Abstract
Sweat is a biofluid with rich information that can reflect an individual’s state of health or activity. But the real-time in situ sweat sensors lack the ability of long-term monitoring. Against this background, this article provides a holistic review on the necessary process and methods for sweat sensing, including sweat collection, composition analysis, energy supply, and data processing. The impacts of the environment in stimulating sweat production, providing energy supply, and intelligent health monitoring are discussed. Based on the review of previous endeavors, the future development in material, structure and artificial intelligence application of long-term sweat monitoring is envisioned.
Similar content being viewed by others
Introduction
Personal health monitoring plays a vital role in daily healthcare and chronic disease management. To monitor physical health, researchers have tried various methods to acquire vital signals from the human body1,2. A growing field in this area is wearable sensing devices, which aim to obtain long-term information regarding personal health3,4,5. Wearable sensor devices can be placed on different positions of the human body, such as the wrists, chest, forehead, waist and soles of the feet. In recent years, wearable sensors have shown increasing promise for use in broad applications regarding human health monitoring6 and medical diagnosis7,8.
Flexible sensing is one of the most promising technologies for health monitoring devices.9,10,11,12,13,14,15 The typical configuration of flexible wearable sensors is include biosensors in combination with wearable technology16. The characteristics of biosensors, such as high specificity, high efficiency, low cost, low power requirements and convenience, make them promising in wearable applications, especially for sweat, saliva, interstitial fluid and other biological fluids. Among the biological fluids, sweat contains an abundance of relevant biomarkers, such as ions and metabolites, that can be leveraged for tracking the user’s metabolism, fatigue, and stress17,18,19,20, a potentially essential metabolic pathway of the human21,22,23,24. The abundant ions and metabolites in sweat are closely related to human health. At the same time, testing sweat is noninvasive and sweat can be the best monitoring substance for in vitro monitoring. The focus of research for testing sweat is usually in device design25,26,27,28,29,30,31, health applications32,33,34,35,36,37,38, or sports management39,40,41.
Despite the research to date, a wearable sweat sensor is still far from being realized for commercial health monitoring. The core issues requiring attention relate to long-term monitoring, including the mechanism for collection and analysis of sweat, the supply and management of energy to the device, and the establishment of the relationship between monitoring data and human health. Research has been aimed at sweat collection42,43,44,45,46,47, self-powered devices48,49,50,51 and applying artificial intelligence to monitors52,53,54. This review provides an overview of current developments in wearable sweat sensors through bibliometric analysis, paying attention especially to long-term stability. On this basis, sweat analysis, including production and collection, energy supply and storage, and the algorithms and models of intelligent monitoring are discussed.
Bibliometrics analysis of wearable sweat sensors
Searching on the subject of ‘wearable sweat sensor’ on the Web of Science website identified the presence of relevant research since 2005, but only a few researchers focused on sweat sensing. An early sweat analysis device was reported by Morris55. The device passively collects sweat based on textiles and performs pH analysis in a predefined channel, which preliminarily verified the feasibility of wearable sweat sensor applications. A critical turning points occurred in 2016, when Gao56 published research on a fully integrated array sweat sensor in Nature that simultaneously and selectively measured sweat metabolites and electrolytes without requiring external analysis57. This significant work attracted the attention of many researchers and in the past five years, research on wearable sweat sensors has grown rapidly.
Using VOSviewer, we created a network visualization map of the popular keywords from our search (Fig. 1). In the map, the larger the circular area, the higher the frequency of occurrence for that keyword. The core keywords observed are ‘wearable,’ ‘sensor,’ ‘biosensor’ and ‘sweat.’ Other keywords fall into several categories, including parameters (such as ‘pH,’ ‘sodium,’ ‘glucose,’ and ‘lactate’) and vocabulary related to devices (such as ‘skin,’ ‘array,’ and ‘platform’). Clustered results are discussed in turn below.
In the first cluster, colored red, the main keyword is ‘wearable.’ The most popular research directions, as indicated by cluster device keywords, are ‘electrochemical sensors,’ ‘flexible sensors,’ and ‘mouthguard biosensors.’ In this cluster, ‘pH’ also has high frequency, reflecting its importance as a parameter of sweat. The second cluster, colored green, has the main keyword ‘sensor.’ The next most frequent word was ‘glucose,’ a reflection of the abundant research on glucose monitoring in blood, sweat and interstitial fluid (ISF). Moreover, ‘lactate’ and ‘sodium’ also showed up as common research keywords. The sensor cluster also includes various sensing platforms, such as ‘patches,’ ‘electronics’ and ‘microfluidics.’ The third cluster, colored yellow, has the main keyword ‘sweat.’ This cluster includes keywords related to pressure monitoring, sensor arrays, and bio-electrochemical sensing, along with ‘performance.’ In the fourth cluster, colored blue, the main keyword is ‘biosensor’ and the associated keywords focus on preparation methods. Materials such as ‘graphene’ and ‘nanoparticles’ are common keywords. The last cluster is colored purple and mainly involves cortisol and stress monitoring. While few studies exist to date, the exploration of mental monitoring is a noteworthy and exciting area of study.
The process of monitoring sweat
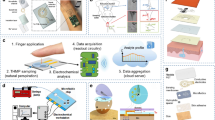
Figure 2 gives an overview of the process of sweat monitoring, including sweat secretion, collection, sensing, transmission, and analysis. In all parts of the process, researchers are concerned with components in sweat that can be detected, how to achieve accurate monitoring with a small sample volume and how to design sensitive sensor devices. The question of what to measure and how to measure it is gradually being answered.
Benefits and challenges of measuring sweat
Sweat has several beneficial characteristics as an indicator of health: collection of sweat is non-invasive, sweat has a fast update rate, and sweat is rich in ions and metabolites for analysis. With long-term sweat monitoring, construction of a matching health monitoring model will be possible. A recent increase in fabric-based wearable sensing technology has enabled continuous monitoring for longer periods (more than a day).
Whether the flexible sensor device has enough life and stability to meet long-term operational needs remains uncertain. Factors affecting long-term monitoring are diverse and include environmental contamination, equipment reliability, biocompatibility, energy supply, and system design. Environmental contamination can include cumulative errors compounded during data collection and contaminants getting mixed in with sweat during collection. Equipment reliability issues most frequently reflect life span limitations, along with tensile and deformation properties of the electrochemical sensor device after repeated cycles. Most existing flexible sensor devices can meet the requirements related to these issues, with the caveat that the service life of an enzyme sensor is related to temperature and pH. Biocompatibility problems mostly reflect issues in the comfort of wearing the sensor for a long time. For example, the wearer may struggle if there is a lack of air permeability, changes in temperature and humidity, or possible allergic or stress reactions; these need to be avoided through the appropriate selection of materials. For energy supply, the key factor is ensuring that the battery capacity can meet the requirements. If continuous use for a longer time is considered, the reliability and stability of a self-powered device are critical. Finally, for the overall design of the sensor system, it is necessary to consider the sensor interaction with the human body and the environment when the sensor is worn for a long time.
Another set of challenges regard the feasibility of the sensor system. Sweat generation can be accomplished through exercise activity, thermal heating, stress or iontophoretic stimulation38. Usually, the sweat we analyze is generated by exercise, but this sweat may have completely different composition characteristics from sweat caused by other factors. Furthermore, there is still sweat metabolism in the human body at rest. To achieve a longer-term wearable continuous monitoring, sensitive sweat collection and analysis under low metabolic rates will be indispensable. Moreover, once long-term monitoring can be achieved, the management and supply of energy presents more challenges. Most current wearable sweat sensor devices have a short operational time, indicating that batteries for power supply are not the best choice. A self-powered device could bring many advantages. Meanwhile, long-term monitoring data could elucidate the relationship between the health status and sweat characteristics.
