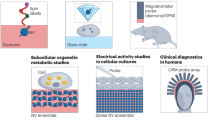
Abstract
Capabilities in real-time monitoring of internal physiological processes could inform pharmacological drug-delivery schedules, surgical intervention procedures and the management of recovery and rehabilitation. Current methods rely on external imaging techniques or implantable sensors, without the ability to provide continuous information over clinically relevant timescales, and/or with requirements in surgical procedures with associated costs and risks. Here, we describe injectable classes of photonic devices, made entirely of materials that naturally resorb and undergo clearance from the body after a controlled operational lifetime, for the spectroscopic characterization of targeted tissues and biofluids. As an example application, we show that the devices can be used for the continuous monitoring of cerebral temperature, oxygenation and neural activity in freely moving mice. These types of devices should prove useful in fundamental studies of disease pathology, in neuroscience research, in surgical procedures and in monitoring of recovery from injury or illness.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information files. The raw and analysed datasets generated during the study are available for research purposes from the corresponding author on reasonable request.
References
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Lo, E. H., Dalkara, T. & Moskowitz, M. A. Mechanisms, challenges and opportunities in stroke. Nat. Rev. Neurosci. 4, 399–414 (2003).
DeBerardinis, R. J. & Chandel, N. S. Fundamentals of cancer metabolism. Sci. Adv. 2, e1600200 (2016).
Tai, L.-C. et al. Methylxanthine drug monitoring with wearable sweat sensors. Adv. Mater. 30, 1707442 (2018).
Kim, H. et al. Single-neuronal cell culture and monitoring platform using a fully transparent microfluidic DEP device. Sci. Rep. 8, 13194 (2018).
Bettinger, C. J. Recent advances in materials and flexible electronics for peripheral nerve interfaces. Bioelectron. Med. 4, 6 (2018).
Hamaoka, T., McCully, K. K., Niwayama, M. & Chance, B. The use of muscle near-infrared spectroscopy in sport, health and medical sciences: recent developments. Phil. Trans. R. Soc. A Math. Phys. Eng. Sci. 369, 4591–4604 (2011).
Ferrari, M. & Quaresima, V. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage 63, 921–935 (2012).
Lloyd-Fox, S., Blasi, A. & Elwell, C. E. Illuminating the developing brain: the past, present and future of functional near infrared spectroscopy. Neurosci. Biobehav. Rev. 34, 269–284 (2010).
Hwang, S.-W. et al. A physically transient form of silicon electronics. Science 337, 1640–1644 (2012).
Kang, S. et al. Bioresorbable silicon electronic sensors for the brain. Nature 530, 71–76 (2016).
Tao, H. et al. Silk-based resorbable electronic devices for remotely controlled therapy and in vivo infection abatement. Proc. Natl Acad. Sci. USA 111, 17385–17389 (2014).
Luo, M., Martinez, A. W., Song, C., Herrault, F. & Allen, M. G. A microfabricated wireless RF pressure sensor made completely of biodegradable materials. J. Micro. Syst. 23, 4–13 (2014).
Lu, L. et al. Biodegradable monocrystalline silicon photovoltaic microcells as power supplies for transient biomedical implants. Adv. Energy Mater. 8, 1703035 (2018).
Bai, W. et al. Flexible transient optical waveguides and surface-wave biosensors constructed from monocrystalline silicon. Adv. Mater. 30, 1801584 (2018).
Nizamoglu, S. et al. Bioabsorbable polymer optical waveguides for deep-tissue photomedicine. Nat. Commun. 7, 10374 (2016).
Yu, K. J. et al. Bioresorbable silicon electronics for transient spatiotemporal mapping of electrical activity from the cerebral cortex. Nat. Mater. 15, 782–791 (2016).
Fang, H. et al. Capacitively coupled arrays of multiplexed flexible silicon transistors for long-term cardiac electrophysiology. Nat. Biomed. Eng. 1, 0038 (2017).
Son, D. et al. Bioresorbable electronic stent integrated with therapeutic nanoparticles for endovascular diseases. ACS Nano 9, 5937–5946 (2015).
