Abstract
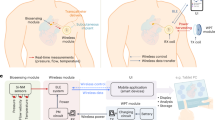
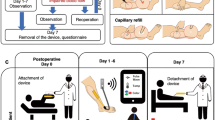
The ability to monitor blood flow is critical to patient recovery and patient outcomes after complex reconstructive surgeries. Clinically available wired implantable monitoring technology requires careful fixation for accurate detection and needs to be removed after use. Here, we report the design of a pressure sensor, made entirely of biodegradable materials and based on fringe-field capacitor technology, for measuring arterial blood flow in both contact and non-contact modes. The sensor is operated wirelessly through inductive coupling, has minimal hysteresis, fast response times, excellent cycling stability, is highly robust, allows for easy mounting and eliminates the need for removal, thus reducing the risk of vessel trauma. We demonstrate the operation of the sensor with a custom-made artificial artery model and in vivo in rats. This technology may be advantageous in real-time post-operative monitoring of blood flow after reconstructive surgery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information. Raw data generated for this study are available from the corresponding author on reasonable request.
References
Regenbogen, S. E. et al. Costs and consequences of early hospital discharge after major inpatient surgery in older adults. JAMA Surg. 152, e170123 (2017).
Smit, J. M. Advancements in free flap monitoring in the last decade: a critical review. Plast. Reconstr. Surg. 125, 177–185 (2010).
Lin, S. J. et al. Tissue oximetry monitoring in microsurgical breast reconstruction decreases flap loss and improves rate of flap salvage. Plast. Reconstr. Surg. 127, 1080–1085 (2011).
Swartz, W. M., Jones, N. F., Cherup, L. & Klein, A. Direct monitoring of microvascular anastomoses with the 20-MHz ultrasonic Doppler probe: an experimental and clinical study. Plast. Reconstr. Surg. 81, 149–161 (1988).
Bill, T. J., Foresman, P. A., Rodeheaver, G. T. & Drake, D. B. Fibrin sealant: a novel method of fixation for an implantable ultrasonic microDoppler probe. J. Reconstr. Microsurg. 17, 257–262 (2001).
Whitaker, I. S., Smit, J. M. & Acosta, R. A simple method of implantable Doppler cuff attachment: experience in 150 DIEP breast reconstructions. J. Plast. Reconstr. Aesthet. Surg. 61, 1251–1252 (2008).
Smit, J. M. et al. Post operative monitoring of microvascular breast reconstructions using the implantable Cook–Swartz Doppler system: a study of 145 probes & technical discussion. J. Plast. Reconstr. Aest. Surg. 62, 1286–1292 (2009).
Ruhhammer, J. et al. Magnetic sensor for monitoring of arterial strain. In 2013 Transducers & Eurosensors XXVII (2013).
Seres, L., Makula, E., Morvay, Z. & Borbely, L. Color Doppler ultrasound for monitoring free flaps in the head and neck region. J. Craniofac. Surg. 13, 75–78 (2002).
Few, J. W., Corral, C. J., Fine, N. A. & Dumanian, G. A. Monitoring buried head and neck free flaps with high-resolution colorduplex ultrasound. Plast. Reconstr. Surg. 108, 709–712 (2001).
Stone, C. A., Dubbins, P. A. & Morris, R. J. Use of colour duplex Doppler imaging in the postoperative assessment of buried free flaps. Microsurgery 21, 223–227 (2001).
Schon, R. et al. Color duplex sonography for the monitoring of vascularized free bone flaps. Otolaryngol. Head Neck Surg. 129, 71–76 (2003).
Scheufler, O., Exner, K. & Andresen, R. Investigation of TRAM flap oxygenation and perfusion by near-infrared reflection spectroscopy and color-coded duplex sonography. Plast. Reconstr. Surg. 113, 141–155 (2004).
Irwin, M. S., Thorniley, M. S., Dore, C. J. & Green, C. J. Near infra-red spectroscopy: a non-invasive monitor of perfusion and oxygenation within the microcirculation of limbs and flaps. Br. J. Plast. Surg. 48, 14–22 (1995).
Repez, A., Oroszy, D. & Arnez, Z. M. Continuous postoperative monitoring of cutaneous free flaps using near infrared spectroscopy. J. Plast. Reconstr. Aesthet. Surg. 61, 71–77 (2008).
Cai, Z. G. et al. Evaluation of near infrared spectroscopy in monitoring postoperative regional tissue oxygen saturation for fibular flaps. J. Plast. Reconstr. Aesthet. Surg. 61, 289–296 (2008).
Delgado, J. M., DeFeudis, F. V., Roth, R. H., Ryugo, D. K. & Mitruka, B. M. Dialytrode for long term intracerebral perfusion in awake monkeys. Arch. Int. Pharmacodynam. Ther. 198, 9–21 (1972).
