Abstract
The Global Initiative for Asthma recommends a stepwise approach to adjust asthma treatment to the needs of individual patients; inhaled corticosteroids (ICS) remain the core pharmacological treatment. However, many patients remain poorly controlled, and evidence-based algorithms to decide on the best order and rationale for add-on therapies are lacking. We explore the challenges of asthma management in primary care and review outcomes from randomised controlled trials and meta-analyses comparing the long-acting muscarinic antagonist (LAMA) tiotropium with long-acting β2-agonists (LABAs) or leukotriene receptor antagonists (LTRAs) as add-on to ICS in patients with asthma. In adults, LAMAs and LABAs provide a greater improvement in lung function than LTRAs as add-on to ICS. In children, results were positive and comparable between therapies, but data are scarce. This information could aid decision-making in primary care, supporting the use of add-on therapy to ICS to help improve lung function, control asthma symptoms and prevent exacerbations.
Similar content being viewed by others
Introduction
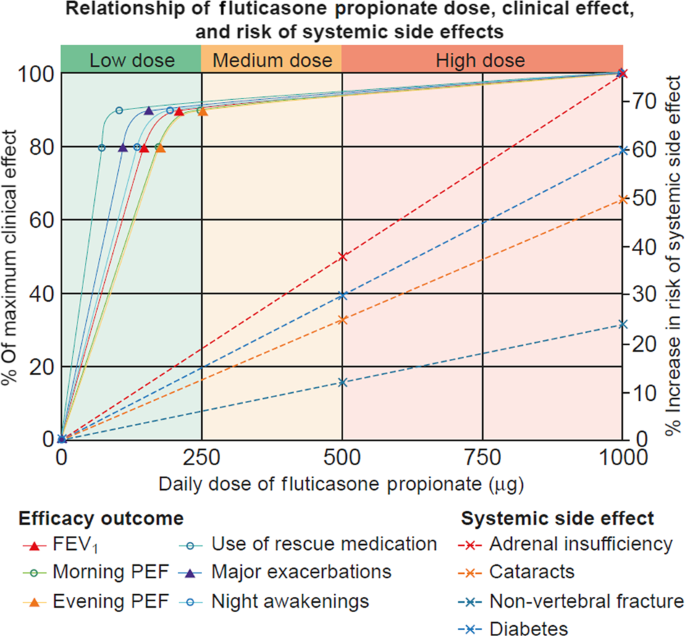
Asthma is a serious global health issue that affects all age groups, with a reported 339 million sufferers worldwide, presenting a number of challenges for primary care physicians1. Many patients with asthma remain symptomatic, despite treatment, for multiple different reasons2,3,4,5,6. It has been suggested that patients may overestimate and thus inaccurately report their level of disease control, because they accept and tolerate a certain level of symptoms, assuming them to be an inevitable consequence of asthma7,8. Physicians may underestimate the prevalence and severity of symptoms and overestimate the degree to which the patient’s asthma is controlled, meaning the patient may not receive adequate medication to achieve control of their disease2,7. Reducing asthma symptoms and future risk through correct add-on therapy and management in patients who remain uncontrolled despite treatment is a major challenge for those working in both secondary and primary care. The Global Initiative for Asthma (GINA) strategy recommends a stepwise approach to asthma management in order to achieve symptom control and prevent future risks, including exacerbations, loss of lung function, and side effects of medication (Fig. 1)9. Inhaled corticosteroids (ICS) are considered an effective long-term controller treatment in the management of asthma10. However, if asthma remains uncontrolled despite medium-dose ICS, increasing the dose of ICS may not be appropriate due to an increased risk of local and systemic side effects and variation in individual ICS dose-responsiveness between patients. In addition, most of the clinical benefit of ICS use is seen at low doses. Add-on treatments may therefore be required9. In addition, evidence suggests that the ICS dose–response curve is relatively flat, with 80–90% of the maximum achievable therapeutic effect in adult asthma obtained at 200–250 μg of fluticasone propionate or equivalent (Fig. 2); therefore, addition of an add-on therapy may be considered to be a more effective and safer treatment strategy11,12,13.
© 2020, Global Initiative for Asthma, reproduced with permission. FEV1 forced expiratory volume in 1s, GINA Global Initiative for Asthma, ICS inhaled corticosteroid, Ig immunoglobulin, IL interleukin, LABA long-acting β2-agonist, LTRA leukotriene receptor antagonist, OCS oral corticosteroid, SABA short-acting β2-agonist.
Reprinted with permission from the American Thoracic Society. Copyright © 2020 American Thoracic Society. Beasley et al.12. FEV1 forced expiratory volume in 1 s, PEF peak expiratory flow.
Long-acting β2-agonist (LABA; e.g. salmeterol, formoterol, vilanterol, indicaterol14,15,16), leukotriene receptor antagonist (LTRA; e.g. montelukast and zafirlukast [discontinued]15,17) and long-acting muscarinic antagonist (LAMA; tiotropium18 [the only LAMA currently indicated for use in patients with asthma]) add-on controller therapies have been shown to improve lung function and asthma control and reduce exacerbations in asthma patients, have safety profiles similar to placebo, and are currently indicated for use in patients with asthma. More information on these classes of agents, including indications and their modes of action, is detailed in Table 1.
With multiple add-on therapies available for the management of asthma, there have been several systematic reviews published that evaluate the efficacy and safety of add-on therapies compared with either placebo or another add-on therapy14,15,16,17,19,20. However, none compare LABA, LTRA and LAMA as add-on treatments to ICS in a single consolidated review, and there are no head-to-head trials evaluating all three treatments within the same trial. Here we systematically analyse and review the literature to explore the challenges of asthma management, the impact of poor asthma control on patients’ lives and compare outcomes from published studies. We examine the effect of three add-on treatments on lung function, asthma control, exacerbations and safety, with the aim of assisting primary care physicians in selecting the most appropriate add-on treatment to ICS.
