, B. J. A plasmonic chip for biomarker discovery and diagnosis of type 1 diabetes. Nat. Med. 20, 948–953 (2014).
Zhang, B. et al. Plasmonic micro-beads for fluorescence enhanced, multiplexed protein detection with flow cytometry. Chem. Sci. 5, 4070–4075 (2014).
Steward, M. W. & Lew, A. M. The importance of antibody affinity in the performance of immunoassays for antibody. J. Immunol. Methods 78, 173–190 (1985).
Roth, S. et al. Photobleaching: improving the sensitivity of fluorescence‐based immunoassays by photobleaching the autofluorescence of magnetic beads (Small 3/2019). Small 15, 1970016 (2019).
Zhang, P. et al. Ultrasensitive detection of circulating exosomes with a 3D-nanopatterned microfluidic chip. Nat. Biomed. Eng. 3, 438–451 (2019).
Espina, V. et al. Protein microarray detection strategies: focus on direct detection technologies. J. Immunol. Methods 290, 121–133 (2004).
Chen, Z. et al. Protein microarrays with carbon nanotubes as multicolor Raman labels. Nat. Biotechnol. 26, 1285–1292 (2008).
Luan, J. et al. Add-on plasmonic patch as a universal fluorescence enhancer. Light. Sci. Appl. 7, 29 (2018).
Reisch, A. et al. Collective fluorescence switching of counterion-assembled dyes in polymer nanoparticles. Nat. Commun. 5, 4089 (2014).
Hu, J. et al. Sensitive and quantitative detection of C-reaction protein based on immunofluorescent nanospheres coupled with lateral flow test strip. Anal. Chem. 88, 6577–6584 (2016).
Huang, L. et al. Brilliant pitaya‐type silica colloids with central–radial and high‐density quantum dots incorporation for ultrasensitive fluorescence immunoassays. Adv. Funct. Mater. 28, 1705380 (2018).
Reisch, A. & Klymchenko, A. S. Fluorescent polymer nanoparticles based on dyes: seeking brighter tools for bioimaging. Small 12, 1968–1992 (2016).
Shulov, I. et al. Fluorinated counterion-enhanced emission of rhodamine aggregates: ultrabright nanoparticles for bioimaging and light-harvesting. Nanoscale 7, 18198–18210 (2015).
Melnychuk, N. & Klymchenko, A. S. DNA-functionalized dye-loaded polymeric nanoparticles: ultrabright FRET platform for amplified detection of nucleic acids. J. Am. Chem. Soc. 140, 10856–10865 (2018).
Xu, Z. et al. Broad‐spectrum tunable photoluminescent nanomaterials constructed from a modular light‐harvesting platform based on macrocyclic amphiphiles. Adv. Mater. 28, 7666–7671 (2016).
Kasha, M., Rawls, H. & El-Bayoumi, M. A. The exciton model in molecular spectroscopy. Pure Appl. Chem. 11, 371–392 (1965).
Méallet-Renault, R. et al. Fluorescent nanoparticles as selective Cu (II) sensors. Photochem. Photobiol. Sci. 5, 300–310 (2006).
Trofymchuk, K., Reisch, A., Shulov, I., Mély, Y. & Klymchenko, A. S. Tuning the color and photostability of perylene diimides inside polymer nanoparticles: towards biodegradable substitutes of quantum dots. Nanoscale 6, 12934–12942 (2014).
Trofymchuk, K. et al. Giant light-harvesting nanoantenna for single-molecule detection in ambient light. Nat. Photon. 11, 657–663 (2017).
Holmberg, A. et al. The biotin–streptavidin interaction can be reversibly broken using water at elevated temperatures. Electrophoresis 26, 501–510 (2005).
Chen, H., Shao, L., Li, Q. & Wang, J. Gold nanorods and their plasmonic properties. Chem. Soc. Rev. 42, 2679–2724 (2013).
Huang, X., Neretina, S. & El‐Sayed, M. A. Gold nanorods: from synthesis and properties to biological and biomedical applications. Adv. Mater. 21, 4880–4910 (2009).
Dreaden, E. C., Alkilany, A. M., Huang, X., Murphy, C. J. & El-Sayed, M. A. The golden age: gold nanoparticles for biomedicine. Chem. Soc. Rev. 41, 2740–2779 (2012).
Ge, S., Kojio, K., Takahara, A. & Kajiyama, T. Bovine serum albumin adsorption onto immobilized organotrichlorosilane surface: influence of the phase separation on protein adsorption patterns. J. Biomater. Sci. Polym. Ed. 9, 131–150 (1998).
Ashitate, Y., Tanaka, E., Stockdale, A., Choi, H. S. & Frangioni, J. V. Near-infrared fluorescence imaging of thoracic duct anatomy and function in open surgery and video-assisted thoracic surgery. J. Thorac. Cardiovasc. Surg. 142, 31–38 (2011).
