Abstract
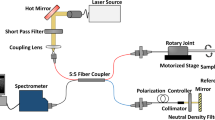
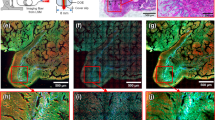
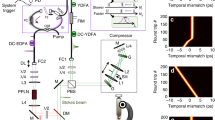
The complex nature of atherosclerosis demands high-resolution approaches to identify subtle thrombogenic lesions and define the risk of plaque rupture. Here, we report the proof-of-concept use of a multimodal scanning fibre endoscope (SFE) consisting of a single optical fibre scanned by a piezoelectric drive that illuminates tissue with red, blue and green laser beams, and that digitally reconstructs images at 30 Hz with high resolution and large fields of view. By combining laser-induced reflectance and fluorescence emission of intrinsic fluorescent constituents in arterial tissues, the SFE allowed us to co-generate endoscopic videos with a label-free biochemical map to derive a morphological and spectral classifier capable of discriminating early, intermediate, advanced and complicated atherosclerotic plaques. We demonstrate the capability of scanning fibre angioscopy for the molecular imaging of vulnerable atherosclerosis by targeting proteolytic activity with a fluorescent probe activated by matrix metalloproteinases. We also show that the SFE generates high-quality spectral images in vivo in an animal model with medium-sized arteries. Multimodal laser-based angioscopy could become a platform for the diagnosis, prognosis, and image-guided therapy of atherosclerosis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Estol, C. J. Dr C. Miller Fisher and the history of carotid artery disease. Stroke 27, 559–566 (1996).
North American Symptomatic Carotid Endarterectomy Trial Collaborators (NASCET). Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N. Engl. J. Med. 325, 445–453 (1991).
Boyle, J. J. Macrophage activation in atherosclerosis: pathogenesis and pharmacology of plaque rupture. Curr. Vasc. Pharmacol. 3, 63–68 (2005).
Howard, D. P. et al. Symptomatic carotid atherosclerotic disease: correlations between plaque composition and ipsilateral stroke risk. Stroke 46, 182–189 (2015).
Ross, R. Atherosclerosis — an inflammatory disease. N. Engl. J. Med. 340, 115–126 (1999).
Falk, E., Shah, P. K. & Fuster, V. Coronary plaque disruption. Circulation 92, 657–671 (1995).
Fisher, M. et al. Carotid plaque pathology: thrombosis, ulceration, and stroke pathogenesis. Stroke 36, 253–257 (2005).
Sloop, G. D. Atherosclerosis — an inflammatory disease. N. Engl. J. Med. 340, 1928–1929 (1999).
Brinjikji, W. et al. Contemporary carotid imaging: from degree of stenosis to plaque vulnerability. J. Neurosurg. 124, 27–42 (2016).
Edwards, J. H., Kricheff, II, Riles, T. & Imparato, A. Angiographically undetected ulceration of the carotid bifurcation as a cause of embolic stroke. Radiology 132, 369–373 (1979).
Saam, T., et al. The vulnerable, or high-risk, atherosclerotic plaque: noninvasive MR imaging for characterization and assessment. Radiology 244, 64–77 (2007).
ten Kate, G. L. et al. Noninvasive imaging of the vulnerable atherosclerotic plaque. Curr. Probl. Cardiol. 35, 556–591 (2010).
Mulder, W. J., Jaffer, F. A., Fayad, Z. A. & Nahrendorf, M. Imaging and nanomedicine in inflammatory atherosclerosis. Sci. Transl. Med. 6, 3005101 (2014).
Sanz, J. & Fayad, Z. A. Imaging of atherosclerotic cardiovascular disease. Nature 451, 953–957 (2008).
Lee, C. M. et al. Scanning fiber endoscopy with highly flexible, 1 mm catheterscopes for wide-field, full-color imaging. J. Biophoton. 3, 385–407 (2010).
Tarkin, J. M., Joshi, F. R. & Rudd, J. H. PET imaging of inflammation in atherosclerosis. Nat. Rev. Cardiol. 11, 443–457 (2014).
Krejza, J. et al. Carotid artery diameter in men and women and the relation to body and neck size. Stroke 37, 1103–1105 (2006).
Lenglet, S. et al. Molecular imaging of matrix metalloproteinases in atherosclerotic plaques. Thromb. Haemost. 107, 409–416 (2012).
Tarkin, J. M. et al. Imaging atherosclerosis. Circ. Res. 118, 750–769 (2016).
Virmani, R., Ladich, E. R., Burke, A. P. & Kolodgie, F. D. Histopathology of carotid atherosclerotic disease. Neurosurgery 59, S3–S13 (2006).
Ishibashi, F., Aziz, K., Abela, G. S. & Waxman, S. Update on coronary angioscopy: review of a 20-year experience and potential application for detection of vulnerable plaque. J. Interv. Cardiol. 19, 17–25 (2006).
Mizuno, K. & Takano, M. Coronary Angioscopy (Springer, 2015).
