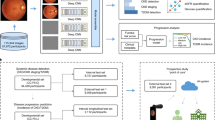
Abstract
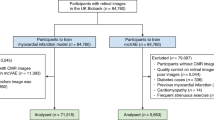
Retinal blood vessels provide information on the risk of cardiovascular disease (CVD). Here, we report the development and validation of deep-learning models for the automated measurement of retinal-vessel calibre in retinal photographs, using diverse multiethnic multicountry datasets that comprise more than 70,000 images. Retinal-vessel calibre measured by the models and by expert human graders showed high agreement, with overall intraclass correlation coefficients of between 0.82 and 0.95. The models performed comparably to or better than expert graders in associations between measurements of retinal-vessel calibre and CVD risk factors, including blood pressure, body-mass index, total cholesterol and glycated-haemoglobin levels. In retrospectively measured prospective datasets from a population-based study, baseline measurements performed by the deep-learning system were associated with incident CVD. Our findings motivate the development of clinically applicable explainable end-to-end deep-learning systems for the prediction of CVD on the basis of the features of retinal vessels in retinal photographs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. The deidentified individual-participant data and data on the evaluation of retinal photographs used in the SIVA-DLS are available on request from the corresponding author or C.Y. Cheung (e-mail: carolcheung@cuhk.edu.hk).
Code availability
The custom code is currently available only on request because it is under a patent examination process.
References
Williams, B. et al. British Hypertension Society guidelines for hypertension management 2004 (BHS-IV): summary. BMJ 328, 634–640 (2004).
Chobanian, A. V. et al. The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure: the JNC 7 report. JAMA 289, 2560–2572 (2003).
Mansia, G. et al. 2007 ESH-ESC guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Blood Press. 16, 135–232 (2007).
Hypertension in Adults: Diagnosis and Management Clinical guideline [CG127], vol. 2019 (National Institute of Health and Clinical Excellence, 2011).
Wong, T. Y. & Mitchell, P. Hypertensive retinopathy. N. Engl. J. Med 351, 2310–2317 (2004).
Liew, G., Wang, J. J., Mitchell, P. & Wong, T. Y. Retinal vascular imaging: a new tool in microvascular disease research. Circ. Cardiovasc. Imaging 1, 156–161 (2008).
Wong, T. Y. et al. Retinal microvascular abnormalities and their relationship with hypertension, cardiovascular disease, and mortality. Surv. Ophthalmol. 46, 59–80 (2001).
Cheung, C. Y., Ikram, M. K., Chen, C. & Wong, T. Y. Imaging retina to study dementia and stroke. Prog. Retin. Eye Res. 57, 89–107 (2017).
Cheung, C. Y., Ikram, M. K., Sabanayagam, C. & Wong, T. Y. Retinal microvasculature as a model to study the manifestations of hypertension. Hypertension 60, 1094–1103 (2012).
Wang, S. B. et al. A spectrum of retinal vasculature measures and coronary artery disease. Atherosclerosis 268, 215–224 (2018).
Hubbard, L. D. et al. Methods for evaluation of retinal microvascular abnormalities associated with hypertension/sclerosis in the Atherosclerosis Risk in Communities Study. Ophthalmology 106, 2269–2280 (1999).
Wong, T. Y. et al. Computer-assisted measurement of retinal vessel diameters in the Beaver Dam Eye Study: methodology, correlation between eyes, and effect of refractive errors. Ophthalmology 111, 1183–1190 (2004).
Cheung, C. Y. et al. A new method to measure peripheral retinal vascular caliber over an extended area. Microcirculation 17, 495–503 (2010).
McGrory, S. et al. Towards standardization of quantitative retinal vascular parameters: comparison of SIVA and VAMPIRE measurements in the Lothian birth cohort 1936. Transl. Vis. Sci. Technol. 7, 12 (2018).
Cheung, C. Y., Ikram, M. K., Klein, R. & Wong, T. Y. The clinical implications of recent studies on the structure and function of the retinal microvasculature in diabetes. Diabetologia 58, 871–885 (2015).
Sun, C., Wang, J. J., Mackey, D. A. & Wong, T. Y. Retinal vascular caliber: systemic, environmental, and genetic associations. Surv. Ophthalmol. 54, 74–95 (2009).
Ding, J. et al. Retinal vascular caliber and the development of hypertension: a meta-analysis of individual participant data. J. Hypertens. 32, 207–215 (2014).