Results
Search results
The literature search identified 14 relevant publications that met the inclusion criteria for this review: 2 Cochrane reviews and 12 additional randomised controlled trials (RCTs) that were not included within the Cochrane reviews.
-
The search strings for LABA studies generated 164 publications, of which 1 meta-analysis and 4 additional RCTs met the criteria for inclusion in this review16,21,22,23.
-
The search strings for LTRA studies generated 54 publications, of which 3 RCTs met the criteria for inclusion in this review24,25,26.
-
The search strings for LAMA studies generated 106 publications, of which 8 RCTs met the criteria for inclusion in this review21,22,27,28,29,30,31.
Asthma control in adult patients
LABA (salmeterol) significantly improved asthma control when added to ICS compared with placebo (measured by Asthma Control Questionnaire [ACQ])21. The LTRA (montelukast) did not have an effect on asthma control when compared with placebo (measured by ACQ) (Table 2)32. Data for LAMA (tiotropium) are more varied (Table 2)21,27,28,29,30,31. Paggiaro et al. reported that there was no difference of effect between tiotropium (5 µg and 2.5 µg) and placebo on ACQ score. Four papers compared the effect of LABAs directly with tiotropium. There was no significant difference between LABAs and tiotropium on asthma control as measured by ACQ (Table 2)21,33,34. One study included difference in Mini-Asthma Quality of Life Questionnaire (Mini-AQLQ) response scores as a secondary efficacy endpoint. At study endpoint at 16 weeks, salmeterol (50 µg) significantly improved overall Mini-AQLQ score compared with placebo, but there was no significant difference in response scores between the tiotropium (5 µg) and placebo groups. When directly compared, there was no difference in treatment response between salmeterol (50 µg) and tiotropium (5 µg) at study endpoint22. No studies compared asthma control, measured by ACQ, in LABA vs LTRA or tiotropium vs LTRA.
Lung function in adult patients
LABAs significantly improved forced expiratory volume in 1 s (FEV1), morning and evening peak expiratory flow (PEF) (salmeterol/formoterol) and forced vital capacity (FVC) (salmeterol) compared with placebo or ICS alone (Figs. 3–5)16,21,22,23. The literature reporting the effect of LTRAs as add-on to ICS on lung function is varied (Figs. 3–5), suggesting no beneficial effect of montelukast on FEV1 or FVC compared with ICS alone24. Results for the effect of montelukast on PEF are conflicting. Only one study of zafirlukast (now discontinued) was identified, with the authors reporting significant improvements in FEV1 and both morning and evening PEF compared with placebo26.
ALAACRC American Lung Association Asthma Clinical Research Centers, AUC area under curve, BID twice daily, CI confidence interval, FEV1 forced expiratory volume in 1 s, LABA long-acting β2-agonist, LAMA long-acting muscarinic agonist, LTRA leukotriene receptor antagonist, NR not reported, NS non-significant, QD once daily.
ALAACRC American Lung Association Asthma Clinical Research Centers, AUC area under curve, BID twice daily, CI confidence interval, FVC forced vital capacity, LABA long-acting β2-agonist, LAMA long-acting muscarinic agonist, LTRA leukotriene receptor antagonist, NR not reported, NS non-significant, QD once daily.
Of the seven available studies of LAMAs (tiotropium), all but two trial arms from two studies reported significant improvements in FEV1 (peak, trough and area under the curve [AUC]) and FVC compared with placebo (Figs. 3, 5)21,22,27,28,29,30,31. In all the published studies in adults21,22,27,28,29,30,31, tiotropium significantly improved morning and evening PEF compared with placebo or ICS alone, except for one tiotropium 2.5 µg trial arm, which reported no significant difference between tiotropium and placebo on trough PEF response (Fig. 4)29.
A systematic review comparing the use of LABAs with LTRAs as add-on to ICS reported that LABAs (salmeterol or formoterol) have a significantly greater effect on FEV1 and PEF compared with LTRAs (montelukast or zafirlukast) (Figs. 3, 4)15. There are no data available comparing the effect of LABAs and LTRAs on FVC. Four papers compared the effect of tiotropium with the LABAs salmeterol and formoterol on lung function parameters (Figs. 3, 4)33.
Exacerbations in adult patients
LABAs provided numerical improvement in the number of patients reporting at least one exacerbation compared with placebo (Table 2)21,22,23. Data for LTRAs (montelukast and zafirlukast [now discontinued]) are varied (Table 2). LAMA (tiotropium) both significantly and non-significantly reduced the number of patients experiencing at least one exacerbation (Table 2)27. A meta-analysis comparing LABA with LTRA reported a 2% reduction in risk of exacerbations in patients using LABA+ICS vs LTRA+ICS combination therapy (Table 2)15. Both tiotropium and LABAs had a comparable effect on the risk of exacerbations21,22,33,34.
Safety in adult patients
Overall, comparable proportions of patients report adverse events (AEs) with LABA, LTRA and tiotropium treatment as add-on to ICS with both placebo and with one another (Table 2)15,21,22,23,25,26,27,28,29,30,31,34.
Efficacy and safety of LABAs, LTRAs and LAMAs in paediatric patients
A recent systematic review by Vogelberg et al. compared the efficacy and safety of LABAs, LTRAs and LAMAs (tiotropium) in paediatric patients aged 4–17 years with asthma35. LABA treatment as add-on to ICS improved lung function when compared with placebo, as measured by FEV1 and FEV1 % predicted. There was no difference in risk of exacerbations requiring oral corticosteroid (OCS) between LABAs plus ICS compared with ICS alone, although it should be noted that not all trials were powered to assess exacerbations. The proportion of patients experiencing AEs or serious AEs (SAEs) with the addition of LABA to ICS was broadly similar35. An additional RCT of 512 patients aged 5–12 years with persistent asthma reported improvements in lung function and asthma control, and no differences in risk of exacerbations and AEs, in patients receiving LABAs (formoterol) compared with those receiving placebo as add-on to ICS36. However, in a systematic review comparing LABA plus ICS vs higher-dose ICS in children with asthma, combination therapy led to a trend towards an increased risk of oral steroid-treated exacerbations and hospital admissions16.
