Abstract
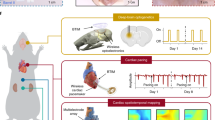
Advanced capabilities in electrical recording are essential for the treatment of heart-rhythm diseases. The most advanced technologies use flexible integrated electronics; however, the penetration of biological fluids into the underlying electronics and any ensuing electrochemical reactions pose significant safety risks. Here, we show that an ultrathin, leakage-free, biocompatible dielectric layer can completely seal an underlying array of flexible electronics while allowing for electrophysiological measurements through capacitive coupling between tissue and the electronics, without the need for direct metal contact. The resulting current-leakage levels and operational lifetimes are, respectively, four orders of magnitude smaller and between two and three orders of magnitude longer than those of other flexible-electronics technologies. Systematic electrophysiological studies with normal, paced and arrhythmic conditions in Langendorff hearts highlight the capabilities of the capacitive-coupling approach. These advances provide realistic pathways towards the broad applicability of biocompatible, flexible electronic implants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Thomas, C., Springer, P., Loeb, G., Berwald-Netter, Y. & Okun, L. A miniature microelectrode array to monitor the bioelectric activity of cultured cells. Exp. Cell Res. 74, 61–66 (1972).
Pertsov, A. M., Davidenko, J. M., Salomonsz, R., Baxter, W. T. & Jalife, J. Spiral waves of excitation underlie reentrant activity in isolated cardiac muscle. Circ. Res. 72, 631–650 (1993).
Sprössler, C., Denyer, M., Britland, S., Knoll, W. & Offenhäusser, A. Electrical recordings from rat cardiac muscle cells using field-effect transistors. Phys. Rev. E 60, 2171–2176 (1999).
Camelliti, P. et al. Adult human heart slices are a multicellular system suitable for electrophysiological and pharmacological studies. J. Mol. Cell. Cardiol. 51, 390–398 (2011).
Huys, R. et al. Single-cell recording and stimulation with a 16k micro-nail electrode array integrated on a 0.18 μm CMOS chip. Lab Chip 12, 1274–1280 (2012).
Zhang, X., Tai, J., Park, J. & Tai, Y.-C. Flexible MEA for adult zebrafish ECG recording covering both ventricle and atrium. In Proc. IEEE 27th Int. Conf. Micro Electro Mechanical Systems (MEMS) 841-844 (IEEE, 2014).
Friedman, P. A. Novel mapping techniques for cardiac electrophysiology. Heart 87, 575–582 (2002).
Kim, D.-H. et al. Materials for multifunctional balloon catheters with capabilities in cardiac electrophysiological mapping and ablation therapy. Nat. Mater. 10, 316–323 (2011).
Kim, D.-H. et al. Electronic sensor and actuator webs for large-area complex geometry cardiac mapping and therapy. Proc. Natl Acad. Sci. USA 109, 19910–19915 (2012).
Xu, L. et al. 3D multifunctional integumentary membranes for spatiotemporal cardiac measurements and stimulation across the entire epicardium. Nat. Commun. 5, 3329 (2014).
Viventi, J. et al. A conformal, bio-interfaced class of silicon electronics for mapping cardiac electrophysiology. Sci. Transl. Med. 2, 24ra22 (2010).
Viventi, J. et al. Flexible, foldable, actively multiplexed, high-density electrode array for mapping brain activity in vivo. Nat. Neurosci. 14, 1599–1605 (2011).
Laks, M. M., Arzbaecher, R., Bailey, J. J., Geselowitz, D. B. & Berson, A. S. Recommendations for safe current limits for electrocardiographs a statement for healthcare professionals from the Committee on Electrocardiography, American Heart Association. Circulation 93, 837–839 (1996).
Swerdlow, C. D. et al. Cardiovascular collapse caused by electrocardiographically silent 60-Hz intracardiac leakage current implications for electrical safety. Circulation 99, 2559–2564 (1999).
Beech, I. B. & Sunner, J. Biocorrosion: towards understanding interactions between biofilms and metals. Curr. Opin. Biotechnol. 15, 181–186 (2004).
Bowman, L. & Meindl, J. D. The packaging of implantable integrated sensors. IEEE Trans. Biomed. Eng. 33, 248–255 (1986).
Liu, X. et al. Stability of the interface between neural tissue and chronically implanted intracortical microelectrodes. IEEE Trans. Rehab. Eng. 7, 315–326 (1999).
Bazaka, K. & Jacob, M. V. Implantable devices: issues and challenges. Electronics 2, 1–34 (2012).
Someya, T. et al. Conformable, flexible, large-area networks of pressure and thermal sensors with organic transistor active matrixes. Proc. Natl Acad. Sci. USA 102, 12321–12325 (2005).
Lacour, S. P., Jones, J., Wagner, S., Li, T. & Suo, Z. Stretchable interconnects for elastic electronic surfaces. Proc. IEEE 93, 1459–1467 (2005).