Rojdmark, J., Blomqvist, L., Malm, M., Adams-Ray, B. & Ungerstedt, U. Metabolism in myocutaneous flaps studied by in situ microdialysis. Scand. J. Plast. Reconstr. Surg. Hand Surg. 32, 27–34 (1998).
Goodman, J. C. et al. Extracellular lactate and glucose alterations in the brain after head injury measured by microdialysis. Crit. Care Med. 27, 1965–1973 (1999).
Setala, L. et al. Microdialysis detects postoperative perfusion failure in microvascular flaps. J. Reconstr. Microsurg. 22, 87–96 (2006).
Holzle, F., Loeffelbein, D. J., Nolte, D. & Wolff, K. D. Free flap monitoring using simultaneous non-invasive laser Doppler flowmetry and tissue spectrophotometry. J. Craniomaxillofac. Surg. 34, 25–33 (2006).
Heller, L., Levin, L. S. & Klitzman, B. Laser Doppler flowmeter monitoring of free-tissue transfers: blood flow in normal and complicated cases. Plast. Reconstr. Surg. 107, 1739–1745 (2001).
Yuen, J. C. & Feng, Z. Monitoring free flaps using the laser Doppler flowmeter: five-year experience. Plast. Reconstr. Surg. 105, 55–61 (2000).
Hallock, G. G. A. A “true” false-negative misadventure in free flap monitoring using laser Doppler flowmetry. Plast. Reconstr. Surg. 110, 1609–1611 (2002).
Fu, R. et al. Implantable and biodegradable poly(l‐lactic acid) fibers for optical neural interfaces. Adv. Opt. Mater. 6, 1870010 (2018).
Yu, K. J. et al. Bioresorbable silicon electronics for transient spatiotemporal mapping of electrical activity from the cerebral cortex. Nat. Mater. 15, 782–791 (2016).
Lee, G. et al. Fully biodegradable microsupercapacitor for power storage in transient electronics, advanced energy. Adv. Energy Mater. 7, 1700157 (2017).
Mahajan, B. K., Yu, X., Shou, W., Pan, H. & Huang, X. Mechanically milled irregular zinc nanoparticles for printable bioresorbable electronics. Small 13, 1700065 (2017).
Chen, C. et al. Transient micromotors that disappear when no longer needed. ACS Nano 10, 10389–10396 (2016).
Um, G. T. et al. Implantable Cook–Swartz Doppler probe versus Synovis Flow Coupler for the postoperative monitoring of free flap breast reconstruction. J. Plast. Reconstr. Aesthet. Surg. 67, 960–966 (2014).
Chen, K. T. et al. Timing of presentation of the first signs of vascular compromise dictates the salvage outcome of free flap transfers. Plast. Reconstr. Surg. 120, 187–195 (2007).
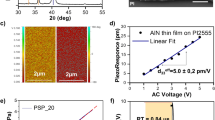
Boutry, C. M. et al. A ensitive and biodegradable pressure sensor array for cardiovascular monitoring. Adv. Mater. 27, 6954–6961 (2015).
Holland, S. J., Tighe, B. J. & Gould, P. L. Polymers for biodegradable medical devices. 1. The potential of polyesters as controlled macromolecular release systems. J. Control. Release 4, 155–180 (1985).
Kelly, P. & Atkins, T. W. in Microencapsulation of Drugs (Harwood Academic Publishers, 1992).
Wang, Y., Ameer, G. A., Sheppard, B. J. & Langer, R. A tough biodegradable elastomer. Nat. Biotechnol. 20, 602–606 (2002).
Tran, R. T. et al. Synthesis and characterization of a biodegradable elastomer featuring a dual crosslinking mechanism. Soft Matter 6, 2449–2461 (2010).
Zhang, B. et al. Biodegradable scaffold with built-in vasculature for organ-on-a-chip engineering and direct surgical anastomosis. Nat. Mater. 15, 669–678 (2016).
Davenport-Huyer, L. et al. Highly elastic and moldable polyester biomaterial for cardiac tissue engineering applications. ACS Biomater. Sci. Eng. 2, 780–788 (2016).
Boutry, C. M. et al. A stretchable and biodegradable strain and pressure sensor for orthopedic application. Nat. Electron. 1, 314–321 (2018).
Purnama, A., Hermawan, H. & Mantovani, D. Biodegradable metal stents: a focused review on materials and clinical studies. J. Biomater. Tissue Eng. 4, 868–874 (2014).
Zheng, Y. F., Gu, X. N. & Witte, F. Biodegradable metals. Mater. Sci. Eng. R 77, 1–34 (2014).
Yin, L. et al. Dissolvable metals for transient electronics. Adv. Funct. Mater. 24, 645–658 (2014).
Chen, L. Y. et al. Continuous wireless pressure monitoring and mapping with ultra-small passive sensors for health monitoring and critical care. Nat. Commun. 5, 5028 (2014).
Chen, H. & Lou, A. A study of RF power attenuation in bio-tissues. J. Med. Biol. Eng. 24, 141–146 (2004).