For LTRA (montelukast), a study by Simons et al. described a greater improvement from baseline FEV1 in patients receiving montelukast compared with placebo37. In addition, a systematic review found an improvement in baseline FEV1 and FEV1 % predicted in patients receiving ICS plus montelukast compared with those receiving ICS plus placebo, but these differences were not significant19. There was no difference between montelukast and placebo as add-on to ICS in the risk of exacerbations19. Limited available data suggest that the proportion of patients experiencing AEs with the addition of montelukast to ICS is comparable with those receiving placebo as add-on to ICS37.
Tiotropium improved FEV1 and FEV1 % predicted as add-on to ICS with or without additional controller therapies21,27,28,38,39,40,41,42. The proportion of paediatric patients with exacerbations requiring OCS was low in all studies included within the review by Vogelberg et al.35. The review authors also concluded that there was no increase in the number of patients with AEs or SAEs with tiotropium compared with placebo as add-on to ICS or add-on to ICS plus other controllers15,21,22,23,25,26,27,28,29,30,31,34. An additional study of 102 patients aged 1–5 years with persistent asthma symptoms reported similar findings, with the number of patients reporting AEs similar in those who received tiotropium as add-on to ICS to those who received placebo as add-on42.
There were fewer published studies on the efficacy and safety of LABAs, LTRAs and LAMAs as add-on to ICS in patients aged <5 years compared with studies in older age groups42,43,44,45. An RCT of 12 patients with asthma aged 2–5 years reported that LABA (formoterol) as add-on to ICS provided rapid and sustained bronchodilation for ≥8 h compared with placebo45. A 12-week RCT of 689 patients with persistent asthma (≥3 episodes of asthma symptoms during the previous year) aged 2–5 years reported that LTRA (montelukast) as add-on to ICS (in at least 50% of participants) improved multiple parameters of asthma control, including daytime and overnight asthma symptoms and the percentage of days without asthma symptoms or asthma compared with placebo. There were no reported differences in the frequency of reported AEs44. Similarly, Bisgaard et al. reported that, in patients aged 2–5 years with intermittent asthma, montelukast significantly reduces the rate of asthma exacerbations and delayed the median time to first exacerbation compared with placebo over 12 months. However, patients in this trial did not receive montelukast as add-on to ICS43. A 12-week RCT of 102 children aged 1–5 years by Vrijlandt et al. reported that tiotropium as add-on to ICS with or without additional controller medications was associated with fewer reported AEs or asthma exacerbations compared with placebo. There was no significant difference in adjusted weekly mean combined daytime asthma symptom score between baseline and Week 12 between the tiotropium and placebo groups42.
Discussion
The long-term aims of asthma management are symptom control, reduction of the future risk of exacerbations and airflow limitation, while at the same time minimising treatment side effects9. Although major advances have been made in asthma treatment and management, there still remain many patients who have poor asthma control and maintain the potential risk of worsening of their symptoms, as well as an increased risk of exacerbations, and unscheduled urgent and emergency care visits and hospitalisations46,47. For adults, adolescents and children, there is a need for effective add-on treatments as an alternative to increasing the ICS dose alone, as long-term, high-dose ICS use is associated with an increased risk of side effects9.
The findings from this literature review suggest that LABAs, LTRAs and tiotropium have similar safety profiles in both adult and paediatric populations (Table 2). Therefore, comparing the reported efficacy of the three add-on treatments in each patient population could assist with decision-making. Greater improvements in lung function have been reported with LABAs and LAMAs vs LTRAs in adults (Figs. 3–5). In addition, there appears to be greater improvements in asthma control and exacerbations with LABAs and LAMAs as add-on therapies than with LTRAs in this population.
Much of the available evidence for asthma management is based on research carried out in adults, which leads to a greater restriction of licensing of medication in children48, creating additional difficulties in selecting the most appropriate treatment option for paediatric patients with asthma49. Despite advances in care, asthma still presents a burden within this population, with many children remaining symptomatic and uncontrolled50. Data in patients aged <18 years are currently limited due to inherent difficulties in the study of this population; however, available evidence suggests that LABAs and tiotropium have comparable effects with respect to lung function, asthma control and exacerbations. Of the LABAs, LTRAs and LAMAs reviewed here, the LTRA montelukast is the only add-on treatment that is indicated for use in patients aged <4 years (as a chewable tablet51) and the only add-on treatment recommended for use in patients aged ≤5 years as an optional controller treatment9. Clinical trials of LTRAs in children aged ≤5 years have not demonstrated any safety concerns52. However, in 2020, the U.S. Food and Drug Administration (FDA) determined that a boxed warning for the LTRA montelukast was appropriate due to the risk of mental health side effects, and advised that healthcare professionals (HCPs) consider the benefits and risks of mental health side effects before prescribing montelukast53. Despite less published evidence regarding use of add-on therapies in paediatric patients (aged <18 years) than in adult patients (aged ≥18 years), current available data suggest that all three add-on therapies have comparable safety profiles, with LABAs and LAMAs providing greater improvements in lung function than LTRAs. When selecting the most appropriate add-on therapy for paediatric patients, it is important to consider the reported efficacy, safety data and subsequent post-marketing safety warnings (if applicable) and the indications of these add-on therapies, as not all are appropriate for all age ranges (Table 1).
