Abstract
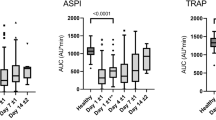
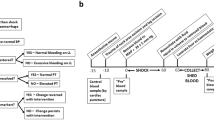
During extracorporeal blood purification, anticoagulants are administered to prevent thrombogenesis. However, haemorrhagic complications owing to near-complete inactivation of blood coagulation and delayed recovery of haemostasis pose serious risks to patients. Here, we show in vitro and in beagle dogs that hydrogel microspheres that adsorb the coagulation factors VIII, IX and XI provide transient blood thinning when placed in the extracorporeal circuit before blood purification. The microspheres inhibited the activities of the coagulation factors by levels (~8–30%) similar to those occurring in mild haemophilia. On its reintroduction into the animal, the purified pseudo-haemophilic blood favoured faster recovery of haemostasis. The transient blood-thinning strategy may increase the safety of clinical blood-purification procedures.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its SupplementaryInformation. The raw and analysed datasets generated during the study are too large to be publicly shared; however, they are available for research purposes from the corresponding authors upon reasonable request.
References
Neri, M. et al. Nomenclature for renal replacement therapy in acute kidney injury: basic principles. Critical Care https://doi.org/10.1186/s13054-016-1489-9 (2016).
Eckardt, K.-U. et al. Evolving importance of kidney disease: from subspecialty to global health burden. Lancet 382, 158–169 (2013).
Zoccali, C. et al. The systemic nature of CKD. Nat. Rev. Nephrol. 13, 344–358 (2017).
Carpentier, B., Gautier, A. & Legallais, C. Artificial and bioartificial liver devices: present and future. Gut 58, 1690–1702 (2009).
Holubek, W. J., Hoffman, R. S., Goldfarb, D. S. & Nelson, L. S. Use of hemodialysis and hemoperfusion in poisoned patients. Kidney Int. 74, 1327–1334 (2008).
Ronco, C. et al. Renal replacement therapy in acute kidney injury: controversy and consensus. Critical Care https://doi.org/10.1186/s13054-015-0850-8 (2015).
Mackman, N. Triggers, targets and treatments for thrombosis. Nature 451, 914–918 (2008).
Davenport, A. What are the anticoagulation options for intermittent hemodialysis? Nat. Rev. Nephrol. 7, 499–508 (2011).
Caruana, R. J., Raja, R. M., Bush, J. V., Kramer, M. S. & Goldstein, S. J. Heparin free dialysis: comparative data and results in high risk patients. Kidney Int. 31, 1351–1355 (1987).
Levine, M. et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N. Engl. J. Med. 334, 677–681 (1996).
Parasa, S., Navaneethan, U., Sridhar, A. R. M., Venkatesh, P. G. K. & Olden, K. End-stage renal disease is associated with worse outcomes in hospitalized patients with peptic ulcer bleeding. Gastrointest. Endosc. 77, 609–616 (2013).
Cheung, J., Yu, A., LaBossiere, J., Zhu, Q. & Fedorak, R. N. Peptic ulcer bleeding outcomes adversely affected by end-stage renal disease. Gastrointest. Endosc. 71, 44–49 (2010).
Rath, C. E., Mailliard, J. A. & Schreiner, G. E. Bleeding tendency in uremia. N. Engl. J. Med. 257, 808–811 (1957).
Lu, G. M. et al. A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat. Med. 19, 446–44 (2013).
Crowther, M. A. & Warkentin, T. E. Bleeding risk and the management of bleeding complications in patients undergoing anticoagulant therapy: focus on new anticoagulant agents. Blood 111, 4871–4879 (2008).
Ainle, F. N. et al. Protamine sulfate down-regulates thrombin generation by inhibiting factor V activation. Blood 114, 1658–1665 (2009).
Shenoi, R. A. et al. Affinity-based design of a synthetic universal reversal agent for heparin anticoagulants. Sci. Transl Med. https://doi.org/10.1126/scitranslmed.3009427 (2014).
Kindgen-Milles, D., Brandenburger, T. & Dimski, T. Regional citrate anticoagulation for continuous renal replacement therapy. Curr. Opin. Crit. Care 24, 450–454 (2018).
Kreuzer, M. et al. Regional citrate anticoagulation is safe in intermittent high-flux haemodialysis treatment of children and adolescents with an increased risk of bleeding. Nephrol. Dialysis Transplant. 25, 3337–3342 (2010).