Karliczek, A. et al. Intraoperative assessment of microperfusion with visible light spectroscopy for prediction of anastomotic leakage in colorectal anastomoses. Color. Dis. 12, 1018–1025 (2010).
Bogomolov, A. et al. Development and testing of an LED-based near-infrared sensor for human kidney tumor diagnostics. Sensors 17, 1914 (2017).
Yun, S. H. & Kwok, S. J. J. Light in diagnosis, therapy and surgery. Nat. Biomed. Eng. 1, 0008 (2017).
Du, Q. et al. Chip-scale broadband spectroscopic chemical sensing using an integrated supercontinuum source in a chalcogenide glass waveguide. Photonics Res. 6, 506–510 (2018).
Warden, M. R., Cardin, J. A. & Deisseroth, K. Optical neural interfaces. Annu. Rev. Biomed. Eng. 16, 103–129 (2014).
Kang, S.-K. et al. Dissolution behaviors and applications of silicon oxides and nitrides in transient electronics. Adv. Funct. Mater. 24, 4427–4434 (2014).
Fu, R. et al. Implantable and biodegradable poly(l-lactic acid) fibers for optical neural interfaces. Adv. Opt. Mater. 6, 1700941 (2018).
Lee, Y. K. et al. Dissolution of monocrystalline silicon nanomembranes and their use as encapsulation layers and electrical interfaces in water-soluble electronics. ACS Nano 11, 12562–12572 (2017).
Yin, L. et al. Dissolvable metals for transient electronics. Adv. Funct. Mater. 24, 645–658 (2014).
Yin, L. et al. Mechanisms for hydrolysis of silicon nanomembranes as used in bioresorbable electronics. Adv. Mater. 27, 1857–1864 (2015).
Kang, S. K. et al. Dissolution chemistry and biocompatibility of silicon- and germanium-based semiconductors for transient electronics. ACS Appl. Mater. Interfaces 7, 9297–9305 (2015).
Brown, J. Q., Vishwanath, K., Palmer, G. M. & Ramanujam, N. Advances in quantitative UV-visible spectroscopy for clinical and pre-clinical application in cancer. Curr. Opin. Biotechnol. 20, 119–131 (2009).
Menon, L. et al. Transferred flexible three-color silicon membrane photodetector arrays. IEEE Photonics J. 7, 1–6 (2015).
Liu, C. et al. High performance, biocompatible dielectric thin-film optical filters integrated with flexible substrates and microscale optoelectronic devices. Adv. Opt. Mater. 6, 1800146 (2018).
Macleod, H. A. & Hugh, A. Thin-Film Optical Filters (CRC Press/Taylor & Francis, 2010).
Chen, T.-W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Besser, R. S., Louris, P. J. & Musket, R. G. Chemical etch rate of plasma-enhanced chemical vapor deposited SiO2 films. J. Electrochem. Soc. 144, 2859–2864 (1997).
Kraitl, J., Timm, U. & Ewald, H. Non-invasive measurement of blood and tissue parameters based on VIS-NIR spectroscopy. In Proc. SPIE 8591 (SPIE, 2013).
Lu, G. & Fei, B. Medical hyperspectral imaging: a review. J. Biomed. Opt. 19, 010901 (2014).
Ciurczak, E. W. & Igne, B. Pharmaceutical and Medical Applications of Near-Infrared Spectroscopy 2nd edn (CRC Press, 2014).
Durduran, T., Choe, R., Baker, W. B. & Yodh, A. G. Diffuse optics for tissue monitoring and tomography. Rep. Prog. Phys. 73, 076701 (2010).
Mrozek, S., Vardon, F. & Geeraerts, T. Brain temperature: physiology and pathophysiology after brain injury. Anesthesiol. Res. Pract. 2012, 989487 (2012).
Wang, H. et al. Brain temperature and its fundamental properties: a review for clinical neuroscientists. Front. Neurosci. 8, 307 (2014).
Ho, C. L., Wang, C. M., Lee, K. K., Ng, I. & Ang, B. T. Cerebral oxygenation, vascular reactivity, and neurochemistry following decompressive craniectomy for severe traumatic brain injury. J. Neurosurg. 108, 943–949 (2008).