When stepping up asthma therapy and considering add-on therapy, it is important to review the options available, to involve patients in decisions about their treatment and to keep a dialogue between patients and HCPs46. An up-to-date individualised asthma action plan can help to keep a record of any attempted treatment approaches and help the patient to self-manage54. The action plans should be discussed and agreed with patients and reviewed at regular intervals to make sure that they remain up to date and are fit for purpose54.
Poor asthma control leads to unfavourable outcomes, more frequent exacerbations, irreversible loss of lung function and even asthma-related deaths. Add-on therapy with LABA, LAMA or LTRA should be considered when asthma symptoms remain uncontrolled with at least medium–high ICS. Primary care practitioners can and should regularly assess symptom control following assessment of adherence, triggers, device technique and comorbidities. Therapy should be stepped up as recommended in GINA guidelines in order to attain optimal control, considering individual symptoms, lung function, comorbidities, inhaler technique, adherence and patient preference as important parameters for a personalised choice.
In conclusion, in adults, LAMAs and LABAs appear to provide a greater improvement in lung function than LTRAs as add-on to ICS, although there are no individual studies that directly compare LAMAs with LTRAs. LAMAs appear to be an effective alternative to LABAs for attaining asthma control, optimising lung function and preventing exacerbations, with a possible higher lung function benefit of LAMAs compared with LABAs. Data in patients aged <18 years are currently limited due to inherent difficulties in the study of this population. Current available evidence from clinical trials suggests that LABAs, LTRAs and LAMAs have comparable safety profiles, with LABAs and LAMAs providing greater improvements in lung function than LTRAs, yet it should be noted the FDA have advised a boxed warning for the LTRA montelukast due to risk of mental health side effects. Asthma should be treated in accordance with current guidelines, with regular checks made to ensure symptoms are controlled, as well as ensuring optimal strategies are in place to prevent exacerbations and achieve best lung function. If control is not achieved, treatment should be stepped up, ensuring that factors that may influence control (such as adherence, administration technique, allergic triggers and comorbidities) are addressed for each individual patient.
Methods
Our literature search was conducted in PubMed. For the comparison of add-on therapies, we identified RCTs and meta-analyses that compared the LAMA tiotropium with LABAs or LTRAs (directly or with placebo) as add-on to ICS in patients with asthma.
Data from RCTs of ≥4 weeks’ duration in all patients with asthma of all age groups, reporting change in FEV1, asthma control, exacerbations and AEs were included. Data were extracted from published manuscripts and publicly available online data. We checked the reference lists of the systematic reviews for references with any additional data for endpoints that were not described in the systematic reviews and to ensure that all trials met the inclusion criteria77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116. Search strings are detailed in Supplementary Methods.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
References
Global Asthma Network. The Global Asthma Report 2018. http://www.globalasthmareport.org/Global%20Asthma%20Report%202018.pdf (2018).
Chapman, K. R., Boulet, L. P., Rea, R. M. & Franssen, E. Suboptimal asthma control: prevalence, detection and consequences in general practice. Eur. Respir. J. 31, 320–325 (2008).
Peters, S. P., Ferguson, G., Deniz, Y. & Reisner, C. Uncontrolled asthma: a review of the prevalence, disease burden and options for treatment. Respir. Med. 100, 1139–1151 (2006).
Sapra, S. J., Broder, M. S. & Chang, E. Alignment with the revised NHLBI 2007 Asthma Guidelines, Expert Panel Report 3 (EPR 3) in a large payer database. J. Allergy Clin. Immunol. 123, S117 (2009).
Stempel, D. A., McLaughin, T. P., Stanford, R. H. & Fuhlbrigge, A. L. Patterns of asthma control: a 3-year analysis of patient claims. J. Allergy Clin. Immunol. 115, 935–939 (2005).
Rabe, K. F. et al. Worldwide severity and control of asthma in children and adults: the global asthma insights and reality surveys. J. Allergy Clin. Immunol. 114, 40–47 (2004).
Horne, R. et al. Can asthma control be improved by understanding the patient’s perspective? BMC Pulm. Med. 7, 8 (2007).
Kritikos, V. et al. A multinational observational study identifying primary care patients at risk of overestimation of asthma control. NPJ Prim. Care Respir. Med. 29, 43 (2019).
Global Initiative for Asthma. Global strategy for asthma management and prevention (2020 report) https://ginasthma.org/wp-content/uploads/2020/04/GINA-2020-full-report_-final-_wms.pdf (2020).
Kew, K. M., Quinn, M., Quon, B. S. & Ducharme, F. M. Increased versus stable doses of inhaled corticosteroids for exacerbations of chronic asthma in adults and children. Cochrane Database Syst. Rev. CD007524 (2016).
Jackson, D. J. et al. Quintupling inhaled glucocorticoids to prevent childhood asthma exacerbations. N. Engl. J. Med. 378, 891–901 (2018).
Beasley, R. et al. Inhaled corticosteroid therapy in adult asthma. Time for a new therapeutic dose terminology. Am. J. Respir. Crit. Care Med. 199, 1471–1477 (2019).
Lemanske, R. F. Jr et al. Step-up therapy for children with uncontrolled asthma receiving inhaled corticosteroids. N. Engl. J. Med. 362, 975–985 (2010).
Chauhan, B. F., Chartrand, C., Ni Chroinin, M., Milan, S. J. & Ducharme, F. M. Addition of long-acting beta2-agonists to inhaled corticosteroids for chronic asthma in children. Cochrane Database Syst. Rev. CD007949 (2015).
Chauhan, B. F. & Ducharme, F. M. Addition to inhaled corticosteroids of long-acting beta2-agonists versus anti-leukotrienes for chronic asthma. Cochrane Database Syst. Rev. CD003137 (2014).
Ducharme, F. M., Ni Chroinin, M., Greenstone, I. & Lasserson, T. J. Addition of long-acting beta2-agonists to inhaled corticosteroids versus same dose inhaled corticosteroids for chronic asthma in adults and children. Cochrane Database Syst. Rev. 5, CD005535 (2010).