Sweat sampling and control
Sweat collection and control are prerequisites for its analysis58. Sweat collection is more complicated than standard body fluid analysis, such as blood analysis or urinalysis. For example, blood is plentiful in the human body and blood vessels are spread throughout the body, making blood sample acquisition relatively simple59. In contrast, sweat is produced only under appropriate environmental conditions. In addition, although sweat glands are distributed in various body parts, they are small and often attached to the skin surface, so that sweat produced from them easily evaporates. Sweat secretion rates can be lower than 10 nL min−1 cm−2 at low secretion sites such as an arm or leg, but can reach 100 nL min−1 cm−2 at high secretion areas like the palm and foot5. It is also not currently clear if the composition of sweat produced by different stimuli differs: high temperature, strenuous exercise and mood swings are all triggers for sweating. In some application scenarios, the goal is to monitor sweat continuously and the amount of sweat may not meet monitoring needs. In such circumstances, iontophoresis, whereby sweat glands are stimulated through delivery of an agonist using a low electrical current, is an useful method to obtain sufficient sweat60,61. However, sweat composition obtained by iontophoresis differs from the composition of sweat spontaneously produced by the human body. Figure 3 shows examples of sweat collecting and control methods.
(a) A sweat-collecting patch that can collect sweat efficiently for fast and continuous healthcare monitoring. Ref. 63, Copyright 2021, Advanced Materials. (b) A hydrophobic/superhydrophilic Janus polyester/nitrocellulose textile embedded with a conical micropore array that has a hydrophilic inner surface. Ref. 64, Copyright 2019, Advanced Materials. (c) A thin, soft, skin-like microfluidic platform that bonds to the skin to allow for the collection and storage of sweat in an interconnected set of micro reservoirs. Ref. 69, Copyright 2017, Advanced Healthcare Materials.
Early research focused on the ionic components in sweat, such as pH and sodium. Due to the lack of appropriate in-situ analysis methods, they had to do analysis using in vitro sweat collection. Bandodkar et al.62 achieved in situ sweat sensing in 2013, using a whole body-washdown method since local sweat collection cannot fully reflect human sweat. For the procedure, the subject is exercised in a closed environment. The weight difference before and after exercise is used to quantify the amount of sweat produced and the subject, the clothing worn, and the sports equipment are all washed with deionized water and collected. The composition of sweat is then calculated. This rigorous and standard method is the gold standard for sweat analysis. However, it is too complicated and cumbersome for regular use and thus subsequent researchers have looked for methods to collect and analyze local sweat in vitro. The methods used include sweat-absorbent patches, armbands and microcatheters. However, all methods have the problem that sweat is lost during the in vitro analysis after collection. Further, such collection methods cannot be accurately time-stamped, making it difficult to match the results of the sweat analysis to the state of the subject.
With the development of micro-, nano- and wearable technology, researchers have begun to try smaller and more precise sweat collection and analysis. At present, such methods can be divided into two categories. The first category focuses on the real-time performance of sweat composition analysis - focusing on sweat renewal rate as opposed to sweat loss. The core parameter in this category is thus the refresh rate of sweat. The second category pursues highly accurate sweat collection and analysis using microchannels and other means to collect and control sweat for subsequent high-throughput sweat analysis. The advantage to this method is that a closed sweat collection system can effectively avoid external contamination.
An important component of precise sweat collection is the control of sweat entering the device. Hydrophilicity and hydrophobicity control is a common method for controlling the movement of sweat. Son et al.63 created a patch that uses cactus-spine-inspired wedge-shaped wettability patterned channels on a hierarchical microstructure/nanostructure surface. Dai et al.64 modified textiles with superhydrophilic/superhydrophobic conditions to drive the directional flow of sweat. He et al.65 demonstrated a flexible and skin-mounted band that combines superhydrophobic and superhydrophilic microarrays with nondendritic colorimetric biosensors for in situ sweat sampling and analysis. They also prepared an intelligent Janus textile band based on a contact pump model to enable the transport of sweat from the body surface to the hydrophilic electrode side66. In addition, the team designed devices to monitor the comfort of hot and humid environments based on the characteristics of Janus materials67.
The design of mechanical structures such as micro-pumps and micro-valves is an alternative way to successfully control sweat. Choi et al.68,69 focused on a structure, termed capillary bursting valves (CBV). They designed valves with different angles to achieve selective flow of sweat under specific contact angle conditions. CBV are passive valves that do not require external control.
With the development of micro-nano processing technology, the technology for local sweat collection is expected to become more mature, realizing a more precise collection of local sweat. At the same time, local sweat differences are likely occur based on factors such as different parts of the human body, different diets, and climate. Therefore, distributed multi-point sweat collection and joint analyses will be necessary in the future.
Sweat composition and application prospects
The flexible sweat sensor can be used in humans for health monitoring, special disease screening, or emotional stress testing70. The components of sweat fall into three categories: electrolytes (ions), metabolites, and xenobiotics. Different components can reflect different health conditions.
Common ions found in sweat include sodium, chlorine, potassium, ammonium, calcium and hydrogen. Although the concentration of ions in sweat is much lower than in blood, they can still reflect the state of the human. Among them, chlorine was used in human health monitoring. When patients with cystic fibrosis were discovered to have higher sweat chloride ion concentrations, monitoring the chloride ion concentration after stimulating sweat through iontophoresis or by directly collecting sweat became a recognized monitoring method.
The ions in sweat are thought to reflect the level of water and salt metabolism in the human body. Excessive loss of sodium and potassium can lead to hyponatremia, muscle cramps, and dehydration. Hydrogen, known as pH, is also an important physiological index parameter that can be used for other parameters.
Sensors to detect ions in sweat can be divided into two general categories: electrochemical and colorimetric. Typically, devices using colorimetry have smaller structures and better flexibility. In comparison, electrochemical-based sensors require further improvement in terms of device flexibility and skin compatibility but have good performance. The long-term monitoring capability and signal acquisition capability of electrochemical based sensor are suitable for longer-term monitoring. For example, Wang et al.71 tested a flexible, self-healable, adhesive and wearable hydrogel patch for sweat colorimetric detection, and showed the good conformality of the patch to the skin. They used a cell phone to analyze the data, which could not provide real-time dynamic monitoring of sweat.
Electrolytes
The monitoring of ions depends on the construction of ion-selective membranes; the selectivity and sensitivity of these membranes are vital parameters when developing sensors. Common ions detected in sweat are outlined below.
pH
It is generally believed that the pH value of human sweat is in the range of 4.2-7.8. Although current research has not found that pH value of sweat alone can reflect the characteristics of health or disease, pH value is an essential reference. As shown in Fig. 4a, Yang et al.72 created a flexible and wearable patch consisting of a charge-coupled device (CCD) pH sensor that can measure a person’s sweat with high sensitivity. Nyein et al.73 demonstrated a wearable electrochemical device for continuous monitoring of ionized calcium and pH of body fluids using disposable and flexible array sensors that interface with flexible printed circuit board(FPCB) (Fig. 4b).
a A flexible charge-coupled device can be used to create a sensor capable of measuring the pH of sweat with high sensitivity. Ref. 72, Copyright 2018, Nature Electronics. b A wearable electrochemical device for continuous monitoring of ionized calcium and pH of body fluid. Ref. 73, Copyright 2016, ACS Nano. c Illustration of WPIS based on a bracelet that was modified with the working/indicator electrode (WE) and a reference electrode (RE) to provide a WPIS. Ref. 77, Copyright 2018, Trends in Analytical Chemistry. d An integrated device to monitor the physiological concentration of two cations, ammonium (NH4+) and calcium (Ca2+). Ref. 78, Copyright 2019 Advanced Healthcare Materials.
