Abstract
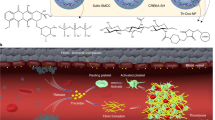
Limited intratumoural perfusion and nanoparticle retention remain major bottlenecks for the delivery of nanoparticle therapeutics into tumours. Here, we show that polymer–lipid–peptide nanoparticles delivering the antiplatelet antibody R300 and the chemotherapeutic agent doxorubicin can locally deplete tumour-associated platelets, thereby enhancing vascular permeability and augmenting the accumulation of the nanoparticles in tumours. R300 is specifically released in the tumour on cleavage of the lipid–peptide shell of the nanoparticles by matrix metalloprotease 2, which is commonly overexpressed in tumour vascular endothelia and stroma, thus facilitating vascular breaches that enhance tumour permeability. We also show that this strategy leads to substantial tumour regression and metastasis inhibition in mice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
02 August 2017
In the version of the Supplementary Information originally published, in Supplementary Fig. 8a, in the bottom row, the left-most image (‘Control’) was not the correct image; this has now been replaced.
References
Iyer, A. K., Khaled, G., Fang, J. & Maeda, H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov. Today 11, 812–818 (2006).
Maeda, H., Nakamura, H. & Fang, J. The EPR effect for macromolecular drug delivery to solid tumors: improvement of tumor uptake, lowering of systemic toxicity, and distinct tumor imaging in vivo. Adv. Drug Deliv. Rev. 65, 71–79 (2013).
Nichols, J. W. & Bae, Y. H. EPR: evidence and fallacy. J. Control. Release 190, 451–464 (2014).
Albanese, A., Tang, P. S. & Chan, W. C. W. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu. Rev. Biomed. Eng. 14, 1–16 (2012).
Meng, H. et al. Use of size and a copolymer design feature to improve the biodistribution and the enhanced permeability and retention effect of doxorubicin-loaded mesoporous silica nanoparticles in a murine xenograft tumor model. ACS Nano 5, 4131–4144 (2011).
Zhao, X. et al. Co-delivery of HIF1α siRNA and gemcitabine via biocompatible lipid-polymer hybrid nanoparticles for effective treatment of pancreatic cancer. Biomaterials 46, 13–25 (2015).
Grodzinski, P. & Farrell, D. Future opportunities in cancer nanotechnology—NCI Strategic Workshop report. Cancer Res. 74, 1307–1310 (2014).
Prabhakar, U. et al. Challenges and key considerations of the enhanced permeability and retention effect for nanomedicine drug delivery in oncology. Cancer Res. 73, 2412–2417 (2013).
Ruenraroengsak, P., Cook, J. M. & Florence, A. T. Nanosystem drug targeting: facing up to complex realities. J. Control. Release 141, 265–276 (2010).
Maeda, H. Nitroglycerin enhances vascular blood flow and drug delivery in hypoxic tumor tissues: analogy between angina pectoris and solid tumors and enhancement of the EPR effect. J. Control. Release 142, 296–298 (2010).
Nagamitsu, A., Greish, K. & Maeda, H. Elevating blood pressure as a strategy to increase tumor-targeted delivery of macromolecular drug SMANCS: cases of advanced solid tumors. Jpn. J. Clin. Oncol. 39, 756–766 (2009).
Noguchi, A. et al. Enhanced tumor localization of monoclonal antibody by treatment with kininase II inhibitor and angiotensin II. Jpn J. Cancer Res. 83, 240–243 (1992).
Seki, T., Fang, J. & Maeda, H. Enhanced delivery of macromolecular antitumor drugs to tumors by nitroglycerin application. Cancer Sci. 100, 2426–2430 (2009).
Cabral, H. et al. Accumulation of sub-100 nm polymeric micelles in poorly permeable tumours depends on size. Nat. Nanotech. 6, 815–823 (2011).
Kano, M. R. et al. Improvement of cancer-targeting therapy, using nanocarriers for intractable solid tumors by inhibition of TGF-beta signaling. Proc. Natl Acad. Sci. USA 104, 3460–3465 (2007).
Maeda, H., Tsukigawa, K. & Fang, J. A retrospective 30 years after discovery of the enhanced permeability and retention effect of solid tumors: next-generation chemotherapeutics and photodynamic therapyproblems, solutions, and prospects. Microcirculation 23, 173–182 (2016).