Ducharme, F., Schwartz, Z., Hicks, G. & Kakuma, R. Addition of anti-leukotriene agents to inhaled corticosteroids for chronic asthma. Cochrane Database Syst. Rev. CD003133 (2004).
Anderson, D. E., Kew, K. M. & Boyter, A. C. Long-acting muscarinic antagonists (LAMA) added to inhaled corticosteroids (ICS) versus the same dose of ICS alone for adults with asthma. Cochrane Database Syst. Rev. CD011397 (2015).
Castro-Rodriguez, J. A. & Rodrigo, G. J. The role of inhaled corticosteroids and montelukast in children with mild-moderate asthma: results of a systematic review with meta-analysis. Arch. Dis. Child. 95, 365–370 (2010).
Sobieraj, D. M. et al. Association of inhaled corticosteroids and long-acting muscarinic antagonists with asthma control in patients with uncontrolled, persistent asthma: a systematic review and meta-analysis. JAMA 319, 1473–1484 (2018).
Kerstjens, H. A. et al. Tiotropium or salmeterol as add-on therapy to inhaled corticosteroids for patients with moderate symptomatic asthma: two replicate, double-blind, placebo-controlled, parallel-group, active-comparator, randomised trials. Lancet Respir. Med. 3, 367–376 (2015).
Bateman, E. D. et al. Tiotropium is noninferior to salmeterol in maintaining improved lung function in B16-Arg/Arg patients with asthma. J. Allergy Clin. Immunol. 128, 315–322 (2011).
Paggiaro, P. et al. High strength extrafine pMDI beclometasone/formoterol (200/6 μg) is effective in asthma patients not adequately controlled on medium-high dose of inhaled corticosteroids. BMC Pulm. Med. 16, 180 (2016).
Djukanovic, R. et al. Montelukast added to fluticasone propionate does not alter inflammation or outcomes. Respir. Med. 104, 1425–1435 (2010).
Vaquerizo, M. J. et al. Effect of montelukast added to inhaled budesonide on control of mild to moderate asthma. Thorax 58, 204–210 (2003).
Virchow, J. C. Jr, Prasse, A., Naya, I., Summerton, L. & Harris, A. Zafirlukast improves asthma control in patients receiving high-dose inhaled corticosteroids. Am. J. Respir. Crit. Care Med. 162, 578–585 (2000).
Kerstjens, H. A. et al. Tiotropium in asthma poorly controlled with standard combination therapy. N. Engl. J. Med. 367, 1198–1207 (2012).
Paggiaro, P. et al. The effect of tiotropium in symptomatic asthma despite low- to medium-dose inhaled corticosteroids: a randomized controlled trial. J. Allergy Clin. Immunol. Pract. 4, 104–113 (2016). e102.
Ohta, K. et al. Long-term once-daily tiotropium Respimat(R) is well tolerated and maintains efficacy over 52 weeks in patients with symptomatic asthma in Japan: a randomised, placebo-controlled study. PLoS ONE 10, e0124109 (2015).
Beeh, K. M. et al. Tiotropium Respimat(R) in asthma: a double-blind, randomised, dose-ranging study in adult patients with moderate asthma. Respir. Res. 15, 61 (2014).
Timmer, W. et al. Once-daily tiotropium Respimat((R)) 5 mug is an efficacious 24-h bronchodilator in adults with symptomatic asthma. Respir. Med. 109, 329–338 (2015).
American Lung Association Asthma Clinical Research Centers. Clinical trial of low-dose theophylline and montelukast in patients with poorly controlled asthma. Am. J. Respir. Crit. Care Med. 175, 235–242 (2007).
Peters, S. P. et al. Tiotropium bromide step-up therapy for adults with uncontrolled asthma. N. Engl. J. Med. 363, 1715–1726 (2010).
Wechsler, M. E. et al. Anticholinergic vs long-acting beta-agonist in combination with inhaled corticosteroids in black adults with asthma: the BELT randomized clinical trial. JAMA 314, 1720–1730 (2015).
Vogelberg, C. et al. A comparison of tiotropium, long-acting β2-agonists and leukotriene receptor antagonists on lung function and exacerbations in paediatric patients with asthma. Respir. Res. 21, 19 (2020).
Płoszczuk, A., Bosheva, M., Spooner, K., McIver, T. & Dissanayake, S. Efficacy and safety of fluticasone propionate/formoterol fumarate in pediatric asthma patients: a randomized controlled trial. Ther. Adv. Respir. Dis. 12, 1–15 (2018).
Simons, F. E. et al. Montelukast added to budesonide in children with persistent asthma: a randomized, double-blind, crossover study. J. Pediatr. 138, 694–698 (2001).
Hamelmann, E. et al. A randomised controlled trial of tiotropium in adolescents with severe symptomatic asthma. Eur. Respir. J. 49, 1601100 (2017).
Hamelmann, E. et al. Tiotropium add-on therapy in adolescents with moderate asthma: a 1-year randomized controlled trial. J. Allergy Clin. Immunol. 138, 441–450 (2016). e448.
Szefler, S. J. et al. A phase III randomized controlled trial of tiotropium add-on therapy in children with severe symptomatic asthma. J. Allergy Clin. Immunol. 140, 1277–1287 (2017).
Vogelberg, C. et al. Tiotropium add-on therapy improves lung function in children with symptomatic moderate asthma. J. Allergy Clin. Immunol. Pract. 6, 2160.e9–2162.e9 (2018).
Vrijlandt, E. J. L. E. et al. Safety and efficacy of tiotropium in children aged 1–5 years with persistent asthmatic symptoms: a randomised, double-blind, placebo-controlled trial. Lancet Respir. Med. 6, 127–137 (2018).
Bisgaard, H. et al. Montelukast reduces asthma exacerbations in 2- to 5-year-old children with intermittent asthma. Am. J. Respir. Crit. Care Med. 171, 315–322 (2005).