Tian, B. et al. Three-dimensional, flexible nanoscale field-effect transistors as localized bioprobes. Science 329, 830–834 (2010).
Takei, K. et al. Nanowire active-matrix circuitry for low-voltage macroscale artificial skin. Nat. Mater. 9, 821–826 (2010).
Schwartz, G. et al. Flexible polymer transistors with high pressure sensitivity for application in electronic skin and health monitoring. Nat. Commun. 4, 1859 (2013).
Wu, W., Wen, X. & Wang, Z. L. Taxel-addressable matrix of vertical-nanowire piezotronic transistors for active and adaptive tactile imaging. Science 340, 952–957 (2013).
Khodagholy, D. et al. NeuroGrid: recording action potentials from the surface of the brain. Nat. Neurosci. 18, 310–315 (2015).
Fromherz, P., Offenhäusser, A., Vetter, T. & Weis, J. A neuron-silicon junction: a Retzius cell of the leech on an insulated-gate field-effect transistor. Science 252, 1290–1293 (1991).
Zeck, G. & Fromherz, P. Noninvasive neuroelectronic interfacing with synaptically connected snail neurons immobilized on a semiconductor chip. Proc. Natl Acad. Sci. USA 98, 10457–10462 (2001).
Chi, Y. M., Jung, T.-P. & Cauwenberghs, G. Dry-contact and noncontact biopotential electrodes: methodological review. IEEE Rev. Biomed. Eng. 3, 106–119 (2010).
Spira, M. E. & Hai, A. Multi-electrode array technologies for neuroscience and cardiology. Nat. Nanotech. 8, 83–94 (2013).
Berdondini, L. et al. Active pixel sensor array for high spatio-temporal resolution electrophysiological recordings from single cell to large scale neuronal networks. Lab Chip 9, 2644–2651 (2009).
Eversmann, B. et al. A 128× 128 CMOS biosensor array for extracellular recording of neural activity. IEEE J. Solid-State Circ. 38, 2306–2317 (2003).
Bakkum, D. J. et al. Tracking axonal action potential propagation on a high-density microelectrode array across hundreds of sites. Nat. Commun. 4, 21821 (2013).
Byers, C. L., Beazell, J. W., Schulman, J. H. & Rostami, A. Hermetically sealed ceramic and metal package for electronic devices implantable in living bodies. US patent US4991582 A (1991).
Zeng, F.-G., Rebscher, S., Harrison, W., Sun, X. & Feng, H. Cochlear implants: system design, integration, and evaluation. IEEE Rev. Biomed. Eng. 1, 115–142 (2008).
Sillay, K. A., Larson, P. S. & Starr, P. A. Deep brain stimulator hardware-related infections: incidence and management in a large series. Neurosurgery 62, 360–367 (2008).
Jeong, J. W. et al. Capacitive epidermal electronics for electrically safe, long-term electrophysiological measurements. Adv. Health. Mater. 3, 642–648 (2014).
Duan, X. et al. Quantification of the affinities and kinetics of protein interactions using silicon nanowire biosensors. Nat. Nanotech. 7, 401–407 (2012).
Fattahi, P., Yang, G., Kim, G. & Abidian, M. R. A review of organic and inorganic biomaterials for neural interfaces. Adv. Mater. 26, 1846–1885 (2014).
Langendorff, O. Untersuchungen am überlebenden Säugethierherzen. Pflügers Archiv Eur. J. Physiol. 61, 291–332 (1895).
Efimov, I. R., Nikolski, V. P. & Salama, G. Optical imaging of the heart. Circ. Res. 95, 21–33 (2004).
Bossaert, L. Fibrillation and defibrillation of the heart. Br. J. Anaesth. 79, 203–213 (1997).
Efimov, I. R., Cheng, Y., Van Wagoner, D. R., Mazgalev, T. & Tchou, P. J. Virtual electrode-induced phase singularity a basic mechanism of defibrillation failure. Circ. Res. 82, 918–925 (1998).
Rogers, J. M. Combined phase singularity and wavefront analysis for optical maps of ventricular fibrillation. IEEE Trans. Biomed. Eng. 51, 56–65 (2004).
Narayan, S. M. et al. Treatment of atrial fibrillation by the ablation of localized sources: CONFIRM (conventional ablation for atrial fibrillation with or without focal impulse and rotor modulation) trial. J. Am. Coll. Cardiol. 60, 628–636 (2012).
Lim, H. S. et al. Noninvasive mapping to guide atrial fibrillation ablation. Cardiac Electrophysiol. Clinics 7, 89–98 (2015).
Bray, M. A., Lin, S. F., Aliev, R. R., Roth, B. J. & Wikswo, J. P. Experimental and theoretical analysis of phase singularity dynamics in cardiac tissue. J. Cardiovasc. Electrophysiol. 12, 716–722 (2001).