Sabanayagam, C. et al. Retinal microvascular calibre and risk of diabetes mellitus: a systematic review and participant-level meta-analysis. Diabetologia 58, 2476–2485 (2015).
Wong, T. Y. et al. Retinal vascular caliber, cardiovascular risk factors, and inflammation: the multi-ethnic study of atherosclerosis (MESA). Invest. Ophthalmol. Vis. Sci. 47, 2341–2350 (2006).
Cheung, N. et al. Retinal arteriolar narrowing and left ventricular remodeling: the multi-ethnic study of atherosclerosis. J. Am. Coll. Cardiol. 50, 48–55 (2007).
De Silva, D. A. et al. The relationship between aortic stiffness and changes in retinal microvessels among Asian ischemic stroke patients. J. Hum. Hypertens. 26, 716–722 (2012).
McGeechan, K. et al. Meta-analysis: retinal vessel caliber and risk for coronary heart disease. Ann. Intern. Med. 151, 404–413 (2009).
McGeechan, K. et al. Prediction of incident stroke events based on retinal vessel caliber: a systematic review and individual-participant meta-analysis. Am. J. Epidemiol. 170, 1323–1332 (2009).
Seidelmann, S. B. et al. Retinal vessel calibers in predicting long-term cardiovascular outcomes: the atherosclerosis risk in communities study. Circulation 134, 1328–1338 (2016).
Wong, T. Y. et al. Retinal arteriolar narrowing and risk of coronary heart disease in men and women. The atherosclerosis risk in communities study. JAMA 287, 1153–1159 (2002).
Wong, T. Y. et al. Retinal microvascular abnormalities and incident stroke: the atherosclerosis risk in communities study. Lancet 358, 1134–1140 (2001).
Ikram, M. K. et al. Retinal vessel diameters and risk of stroke: the Rotterdam study. Neurology 66, 1339–1343 (2006).
Kawasaki, R. et al. Retinal microvascular signs and risk of stroke: the multi-ethnic study of atherosclerosis (MESA). Stroke 43, 3245–3251 (2012).
McGeechan, K. et al. Risk prediction of coronary heart disease based on retinal vascular caliber (from the atherosclerosis risk in communities [ARIC] study). Am. J. Cardiol. 102, 58–63 (2008).
Wang, J. J. et al. Retinal vessel diameter and cardiovascular mortality: pooled data analysis from two older populations. Eur. Heart J. 28, 1984–1992 (2007).
Mutlu, U. et al. Retinal microvasculature is associated with long-term survival in the general adult Dutch population. Hypertension 67, 281–287 (2016).
Cheung, C. Y. et al. Retinal microvascular changes and risk of stroke: the Singapore Malay eye study. Stroke 44, 2402–2408 (2013).
Ho, H. et al. Retinopathy signs improved prediction and reclassification of cardiovascular disease risk in diabetes: a prospective cohort study. Sci. Rep. 7, 41492 (2017).
Wong, T. Y. et al. Quantitative retinal venular caliber and risk of cardiovascular disease in older persons: the cardiovascular health study. Arch. Intern. Med. 166, 2388–2394 (2006).
Wong, T. Y. et al. Retinal microvascular abnormalities and 10-year cardiovascular mortality: a population-based case-control study. Ophthalmology 110, 933–940 (2003).
Rajkomar, A., Dean, J. & Kohane, I. Machine learning in medicine. N. Engl. J. Med. 380, 1347–1358 (2019).
Chen, J. H. & Asch, S. M. Machine learning and prediction in medicine—beyond the peak of inflated expectations. N. Engl. J. Med. 376, 2507–2509 (2017).
Obermeyer, Z. & Emanuel, E. J. Predicting the future—big data, machine learning, and clinical medicine. N. Engl. J. Med. 375, 1216–1219 (2016).
Ting, D. S. W. et al. Development and validation of a deep learning system for diabetic retinopathy and related eye diseases using retinal images from multiethnic populations with diabetes. JAMA 318, 2211–2223 (2017).
Gulshan, V. et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 316, 2402–2410 (2016).
De Fauw, J. et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat. Med. 24, 1342–1350 (2018).
Kermany, D. S. et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell 172, 1122–1131 (2018).
Ran, A. R. et al. Detection of glaucomatous optic neuropathy with spectral-domain optical coherence tomography: a retrospective training and validation deep-learning analysis. Lancet Digit. Health 1, e172–e182 (2019).