Knorr, B. et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 108, E48 (2001).
Nielsen, K. G. & Bisgaard, H. Bronchodilation and bronchoprotection in asthmatic preschool children from formoterol administered by mechanically actuated dry-powder inhaler and spacer. Am. J. Respir. Crit. Care Med. 164, 256–259 (2001).
Buhl, R. & Hamelmann, E. Future perspectives of anticholinergics for the treatment of asthma in adults and children. Ther. Clin. Risk Manag. 15, 473–485 (2019).
Aalbers, R. & Park, H. S. Positioning of long-acting muscarinic antagonists in the management of asthma. Allergy Asthma Immunol. Res. 9, 386–393 (2017).
Cates, C. J. Current challenges in asthma. Br. J. Gen. Pract. 57, 179–180 (2007).
Vogelberg, C. Preschool children with persistent asthmatic symptoms. Ther. Clin. Risk Manag. 15, 451–460 (2019).
Hossny, E. et al. The use of inhaled corticosteroids in pediatric asthma: update. World Allergy Organ. J. 9, 26 (2016).
Accord Healthcare Limited. Montelukast 10 mg film coated tablets. Summary of product characteristics. June 2019. https://www.medicines.org.uk/emc/product/1243/smpc (2019).
Doherty, G. M. Is montelukast effective and well tolerated in the management of asthma in young children?: Part A: Evidence-based answer and summary. Paediatr. Child Health 12, 307–308 (2007).
U.S. Food & Drug Administration. FDA requires Boxed Warning about serious mental health side effects for asthma and allergy drug montelukast (Singulair); advises restricting use for allergic rhinitis. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requires-boxed-warning-about-serious-mental-health-side-effects-asthma-and-allergy-drug (2020).
Pinnock, H. Supported self-management for asthma. Breathe 11, 98–109 (2015).
Merck Sharp & Dohme Limited. Singulair paediatric 4 mg tablets. Summary of product characteristics. October 2018. https://www.medicines.org.uk/emc/product/6500/smpc (2018).
Merck Sharp & Dohme Limited. Singulair paediatric 5 mg chewable tablets. Summary of product characteristics. October 2018. https://www.medicines.org.uk/emc/product/197/smpc (2018).
Boehringer Ingelheim Limited. Spiriva Respimat 2.5 microgram, inhalation solution. Summary of product characteristics. April 2018. https://www.medicines.org.uk/emc/product/407/smpc (2018).
GlaxoSmithKline UK. Serevent Accuhaler. Summary of Product Characteristics. October 2018. https://www.medicines.org.uk/emc/product/848/smpc (2018).
GlaxoSmithKline. Highlights of prescribing information for SEREVENT DISKUS (salmeterol xinafoate inhalation powder), for oral inhalation use. December 2018. https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Serevent_Diskus/pdf/SEREVENT-DISKUS-PI-MG-IFU.PDF (2018).
Merck & Co Inc. Highlights of prescribing information for FORADIL AEROLIZER (formoterol fumarate inhalation powder) for oral inhalation only. November 2012. https://www.merck.com/product/usa/pi_circulars/f/foradil/foradil_pi.pdf (2012).
Novartis Pharmaceuticals UK Ltd. Foradil. Summary of product characteristics. October 2016. https://www.medicines.org.uk/emc/product/1030/smpc (2016).
Price, D. B., Swern, A., Tozzi, C. A., Philip, G. & Polos, P. Effect of montelukast on lung function in asthma patients with allergic rhinitis: analysis from the COMPACT trial. Allergy 61, 737–742 (2006).
Boheringer Ingelheim Pharmaceutics Inc. Prescribing information for Spiriva® Respimat® (tiotropium bromide) inhalation spray, for oral inhalation. March 2019. https://docs.boehringer-ingelheim.com/Prescribing%20Information/PIs/Spiriva%20Respimat/spirivarespimat.pdf (2019).
Merck Sharp & Dohme Limited. Singulair paediatric 4 mg granules. Summary of product characteristics. October 2018. https://www.medicines.org.uk/emc/product/45/smpc (2018).
U.S. Food & Drug Administration. Advair Diskus® (fluticasone propionate/salmeterol): Prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/021077s029lbl.pdf (2008).
AstraZeneca UK Limited. Symbicort Turbohaler 100/6, inhalation powder. Summary of product characteristics. November 2018. https://www.medicines.org.uk/emc/product/1326/smpc (2018).
Generics UK T/A Mylan. Sirdupla 25 microgram/250 microgram per metered dose pressurised inhalation, suspension. Summary of product characteristics. February 2019. https://www.medicines.org.uk/emc/product/2538/smpc (2019).
Sandoz Limited. AirFluSal MDI 25 microgram/250 microgram per actuation pressurised inhalation, suspension. Summary of procuct characteristics. April 2019. https://www.medicines.org.uk/emc/product/8785/smpc (2019).
U.S. Food & Drug Administration. Breo Ellipta® (fluticasone furoate/vilanterol): prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/204275s017lbl.pdf (2019).
U.S. Food & Drug Administration. Dulera® (mometasone/formoterol): prescribing information, https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/022518s021lbl.pdf (2017).
Teva Pharma BV. Aerivio Spiromax 50 microgram/500 microgram inhalation powder. Summary of Product Characteristics. June 2017. https://www.medicines.org.uk/emc/product/2482/smpc (2017).
Teva Pharma BV. DuoResp Spiromax 160 micrograms/4.5 micrograms inhalation powder. Summary of product characteristics. April 2019. https://www.medicines.org.uk/emc/product/3323/smpc (2019).
Chiesi Limited. Fostair 100/6 inhalation solution. Summary of product characteristics. February 2019. https://www.medicines.org.uk/emc/product/6318/smpc (2019).