Liu, C. et al. Fluorescence‐converging carbon nanodots‐hybridized silica nanosphere. Small 12, 4702–4706 (2016).
Flauraud, V. et al. In-plane plasmonic antenna arrays with surface nanogaps for giant fluorescence enhancement. Nano Lett. 17, 1703–1710 (2017).
Tam, F., Goodrich, G. P., Johnson, B. R. & Halas, N. J. Plasmonic enhancement of molecular fluorescence. Nano Lett. 7, 496–501 (2007).
Kinkhabwala, A. et al. Large single-molecule fluorescence enhancements produced by a bowtie nanoantenna. Nat. Photon. 3, 654–657 (2009).
Cai, Y. et al. Photoluminescence of gold nanorods: Purcell effect enhanced emission from hot carriers. ACS Nano 12, 976–985 (2018).
Liu, B. et al. High performance, multiplexed lung cancer biomarker detection on a plasmonic gold chip. Adv. Funct. Mater. 26, 7994–8002 (2016).
Mayer, K. M. & Hafner, J. H. Localized surface plasmon resonance sensors. Chem. Rev. 111, 3828–3857 (2011).
Haes, A. J., Chang, L., Klein, W. L. & Van Duyne, R. P. Detection of a biomarker for Alzheimer’s disease from synthetic and clinical samples using a nanoscale optical biosensor. J. Am. Chem. Soc. 127, 2264–2271 (2005).
Bardhan, R., Grady, N. K., Cole, J. R., Joshi, A. & Halas, N. J. Fluorescence enhancement by Au nanostructures: nanoshells and nanorods. ACS Nano 3, 744–752 (2009).
Khatua, S. et al. Resonant plasmonic enhancement of single-molecule fluorescence by individual gold nanorods. ACS Nano 8, 4440–4449 (2014).
Anger, P., Bharadwaj, P. & Novotny, L. Enhancement and quenching of single-molecule fluorescence. Phys. Rev. Lett. 96, 113002 (2006).
Tian, L. et al. Gold nanocages with built-in artificial antibodies for label-free plasmonic biosensing. J. Mater. Chem. B 2, 167–170 (2014).
Abadeer, N. S., Brennan, M. R., Wilson, W. L. & Murphy, C. J. Distance and plasmon wavelength dependent fluorescence of molecules bound to silica-coated gold nanorods. ACS Nano 8, 8392–8406 (2014).
Mishra, H., Mali, B. L., Karolin, J., Dragan, A. I. & Geddes, C. D. Experimental and theoretical study of the distance dependence of metal-enhanced fluorescence, phosphorescence and delayed fluorescence in a single system. Phys. Chem. Chem. Phys. 15, 19538–19544 (2013).
Yan, B. et al. Engineered SERS substrates with multiscale signal enhancement: nanoparticle cluster arrays. ACS Nano 3, 1190–1202 (2009).
Pierre, M. C. S. & Haes, A. J. Purification implications on SERS activity of silica coated gold nanospheres. Anal. Chem. 84, 7906–7911 (2012).
Bardhan, R., Grady, N. K. & Halas, N. J. Nanoscale control of near‐infrared fluorescence enhancement using Au Nanoshells. Small 4, 1716–1722 (2008).
Thompson, D. K., Huffman, K. M., Kraus, W. E. & Kraus, V. B. Critical appraisal of four IL-6 immunoassays. PLoS ONE 7, e30659 (2012).
Samant, P. P. & Prausnitz, M. R. Mechanisms of sampling interstitial fluid from skin using a microneedle patch. Proc. Natl Acad. Sci. USA 115, 4583–4588 (2018).
Mishra, J. et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365, 1231–1238 (2005).
Han, W. et al. Urinary biomarkers in the early diagnosis of acute kidney injury. Kidney Int. 73, 863–869 (2008).
Munier, M. et al. Physicochemical factors affecting the stability of two pigments: R-phycoerythrin of Grateloupia turuturu and B-phycoerythrin of Porphyridium cruentum. Food Chem. 150, 400–407 (2014).
Yuan, H., Khatua, S., Zijlstra, P., Yorulmaz, M. & Orrit, M. Thousand‐fold enhancement of single‐molecule fluorescence near a single gold nanorod. Angew. Chem. Int. Ed. 52, 1217–1221 (2013).
Ma, Y. et al. Au@ Ag core−shell nanocubes with finely tuned and well-controlled sizes, shell thicknesses, and optical properties. ACS Nano 4, 6725–6734 (2010).
Liu, K.-K., Tadepalli, S., Tian, L. & Singamaneni, S. Size-dependent surface enhanced Raman scattering activity of plasmonic nanorattles. Chem. Mater. 27, 5261–5270 (2015).