Poplin, R. et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat. Biomed. Eng. 2, 158–164 (2018).
He, J. et al. The practical implementation of artificial intelligence technologies in medicine. Nat. Med. 25, 30–36 (2019).
Sharafi, S. M. et al. Vascular retinal biomarkers improves the detection of the likely cerebral amyloid status from hyperspectral retinal images. Alzheimers Dement. 5, 610–617 (2019).
Chan, V. T. T. et al. Using retinal imaging to study dementia. J. Vis. Exp. 129, 56137 (2017).
Welikala, R. A. et al. Automated arteriole and venule classification using deep learning for retinal images from the UK Biobank cohort. Comput. Biol. Med. 90, 23–32 (2017).
Yan, Z., Yang, X. & Cheng, K. T. A three-stage deep learning model for accurate retinal vessel segmentation. IEEE J. Biomed. Health Inf. 23, 1427–1436 (2019).
Oliveira, A., Pereira, S. & Silva, C. A. Retinal vessel segmentation based on fully convolutional neural networks. Expert Syst. Appl. 112, 229–242 (2018).
Welikala, R. A. et al. Automated quantification of retinal vessel morphometry in the UK biobank cohort. In Proc. 2017 Seventh International Conference on Image Processing Theory, Tools and Applications (IPTA) 1–6 (IEEE, 2017).
Maderuelo-Fernandez, J. A. et al. Automatic image analyser to assess retinal vessel calibre (ALTAIR). A new tool to evaluate the thickness, area and length of the vessels of the retina. Int. J. Med. Inf. 136, 104090 (2020).
Gunn, R. M. Ophthalmoscopic evidence of (1) arterial changes associated with chronic renal diseases and (2) of increased arterial tension. Trans. Ophthalmol. Soc. UK 12, 124–125 (1982).
Nguyen, T. T. & Wong, T. Y. Retinal vascular manifestations of metabolic disorders. Trends Endocrinol. Metab. 17, 262–268 (2006).
Wong, T. Y. & Scott, I. U. Clinical practice. Retinal-vein occlusion. N. Engl. J. Med. 363, 2135–2144 (2010).
Sharrett, A. R. et al. Retinal arteriolar diameters and elevated blood pressure: the atherosclerosis risk in communities study. Am. J. Epidemiol. 150, 263–270 (1999).
Wong, T. Y., Klein, R., Klein, B. E., Meuer, S. M. & Hubbard, L. D. Retinal vessel diameters and their associations with age and blood pressure. Invest. Ophthalmol. Vis. Sci. 44, 4644–4650 (2003).
Leung, H. et al. Relationships between age, blood pressure, and retinal vessel diameters in an older population. Invest. Ophthalmol. Vis. Sci. 44, 2900–2904 (2003).
Wong, T. Y. et al. Retinal microvascular abnormalities and blood pressure in older people: the cardiovascular health study. Br. J. Ophthalmol. 86, 1007–1013 (2002).
Kawasaki, R. et al. Cardiovascular risk factors and retinal microvascular signs in an adult Japanese population: the Funagata study. Ophthalmology 113, 1378–1384 (2006).
Ikram, M. K. et al. Are retinal arteriolar or venular diameters associated with markers for cardiovascular disorders? The Rotterdam study. Invest. Ophthalmol. Vis. Sci. 45, 2129–2134 (2004).
Jeganathan, V. S. et al. Effect of blood pressure on the retinal vasculature in a multi-ethnic Asian population. Hypertens. Res. 32, 975–982 (2009).
Ikram, M. K. et al. Retinal vessel diameters and risk of impaired fasting glucose or diabetes: the Rotterdam study. Diabetes 55, 506–510 (2006).
Nguyen, T. T., Wang, J. J. & Wong, T. Y. Retinal vascular changes in pre-diabetes and prehypertension: new findings and their research and clinical implications. Diabetes Care 30, 2708–2715 (2007).
Islam, F. M. et al. Quantitative retinal vascular calibre changes in diabetes and retinopathy: the Singapore Malay eye study. Eye 23, 1719–1724 (2009).
Tsai, A. S. et al. Differential association of retinal arteriolar and venular caliber with diabetes and retinopathy. Diabetes Res. Clin. Pr. 94, 291–298 (2011).
Cheung, C. Y. et al. Retinal vascular geometry in Asian persons with diabetes and retinopathy. J. Diabetes Sci. Technol. 6, 595–605 (2012).