Ho-Tin-Noé, B., Demers, M. & Wagner, D. D. How platelets safeguard vascular integrity. J. Thromb. Haemost. 9, 56–65 (2011).
Ho-Tin-Noé, B., Goerge, T. & Wagner, D. D. Platelets: guardians of tumor vasculature. Cancer Res. 69, 5623–5626 (2009).
Kisucka, J. et al. Platelets and platelet adhesion support angiogenesis while preventing excessive hemorrhage. Proc. Natl Acad. Sci. USA 103, 855–860 (2006).
Gros, A. et al. Single platelets seal neutrophil-induced vascular breaches via GPVI during immune-complex–mediated inflammation in mice. Blood 126, 1017–1026 (2015).
Hillgruber, C. et al. Blocking neutrophil diapedesis prevents hemorrhage during thrombocytopenia. J. Exp. Med. 212, 1255–1266 (2015).
Boulaftali, Y. et al. Platelet ITAM signaling is critical for vascular integrity in inflammation. J. Clin. Invest. 123, 908–916 (2013).
Ho-Tin-Noé, B. et al. Platelet granule secretion continuously prevents intratumor hemorrhage. Cancer Res. 68, 6851–6858 (2008).
Demers, M. et al. Increased efficacy of breast cancer chemotherapy in thrombocytopenic mice. Cancer Res. 71, 1540–1549 (2011).
Demers, M. & Wagner, D. D. Targeting platelet function to improve drug delivery. Oncoimmunology 1, 100–102 (2012).
Bergmeier, W. et al. Structural and functional characterization of the mouse von Willebrand factor receptor GPIb-IX with novel monoclonal antibodies. Blood 95, 886–893 (2000).
Nieswandt, B. et al. Identification of critical antigen-specific mechanisms in the development of immune thrombocytopenic purpura in mice. Blood 96, 2520–2527 (2000).
Brooks, P. C. et al. Localization of matrix metalloproteinase MMP-2 to the surface of invasive cells by interaction with integrin αvβ3. Cell 85, 683–693 (1996).
Gref, R. et al. Biodegradable long-circulating polymeric nanospheres. Science 263, 1600–1603 (1994).
Owens, D. E. & Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Nam, Y. S. et al. New micelle-like polymer aggregates made from PEI–PLGA diblock copolymers: micellar characteristics and cellular uptake. Biomaterials 24, 2053–2059 (2003).
Yu, K. et al. Enhanced delivery of Paclitaxel using electrostatically-conjugated Herceptin-bearing PEI/PLGA nanoparticles against HER-positive breast cancer cells. Int. J. Pharm. 497, 78–87 (2016).
Li, F. et al. Multiple layer-by-layer lipid-polymer hybrid nanoparticles for improved FOLFIRINOX chemotherapy in pancreatic tumor models. Adv. Funct. Mater. 25, 788–798 (2015).
Wang, S., Huang, P. & Chen, X. Hierarchical targeting strategy for enhanced tumor tissue accumulation/retention and cellular internalization. Adv. Mater. 28, 7340–7364 (2016).
Ji, T. et al. An MMP-2 responsive liposome integrating antifibrosis and chemotherapeutic drugs for enhanced drug perfusion and efficacy in pancreatic cancer. ACS Appl. Mater. Interfaces 8, 3438–3445 (2016).
Penate Medina, O. et al. Liposomal tumor targeting in drug delivery utilizing MMP-2- and MMP-9-binding ligands. J. Drug Deliv. 2011, 160515 (2011).
MacKay, A. J. et al. Self-assembling chimeric polypeptide-doxorubicin conjugate nanoparticles that abolish tumours after a single injection. Nat. Mater. 8, 993–999 (2009).
Wang, H., Wu, Y., Zhao, R. & Nie, G. Engineering the assemblies of biomaterial nanocarriers for delivery of multiple theranostic agents with enhanced antitumor efficacy. Adv. Mater. 25, 1616–1622 (2013).
Jang, J. H. et al. Enhancement of the cancer targeting specificity of buforin IIb by fusion with an anionic peptide via a matrix metalloproteinases-cleavable linker. Peptides 32, 895–899 (2011).