Leal-Noval, S. R. et al. Invasive and noninvasive assessment of cerebral oxygenation in patients with severe traumatic brain injury. Intensive Care Med. 36, 1309–1317 (2010).
Toet, M. C., Lemmers, P. M. A., van Schelven, L. J. & van Bel, F. Cerebral oxygenation and electrical activity after birth asphyxia: their relation to outcome. Pediatrics 117, 333–339 (2006).
Brassard, P., Ainslie, P. N. & Secher, N. H. Cerebral oxygenation in health and disease. Front. Physiol. 5, 458 (2014).
Van Bel, F. & Mintzer, J. P. Monitoring cerebral oxygenation of the immature brain: a neuroprotective strategy? Pediatr. Res. 84, 159–164 (2018).
Tye, K. M. & Deisseroth, K. Optogenetic investigation of neural circuits underlying brain disease in animal models. Nat. Rev. Neurosci. 13, 251–266 (2012).
Cui, G. et al. Concurrent activation of striatal direct and indirect pathways during action initiation. Nature 494, 238–242 (2013).
Thrane, A. S. et al. General anesthesia selectively disrupts astrocyte calcium signaling in the awake mouse cortex. Proc. Natl Acad. Sci. USA 109, 18974–18979 (2012).
Siuda, E. R., Al-Hasani, R., McCall, J. G., Bhatti, D. L. & Bruchas, M. R. Chemogenetic and optogenetic activation of Gαs signaling in the basolateral amygdala induces acute and social anxiety-like states. Neuropsychopharmacology 41, 2011–2023 (2016).
Jeong, J.-W. et al. Wireless optofluidic systems for programmable in vivo pharmacology and optogenetics. Cell 162, 662–674 (2015).
Acknowledgements
W.Z. acknowledges support from the Army Research Office under grant W911NF-15-1-0035. This work utilized the Northwestern University Micro/Nano Fabrication Facility, which is partially supported by the Soft and Hybrid Nanotechnology Experimental Resource (NSF ECCS-1542205), Materials Research Science and Engineering Center (NSF DMR-1121262), State of Illinois, Northwestern University and Center for Bio-Integrated Electronics (Simpson Querrey Institute). The Center for Developmental Therapeutics is supported by Cancer Center Support Grant P30 CA060553 from the National Cancer Institute, awarded to the Robert H. Lurie Comprehensive Cancer Center.
Author information
Authors and Affiliations
Contributions
W.B., R.F., J.S., D.L., X.N., Y.P., Z.L., T.H., Y.L., D.W., H.Z., X.S., L.Y., W.Z. and J.A.R. designed and fabricated the devices, and performed the analysis. W.B., I.K., J.S., D.L., X.N., Y.P., I.S. and F.A. performed the animal study. C.R.H. and A.B. performed the computed tomography imaging. W.B., I.K, J.S., D.W., X.N., Q.Y., J.Z., K.M. and M.P. performed the study of bioresorption, biodistribution and toxicity. W.B., J.S., I.K., R.F., W.Z. and J.A.R wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary notes, figures and tables.
Rights and permissions
About this article
Cite this article
Bai, W., Shin, J., Fu, R. et al. Bioresorbable photonic devices for the spectroscopic characterization of physiological status and neural activity. Nat Biomed Eng 3, 644–654 (2019). https://doi.org/10.1038/s41551-019-0435-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0435-y
This article is cited by
-
Digital automation of transdermal drug delivery with high spatiotemporal resolution
Nature Communications (2024)
-
A wireless optoelectronic probe to monitor oxygenation in deep brain tissue
Nature Photonics (2024)
-
Advances in Wireless, Batteryless, Implantable Electronics for Real-Time, Continuous Physiological Monitoring
Nano-Micro Letters (2024)
-
Enabling low-drift flexible perovskite photodetectors by electrical modulation for wearable health monitoring and weak light imaging
Nature Communications (2023)
-
Wireless charging-mediated angiogenesis and nerve repair by adaptable microporous hydrogels from conductive building blocks
Nature Communications (2022)