Ting, D. S. W. et al. Artificial intelligence and deep learning in ophthalmology. Br. J. Ophthalmol. 103, 167–175 (2019).
Wang, F., Kaushal, R. & Khullar, D. Should health care demand interpretable artificial intelligence or accept “black box” medicine? Ann. Intern. Med. 172, 59–60 (2019).
Krittanawong, C. et al. Deep learning for cardiovascular medicine: a practical primer. Eur. Heart J. 40, 2058–2073 (2019).
Kuan, R. Adopting AI in health care will be slow and difficult. Harvard Business Review (18 October 2019).
Rizzoni, D. & Muiesan, M. L. Retinal vascular caliber and the development of hypertension: a meta-analysis of individual participant data. J. Hypertens. 32, 225–227 (2014).
Houben, A., Berendschot, T. & Grassi, G. Retinal microvascular diameters: normative data and their use in clinical hypertension. J. Hypertens. 35, 1573–1574 (2017).
Lee, H. et al. An explainable deep-learning algorithm for the detection of acute intracranial haemorrhage from small datasets. Nat. Biomed. Eng. 3, 173–182 (2019).
Wang, T. J. et al. Multiple biomarkers for the prediction of first major cardiovascular events and death. N. Engl. J. Med 355, 2631–2639 (2006).
Koenig, W., Lowel, H., Baumert, J. & Meisinger, C. C-reactive protein modulates risk prediction based on the Framingham score: implications for future risk assessment: results from a large cohort study in southern Germany. Circulation 109, 1349–1353 (2004).
Wickremasinghe, S. S. et al. Retinal vascular calibre changes after intravitreal bevacizumab or dexamethasone implant treatment for diabetic macular oedema. Br. J. Ophthalmol. 101, 1329–1333 (2017).
Wickremasinghe, S. S., Rogers, S. L., Gillies, M. C., Zhu, M. & Wong, T. Y. Retinal vascular caliber changes after intravitreal triamcinolone treatment for diabetic macular edema. Invest. Ophthalmol. Vis. Sci. 49, 4707–4711 (2008).
Reid, C. M. et al. Aspirin for the prevention of cognitive decline in the elderly: rationale and design of a neuro-vascular imaging study (ENVIS-ion). BMC Neurol. 12, 3 (2012).
Hughes, A. D. et al. Effect of antihypertensive treatment on retinal microvascular changes in hypertension. J. Hypertens. 26, 1703–1707 (2008).
Thom, S. et al. Differential effects of antihypertensive treatment on the retinal microcirculation: an Anglo-Scandinavian cardiac outcomes trial substudy. Hypertension 54, 405–408 (2009).
Knudtson, M. D. et al. Revised formulas for summarizing retinal vessel diameters. Curr. Eye Res. 27, 143–149 (2003).
Yip, W. et al. Comparison of common retinal vessel caliber measurement software and a conversion algorithm. Transl. Vis. Sci. Technol. 5, 11 (2016).
Sabanayagam, C. et al. Singapore Indian eye study-2: methodology and impact of migration on systemic and eye outcomes. Clin. Exp. Ophthalmol. 45, 779–789 (2017).
Foong, A. W. et al. Rationale and methodology for a population-based study of eye diseases in Malay people: the Singapore Malay eye study (SiMES). Ophthalmic Epidemiol. 14, 25–35 (2007).
Lavanya, R. et al. Methodology of the Singapore Indian Chinese cohort (SICC) eye study: quantifying ethnic variations in the epidemiology of eye diseases in Asians. Ophthalmic Epidemiol. 16, 325–336 (2009).
Cheung, C. Y. et al. C-reactive protein and retinal microvascular caliber in a multiethnic Asian population. Am. J. Epidemiol. 171, 206–213 (2010).
Li, X. et al. Racial differences in retinal vessel geometric characteristics: a multiethnic study in healthy Asians. Invest. Ophthalmol. Vis. Sci. 54, 3650–3656 (2013).
Shalev, I. et al. Retinal vessel caliber and lifelong neuropsychological functioning: retinal imaging as an investigative tool for cognitive epidemiology. Psychol. Sci. 24, 1198–1207 (2013).
Cheung, C. Y. et al. Quantitative retinal microvasculature in children using swept-source optical coherence tomography: the Hong Kong children eye study. Br. J. Ophthalmol. 103, 672–679 (2018).
Gopinath, B. et al. Retinal vascular geometry and the prevalence of atrial fibrillation and heart failure in a clinic-based sample. Heart Lung Circ. 28, 1631–1637 (2018).