Sodium
The most common ion in human sweat, sodium is the primary indicator for levels of human water and salt metabolism. Sodium ions can indirectly reflect the sweat rate of the human body and thus also provide a reference for calibration of sweat monitoring. Zhai et al.74 used a vertically aligned mushroom-like gold nanowire to achieve a multiplex in situ analysis of pH, sodium and potassium in sweat. The device performed well even under a 30% strain during stretch-release cycles. At the same time, it was possible to wear the sensor for 30 minutes of in-situ wireless monitoring.
Chlorine
Abnormally high sweat chloride concentration is observed in cystic fibrosis (CF) patients, and sweat chloride analysis has been adopted as the gold standard for cystic fibrosis diagnosis75 Wescor-macrodut is a specific product made for chlorine monitoring.
Potassium
The concentration of potassium in the serum can predict muscle activity, and is also closely related to hypokalemia and hyperkalemia, regardless of cause. However, the current mechanism of potassium ion secretion in sweat is still uncertain, so the final potassium concentration in sweat may be maintained within the plasma potassium concentration range76.
Calcium
Calcium can be used to monitor the status of the liver and bones77. Sweat and blood concentrations of calcium are correlated. As shown in Fig. 4c, A sensor for calcium is based on working electrode and reference electrode output signals, and three feasible sensing mechanisms are known, two of which (a and b in Fig. 4c) correspond to all-solid-state potentiometric sensors, using conductive polymers with high redox capacitance. In the third mechanism (c in Fig. 4c), nanomaterials for double-layer capacitance measure the response mechanism of reference electrode (RE) based on Ag/AgCl layer and polymer reference film. Keene et al.78 reported on ion-selective organic electrochemical transistor (IS-OECT) devices that can monitor the physiological concentration of two cations, ammonium and calcium. As seen in Fig. 4d, the device is small and convenient, has good stability, and also exhibits well characteristics when compared to standard monitoring methods.
Metabolites
The metabolites in sweat are represented by lactic acid and glucose. Unlike ions, these metabolites need to be precisely monitored using enzymes. The specificity of enzymes allows the monitoring of metabolites in sweat to have higher precision, accuracy, and speed. Still, due to the limitations of enzyme activity, the life of the enzyme sensor will be lower than that of the ion-selective sensor. Figure 5 summarizes examples of sensors used for metabolite sensing.
a A real-time noninvasive lactate sensor for human perspiration. Ref. 82, Copyright 2021, Talanta. b An elastic gold fiber-based three-electrode electrochemical platform that can meet the criteria of wearable textile glucose biosensing. Ref. 85, Copyright 2019, Analytical Chemistry. c Multifunction sensor: ultrasensitive sweat UA and Tyr detection, sweat rate estimation, temperature sensing and vital-signs monitoring. Ref. 86, Copyright 2020, Nature Biotechnology. d An integrated wireless graphene-based sweat stress sensing system for dynamic and noninvasive stress hormone analysis. Ref. 89, Copyright 2020, Matter.
Lactate
Lactate is a minor molecular metabolite produced by the body during anaerobic exercise, including high-intensity exercise and metabolism in critically ill patients. Lactic acid is also used to maintain the Purine Nucleotide Cycle (PNC) and the Tricarboxylic Acid Cycle (TCA cycle). These two processes provide energy in a hypoxic environment. However, the secretion mechanism of lactic acid into sweat is complex and still unclear79. Nonetheless, sweat lactic acid can indirectly reflect the physical state of exercise. Compared with the activity of the whole body, sweat lactic acid may have a more direct relationship with the activity of the sweat glands themselves. At present, research teams are exploring the relationship between sweat lactic acid and blood lactic acid after exercise80,81. The concentration range of lactic acid in sweat ranges from ~6 mM to ~100 mM. Wang et al.82 described a textile lactate biosensor. As shown in Fig. 5a, gold fibers are used to fabricate lactate-sensing working electrodes, reference electrodes and counter electrodes and then further weaved into textiles in a standard three-electrode system with a planar layout.
Glucose
Continuous monitoring of blood glucose levels is necessary for diagnosis and treatment of diabetes. A correlation between sweat glucose and blood glucose exists and obtaining blood glucose values by detecting glucose in sweat is thus expected to replace conventional diabetes blood glucose monitoring practices83,84 Zhao et al.85 created an elastic gold fiber-based three-electrode electrochemical platform that meets the criteria for wearable textile glucose biosensing. Figure 5b shows the gold fiber that can be functionalized with Prussian blue and glucose oxidase to obtain a working electrode and modified by Ag/AgCl to serve as the reference electrode. The nonmodified gold fiber can serve as the counter electrode. The device has stable performance even when the stretch rate reaches 200%.
Uric acid (UA)
UA is a risk factor for cardiovascular disease, type 2 diabetes, and renal disease; it is also widely used in clinical settings to manage gout, the most common inflammatory arthritis, which affects tens of millions of people worldwide. Yang et al.86 described a laser-engraved wearable sensor that can rapidly and accurately detect UA and tyro-sine (Tyr) in human sweat in situ. As shown in Fig. 5c, on the graphene substrate, they not only arranged UA and Tyr sensing units, but also included auxiliary signal sensors, such as temperature and sweat rate.
Cortisol
Cortisol is a hormone related to human stress released by hypothalamic-pituitary-adrenal axons87. There are two types of cortisol bound to carrier protein: free cortisol and cortisol. Free cortisol can diffuse through the cell membrane through passive transport. The concentration of cortisol in sweat ranges from 2.21 × 10−5 to 3.86 × 10-4 mM, with the largest concentration typically occurring in the morning. In comparison, the cortisol concentration in the blood is generally 1.24 × 10-4 to 4.0 × 10-4 mM88. This difference may be related to the type II 11-β-hydroxysteroid-dehydrogenase (HSD) in the sweat ducts that converts cortisol into corticosterone. As shown in Fig. 5d, Torrente-Rodríguez et al.89 proposed a highly sensitive, selective and miniaturized mHealth device, based on a laser-enabled flexible graphene sensor, to monitor levels of stress hormones non-invasively; they also discuss the relationship between cortisol and circadian rhythms and stress response.
Xenobiotics
Drug
Drug analysis is another primary function of sweat analysis. It is commonly implemented for doping control, drug abuse testing, forensic investigation, clinical therapeutics, and digital health monitoring. Sources of biological specimens for drug analysis include urine, saliva, hair, sweat, and exhaled breath. Tai et al.90 present a wearable sweatband that can detect the concentration of caffeine in sweat. They selected caffeine as an example methylxanthine drug that can lead to health problems such as coronary syndromes, hypertension and depression.
Alcohol
Human alcohol metabolism has been thoroughly studied. Alcohol is a commonly abused substance and assessing blood alcohol level is key for detecting individuals who cannot safely drive or for monitoring those with alcoholism. Studies have shown a high correlation between the concentrations of alcohol in sweat and blood, which enables continuous noninvasive blood alcohol monitoring to be achieved by monitoring sweat alcohol concentration91,92.
Heavy metals ion
Heavy metal ions are not ordinary human sweat metabolites, but the primary way to metabolize heavy metal ions is through sweat. Therefore, in the face of possible heavy metal ion poisoning, a wearable sweat sensor to monitor levels can provide an effective diagnostic tool.
Multichannel sweat composition analysis has become a relatively mature technology, but the perceptual parameters required differ according to the specific health monitoring task. For example, the target of diabetes monitoring could be glucose and the exercise-related targets could be lactic acid, sodium and potassium ions. Deciding on the appropriate parameter to monitor, however, depends on the medical data to provide sufficient theoretical analysis to not only identify the parameter, but also to provide quantitative data regarding normal levels. Unfortunately, medical knowledge may not be able to provide such information. Another possibility is to use portable, fully integrated wearable sweat sensors to monitor the human body, and then use data analysis to judge the correlation between sweat components and health goals. The challenges with this are that the targets that can be monitored are limited and screening accuracy with wearable sensors is unlikely to match that required of special medical devices. Therefore, in the case of insufficient monitoring accuracy, wearable sensors can still only provide rough trend information of health targets.