Evenepoel, P., Maes, B. & Vanwalleghem, J. Regional citrate anticoagulation for hemodialysis using a conventional calcium-containing dialysate. Am. J. Kidney Dis. 39, 315–323 (2002).
Ji, H. et al. Reinforced-concrete structured hydrogel microspheres with ultrahigh mechanical strength, restricted water uptake, and superior adsorption capacity. ACS Sustain. Chem. Eng. 6, 5950–5958 (2018).
He, C. et al. Heparin-based and heparin-inspired hydrogels: size-effect, gelation and biomedical applications. J. Mater. Chem. B 7, 1186–1208 (2019).
Paluck, S. J., Nguyen, T. H. & Maynard, H. D. Heparin-mimicking polymers: synthesis and biological applications. Biomacromolecules 17, 3417–3440 (2016).
Song, X. et al. Design of carrageenan-based heparin-mimetic gel beads as self-anticoagulant hemoperfusion adsorbents. Biomacromolecules 19, 1966–1978 (2018).
Zhao, C., Xue, J., Ran, F. & Sun, S. Modification of polyethersulfone membranes—a review of methods. Prog. Mater. Sci. 58, 76–150 (2013).
Niewiarowski, S. & Thomas, D. P. Platelet factor 4 and adenosine diphosphate release during human platelet aggregation. Nature 222, 1269–126 (1969).
Schmaier, A. H. The contact activation and kallikrein/kinin systems: pathophysiologic and physiologic activities. J. Thrombosis Haemost. 14, 28–39 (2016).
Hong, J., Ekdahl, K. N., Reynolds, H., Larsson, R. & Nilsson, B. A new in vitro model to study interaction between whole blood and biomaterials. Studies of platelet and coagulation activation acid the effect of aspirin. Biomaterials 20, 603–611 (1999).
Flanders, M. M., Crist, R. & Rodgers, G. M. Comparison of five thrombin time reagents. Clin. Chem. 49, 169–172 (2003).
Kanmangne, F. M., Labarre, D., Serne, H. & Jozefowicz, M. Heparin-like activity of insoluble sulphonated polystyrene resins. Part I: influence of the surface density, nature and binding of substituted anionic groups. Biomaterials 6, 297–302 (1985).
Jozefowicz, M. & Jozefonvicz, J. Randomness and biospecificity: random copolymers are capable of biospecific molecular recognition in living systems. Biomaterials 18, 1633–1644 (1997).
Razi, N. et al. Structural and functional properties of heparin analogs obtained by chemical sulfation of Escherichia coli K5 capsular polysaccharide. Biochem. J. 309, 465–472 (1995).
Shworak, N. W., Kobayashi, T., de Agostini, A. & Smits, N. C. in Progress in Molecular Biology and Translational Science: Glycosaminoglycans in Development, Health and Disease Vol. 93 (ed. Zhang, L.) 153–178 (2010).
Li, W., Johnson, D. J. D., Esmon, C. T. & Huntington, J. A. Structure of the antithrombin–thrombin–heparin ternary complex reveals the antithrombotic mechanism of heparin. Nat. Struct. Mol. Biol. 11, 857–862 (2004).
Kalathottukaren, M. T. et al. Alteration of blood clotting and lung damage by protamine are avoided using the heparin and polyphosphate inhibitor UHRA. Blood 129, 1368–1379 (2017).
Guglietta, S. et al. Coagulation induced by C3aR-dependent NETosis drives protumorigenic neutrophils during small intestinal tumorigenesis. Nat. Commun. https://doi.org/10.1038/ncomms11037 (2016).
Engelmann, B. & Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 13, 34–45 (2013).
Hirsh, J., Anand, S. S., Halperin, J. L. & Fuster, V. Guide to anticoagulant therapy: heparin a statement for healthcare professionals from the American Heart Association. Circulation 103, 2994–3018 (2001).
Prince, R. et al. Targeting anticoagulant protein S to improve hemostasis in hemophilia. Blood 131, 1360–1371 (2018).
Peyvandi, F., Garagiola, I. & Young, G. The past and future of haemophilia: diagnosis, treatments, and its complications. Lancet 388, 187–197 (2016).
Hall, J. E. Guyton and Hall Textbook of Medical Physiology (Elsevier Health Sciences, 2015).
Srivastava, A. et al. Guidelines for the management of hemophilia. Haemophilia 19, E1–E47 (2013).
Mannucci, P. M. Desmopressin (DDAVP) in the treatment of bleeding disorders: the first 20 years. Blood 90, 2515–2521 (1997).