Zhang, L., Kharbanda, S., McLeskey, S. W. & Kern, F. G. Overexpression of fibroblast growth factor 1 in MCF-7 breast cancer cells facilitates tumor cell dissemination but does not support the development of macrometastases in the lungs or lymph nodes. Cancer Res. 59, 5023–5029 (1999).
Mahler, M., Miyachi, K., Peebles, C. & Fritzler, M. J. The clinical significance of autoantibodies to the proliferating cell nuclear antigen (PCNA). Autoimmun. Rev. 11, 771–775 (2012).
Mailand, N., Gibbs-Seymour, I. & Bekker-Jensen, S. Regulation of PCNA-protein interactions for genome stability. Nat. Rev. Mol. Cell Biol. 14, 269–282 (2013).
Zhao, T. et al. A transistor-like pH nanoprobe for tumour detection and image-guided surgery. Nat. Biomed. Eng 1, 0006 (2016).
Hu, Q. et al. Relay drug delivery for amplifying targeting signal and enhancing anticancer efficacy. Adv. Mater. 29, 1605803 (2017).
Gabizon, A., Shiota, R. & Papahadjopoulos, D. Pharmacokinetics and tissue distribution of doxorubicin encapsulated in stable liposomes with long circulation times. J. Natl Cancer Inst. 81, 1484–1488 (1989).
Kunjachan, S. et al. Nanoparticle mediated tumor vascular disruption: a novel strategy in radiation therapy. Nano Lett. 15, 7488–7496 (2015).
Zhen, M. et al. A highly efficient and tumor vascular-targeting therapeutic technique with size-expansible gadofullerene nanocrystals. Sci. China Mater. 58, 799–810 (2015).
Acknowledgements
The authors thank the cyclotron teams from the Department of Nuclear Medicine, Peking University Cancer Hospital and Institute for the 64Cu production. This work was supported by grants from the National Basic Research Plan of China (2016YFA0201600 and 2013CB932701), National Distinguished Young Scientist Program (31325010), Innovation Group of the National Natural Science Foundation of China (11621505), National Natural Science Foundation of China (31200752, 31661130152, 31470969, 21373067 and 51673051), Frontier Research Program of the Chinese Academy of Sciences (QYZDJ-SSW-SLH022), Beijing Municipal Science and Technology Commission (Z161100000116035), Beijing Nova Program (Z171100001117010), Beijing Natural Science Foundation (7172164) and Youth Innovation Promotion Association of the Chinese Academy of Sciences (2017056).
Author information
Authors and Affiliations
Contributions
S.Li, Y.Zhang and G.N. conceived and designed the experiments. J.Wang, S.Li, Y.Zhang, Y.Zhao, T.J., F.L., Xiao Zhao, X.Y., S.Liu and J.L. performed the experiments. S.Li, Y.Zhang, J.Wang, Y.Zhao, Y.D., Xiaozheng Zhao, R.Z. and G.N. collected and analysed the data. A.K.W., G.J.A., J.Wei and Z.L. provided suggestions and technical support on the project and revised the manuscript. G.N. supervised the project. S.Li, J.Wang, Y.Zhang, Y.Zhao and G.N. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corrected online: Author correction 2 August 2017
A correction to this article is available online at https://doi.org/10.1038/s41551-017-0125-6.
Electronic supplementary material
Supplementary Information
Supplementary figures and tables (PDF 4.91 MB)
Rights and permissions
About this article
Cite this article
Li, S., Zhang, Y., Wang, J. et al. Nanoparticle-mediated local depletion of tumour-associated platelets disrupts vascular barriers and augments drug accumulation in tumours. Nat Biomed Eng 1, 667–679 (2017). https://doi.org/10.1038/s41551-017-0115-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-017-0115-8
This article is cited by
-
The dynamic role of platelets in cancer progression and their therapeutic implications
Nature Reviews Cancer (2024)
-
Anti-lymphangiogenesis for boosting drug accumulation in tumors
Signal Transduction and Targeted Therapy (2024)
-
Platelets for cancer treatment and drug delivery
Clinical and Translational Oncology (2022)
-
Responsive and activable nanomedicines for remodeling the tumor microenvironment
Nature Protocols (2021)
-
Multifunctional biomolecule nanostructures for cancer therapy
Nature Reviews Materials (2021)