Sustainable energy supply system
For wearable sensors, long-term continuous monitoring is required to obtain sufficient data to accurately measure human health. Therefore, energy supply for the sensor is vital. Long-term monitoring requires the establishment of a complete data perception, storage, and calculation systems, all of which require energy. The most straightforward energy source is additional functional components, such as button batteries or small lithium batteries. However, traditional batteries are not light and thin enough to meet the needs of flexible wearables. Existing technologies include battery-less systems such as near-field communication (NFC) devices93,94 and flexible battery devices, such as supercapacitor flexible batteries26,95 and self-powered devices50,96.
A battery-free system is a simpler and more stable structure with a lower cost. NFC devices have performed well in studies93,94, because the passive data transmission mode fits well with the needs of battery-free systems. However, this acquisition mode cannot achieve continuous data monitoring. In addition, compared with other communication methods, such as Bluetooth and WiFi, its short interaction distance is limiting. Therefore, it is difficult for a battery-free system to meet the needs of continuous monitoring for an extended period.
Flexible batteries are currently the best technical option for traditional sensors. Intrinsically flexible batteries still have certain technical difficulties, but structurally flexible batteries realized by island structures are relatively easy to implement. Liu97 reviewed the recent development of flexible and stretchable lithium(Li)-ion batteries and supercapacitors. Li-ion batteries have high energy density, high power density and a long life cycle. Breakthroughs in flexibility, stretchability, long-term stability and microscale energy harvesting have been achieved through research on different materials, joint design, fabrication methods and integrated assembly. Wang et al.98 proposed an environmentally friendly and degradable biofuel cell that cleverly uses the kirigami process to design the device structure. It has good electrochemical performance and mechanical flexibility without sacrificing capacity, providing different application possibilities for wearable devices. Peng et al.99 proposed woven lithium-ion fiber batteries, which can be prepared in large quantities and have good stability. These batteries have a capacity retention that reaches 90.5% after 500 charge-discharge cycles, comparable to commercial batteries such as pouch cells. Over 80% capacity was maintained after bending the fiber for 100,000 cycles.
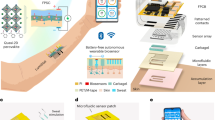
Given the changing nature of batteries, some researchers prefer to find solutions without batteries. The most popular direction is a self-supplied energy system associated with environmental factors. By collecting energy from the environment, the sensor becomes self-sufficient. With proper design, self-powered flexible sensors can meet the long-term monitoring goals of most applications. The correlation between the production of sweat and environmental factors makes it especially feasible to match the self-supply of the environment. Figure 6 shows several energy sources and their corresponding self-powered device design.
a Self-powered devices, a wearable BFC harvester powered by finger touch. Ref. 100, Copyright 2021, Advanced Functional Materials. b The FTENG for harvesting energy from human motion. Ref. 103 Copyright 2015, Biosensors and Bioelectronics. c A flexible and textile-based self-powered sweat sensor based on TENG. Ref. 105, Copyright 2018, Nano Energy. d A scalable, flexible magnetoelectrical clothing generator. Ref. 111, Copyright 2018, Nano Energy.
Some researchers use biofuel cells to power the sensor system100,101,102,103. The commonly used battery type is a lactic acid fuel cell, and lactic acid in sweat can meet the needs of low-power sensors. Wearable biofuel cells (w-BFCs) generate green electricity from energy-dense carbon-neutral fuels via highly efficient bioelectrochemical reactions, delivering good biocompatibility, remarkable environmental sustainability, and exceptional capacity for miniaturization104. Therefore, biofuel cells are one of the most suitable components of self-powered sweat sensors. Sweat contains a variety of small molecular organic substances, such as lactate and glucose, and these substances can be used as energy sources for biofuel cells. A major advantage of the biofuel cell is that it can use the very substances the sensor is detecting, reducing the system’s complexity and ensuring its stability.
Active development of flexible self-supply systems has provided more options for powering flexible sweat sensor. Wearable self-power generation devices include triboelectric nanogenerators (TENG), piezoelectric nanogenerators (PENG) and wearable thermoelectric generators (WTEG)105. Typically, sweat production is accompanied by higher exercise intensity or ambient temperature, providing an energy source for various wearable self-powered devices that can, in turn be used for sweat sensor monitoring.
Xia et al.106 reviewed the application of TENG in the biomedical field, noting that TENG has become the power source for many commercialized medical sensors. TENG devices reported in the literature can supply energy for up to 72 h. While existing self-powered systems can meet the energy requirements of the body surface sensor, research on design is required to achieve a stable supply of energy. Yang et al.107 reviewed the progress of combining textiles with TENG. For textile wearable TENG, lifespan is evaluated in terms of factors such as washability and stretchability. As long as the structure was not damaged, its long-term stability was satisfactory. However, most of the energy sources for TENG fabrics are low-frequency human motion and transformation of the motion to energy for a sensor remains to be studied. Tang et al.108 reviewed the development of paper-based TENGs. Paper is cost effective, flexible, lightweight and biocompatible, making it a promising substrate that can serve as an uesful frame and a suitable material for integrated energy converting devices. Unfortunately, current paper-based self-powered sensors still face many problems, including limited mechanical stability, moisture resistance and lifespan. Further development and research are needed. Fischer et al.109 demonstrated a self-powered paper-based biosensor for glucose monitoring. Their device operating mechanism is based on a glucose/oxygen enzymatic fuel cell. Its advantage lies in its low cost and strong portability.
TENG is also one of the most popular devices in use for self-powered wearable systems. Because its energy supply requires mechanical movement, the relationship between exercise and sweat in human health monitoring makes it a good alternative. Textile TENG is used for monitoring sweat by changing the triboelectric characteristics. Jao et al.110 develop a textile-based TENG to harvest biomechanical energy from human motion and biofluids. Their TENG can be further developed into various kinds of self-powered healthcare sensors for humidity, sweat, and gait phase detection. Although the potential has been verified, more specific selectivity and stability research is required for further exploration. Wang et al.111 demonstrate a scalable-manufactured fabrication of magnetoelectrical clothing generator that enables power generation through arm-swinging movement. The nature of using electromagnetic energy still requires motion, but this innovative idea expands the potential of self-powered devices.
In summary, the use of biofuel cells as a self-powered technology for sweat sensing has been studied and needs to now be tested in the context of applied use. Self-powered devices such as TENG, PENG, and WTEG all show potential in the field of sweat sensors. The use of environmental energy, such as solar or wind energy, is another direction that should be explored. It is worth noting that a hybrid energy supply could further enhance the stability of energy supply112. The coordination of multiple energy supply systems can cover a broader range of sensor device usage requirements and avoid the limitations of a single self-supply system. However, this also brings about challenges in terms of energy management and the complexity of structure.
Intelligent monitoring and prediction analysis
The goal of using wearable sensors is to obtain data to monitor human health. However, due to current technology and data limitations, it is still difficult to directly obtain critical parameters that affect the state of the human body. Most researchers currently study simple heart rate, electrocardiograms, blood oxygen levels, and other simple parameters. The human body is a complex system and a variety of monitorable data is required to establish a comprehensive model of its health.
Artificial intelligence (AI) can be integrated into wearable sensors to improve the applied performance of wearable sensors for health monitoring. The AI biosensor is a rapidly growing area of research. This system contains three main elements: information collection, signal conversion and AI-data processing52. At present, there is no clear relationship between sweat signals and human health parameters; yet many scholars have studied the relationship between sweat sugar and blood sugar. Therefore, it may be possible to use machine learning to calculate and build their relationship model to obtain interpretable results.
