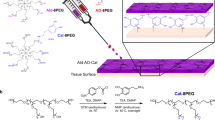
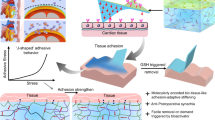
Abstract
Post-operative adhesions form as a result of normal wound healing processes following any type of surgery. In cardiac surgery, pericardial adhesions are particularly problematic during reoperations, as surgeons must release the adhesions from the surface of the heart before the intended procedure can begin, thereby substantially lengthening operation times and introducing risks of haemorrhage and injury to the heart and lungs during sternal re-entry and cardiac dissection. Here we show that a dynamically crosslinked supramolecular polymer–nanoparticle hydrogel, with viscoelastic and flow properties that enable spraying onto tissue as well as robust tissue adherence and local retention in vivo for two weeks, reduces the formation of pericardial adhesions. In a rat model of severe pericardial adhesions, the hydrogel markedly reduced the severity of the adhesions, whereas commercial adhesion barriers (including Seprafilm and Interceed) did not. The hydrogels also reduced the severity of cardiac adhesions (relative to untreated animals) in a clinically relevant cardiopulmonary-bypass model in sheep. This viscoelastic supramolecular polymeric hydrogel represents a promising clinical solution for the prevention of post-operative pericardial adhesions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. The raw and analysed datasets generated during the study are available for research purposes from the corresponding authors on reasonable request.
References
Lauder, C. I., Garcea, G., Strickland, A. & Maddern, G. J. Abdominal adhesion prevention: still a sticky subject? Dig. Surg. 27, 347–358 (2010).
Weibel, M. A. & Majno, G. Peritoneal adhesions and their relation to abdominal surgery. A postmortem study. Am. J. Surg. 126, 345–353 (1973).
DiZerega, G. S. in Peritoneal Surgery (ed. DiZerega, G. S.) 3–37 (Springer, 2000).
Ito, T. et al. The prevention of peritoneal adhesions by in situ cross-linking hydrogels of hyaluronic acid and cellulose derivatives. Biomaterials 28, 975–983 (2007).
Yea, Y. et al. Prevention of peritoneal adhesions with an in situ cross-linkable hyaluronan hydrogel delivering budesonide. J. Control. Release 120, 178–185 (2007).
Hirschelmann, A., Tchartchian, G., Wallwiener, M., Hackethal, A. & De Wilde, R. L. A review of the problematic adhesion prophylaxis in gynaecological surgery. Arch. Gynecol. Obstet. 285, 1089–1097 (2012).
Shahian, D. M. et al. The society of thoracic surgeons 2008 cardiac surgery risk models: part 1—coronary artery bypass grafting surgery. Ann. Thorac. Surg. 88, S2–S22 (2009).
O’Brien, S. M. et al. The Society of thoracic surgeons 2008 cardiac surgery risk models: part 2—isolated valve surgery. Ann. Thorac. Surg. 88, S23–S42 (2009).
Kansara, P. et al. Heart transplantation with and without prior sternotomy: analysis of the united network for organ sharing database. Transpl. Proc. 46, 249–255 (2014).
Hoffman, J. L. et al. The incidence of congenital heart disease. J. Am. Coll. Cardiol. 39, 1890–1900 (2002).
Jacobsa, J. P. et al. Reoperations for pediatric and congenital heart disease: An analysis of the society of thoracic surgeons (STS) congenital heart surgery database. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. Annu. 17, 2–8 (2014).
Sikirica, V. et al. The inpatient burden of abdominal and gynecological adhesiolysis in the US. BMC Surgery 11, 1–9 (2011).
Diamond, M. P., Burns, E. L., Accomando, B., Mian, S. & Holmdahl, L. Seprafilm adhesion barrier: (1) a review of preclinical, animal, and human investigational studies. Gynecol. Surg. 9, 237–245 (2012).
Malm, T., Bowald, S., Bylock, A. & Busch, C. Prevention of postoperative pericardial adhesions by closure of the pericardium with absorbable polymer patches. An experimental study. J. Thorac. Cardiovasc. Surg. 104, 600–607 (1992).
Duncan, D. A. et al. Prevention of postoperative pericardial adhesions with hydrophilic polymer solutions. J. Surg. Res. 45, 44–49 (1987).
Seeger, J. M. et al. Prevention of postoperative pericardial adhesions using tissue-protective solutions. J. Surg. Res. 68, 63–66 (1997).
Hoare, T., Yeo, Y., Bellas, E., Bruggeman, J. P. & Kohane, D. S. Prevention of peritoneal adhesions using polymeric rheological blends. Acta Biomater. 10, 1187–1193 (2014).
Yeo, Y. & Kohane, D. S. Polymers in the prevention of peritoneal adhesions. Eur. J. Pharm. Biopharm. 68, 57–66 (2008).
Grainger, D. A., Meyer, W. R., DeCherney, A. H. & Diamond, M. P. The use of hyaluronic acid polymers to reduce postoperative adhesions. J. Gynecol. Surg. 7, 97–101 (2009).