Donaldson, N. D. & Perkins, T. A. Analysis of resonant coupled coils in the design of radio frequency transcutaneous links. Med. Biol. Eng. Comput. 21, 612–627 (1983).
Bocan, K. N. et al. Multi-disciplinary challenges in tissue modeling for wireless electromagnetic powering: a review. IEEE Sens. J. 17, 6498–6509 (2017).
Cicekcibasi, A. E. et al. The mandibular landmarks about the facial artery and vein with multidetector computed tomography angiography (MDCTA): an anatomical and radiological morphometric study. Int. J. Morphol. 30, 504–509 (2012).
Graf., S. et al. Experimental and clinical validation of arterial diameter waveform and intimal media thickness obtained from B-mode ultrasound image processing. Ultrasound Med. Biol. 25, 1353–1363 (1999).
Zhao, Y. P. et al. Color Doppler sonography of the facial artery. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 93, 195–201 (2002).
Nair, N. R. et al. in Current Developments in Biotechnology and Bioengineering Production, Isolation and Purification of Industrial Products 739–755 (2017).
Hoa, J. S. et al. Wireless power transfer to deep-tissue microimplants. Proc. Natl Acad. Sci. USA 111, 7974–7979 (2014).
Brennan, J. F. et al. Determination of human coronary artery composition by Raman spectroscopy. Circulation 96, 99–105 (1997).
Gamble, G., Zorn, J., Sanders, G., MacMahon, S. & Sharpe, N. Estimation of arterial stiffness, compliance, and distensibility from M-mode ultrasound measurements of the common carotid artery. Stroke 25, 11–16 (1994).
Chortos, A., Liu, J. & Bao, Z. Pursuing prosthetic electronic skin. Nat. Mater. 15, 937–950 (2016).
Acknowledgements
C.M.B. acknowledges postdoctoral fellowship support from the Swiss National Science Foundation (postdoc mobility fellowship no. P2EZP2_152118) and the European Commission (Marie Curie international outgoing fellowship grant no. 622362). L.B. and Y.K. acknowledge the Stanford ChEM-H Postdocs at the Interface seed grant. Part of this work was performed at the Stanford Nano Shared Facilities. A.C.H. acknowledges support from the National Science Foundation Graduate Research Fellowship (grant no. DGE-1147474).
Author information
Authors and Affiliations
Contributions
C.M.B. and L.B. were the main contributors to this work and were responsible for elaboration of the sensor concept, development of new fabrication processes, performing all experiments for investigations into the materials, characterization of devices, data collection for in vitro experiments, data analysis and interpretation, and drafting of the article. L.B. and Y.K. performed the in vivo experiments. Y.K. performed implantation surgeries related to the in vivo studies, which included the design of the in vivo studies, development of the protocols, sensor implantation, operation studies and material biocompatibility studies. Y.K. also worked on data analysis and interpretation. C.V. contributed with the design of the LCR wireless sensor and performed all of the Computer Simulation Technology simulations. H.T. synthesized the biodegradable materials. A.C.H. and R.P. developed a new fabrication set-up for the fabrication of the Mg interconnect and provided critical revisions of the article. S.N. provided the initial Comsol simulations. J.L. and J.Cl. participated in the elaboration of the sensor concept, development of new fabrication processes and fabrication of the sensor. Z.W., J.Ch. and P.M.F. contributed to the design of the in vivo studies, working on the protocols and data interpretation. Y.K., P.M.F. and C.V. also contributed to the drafting and critical revision of the article. Z.B. contributed to the development of the sensor concept, fabrication processes, investigations of the materials, characterization of the devices, data interpretation and critical revisions of the article.
Corresponding author
Ethics declarations
Competing interests
Stanford University has filed a provisional patent application (62750518) related to this technology.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures, table and discussion
Supplementary Video 1
Heart-pulse-rate measurement via external Doppler ultrasound during in vivo characterization
Supplementary Video 2
After 12 weeks of sensor implantation, the rat was able to move without any apparent limb impairment
Rights and permissions
About this article
Cite this article
Boutry, C.M., Beker, L., Kaizawa, Y. et al. Biodegradable and flexible arterial-pulse sensor for the wireless monitoring of blood flow. Nat Biomed Eng 3, 47–57 (2019). https://doi.org/10.1038/s41551-018-0336-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-018-0336-5
This article is cited by
-
Flexible pressure sensors with ultrahigh stress tolerance enabled by periodic microslits
Microsystems & Nanoengineering (2024)
-
Two way workable microchanneled hydrogel suture to diagnose, treat and monitor the infarcted heart
Nature Communications (2024)
-
Kirigami-inspired, three-dimensional piezoelectric pressure sensors assembled by compressive buckling
npj Flexible Electronics (2024)
-
Permanent fluidic magnets for liquid bioelectronics
Nature Materials (2024)
-
Fully bioresorbable hybrid opto-electronic neural implant system for simultaneous electrophysiological recording and optogenetic stimulation
Nature Communications (2024)