White, G. C. et al. Definitions in hemophilia—recommendation of the scientific subcommittee on factor VIII and factor IX of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Thrombosis Haemost. 85, 560–560 (2001).
Gao, A. et al. Linker-free covalent immobilization of heparin, SDF-1 alpha, and CD47 on PTFE surface for antithrombogenicity, endothelialization and anti-inflammation. Biomaterials 140, 201–211 (2017).
Laville, M. et al. Results of the HepZero study comparing heparin-grafted membrane and standard care show that heparin-grafted dialyzer is safe and easy to use for heparin-free dialysis. Kidney Int. 86, 1260–1267 (2014).
Biran, R. & Pond, D. Heparin coatings for improving blood compatibility of medical devices. Adv. Drug Deliv. Rev. 112, 12–23 (2017).
Islam, M. S. et al. Vitamin E-coated and heparin-coated dialyzer membranes for heparin-free hemodialysis: a multicenter, randomized, crossover trial. Am. J. Kidney Dis. 68, 752–762 (2016).
Meng, F. et al. MXene sorbents for removal of urea from dialysate: a step toward the wearable artificial kidney. ACS Nano 12, 10518–10528 (2018).
Davenport, A. et al. A wearable haemodialysis device for patients with end-stage renal failure: a pilot study. Lancet 370, 2005–2010 (2007).
Davenport, A. A wearable dialysis device: the first step to continuous therapy. Nat. Rev. Nephrol. 12, 512–514 (2016).
Song, X., Wang, R., Zhao, W., Sun, S. & Zhao, C. A facile approach towards amino-coated polyethersulfone particles for the removal of toxins. J. Colloid Interface Sci. 485, 39–50 (2017).
Acknowledgements
This work was financially sponsored by the State Key Research Development Programme of China (grant numbers 2016YFC1103000 and 2018YFC1106400), the National Natural Science Foundation of China (grant numbers 51433007, 51673125, 51773127, 51873115, 51803131 and 51803134) and the Science and Technology Program of Sichuan Province (2017SZ0011 and 2019YJ0132). We also gratefully acknowledge the help of H. Wang, of the Analytical and Testing Center at Sichuan University, for the SEM micrographs. Moreover, we thank our laboratory members for their generous help. J.N.K. acknowledges funding from the Canadian Institutes of Health Research and Natural Sciences and Engineering Council of Canada (NSERC). J.N.K. is a recipient of a Michael Smith Foundation for Health Research Career Scholar Award.
Author information
Authors and Affiliations
Contributions
X.S., H.J., W.Z. and C.Z. conceived the early pseudo-haemophilia model concepts and/or designed the experiments. X.S. and H.J. performed initial development work and analysed the data. X.S. and H.J. prepared, characterized and performed the in vitro biocompatibility evaluation of the microspheres. Y.L. and Y.X. designed and conducted the animal experiments. R.Z. and M.T. designed and conducted the studies for the coagulation factors. L.Q., J.N.K., B.S. and Q.W. provided conceptual advice and technical support. X.S., Y.L. and W.Z. drafted the manuscript. W.Z. and C.Z. supervised the study and revised the paper. All authors gave approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature Biomedical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Supplementary information
Supplementary Information
Supplementary methods, figures, tables, scheme, video captions and references.
Supplementary Video 1
Simulated pseudo-haemophilia model.
Supplementary Video 2
Treatment and recovery process in a transfused dog.
Rights and permissions
About this article
Cite this article
Song, X., Ji, H., Li, Y. et al. Transient blood thinning during extracorporeal blood purification via the inactivation of coagulation factors by hydrogel microspheres. Nat Biomed Eng 5, 1143–1156 (2021). https://doi.org/10.1038/s41551-020-00673-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-00673-x
This article is cited by
-
Targeting circulating high mobility group box-1 and histones by extracorporeal blood purification as an immunomodulation strategy against critical illnesses
Critical Care (2023)
-
Self-anticoagulant sponge for whole blood auto-transfusion and its mechanism of coagulation factor inactivation
Nature Communications (2023)
-
Advances in Enhancing Hemocompatibility of Hemodialysis Hollow-Fiber Membranes
Advanced Fiber Materials (2023)
-
Wireless charging-mediated angiogenesis and nerve repair by adaptable microporous hydrogels from conductive building blocks
Nature Communications (2022)
-
Nanozyme-reinforced hydrogel as a H2O2-driven oxygenerator for enhancing prosthetic interface osseointegration in rheumatoid arthritis therapy
Nature Communications (2022)