Figure 7 shows the information exchange between signal monitoring and data processing113,114. The sensors acquire signals and send them to the analysis platform. Data processing has three components: data preprocessing, machine learning algorithms and healthcare models. Data preprocessing refers to the reorganization of complex data into a standardized dataset for analysis. Machine learning establishes the relationship between health characteristics and monitored signals. More specifically, this component can be divided into several stages115. The raw data may include various measured sweat parameters, temperature, heart rate, blood oxygen, blood pressure, and other basic signals. After preprocessing, such as labeling, filtering, segmentation, feature extraction and features selection, key characteristic parameters are retained, and then basic signals and complex health state models can be established by classification. The third component of data processing, healthcare models, is the output from this machine learning model that can provide interpretable results in the medical field.
a Touch-based fingertip sweat sensor with an algorithm that can provide personal blood glucose. Ref. 113, Copyright 2021, American Chemical Society. b Correlation of data acquired from biofuel cell–based glucose and lactate sweat sensors with that acquired from blood glucose and lactate meters. Ref. 114 Copyright 2019, Science Advances.
Existing machine learning algorithms have been used to analyze human physical signals, such as electrocardiogram and heart rate analysis. For example, Quan et al.116 used contact lenses to monitor the glucose content in the tears of diabetics, performed data processing and then combined it with historical data to analyze and predict blood glucose changes (algorithms). When future low glucose levels are predicted, patients are reminded to take interventions (healthcare models).
Machine learning (ML) is defined by computational algorithms designed to extract the required information from data through different probabilistic learning paradigms117. Traditionally, medical research aims to combine ML algorithms with clinical data (for example, age, gender, physical examination results, symptoms, vital signs, laboratory values, imaging variables, test result values) to predict clinical outcomes or find predictive variables for the relationship between data and clinical outcomes. Although wearable device data can be integrated with broader patient data sets to provide additional background information about the patient, raw sensor data can also be used as direct input to ML algorithms to predict clinical outcomes (e.g., physiological or pathophysiological states) or to extract meaningful data features. The general difference between ML algorithms for each of these purposes is based on the type of training data used (labeled or unlabeled) and whether the goal is to predict specific results or learn patterns from the data. These scenarios include supervised (e.g., logistic regression118,119, naive Bayes120,121, decision tree, nearest neighbor120,122, random forest123, discriminant analysis, support vector machine (SVM)120,124, neural network), unsupervised (e.g., clustering algorithm125,126, principal components analysis118,127) or semi-supervised learning paradigms128. A third class of ML algorithms includes those that perform integrated goals; these are algorithms that weigh multiple individual classifiers and combine them to obtain a classifier that is superior to the individual starting classifiers (e.g., naive Bayesian optimal classifier, guided aggregation or bagging, boosting, Bayesian model/parameter averaging, model buckets, stacking)129.
The analysis of blood glucose is a relatively mature applied model, for which physiological model-based algorithms and data-driven algorithms are used. The physiological model of the human body depends on many factors (e.g., gender, age, weight, meals, external environment). In the classic biological model, glucose, insulin, and glucagon must be considered comprehensively. At the same time, change in blood sugar levels has a certain degree of hysteresis, and the change of glucose in sweat depends on, and lags behind, the blood sugar, making it difficult to establish the model. The complexity of the human body further makes the modeling accuracy insufficient. In recent years, data-driven algorithms have been increasingly applied to various model predictions and this can build models even in the case of complex and unknown relationships of information that are more inline with reality.
Discussion
Wearable sweat sensors are widely used in healthcare for monitoring and diagnostic purposes. This article focuses on the development of long-term sweat sensors and reviews recent research on sweat collection, sensor energy supply, and data analysis. Different technologies proposed to achieve longer-term, on-site, real-time monitoring and to improve the integrity and practicability of the sensors are reviewed.
Regarding sweat collection, a shift has occurred from extensive uncontrolled collection to more precise and controlled collection. However, problems of low collection efficiency and poor results still exist. Contamination on the skin surface is a key challenge affecting the accuracy of the device. In addition, specific differences occur in the composition of sweat obtained by different methods used to stimulate perspiration; exploring and identifying these differences is a key area for future research.
Specifically, attention is needed regarding materials used for sweat collection. Traditional microfluidic technology remains a major contender for sweat collection but faces the problem of processing efficiency. The rise of 3D printing may be a solution to this problem. In addition, paper bases and fabrics show considerable application potential. They have high hydrophilicity, a natural advantage for sweat collection, but control remains a problem. In summary, the key lies in the comprehensive consideration of material properties and structural characteristics to design devices that can meet long-term sweat collection and control.
Regarding self-supplied energy for sensors, sweat is a biological fluid with an abundance of small molecules that make it a potential choice for biofuel cells. Both lactic acid and glucose have been demonstrated as potential fuels for the self-powered sensor. At the same time, TENG, which has received much attention recently, has also shown its potential in sweat sensing and monitoring. In addition, because sweat production is strongly coupled to movement and temperature, PENG (piezoelectric nanogenerators) and WTNG (wearable thermoelectric generators) are energy supplies that should be further explored. Current wearable self-powered devices have good compatibility with wearable sweat sensors. Various self-powered technologies provide more possibilities for long-term continuous monitoring of wearable sweat sensors. However, the problem is not only the energy supply of the device itself but also that a single energy supply element cannot meet the demands faced with scenarios that require data transmission. This suggests that hybrid energy supply components and optimized energy management systems are worthy of attention.
Finally, AI biosensors are a promising research direction. The current analysis of sweat data is limited to visual presentation or simple statistical analysis. While limitations remain in the accuracy of existing sensors, obtaining data is only one component of the original intention of sweat sensing and health monitoring. Thoroughly characterizing the relationship between sweat composition and health is the ultimate desired result. Careful consideration must be given to the risk with intelligent monitoring of personal privacy being maintained, especially with noninvasive sweat analysis, which increases the chance of personal information theft. Convenient personal information monitoring is worth looking forward to, but standard ethics and data security are also worthy of attention.
As a summary, the sweat sensor is a potential type of wearable sensor placed on the body surface that can detect human health under non-invasive conditions. This paper summarizes and explores the factors affecting long-term stability in various stages of sweat sensor application. First, for the collection method of sweat, the application of hydrophilic and hydrophobic structures and materials is reviewed. Next, the abundant ions, small molecules and other substances in sweat were reviewed. In addition, the energy supply needs of the sensor system is emphasized and the application prospects of self-powered sensors are discussed. Finally, research progress in combining machine learning techniques and sweat sensing are highlighted. The advantages and disadvantages of various technologies are considered, and future research directions are proposed.
References
Windmiller, J. R. & Wang, J. Wearable Electrochemical Sensors and Biosensors: A Review. Electroanalysis 25, 29–46 (2013).
Chen, Y. et al. Flex. Inorg. Bioelectron. npj Flex. Electron. 4, 1–20 (2020).
Wilson, E. K. Wearable Sweat Sensors. Engineering 5, 359–360 (2019).
Bhide, A., Muthukumar, S. & Prasad, S. CLASP (Continuous lifestyle awareness through sweat platform): A novel sensor for simultaneous detection of alcohol and glucose from passive perspired sweat. Biosens. Bioelectron. 117, 537–545 (2018).
Nyein, H. Y. Y. et al. A wearable patch for continuous analysis of thermoregulatory sweat at rest. Nat. Commun. 12, 1823 (2021).
Lou, Z., Wang, L., Jiang, K., Wei, Z. & Shen, G. Reviews of wearable healthcare systems: Materials, devices and system integration. Mater. Sci. Eng. R. Rep. 140, 100523 (2020).
Burklund, A., Tadimety, A., Nie, Y., Hao, N. & Zhang, J. X. J. Advances in diagnostic microfluidics. in Advances in clinical chemistry 95 1–72 (Elsevier Inc., 2020).