Sawhney, A. S., Pathak, C. P., Van Rensburg, J. J., Dunn, R. C. & Hubbell, J. A. Optimization of photopolymerized bioerodible hydrogel properties for adhesion prevention. Biomed. Mater. Res. 28, 831–838 (1994).
Connors, R. C. et al. Postoperative pericardial adhesion prevention using carbylan-SX in a rabbit model. J. Surg. Res. 140, 237–242 (2007).
Li, L. et al. Biodegradable and injectable in situ cross-linking chitosan-hyaluronic acid based hydrogels for postoperative adhesion prevention. Biomaterials 35, 903–3917 (2014).
Zhu, W. et al. Metal and light free ‘click’ hydrogels for prevention of post-operative peritoneal adhesions. Polym. Chem. 5, 2018–2026 (2014).
Chan, M. et al. Reducing the oxidation level of dextran aldehyde in a chitosan/dextran-based surgical hydrogel increases biocompatibility and decreases antimicrobial efficacy. Int. J. Mol. Sci. 16, 13798–13814 (2015).
Song, L. et al. Peritoneal adhesion prevention with a biodegradable and injectable N,O-carboxymethyl chitosan-aldehyde hyaluronic acid hydrogel in a rat repeated-injury model. Sci. Rep. 6, 37600 (2016).
Yang, Y. et al. A postoperative anti-adhesion barrier based on photoinduced imine-crosslinking hydrogel with tissue-adhesive ability. Acta Biomater. 62, 199–209 (2017).
Banasiewicz, T. et al. Preliminary study with SprayShieldadhesion barrier system in the prevention of abdominal adhesions. Video. Mini. 8, 301–309 (2013).
Napoleone, C. et al. An observational study of CoSeal for the prevention of adhesions in pediatric cardiac surgery. Inter. Cardiovasc. Thorac. Surg. 9, 978–982 (2009).
Haensig, M. et al. Bioresorbable adhesion barrier for reducing the severity of postoperative cardiac adhesions: Focus on REPEL-CV. Med. Devices 4, 17–25 (2011).
Hirschelmann, A. et al. Is patient education about adhesions a requirement in abdominopelvic surgery? Geburtshilfe Frauenheilkd. 72, 299–304 (2012).
Wang, Q. et al. High-water-content mouldable hydrogels by mixing clay and a dendritic molecular binder. Nature 463, 339–343 (2010).
Appel, E. A., Barrio, J., Loh, X. J. & Scherman, O. A. Supramolecular polymeric hydrogels. Chem. Soc. Rev. 41, 6195–6214 (2012).
Rose, S. et al. Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505, 382–385 (2014).
Appel, E. A. et al. Self-assembled hydrogels utilizing polymer–nanoparticle interactions. Nat. Commun. 4, 848–852 (2015).
Rodell, C. B. et al. Shear-thinning supramolecular hydrogels with secondary autonomous covalent crosslinking to modulate viscoelastic properties. Adv. Funct. Mater. 25, 636–644 (2015).
Webber, M. J. et al. Supramolecular biomaterials. Nat. Mater. 15, 13–26 (2015).
Appel, E. A. et al. Supramolecular cross-linked networks via host–guest complexation with cucurbit[8]uril. J. Am. Chem. Soc. 132, 14251–14260 (2010).
Appel, E. A. et al. High-water-content hydrogels from renewable resources through host-guest interactions. J. Am. Chem. Soc. 134, 11767–11773 (2012).
Appel, E. A. et al. Sustained release of proteins from high water content supramolecular hydrogels. Biomaterials 33, 4646–4652 (2012).
Appel, E. A. et al. Activation energies control macroscopic properties of physically crosslinked materials. Angew. Chem. Int. Ed. 53, 10038–10043 (2014).
Appel, E. A. et al. The control of cargo release from physically crosslinked hydrogels by crosslink dynamics. Biomaterials 35, 9897–9903 (2014).
Appel, E. A. & Scherman, O. A. Gluing gels: A nanoparticle solution. Nat. Mater. 13, 231–232 (2014).
Yu, A. C. et al. Scalable manufacturing of biomimetic moldable hydrogels for industrial applications. Proc. Natl Acad. Sci. USA 113, 14255–14260 (2016).
Evans, N. D., Oreffo, R. O., Healy, E., Thurner, P. J. & Man, Y. H. Epithelial mechanobiology, skin wound healing, and the stem cell niche. J. Mech. Behav. Biomed. Mater. 28, 397–409 (2013).
Arung, W., Meurisse, M. & Detry, O. Pathophysiology and prevention of postoperative peritoneal adhesions. World J. Gastroenterol. 17, 4545–4553 (2011).
Alizzi, A. M. et al. Reduction of post-surgical pericardial adhesions using a pig model. Heart Lung Circ. 21, 22–29 (2012).