Onuki, Y., Bhardwaj, U., Papadimitrakopoulos, F. & Burgess, D. J. A review of the biocompatibility of implantable devices: current challenges to overcome foreign body response. J. Diabetes Sci. Technol. 2, 1003–1015 (2008).
Ward, W. K. A review of the foreign-body response to subcutaneously-implanted devices: the role of macrophages and cytokines in biofouling and fibrosis. J. Diabetes Sci. Technol. 2, 768–777 (2008).
Morais, J. M., Papadimitrakopoulos, F. & Burgess, D. J. Biomaterials/tissue interactions: possible solutions to overcome foreign body response. AAPS J. 12, 188–196 (2010).
Vegas, A. J. et al. Combinatorial hydrogel library enables identification of materials that mitigate the foreign body response in primates. Nat. Biotechnol. 34, 345–352 (2016).
Hibbitt, H., Karlsson, B. & Sorensen, P. Abaqus analysis user’s manual v.6.10 (Dassault Systèmes Simulia Corp, 2011).
Shi, Y., Rogers, J. A., Gao, C. & Huang, Y. Multiple neutral axes in bending of a multiple-layer beam with extremely different elastic properties. J. Appl. Mech. 81, 114501 (2014).
Li, L. et al. Integrated flexible chalcogenide glass photonic devices. Nat. Photon. 8, 643–649 (2014).
Su, Y., Li, S., Li, R. & Dagdeviren, C. Splitting of neutral mechanical plane of conformal, multilayer piezoelectric mechanical energy harvester. Appl. Phys. Lett. 107, 041905 (2015).
Fang, H. et al. Dataset for ‘Capacitively coupled arrays of multiplexed flexible silicon transistors for long-term cardiac electrophysiology’. figsharehttps://figshare.com/s/961786fcede5a8703ec5 (2017).
Acknowledgements
This work is supported by the NIH grants R01 HL115415, R01 HL114395 and R21 HL112278, and through the Frederick Seitz Materials Research Laboratory and Center for Microanalysis of Materials at the University of Illinois at Urbana-Champaign. We would like to thank the Micro and Nanotechnology Laboratory and the School of Chemical Sciences Machine Shop at the University of Illinois for help on the device fabrication. J.Z. acknowledges support from a Louis J. Larson Fellowship, Swiegert Fellowship, and H. C. Ting Fellowship from the University of Illinois, Urbana-Champaign. M.T. and J.V. acknowledge the support from the National Science Foundation award CCF 1422914. C.-H.C. and J.V. acknowledge the support from the Army Research Office award W911NF-14-1-0173.
Author information
Authors and Affiliations
Contributions
H.F., K.J.Y., C.G., Z.Y., I.R.E. and J.A.R. designed the research; H.F., K.J.Y., Z.Y., E.S., C.-H.C., J.Z., S.X., S.M.W., Y.Z., S.W.H., D.X. and S.W.C. fabricated the devices and electronics; H.F., C.G., Z.Y. and J.Z. carried out animal experiments; H.F., K.J.Y., C.G., Z.Y., C.-H.C., J.Z., M.T., J.V., G.C. and M.K. performed data analysis; H.F., Z.Y., Y.X. and Y.H. contributed to mechanical simulations; H.F., K.J.Y., C.G., Z.Y., I.R.E. and J.A.R. co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary notes and figures. (PDF 2974 kb)
Supplementary Video 1
A flexible capacitively coupled sensing electronic system on a Langendorff-perfused rabbit heart model. (MP4 4793 kb)
Supplementary Video 2
Voltage data from all electrodes, illustrating the activation pattern of the heart during sinus rhythm. (MP4 6160 kb)
Supplementary Video 3
Voltage data from all electrodes, illustrating the paced activation pattern moving from the apex to the base. (MP4 5493 kb)
Supplementary Video 4
Voltage data from all electrodes, illustrating the activation pattern of the heart during ventricular fibrillation. (MP4 14134 kb)
Rights and permissions
About this article
Cite this article
Fang, H., Yu, K., Gloschat, C. et al. Capacitively coupled arrays of multiplexed flexible silicon transistors for long-term cardiac electrophysiology. Nat Biomed Eng 1, 0038 (2017). https://doi.org/10.1038/s41551-017-0038
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-017-0038
This article is cited by
-
Isolated Perfused Hearts for Cardiovascular Research: An Old Dog with New Tricks
Journal of Cardiovascular Translational Research (2024)
-
Bionic artificial skin with a fully implantable wireless tactile sensory system for wound healing and restoring skin tactile function
Nature Communications (2024)
-
A soft, high-density neuroelectronic array
npj Flexible Electronics (2023)
-
Recent advances in flexible noninvasive electrodes for surface electromyography acquisition
npj Flexible Electronics (2023)
-
Highly conductive tissue-like hydrogel interface through template-directed assembly
Nature Communications (2023)