Seo, S. E. et al. Smartphone with optical, physical, and electrochemical nanobiosensors. J. Ind. Eng. Chem. 77, 1–11 (2019).
Ray, T. R. et al. Bio-integrated wearable systems: A comprehensive review. Chem. Rev. 119, 5461–5533 (2019).
Sempionatto, J. R., Jeerapan, I., Krishnan, S. & Wang, J. Wearable Chemical Sensors: Emerging Systems for On-Body Analytical Chemistry. Anal. Chem. 92, 378–396 (2019).
Chung, M., Fortunato, G. & Radacsi, N. Wearable flexible sweat sensors for healthcare monitoring: A review. J. R. Soc. Interface 16, 20190217 (2019).
Li, S., Ma, Z., Cao, Z., Pan, L. & Shi, Y. Advanced Wearable Microfluidic Sensors for Healthcare Monitoring. Small 16, 1903822 (2020).
Xu, J., Fang, Y. & Chen, J. Wearable biosensors for non-invasive sweat diagnostics. Biosensors 11, 245 (2021).
Cuartero, M., Parrilla, M. & Crespo, G. Wearable Potentiometric Sensors for Medical Applications. Sensors 19, 363 (2019).
Liu, Y., Pharr, M. & Salvatore, G. A. Lab-on-Skin: A Review of Flexible and Stretchable Electronics for Wearable Health Monitoring. ACS Nano 11, 9614–9635 (2017).
Jo, S., Sung, D., Kim, S. & Koo, J. A review of wearable biosensors for sweat analysis. Biomed. Eng. Lett. 11, 117–129 (2021).
Upasham, S., Churcher, N. K. M., Rice, P. & Prasad, S. Sweating out the Circadian Rhythm: A Technical Review. ACS Sens. 6, 659–672 (2021).
Klimuntowski, M., Alam, M. M., Singh, G. & Howlader, M. M. R. Electrochemical Sensing of Cannabinoids in Biofluids: A Noninvasive Tool for Drug Detection. ACS Sens. 5, 620–636 (2020).
Oktavius, A. K. et al. Fully-Conformable Porous Polyethylene Nanofilm Sweat Sensor for Sports Fatigue. IEEE Sens. J. 21, 8861–8867 (2021).
Pali, M. et al. Tracking metabolic responses based on macronutrient consumption: A comprehensive study to continuously monitor and quantify dual markers (cortisol and glucose) in human sweat using WATCH sensor. Bioeng. Transl. Med. 6, e10241 (2021).
Lee, J. W. et al. Soft, thin skin-mounted power management systems and their use in wireless thermography. Proc. Natl Acad. Sci. USA. 113, 6131–6136 (2016).
Yeung, K. K. et al. Recent Advances in Electrochemical Sensors for Wearable Sweat Monitoring: A Review. IEEE Sens. J. 21, 14522–14539 (2021).
Kaya, T. et al. Wearable Sweat Sensors: Background and Current Trends. Electroanalysis 31, 411–421 (2019).
Bandodkar, A. J., Jeang, W. J., Ghaffari, R. & Rogers, J. A. Wearable Sensors for Biochemical Sweat Analysis. Annu. Rev. Anal. Chem. 12, 1–22 (2019).
Kwon, J. Y., Park, H. W., Park, Y., Bin & Kim, N. Potentials of additive manufacturing with smart materials for chemical biomarkers in wearable applications. Int. J. Precis. Eng. Manuf. - Green. Technol. 4, 335–347 (2017).
Manjakkal, L., Yin, L., Nathan, A., Wang, J. & Dahiya, R. Energy Autonomous Sweat-Based Wearable Systems. Adv. Mater. 33, 2100899 (2021).
Sekar, M. et al. Review—Towards Wearable Sensor Platforms for the Electrochemical Detection of Cortisol. J. Electrochem. Soc. 167, 067508 (2020).
Matzeu, G., Florea, L. & Diamond, D. Advances in wearable chemical sensor design for monitoring biological fluids. Sensors Actuators. B Chem. 211, 403–418 (2015).
McCaul, M., Glennon, T. & Diamond, D. Challenges and opportunities in wearable technology for biochemical analysis in sweat. Curr. Opin. Electrochem 3, 46–50 (2017).
Yang, Y. & Gao, W. Wearable and flexible electronics for continuous molecular monitoring. Chem. Soc. Rev. 48, 1465–1491 (2019).
Jin, H., Abu-Raya, Y. S. & Haick, H. Advanced Materials for Health Monitoring with Skin-Based Wearable Devices. Adv. Healthc. Mater. 6, 1700024 (2017).
Li, G. & Wen, D. Wearable biochemical sensors for human health monitoring: sensing materials and manufacturing technologies. J. Mater. Chem. B 8, 3423–3436 (2020).
Fan, R. & Andrew, T. L. Perspective—Challenges in Developing Wearable Electrochemical Sensors for Longitudinal Health Monitoring. J. Electrochem. Soc. 167, 037542 (2020).
Gao, W., Brooks, G. A. & Klonoff, D. C. Wearable physiological systems and technologies for metabolic monitoring. J. Appl. Physiol. 124, 548–556 (2018).
Yokus, B. M. A. & Daniele, M. A. Integrated non-invasive biochemical and biophysical sensing systems for health and performance monitoring: A systems perspective. Biosens. Bioelectron. 184, 113249 (2021).
Wang, X., Liu, Z. & Zhang, T. Flexible Sensing Electronics for Wearable/Attachable Health Monitoring. Small 13, 1602790 (2017).
Hatamie, A. et al. Review—Textile Based Chemical and Physical Sensors for Healthcare Monitoring. J. Electrochem. Soc. 167, 037546 (2020).
Kim, J., Campbell, A. S., de Ávila, B. E. F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
Ray, T. et al. Soft, skin-interfaced wearable systems for sports science and analytics. Curr. Opin. Biomed. Eng. 9, 47–56 (2019).
Ye, S., Feng, S., Huang, L. & Bian, S. Recent Progress in Wearable Biosensors: From Healthcare Monitoring to Sports Analytics. Biosensors 10, 205 (2020).
Kim, S. et al. Soft, skin-interfaced microfluidic systems with integrated immunoassays, fluorometric sensors, and impedance measurement capabilities. Proc. Natl Acad. Sci. USA. 117, 27906–27915 (2020).
Wang, L., Xu, T. & Zhang, X. Multifunctional conductive hydrogel-based flexible wearable sensors. TrAC - Trends Anal. Chem. 134, 116130 (2021).
Rabost-Garcia, G., Farré-Lladós, J. & Casals-Terré, J. Recent Impact of Microfluidics on Skin Models for Perspiration Simulation. Membr. (Basel) 11, 150 (2021).
Yu, H. & Sun, J. Sweat detection theory and fluid driven methods: A review. Nanotechnol. Precis. Eng. 3, 126–140 (2020).
Kalkal, A. et al. Recent advances in 3D printing technologies for wearable (bio)sensors. Addit. Manuf. 46, 102088 (2021).
Sarwar, M., Rodriguez, P. & Li, C. zhong. Sweat-Based in Vitro Diagnostics (IVD): From Sample Collection to Point-of-Care Testing (POCT). J. Anal. Test. 3, 80–88 (2019).
Heikenfeld, J. et al. Accessing analytes in biofluids for peripheral biochemical monitoring. Nat. Biotechnol. 37, 407–419 (2019).
Shetti, N. P. et al. Skin-Patchable Electrodes for Biosensor Applications: A Review. ACS Biomater. Sci. Eng. 6, 1823–1835 (2020).
Wen, F. et al. Advances in chemical sensing technology for enabling the next-generation self-sustainable integrated wearable system in the IoT era. Nano Energy 78, 105155 (2020).