Lassaletta, A. D., Chu, L. M. & Selke, F. W. Effects of alcohol on pericardial adhesion formation in hypercholesterolemic swine. J. Thorac. Cardiovasc. Surg. 143, 953–959 (2012).
Lassaletta, A. D. et al. Mechanism for reduced pericardial adhesion formation in hypercholesterolemic swine supplemented with alcohol. Eur. J. Cardiothorac. Surg. 43, 1058–1064 (2013).
Macarthur, J. W. et al. Preclinical evaluation of the engineered stem cell chemokine stromal cell-derived factor 1α analog in a translational ovine myocardial infarction model. Circ. Res. 114, 650–659 (2014).
Elmadhun, N. Y. et al. Effects of alcohol on postoperative adhesion formation in ischemic myocardium and pericardium. Ann. Thorac. Surg. 104, 545–552 (2017).
MacArthur, J. W. et al. Sustained release of engineered stromal cell-derived factor 1-α from injectable hydrogels effectively recruits endothelial progenitor cells and preserves ventricular function after myocardial infarction. Circulation 128, S79–S86 (2013).
DiZerega, G. S. et al. Peritoneal repair and post-surgical adhesion formation. Hum. Reprod. 6, 547–555 (2001).
Yashiharu, K. et al. Pharmacokinetics and biodisposition of poly(vinyl alcohol) in rats and mice. Drug Metab. Pharmacokinet. 20, 435–442 (2005).
Acknowledgements
This work was made possible by the financial support from the Stanford BIOX Interdisciplinary Initiatives Program Seed Grant (E.A.A. and Y.J.W.), the Stanford-Coulter Translational Research Grant (E.A.A. and Y.J.W.), the National Institutes of Health (R01HL089315-01, Y.J.W.), the American Heart Association postdoctoral fellowship (H.W. and M.J.P.) and predoctoral fellowship (L.M.S.), the National Science Foundation AGEP California Alliance Postdoctoral Fellowship (H.L.H.) and Graduate Research Fellowship Program (DGE-1147470, L.M.S., A.N.S. and G.A.), the Stanford Interdisciplinary Graduate Fellowship (L.M.S.), and the American Association for Thoracic Surgery Summer Intern Scholarship (K.M.W.). The authors thank the Bogyo laboratory for the use of the Pearl instrument, the Stanford Veterinary Services Center for assistance with ovine surgeries and the Stanford Animal Histology Services for assistance with histology.
Author information
Authors and Affiliations
Contributions
L.M.S., A.N.S., A.C.Y., H.W., M.J.P., A.A.A.S., Y.J.W. and E.A.A. designed experiments; L.M.S., H.W., H.J.L., A.D.T., H.L.H., G.A., K.P.T. and E.A.A. conducted experiments; J.M.F., H.J.L., A.D.T., A.E., K.M.W., C.E.H., K.J.J., M.J.P., S.W.B., B.C., C.M., F.G., H.B. and M.M. assisted with ovine surgeries; L.M.S., A.N.S., Y.T., Y.J.W. and E.A.A. analysed data; and L.M.S., Y.J.W. and E.A.A. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures and video captions.
Supplementary Video 1
An untreated heart in a rodent pericardial-adhesion model
Supplementary Video 2
Seprafilm-treated heart in a rodent pericardial-adhesion model
Supplementary Video 3
Interceed-treated heart in a rodent pericardial-adhesion model
Supplementary Video 4
PNP-2:10-hydrogel-treated heart in a rodent pericardial-adhesion model
Supplementary Video 5
PNP-1:10-hydrogel-treated heart in a rodent pericardial-adhesion model
Supplementary Video 6
PNP-1:5-hydrogel-treated heart in a rodent pericardial-adhesion model
Supplementary Video 7
PNP-1:1-hydrogel-treated heart in a rodent pericardial-adhesion model
Supplementary Video 8
PNP-0.2:10-hydrogel-treated heart in a rodent pericardial-adhesion model
Supplementary Video 9
Spraying the PNP-1:10-hydrogel treatment onto the epicardial surface following epicardial abrasion
Rights and permissions
About this article
Cite this article
Stapleton, L.M., Steele, A.N., Wang, H. et al. Use of a supramolecular polymeric hydrogel as an effective post-operative pericardial adhesion barrier. Nat Biomed Eng 3, 611–620 (2019). https://doi.org/10.1038/s41551-019-0442-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0442-z
This article is cited by
-
Neutralization of excessive levels of active TGF-β1 reduces MSC recruitment and differentiation to mitigate peritendinous adhesion
Bone Research (2023)
-
Chronological adhesive cardiac patch for synchronous mechanophysiological monitoring and electrocoupling therapy
Nature Communications (2023)
-
Cell barrier function of resident peritoneal macrophages in post-operative adhesions
Nature Communications (2021)
-
Physical networks from entropy-driven non-covalent interactions
Nature Communications (2021)
-
Preventing post-surgical cardiac adhesions with a catechol-functionalized oxime hydrogel
Nature Communications (2021)