Orion Pharma (UK) Limited. Fobumix Easyhaler 160/4.5 inhalation powder. Summary of product characteristics. December 2017. https://www.medicines.org.uk/emc/product/8706/smpc (2017).
Napp Pharmaceuticals Limited. Flutiform 125 microgram/5 microgram per actuation pressurised inhalation, suspension. Summary of product characteristics. January 2019. https://www.medicines.org.uk/emc/product/7649/smpc (2019).
Szefler, S. J., Carlsson, L. G., Uryniak, T. & Baker, J. W. Budesonide inhalation suspension versus montelukast in children aged 2 to 4 years with mild persistent asthma. J. Allergy Clin. Immunol. Pract. 1, 58–64 (2013).
Demoly, P., Annunziata, K., Gubba, E. & Adamek, L. Repeated cross-sectional survey of patient-reported asthma control in Europe in the past 5 years. Eur. Respir. Rev. 21, 66–74 (2012).
Fuhlbrigge, A. et al. The status of asthma control in the U.S. adult population. Allergy Asthma Proc. 30, 529–533 (2009).
Reddel, H. K., Sawyer, S. M., Everett, P. W., Flood, P. V. & Peters, M. J. Asthma control in Australia: a cross-sectional web-based survey in a nationally representative population. Med. J. Aust. 202, 492–496 (2015).
Nunes, C., Pereira, A. M. & Morais-Almeida, M. Asthma costs and social impact. Asthma Res. Pract. 3, 1 (2017).
Soriano, J. B. et al. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir. Med. 5, 691–706 (2017).
Allen, D., Hines, E. W., Pazdernik, V., Konecny, L. T. & Breitenbach, E. Four-year review of presenteeism data among employees of a large United States health care system: a retrospective prevalence study. Hum. Resour. Health 16, 59 (2018).
Sullivan, P. W. et al. The relationship between asthma, asthma control and economic outcomes in the United States. J. Asthma 51, 769–778 (2014).
McGeachie, M. J. et al. Patterns of growth and decline in lung function in persistent childhood asthma. N. Engl. J. Med. 374, 1842–1852 (2016).
Price, D., Fletcher, M. & van der Molen, T. Asthma control and management in 8,000 European patients: the REcognise Asthma and LInk to Symptoms and Experience (REALISE) survey. NPJ Prim. Care Respir. Med. 24, 14009 (2014).
Wechsler, M. E. Managing asthma in primary care: putting new guideline recommendations into context. Mayo Clin. Proc. 84, 707–717 (2009).
De Simoni, A., Horne, R., Fleming, L., Bush, A. & Griffiths, C. What do adolescents with asthma really think about adherence to inhalers? Insights from a qualitative analysis of a UK online forum. BMJ Open 7, e015245 (2017).
FitzGerald, J. M., Boulet, L. P., McIvor, R. A., Zimmerman, S. & Chapman, K. R. Asthma control in Canada remains suboptimal: The Reality of Asthma Control (TRAC) study. Can. Respir. J. 13, 253–259 (2006).
Thomas, M., Leather, D. & Price, D. High-dose inhaled corticosteroids and add-on therapy use in adults with asthma in the UK in 2003: an observational study. Prim. Care Respir. J. 15, 166–172 (2006).
Thomas, M., Murray-Thomas, T., Fan, T., Williams, T. & Taylor, S. Prescribing patterns of asthma controller therapy for children in UK primary care: a cross-sectional observational study. BMC Pulm. Med. 10, 29 (2010).
Hamelmann, E., von Mutius, E., Bush, A. & Szefler, S. J. Addressing the risk domain in the long-term management of pediatric asthma. Pediatr. Allergy Immunol. 31, 233–242 (2020).
Levy, M. L. et al. Summary of the 2008 BTS/SIGN British Guideline on the management of asthma. Prim. Care Respir. J. 18, S1–S16 (2009).
Kaplan, A. & Price, D. Treatment adherence in adolescents with asthma. J. Asthma Allergy 13, 39–49 (2020).
Koster, E. S., Philbert, D., de Vries, T. W., van Dijk, L. & Bouvy, M. L. “I just forget to take it”: asthma self-management needs and preferences in adolescents. J. Asthma 52, 831–837 (2015).
Shimoda, T., Obase, Y., Kishikawa, R. & Iwanaga, T. Influence of cigarette smoking on airway inflammation and inhaled corticosteroid treatment in patients with asthma. Allergy Asthma Proc. 37, e50–e58 (2016).
Boulet, L.-P. & Nair, P. Inhaled corticosteroids and adult asthma. Am. J. Respir. Crit. Care Med. 200, 1556–1557 (2019).
Global Initiative for Asthma. Difficult-to-treat & severe asthma in adolescent and adult patients – diagnosis and management. https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf (2019).
Turner, S. et al. Long-acting β-agonist in combination or separate inhaler as step-up therapy for children with uncontrolled asthma receiving inhaled corticosteroids. J. Allergy Clin. Immunol. Pract. 5, 99–106 (2017). e103.
Virchow, J. C. et al. Single inhaler extrafine triple therapy in uncontrolled asthma (TRIMARAN and TRIGGER): two double-blind, parallel-group, randomised, controlled phase 3 trials. Lancet 394, 1737–1749 (2019).
Rodrigo, G. J. & Castro-Rodriguez, J. A. Safety of long-acting beta agonists for the treatment of asthma: clearing the air. Thorax 67, 342–349 (2012).
Cazzola, M., Page, C. P., Rogliani, P. & Matera, M. G. Beta2-agonist therapy in lung disease. Am. J. Respir. Crit. Care Med. 187, 690–696 (2013).
Busse, W. W. et al. Combined analysis of asthma safety trials of long-acting β2-agonists. N. Engl. J. Med. 378, 2497–2505 (2018).
Xia, Y. et al. Safety of long-acting beta agonists and inhaled corticosteroids in children and adolescents with asthma. Ther. Adv. Drug Saf. 4, 254–263 (2013).