Tanemura, H. et al. Angioscopic observation during carotid angioplasty with stent placement. Am. J. Neuroradiol. 26, 1943–1948 (2005).
Uchida, Y. Coronary Angioscopy (Wiley, 2001).
White, J. V. & Eid, I. Diagnostic and interventional angioscopy. Surg. Clin. North. Am. 78, 539–559 (1998).
Uchida, Y. et al. Detection of vulnerable coronary plaques by color fluorescent angioscopy. Cardiovasc. Imaging 3, 398–408 (2010).
Uchida, Y. Recent advances in coronary angioscopy. J. Cardiol. 57, 18–30 (2011).
Arakawa, K. et al. Fluorescence analysis of biochemical constituents identifies atherosclerotic plaque with a thin fibrous cap. Arterioscler. Thromb. Vasc. Biol. 22, 1002–1007 (2002).
Marcu, L. et al. Detection of rupture-prone atherosclerotic plaques by time-resolved laser-induced fluorescence spectroscopy. Atherosclerosis 204, 156–164 (2009).
Bartorelli, A. L. et al. In vivo human atherosclerotic plaque recognition by laser-excited fluorescence spectroscopy. J. Am. Coll. Cardiol. 17, 160B–168B (1991).
Otsuka, F. et al. Clinical classification of plaque morphology in coronary disease. Nat. Rev. Cardiol. 11, 379–389 (2014).
Tearney, G. J. et al. Three-dimensional coronary artery microscopy by intracoronary optical frequency domain imaging. Cardiovasc. Imaging 1, 752–761 (2008).
van Soest, G., et al. Pitfalls in plaque characterization by OCT: image artifacts in native coronary arteries. Cardiovasc. Imaging 4, 810–813 (2011).
Farooq, V. et al. New insights into the coronary artery bifurcation hypothesis-generating concepts utilizing 3-dimensional optical frequency domain imaging. Cardiovasc. Interv. 4, 921–931 (2011).
Okamura, T., Fujimura, T. & Yano, M. Three-dimensional reconstruction of optical coherence tomography for improving bifurcation stenting. J. Cardiol. Cases 13, 137–138 (2016).
Iakovou, I. et al. Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 293, 2126–2130 (2005).
McVeigh, P. Z. et al. High-resolution angioscopic imaging during endovascular neurosurgery. Neurosurgery 75, 171–180 (2014).
Wiethoff, A. et al. Molecular imaging of thrombosis. Curr. Cardiovasc Imag. Rep. 3, 34–41 (2010).
Bang, O. Y. et al. Frequency and mechanisms of stroke recurrence after cryptogenic stroke. Ann. Neurol. 54, 227–234 (2003).
Joshi, B. P. et al. Multispectral endoscopic imaging of colorectal dysplasia in vivo. Gastroenterology 143, 1435–1437 (2012).
Finn, A. V. et al. Concept of vulnerable/unstable plaque. Arterioscler. Thromb. Vasc. Biol. 30, 1282–1292 (2010).
Newby, A. C. Metalloproteinases and vulnerable atherosclerotic plaques. Trends Cardiovasc. Med. 17, 253–258 (2007).
Bourantas, C. V. et al. Hybrid intravascular imaging: recent advances, technical considerations, and current applications in the study of plaque pathophysiology. Eur. Heart J. http://dx.doi.org/10.1093/eurheartj/ehw097 (2016).
Jaffer, F. A. & Libby, P. in Imaging Coronary Atherosclerosis (ed. Nicholls, S. J. ) 187–202 (Springer, 2014).
Ughi, G. J. et al. Clinical characterization of coronary atherosclerosis with dual-modality OCT and near-infrared autofluorescence imaging. Cardiovasc. Imaging 3, 00038–00033 (2016).
Arbab-Zadeh, A. & Fuster, V. The myth of the “vulnerable plaque”: transitioning from a focus on individual lesions to atherosclerotic disease burden for coronary artery disease risk assessment. J. Am. Coll. Cardiol. 65, 846–855 (2015).
Kim, D. E. et al. Protease imaging of human atheromata captures molecular information of atherosclerosis, complementing anatomic imaging. Arterioscler. Thromb. Vasc. Biol. 30, 449–456 (2010).
Kataiwa, H. et al. Safety and usefulness of non-occlusion image acquisition technique for optical coherence tomography. Circ. J. 72, 1536–1537 (2008).
Biamino, G. The excimer laser: science fiction fantasy or practical tool? J. Endovasc. Ther. 11, II207–II222 (2004).
Abela, G. S. et al. Laser recanalization of occluded atherosclerotic arteries in vivo and in vitro. Circulation 71, 403–411 (1985).
Karsch, K. R. & Haase, K. K. Coronary Laser Angioplasty: An Update (Springer-Verlag, 1991).
Leon, M. B. et al. Human arterial surface fluorescence: atherosclerotic plaque identification and effects of laser atheroma ablation. J. Am. Coll. Cardiol. 12, 94–102 (1988).