Shitanda, I. & Tsujimura, S. Toward self-powered real-Time health monitoring of body fluid components based on improved enzymatic biofuel cells. JPhys Energy 3, 032002 (2021).
Rao, J. et al. Recent Progress in Self-Powered Skin. Sens. Sens. 19, 2763 (2019).
Jin, X., Liu, C., Xu, T., Su, L. & Zhang, X. Artificial intelligence biosensors: Challenges and prospects. Biosens. Bioelectron. 165, 112412 (2020).
Nguyen, B., Coelho, Y., Bastos, T. & Krishnan, S. Trends in human activity recognition with focus on machine learning and power requirements. Mach. Learn. Appl 5, 100072 (2021).
Witt, D. R., Kellogg, R. A., Snyder, M. P. & Dunn, J. Windows into human health through wearables data analytics. Curr. Opin. Biomed. Eng. 9, 28–46 (2019).
Morris, D. et al. Bio-sensing textile based patch with integrated optical detection system for sweat monitoring. Sensors Actuators. B Chem. 139, 231–236 (2009).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Heikenfeld, J. Non-invasive Analyte Access and Sensing through Eccrine Sweat: Challenges and Outlook circa 2016. Electroanalysis 28, 1242–1249 (2016).
Liu, C., Xu, T., Wang, D. & Zhang, X. The role of sampling in wearable sweat sensors. Talanta 212, 120801 (2020).
Tankasala, D. & Linnes, J. C. Noninvasive glucose detection in exhaled breath condensate. Transl. Res. 213, 1–22 (2019).
Emaminejad, S. et al. Autonomous sweat extraction and analysis applied to cystic fibrosis and glucose monitoring using a fully integrated wearable platform. Proc. Natl Acad. Sci. USA. 114, 4625–4630 (2017).
Kim, J. et al. Noninvasive Alcohol Monitoring Using a Wearable Tattoo-Based Iontophoretic-Biosensing System. ACS Sens. 1, 1011–1019 (2016).
Bandodkar, A. J. et al. Epidermal tattoo potentiometric sodium sensors with wireless signal transduction for continuous non-invasive sweat monitoring. Biosens. Bioelectron. 54, 603–609 (2014).
Son, J. et al. Cactus-Spine-Inspired Sweat-Collecting Patch for Fast and Continuous Monitoring of Sweat. Adv. Mater. 33, 2102740 (2021).
Dai, B. et al. Bioinspired Janus Textile with Conical Micropores for Human Body Moisture and Thermal Management. Adv. Mater. 31, 1904113 (2019).
He, X. et al. Flexible and Superwettable Bands as a Platform toward Sweat Sampling and Sensing. Anal. Chem. 91, 4296–4300 (2019).
He, X. et al. Integrated Smart Janus Textile Bands for Self-Pumping Sweat Sampling and Analysis. ACS Sens. 5, 1548–1554 (2020).
He, X., Fan, C., Xu, T. & Zhang, X. Biospired Janus Silk E-Textiles with Wet-Thermal Comfort for Highly Efficient Biofluid Monitoring. Nano Lett. 21, 8880–8887 (2021).
Choi, J. et al. Soft, skin-mounted microfluidic systems for measuring secretory fluidic pressures generated at the surface of the skin by eccrine sweat glands. Lab Chip 17, 2572–2580 (2017).
Choi, J., Kang, D., Han, S., Kim, S. B. & Rogers, J. A. Thin, Soft, Skin-Mounted Microfluidic Networks with Capillary Bursting Valves for Chrono-Sampling of Sweat. Adv. Healthc. Mater. 6, 1601355 (2017).
Samson, C. & Koh, A. Stress Monitoring and Recent Advancements in Wearable Biosensors. Front. Bioeng. Biotechnol. 8, 1037 (2020).
Wang, L., Xu, T., He, X. & Zhang, X. Flexible, self-healable, adhesive and wearable hydrogel patch for colorimetric sweat detection. J. Mater. Chem. C. 9, 14938–14945 (2021).
Yang, Y. & Gao, W. Wearable pH sensing beyond the Nernst limit. Nat. Electron. 1, 580–581 (2018).
Nyein, H. Y. Y. et al. A Wearable Electrochemical Platform for Noninvasive Simultaneous Monitoring of Ca2+ and pH. ACS Nano 10, 7216–7224 (2016).
Zhai, Q. et al. Vertically Aligned Gold Nanowires as Stretchable and Wearable Epidermal Ion-Selective Electrode for Noninvasive Multiplexed Sweat Analysis. Anal. Chem. 92, 4647–4655 (2020).
O’Sullivan, B. P. & Freedman, S. D. Cystic fibrosis. Lancet 373, 1891–1904 (2009).
Baker, L. B. Sweating Rate and Sweat Sodium Concentration in Athletes: A Review of Methodology and Intra/Interindividual Variability. Sport. Med. 47, 111–128 (2017).
Parrilla, M., Cuartero, M. & Crespo, G. A. Wearable potentiometric ion sensors. TrAC - Trends Anal. Chem. 110, 303–320 (2019).
Keene, S. T. et al. Wearable Organic Electrochemical Transistor Patch for Multiplexed Sensing of Calcium and Ammonium Ions from Human Perspiration. Adv. Healthc. Mater. 8, e1901321 (2019).
Jia, W. et al. Electrochemical tattoo biosensors for real-time noninvasive lactate monitoring in human perspiration. Anal. Chem. 85, 6553–6560 (2013).
Alvear-Ordenes, I., García-López, D., De Paz, J. A. & González-Gallego, J. Sweat Lactate, Ammonia, and Urea in Rugby Players. Int. J. Sports Med. 26, 632–637 (2005).
Lamont, L. S. Sweat lactate secretion during exercise in relation to women’s aerobic capacity. J. Appl. Physiol. 62, 194–198 (1987).
Wang, R., Zhai, Q., An, T., Gong, S. & Cheng, W. Stretchable gold fiber-based wearable textile electrochemical biosensor for lactate monitoring in sweat. Talanta 222, 121484 (2021).
Zhou, W. et al. Recent advances in flexible sweat glucose biosensors. J. Phys. D. Appl. Phys. 54, 423001 (2021).
Yunos, M. F. A. M. & Nordin, A. N. Non-invasive glucose monitoring devices: A review. Bull. Electr. Eng. Inform. 9, 2609–2618 (2020).
Zhao, Y. et al. Highly Stretchable and Strain-Insensitive Fiber-Based Wearable Electrochemical Biosensor to Monitor Glucose in the Sweat. Anal. Chem. 91, 6569–6576 (2019).
Yang, Y. et al. A laser-engraved wearable sensor for sensitive detection of uric acid and tyrosine in sweat. Nat. Biotechnol. 38, 217–224 (2020).
Russell, E., Koren, G., Rieder, M. & Van Uum, S. H. M. The detection of cortisol in human sweat: Implications for measurement of cortisol in hair. Ther. Drug Monit. 36, 30–34 (2014).
Raul, J. S., Cirimele, V., Ludes, B. & Kintz, P. Detection of physiological concentrations of cortisol and cortisone in human hair. Clin. Biochem. 37, 1105–1111 (2004).
Torrente-Rodríguez, R. M. et al. Investigation of Cortisol Dynamics in Human Sweat Using a Graphene-Based Wireless mHealth System. Matter 2, 921–937 (2020).
Tai, L. C. et al. Methylxanthine Drug Monitoring with Wearable Sweat Sensors. Adv. Mater. 30, 1707442 (2018).
Gamella, M. et al. A novel non-invasive electrochemical biosensing device for in situ determination of the alcohol content in blood by monitoring ethanol in sweat. Anal. Chim. Acta 806, 1–7 (2014).
Davis-Martin, R. E., Alessi, S. M. & Boudreaux, E. D. Alcohol Use Disorder in the Age of Technology: A Review of Wearable Biosensors in Alcohol Use Disorder Treatment. Front. Psychiatry 12, 642813 (2021).