Dixit, C. K. & Aguirre, G. R. Protein microarrays with novel microfluidic methods: current advances. Microarrays 3, 180–202 (2014).
Jain, P. K., Lee, K. S., El-Sayed, I. H. & El-Sayed, M. A. Calculated absorption and scattering properties of gold nanoparticles of different size, shape, and composition: applications in biological imaging and biomedicine. J. Phys. Chem. B 110, 7238–7248 (2006).
De La Rica, R. & Stevens, M. M. Plasmonic ELISA for the ultrasensitive detection of disease biomarkers with the naked eye. Nat. Nanotechnol. 7, 821–824 (2012).
Deng, W., Xie, F., Baltar, H. T. & Goldys, E. M. Metal-enhanced fluorescence in the life sciences: here, now and beyond. Phys. Chem. Chem. Phys. 15, 15695–15708 (2013).
Lee, K.-S. & El-Sayed, M. A. Dependence of the enhanced optical scattering efficiency relative to that of absorption for gold metal nanorods on aspect ratio, size, end-cap shape, and medium refractive index. J. Phys. Chem. B 109, 20331–20338 (2005).
Gole, A. & Murphy, C. J. Azide-derivatized gold nanorods: functional materials for “click” chemistry. Langmuir 24, 266–272 (2008).
Tebbe, M., Kuttner, C., Männel, M., Fery, A. & Chanana, M. Colloidally stable and surfactant-free protein-coated gold nanorods in biological media. ACS Appl. Mater. Interfaces 7, 5984–5991 (2015).
Nedrebø, T., Reed, R. K., Jonsson, R., Berg, A. & Wiig, H. Differential cytokine response in interstitial fluid in skin and serum during experimental inflammation in rats. J. Physiol. 556, 193–202 (2004).
Acknowledgements
We acknowledge support from National Science Foundation (award nos. CBET-1512043 and CBET-1900277), National Institutes of Health (R01DE027098 and R01CA141521), Center for Multiple Myeloma Nanotherapy (U54 CA199092) and a grant from the Barnes-Jewish Hospital Research Foundation (no. 3706). We thank K. Naegle for providing access to a LI-COR Odyssey CLx scanner, L. Setton for the flow cytometer, J. Rudra and T. Pietka for Luminex readers, and the Nano Research Facility (NRF) and Institute of Materials Science and Engineering (IMSE) at Washington University for providing access to electron microscopy facilities. We also thank G. Genin for inspiring discussions and suggestions.
Author information
Authors and Affiliations
Contributions
S.S. and J.L. designed the project and experiments. J.L. synthesized plasmonic-fluors. A.S. and J.L. designed and performed the flow cytometry experiments of SK-BR-3 and BMDCs. R.G. performed the AFM and TEM characterization. Z.W. performed the immunocytochemistry experiment of SK-BR-3 cells and B.X. did the confocal imaging of the cells. P.R., S.C. and H.G.D. performed the SEM characterizations. R.T. performed the fluorescence lifetime measurements and analysed the data. S.A. helped to design the lifetime measurement experiments and analysed the data. J.J.M. helped to design the kidney disease-related experiments and provided the kidney disease patient samples. S.S. and J.L. wrote the paper. All authors reviewed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.L., J.J.M. and S.S. are inventors on a provisional patent related to this technology. The technology has been licensed by the Office of Technology Management at Washington University in St. Louis to Auragent Bioscience LLC, which is developing plasmonic-fluor products. J.L., J.J.M. and S.S. are co-founders/shareholders of Auragent Bioscience LLC. These potential conflicts of interest have been disclosed and are being managed by Washington University in St. Louis. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, figures, tables and references.
Rights and permissions
About this article
Cite this article
Luan, J., Seth, A., Gupta, R. et al. Ultrabright fluorescent nanoscale labels for the femtomolar detection of analytes with standard bioassays. Nat Biomed Eng 4, 518–530 (2020). https://doi.org/10.1038/s41551-020-0547-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-0547-4
This article is cited by
-
Microneedle patch-based enzyme-linked immunosorbent assay to quantify protein biomarkers of tuberculosis
Biomedical Microdevices (2024)
-
Ultrasensitive lateral-flow assays via plasmonically active antibody-conjugated fluorescent nanoparticles
Nature Biomedical Engineering (2023)
-
Extending the dynamic range of biomarker quantification through molecular equalization
Nature Communications (2023)
-
Large-scale controllable fabrication of aluminum nanobowls for surface plasmon-enhanced fluorescence
Nano Research (2023)
-
Optimization of Structural and Optical Parameters of Plasmon Silver Films for IgG-FITC Fluorescent Analysis
Journal of Applied Spectroscopy (2023)