Kirshenbaum, M. R. & Seibel, E. J. Delivery of single-mode and multi-mode therapeutic laser light using a single and dual cladding optical fiber for a scanning fiber endoscope. Proc. SPIE 7894, 789413 (2011).
Woldetensae, M. H. et al. Fluorescence image-guided photodynamic therapy of cancer cells using a scanning fiber endoscope. Proc. SPIE 8576, 85760L (2013).
Deckelbaum, L. I. in Lasers in Cardiovascular Medicine and Surgery: Fundamentals and Techniques (ed. Abela, G. S. ) 153–165 (Kluwer, 1990).
Dippel, E. J. et al. Randomized controlled study of excimer laser atherectomy for treatment of femoropopliteal in-stent restenosis: initial results from the EXCITE ISR trial (EXCImer Laser Randomized Controlled Study for Treatment of FemoropopliTEal In-Stent Restenosis). Cardiovasc. Interv. 8, 92–101 (2015).
Fifi, J. T. et al. Complications of modern diagnostic cerebral angiography in an academic medical center. J. Vasc. Interv. Radiol. 20, 442–447 (2009).
Crouse, J. R. et al. Risk factors for extracranial carotid artery atherosclerosis. Stroke 18, 990–996 (1987).
Yang, C. et al. Target-to-background enhancement in multispectral endoscopy with background autofluorescence mitigation for quantitative molecular imaging. J. Biomed. Opt. 19, 076014 (2014).
Miller, S. J. et al. Targeted detection of murine colonic dysplasia in vivo with flexible multispectral scanning fiber endoscopy. J. Biomed. Opt. 17, 021103 (2012).
Zhang, L. et al. Trimodal detection of early childhood caries using laser light scanning and fluorescence spectroscopy: clinical prototype. J. Biomed. Opt. 18, 111412 (2013).
Yang, C., Hou, V., Nelson, L. Y. & Seibel, E. J. Color-matched and fluorescence-labeled esophagus phantom and its applications. J. Biomed. Opt. 18, 026020 (2013).
Wang, T. D. et al. Mathematical model of fluorescence endoscopic image formation. Appl. Opt. 37, 8103–8111 (1998).
Savastano, L. et al. Diagnostic and interventional optical angioscopy in ex vivo carotid arteries. Oper. Neurosurg. http://dx.doi.org/10.1093/ons/opw002 (2016).
Stary, H. C. et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 92, 1355–1374 (1995).
Virmani, R. et al. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 20, 1262–1275 (2000).
Acknowledgements
This work was supported by Cerebrovascular Research Award, Joint Section on Cerebrovascular Surgery of the American Association of Neurological Surgeons and Congress of Neurological Surgeons, and the National Institutes of Health (NIH) U54 CA163059 (E.J.S. and T.D.W.), R01 EB016457 (E.J.S.), R01 HL129778 and R01 HL117491 (Y.E.C.), and R01CA200007 (E.J.S. and T.D.W.). The authors thank D. French, C. Prescott, D. Griffiths and J. Jentzen for their expertise and technical assistance in obtaining human cadaveric specimens. We are also grateful to M. Foldenauer for artwork assistance, J. Diaz for his endovascular expertise, and H. Wagner for editorial assistance.
Author information
Authors and Affiliations
Contributions
L.E.S. designed and performed the experiments and wrote the manuscript. Q.Z., A.S., K.V., C.M.-Z., D.G., J.M. and L.Z. analysed and processed the data. M.M.W. contributed to the design of experiments. A.P. and B.G.T. contributed to the design of experiments and provided surgical specimens. J.X., J.Z. and Y.E.C. contributed with the animal model and experiments. E.J.S. developed SFE technology and contributed to preparation of the manuscript. T.D.W. contributed to experiment design and manuscript preparation, and supervised the overall project. All authors read and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.J.S. participates in royalty sharing with his employer, the University of Washington, which has ownership of patents that may gain or lose financially through this publication. The remaining authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures, tables and video legends. (PDF 407 kb)
Supplementary Video 1
Ex vivo spectral video angioscopy of a normal artery. (MP4 17698 kb)
Supplementary Video 2
Ex vivo spectral video angioscopy of an early lesion. (MP4 20672 kb)
Supplementary Video 3
Ex vivo spectral video angioscopy of an intermediate lesion. (MP4 18921 kb)
Supplementary Video 4
Ex vivo spectral video angioscopy of an advanced lesion. (MP4 21564 kb)
Supplementary Video 5
Ex vivo spectral video angioscopy of complicated plaques. (MP4 22770 kb)
Supplementary Video 6
In vivo spectral video angioscopy. (MP4 25427 kb)
Rights and permissions
About this article
Cite this article
Savastano, L., Zhou, Q., Smith, A. et al. Multimodal laser-based angioscopy for structural, chemical and biological imaging of atherosclerosis. Nat Biomed Eng 1, 0023 (2017). https://doi.org/10.1038/s41551-016-0023
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-016-0023