Sun, Z. et al. OCT angiography metrics predict progression of diabetic retinopathy and development of diabetic macular edema: a prospective study. Ophthalmology 126, 1675–1684 (2019).
Li, L. J. et al. Gestational retinal microvasculature and the risk of 5 year postpartum abnormal glucose metabolism. Diabetologia 60, 2368–2376 (2017).
Nguyen, H. V. et al. Cost-effectiveness of a national telemedicine diabetic retinopathy screening program in Singapore. Ophthalmology 123, 2571–2580 (2016).
Yip, W. et al. Reliability and determinants of retinal vessel oximetry measurements in healthy eyes. Invest. Ophthalmol. Vis. Sci. 55, 7104–7110 (2014).
Wang, Y. X., Zhang, J. S., You, Q. S., Xu, L. & Jonas, J. B. Ocular diseases and 10-year mortality: the Beijing eye study 2001/2011. Acta Ophthalmol. 92, e424–e428 (2014).
Chua, S. Y. L. et al. Cohort profile: design and methods in the eye and vision consortium of UK Biobank. BMJ Open 9, e025077 (2019).
Rhee, E. J., Chung, P. W., Wong, T. Y. & Song, S. J. Relationship of retinal vascular caliber variation with intracranial arterial stenosis. Microvasc. Res. 108, 64–68 (2016).
Al-Fiadh, A. H. et al. Usefulness of retinal microvascular endothelial dysfunction as a predictor of coronary artery disease. Am. J. Cardiol. 115, 609–613 (2015).
Xu, D., Lee, M. L. & Hsu, W. Propagation mechanism for deep and wide neural networks. In Proc. 30th IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR) 9220–9228 (IEEE/CVF, 2019).
Nagelkerke, N. J. D. A note on a general definition of the coefficient of determination. Biometrika 78, 691–692 (1991).
Acknowledgements
We thank all of the staff at the SNEC Ocular Reading Centre (SORC) for their contribution to this study. We acknowledge funding support from Singapore Ministry of Health’s National Medical Research Council (NMRC) grants OFLCG/001/2017, NMRC/STaR/003/2008, NMRC/STaR/0016/2013 and NMRC/CIRG/1371/2013. This research is also supported by the National Research Foundation, Singapore, under its AI Singapore Programme (AISG Award no. AISG-GC-2019-001). Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not reflect the views of the National Research Foundation, Singapore.
Author information
Authors and Affiliations
Contributions
C.Y. Cheung, D.X., W.H., M.L.L. and T.Y.W. contributed to study conception and design. D.X., W.H. and M.L.L. coded and optimized the DLS. C.Y. Cheung and M.Y. analysed the data. C.Y. Cheung and T.Y.W. contributed to the initial draft of the manuscript. C.Y. Cheung and T.Y.W. had full access to the data, vouch for the integrity of the data and the adherence to the study protocol, and were responsible for the decision to submit the manuscript. All of the authors contributed to data collection and interpretation, and revision of the manuscript for important content.
Corresponding author
Ethics declarations
Competing interests
C.Y. Cheung, D.X., W.H., M.L.L. and T.Y.W. are filing a patent (World Intellectual Property Organization International Bureau; International publication no. WO 2020/167251) for the DLS described in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures and tables.
Rights and permissions
About this article
Cite this article
Cheung, C.Y., Xu, D., Cheng, CY. et al. A deep-learning system for the assessment of cardiovascular disease risk via the measurement of retinal-vessel calibre. Nat Biomed Eng 5, 498–508 (2021). https://doi.org/10.1038/s41551-020-00626-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-00626-4
This article is cited by
-
Remnant cholesterol is correlated with retinal vascular morphology and diabetic retinopathy in type 2 diabetes mellitus: a cross-sectional study
Lipids in Health and Disease (2024)
-
Relationship between psycho-physiological indicators and task performance under various indoor space designs for telecommuting environment by introducing mixed-reality
Scientific Reports (2024)
-
AI-integrated ocular imaging for predicting cardiovascular disease: advancements and future outlook
Eye (2024)
-
Res-Unet based blood vessel segmentation and cardio vascular disease prediction using chronological chef-based optimization algorithm based deep residual network from retinal fundus images
Multimedia Tools and Applications (2024)
-
Vascularized organoid-on-a-chip: design, imaging, and analysis
Angiogenesis (2024)