Cao, Z. et al. Near-Field Communication. Sens. Sens. 19, 3947 (2019).
Xu, G. et al. Smartphone-based battery-free and flexible electrochemical patch for calcium and chloride ions detections in biofluids. Sens. Actuators B Chem. 297, 126743 (2019).
Bandodkar, A. J. et al. Soft, stretchable, high power density electronic skin-based biofuel cells for scavenging energy from human sweat. Energy Environ. Sci. 10, 1581–1589 (2017).
Reid, R. C. & Mahbub, I. Wearable self-powered biosensors. Curr. Opin. Electrochem 19, 55–62 (2020).
Liu, W., Song, M.-S., Kong, B. & Cui, Y. Flexible and Stretchable Energy Storage: Recent Advances and Future Perspectives. Adv. Mater. 29, 1603436 (2017).
Wang, Z. et al. Fully transient stretchable fruit‐based battery as safe and environmentally friendly power source for wearable electronics. EcoMat 3, e12073 (2021).
He, J. et al. Scalable production of high-performing woven lithium-ion fibre batteries. Nature 597, 57–63 (2021).
Yin, L. et al. A passive perspiration biofuel cell: High energy return on investment. Joule 5, 1888–1904 (2021).
Heo, S. Y. et al. Wireless, battery-free, flexible, miniaturized dosimeters monitor exposure to solar radiation and to light for phototherapy. Sci. Transl. Med. 10, eaau1643 (2018).
Han, S. et al. Battery-free, wireless sensors for full-body pressure and temperature mapping. Sci. Transl. Med. 10, eaan4950 (2018).
Song, Y. et al. Wireless battery-free wearable sweat sensor powered by human motion. Sci. Adv. 6, eaay9842 (2020).
Wu, H., Zhang, Y., Kjøniksen, A. L., Zhou, X. X. & Zhou, X. X. Wearable Biofuel Cells: Advances from Fabrication to Application. Adv. Funct. Mater. 2103976, 1–38 (2021).
Yuan, J. & Zhu, R. A fully self-powered wearable monitoring system with systematically optimized flexible thermoelectric generator. Appl. Energy 271, 115250 (2020).
Xia, X., Liu, Q., Zhu, Y. & Zi, Y. Recent advances of triboelectric nanogenerator based applications in biomedical systems. EcoMat 2, e12049 (2020).
Yang, B., Xiong, Y., Ma, K., Liu, S. & Tao, X. Recent advances in wearable textile‐based triboelectric generator systems for energy harvesting from human motion. EcoMat 2, e12054 (2020).
Tang, Q., Guo, H., Yan, P. & Hu, C. Recent progresses on paper‐based triboelectric nanogenerator for portable self‐powered sensing systems. EcoMat 2, e12060 (2020).
Fischer, C., Fraiwan, A. & Choi, S. A 3D paper-based enzymatic fuel cell for self-powered, low-cost glucose monitoring. Biosens. Bioelectron. 79, 193–197 (2016).
Jao, Y. T. et al. A textile-based triboelectric nanogenerator with humidity-resistant output characteristic and its applications in self-powered healthcare sensors. Nano Energy 50, 513–520 (2018).
Wang, R. et al. Magnetoelectrical Clothing Generator for High-Performance Transduction from Biomechanical Energy to Electricity. Adv. Funct. Mater. 2107682, 1–10 (2021).
García Núñez, C., Manjakkal, L. & Dahiya, R. Energy autonomous electronic skin. npj Flex. Electron 3, 1 (2019).
Sempionatto, J. R., Moon, J.-M. & Wang, J. Touch-Based Fingertip Blood-Free Reliable Glucose Monitoring: Personalized Data Processing for Predicting Blood Glucose Concentrations. ACS Sens. 6, 1875–1883 (2021).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Kamiŝalić, A., Fister, I., Turkanović, M. & Karakatiĉ, S. Sensors and functionalities of non-invasive wrist-wearable devices: A review. Sens. (Switz.) 18, 1714 (2018).
Quan, T. M. et al. AI-Based Edge-Intelligent Hypoglycemia Prediction System Using Alternate Learning and Inference Method for Blood Glucose Level Data with Low-periodicity. in 2019 IEEE International Conference on Artificial Intelligence Circuits and Systems (AICAS) 201–206 (IEEE, 2019). https://doi.org/10.1109/AICAS.2019.8771604.
Yu, Y. et al. An automatic and non-invasive physical fatigue assessment method for construction workers. Autom. Constr. 103, 1–12 (2019).
Bordbar, M. M., Barzegar, H., Tashkhourian, J., Bordbar, M. & Hemmateenejad, B. A non-invasive tool for early detection of acute leukemia in children using a paper-based optoelectronic nose based on an array of metallic nanoparticles. Anal. Chim. Acta 1141, 28–35 (2021).
Kumar, P. M. & Devi Gandhi, U. A novel three-tier Internet of Things architecture with machine learning algorithm for early detection of heart diseases. Comput. Electr. Eng. 65, 222–235 (2018).
Otoom, M., Otoum, N., Alzubaidi, M. A., Etoom, Y. & Banihani, R. An IoT-based framework for early identification and monitoring of COVID-19 cases. Biomed. Signal Process. Control 62, 102149 (2020).
Syed, L., Jabeen, S., S., M. & Alsaeedi, A. Smart healthcare framework for ambient assisted living using IoMT and big data analytics techniques. Futur. Gener. Comput. Syst. 101, 136–151 (2019).
Veeralingam, S., Khandelwal, S. & Badhulika, S. AI/ML-Enabled 2-D - RuS 2 Nanomaterial-Based Multifunctional, Low Cost, Wearable Sensor Platform for Non-Invasive Point of Care Diagnostics. IEEE Sens. J. 20, 8437–8444 (2020).
Chen, A. et al. Machine-learning enabled wireless wearable sensors to study individuality of respiratory behaviors. Biosens. Bioelectron. 173, 112799 (2021).
Kanoga, S., Hoshino, T. & Asoh, H. Semi-supervised style transfer mapping-based framework for sEMG-based pattern recognition with 1- or 2-DoF forearm motions. Biomed. Signal Process. Control 68, 102817 (2021).
Tummers, J., Catal, C., Tobi, H., Tekinerdogan, B. & Leusink, G. Coronaviruses and people with intellectual disability: an exploratory data analysis. J. Intellect. Disabil. Res. 64, 475–481 (2020).
Gondalia, A. et al. IoT-based Healthcare Monitoring System for War Soldiers using. Mach. Learn. Procedia Comput. Sci. 133, 1005–1013 (2018).
Tang, B. et al. Kinetic investigation into pH-dependent color of anthocyanin and its sensing performance. Dye. Pigment. 170, 107643 (2019).
Lecun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Aziz, O. et al. Validation of accuracy of SVM-based fall detection system using real-world fall and non-fall datasets. PLoS One 12, e0180318 (2017).
Acknowledgements
This study was financially supported by the State Key Laboratory of Mechanical System and Vibration Project (Grant no. MSVZD202108) and the open project of National Research Center for Translational Medicine, Shanghai (Project no. TMSK-2021-146). We also thanks to the Base for Interdisciplinary Innovative Talent Training.
Author information
Authors and Affiliations
Contributions
P.F.Y., G.F.W., F.W.H. and Z.N.Z. conceptualized the work. P.F.Y. wrote the original draft; A.L. and Z.N.Z. reviewed and edited the manuscript. G.F.W. and Z.N.Z. provided overall supervision of the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, P., Wei, G., Liu, A. et al. A review of sampling, energy supply and intelligent monitoring for long-term sweat sensors. npj Flex Electron 6, 33 (2022). https://doi.org/10.1038/s41528-022-00165-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41528-022-00165-9