Khalid, F., Aftab, A. & Khatri, S. The association between leukotriene-modifying agents and suicidality: a review of literature. Psychosomatics 59, 19–27 (2018).
Kerstjens, H. A. et al. Tiotropium improves lung function, exacerbation rate, and asthma control, independent of baseline characteristics including age, degree of airway obstruction, and allergic status. Respir. Med. 117, 198–206 (2016).
Casale, T. B. et al. Tiotropium Respimat add-on is efficacious in symptomatic asthma, independent of T2 phenotye. J. Allergy Clin. Immunol. Pract. 6, 923–935 (2018).
Szefler, S. J. et al. Tiotropium is efficacious in 6- to 17-year-olds with asthma, independent of T2 phenotype. J. Allergy Clin. Immunol. Pract. 7, 2286–2295 (2019).
Hamelmann, E. & Szefler, S. J. Efficacy and safety of tiotropium in children and adolescents. Drugs 78, 327–338 (2018).
Aalbers, R., Vogelmeier, C. & Kuna, P. Achieving asthma control with ICS/LABA: a review of strategies for asthma management and prevention. Respir. Med. 111, 1–7 (2016).
Lin, J. et al. Symbicort® Maintenance and Reliever Therapy (SMART) and the evolution of asthma management within the GINA guidelines. Expert Rev. Respir. Med. 12, 191–202 (2018).
Dahl, R. Systemic side effects of inhaled corticosteroids in patients with asthma. Respir. Med. 100, 1307–1317 (2006).
Gruffydd-Jones, K. Unmet needs in asthma. Ther. Clin. Risk Manag. 15, 409–421 (2019).
Novartis Pharmaceuticals UK Ltd. Xolair 150 mg Solution for Injection. Summary of product characteristics. January 2019. https://www.medicines.org.uk/emc/product/4725/smpc (2019).
GlaxoSmithKline UK. Nucala 100 mg powder for solution for injection. Summary of product characteristics. June 2019. https://www.medicines.org.uk/emc/product/1938/smpc (2019).
AstraZeneca UK Limited. Fasenra 30 mg solution for injection in pre-filled syringe. Summary of product characteristics. July 2019. https://www.medicines.org.uk/emc/product/8918/smpc (2019).
Teva Pharma Ltd. Cinqaero (reslizumab) 10 mg/mL concentrate for solution for infusion. Summary of product characteristics. https://www.medicines.org.uk/emc/product/4370/smpc (2018).
Acknowledgements
Medical writing assistance, in the form of the preparation and revision of the draft manuscript, was supported financially by Boehringer Ingelheim and provided by Rosie Robson of MediTech Media under the authors’ conceptual direction and based on feedback from the authors. Boehringer Ingelheim was given the opportunity to review the manuscript for factual accuracy only.
Author information
Authors and Affiliations
Contributions
The authors take full responsibility for the scope, direction, content and editorial decisions relating to the manuscript, were involved at all stages of development and have approved the submitted manuscript. A.K. is a guarantor of this work.
Corresponding author
Ethics declarations
Competing interests
A.K. is on the advisory board and/or speakers bureau for AstraZeneca, Behring, Boehringer Ingelheim, Covis, Grifols, GSK, Merck Frosst, Novartis, Novo Nordisk, Pfizer, Purdue, Sanofi, Teva and Trudell; and is a member of Health Canada Section on Allergy and Respiratory Therapeutics; co-chair of Ontario Health Quality, Ontario Quality Standards for COPD; member of Ontario Health Quality, Ontario Quality Standards Asthma Chairperson, Family Physician Airways Group of Canada; honorary professor of Respiratory Medicine of the Observational and Pragmatic Research, Singapore; vice president of the Respiratory Effectiveness Group and medical lead at Lung Health Portal, MD Briefcase. J.M.F. is on the advisory board and/or speakers bureau for AstraZeneca, Boehringer Ingelheim, GSK, Novartis, Regeneron, Sanofi and Theravance; receives peer-review funding from AllerGen, NIH, and CIHR; research funding from AstraZeneca, GSK, Novartis and Sanofi-Regeneron, all paid directly to UBC; is a member of the steering committee for the International Severe Asthma Registry and PI for Canadian Severe Asthma Registry and is a member of the GINA Science and Executive Committees. R.B. reports grants to Mainz University and personal fees from Boehringer Ingelheim, GlaxoSmithKline, Novartis and Roche, as well as personal fees from AstraZeneca, Chiesi, Cipla, Sanofi and Teva, and is a member of the GINA Science Committee. C.V. is on the advisory board and/or speakers bureau for ALK, AllergoPharma, Bencard, Boehringer Ingelheim, DBV Technology, HAL Allergy, Novartis and Sanofi; is President of the Society of Pediatric Allergology and Environmental Medicine (GPA); he is co-author of the German National Management Guideline for Asthma (NVL Asthma) and he is receiving grants from the German Research Foundation (DFG). E.H. is on the advisory board and/or speakers bureau for ALK, AllergoPharma, Bencard, Boehringer Ingelheim, GSK, Novartis, Nutricia, Sanofi and Stallergenes; is President of the German Asthma Net (GAN) e.V., Vice-President of the German Allergy Society (DGAKI) and co-chair of the asthma section of the German Pediatric Pneumology Society (GPP); he is coordinator of the German guideline for severe asthma in children and co-author of the German National Management Guideline for Asthma (NVL Asthma) and he is receiving grants from the German Ministry of Education and Research (BMBF).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kaplan, A., FitzGerald, J.M., Buhl, R. et al. Comparing LAMA with LABA and LTRA as add-on therapies in primary care asthma management. npj Prim. Care Respir. Med. 30, 50 (2020). https://doi.org/10.1038/s41533-020-00205-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41533-020-00205-9