Abstract
Use of gonadotropin-releasing hormone (GnRH) agonists has been widely adopted to provide reversible ovarian function suppression for pre-menopausal breast cancer patients who are also receiving aromatase inhibitor or tamoxifen therapy based on results of 25 randomized trials representing almost 15,000 women demonstrating a survival benefit with this approach. Past clinical trials designed to establish the efficacy of GnRH agonists have monitored testosterone in the prostate cancer setting and estradiol in the breast cancer setting. We explore the merits of various biomarkers including estradiol, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) and their utility for informing GnRH agonist treatment decisions in breast cancer. Estradiol remains our biomarker of choice in ensuring adequate ovarian function suppression with GnRH agonist therapy among pre-menopausal women with breast cancer. We recommend future trials to continue to focus on estradiol levels as the primary endpoint, as they have in the past.
Similar content being viewed by others
Introduction
Approximately 20% of breast cancers in the United States occur in pre-menopausal women1,2. Preservation of ovarian function is an important consideration among these young patients who would have otherwise had many viable childbearing years3. Ovarian function suppression added to endocrine therapy is one strategy used to prevent premature ovarian insufficiency, impaired fertility, and early menopause while improving the menses recovery rate and potential for pregnancy after chemotherapy3,4,5. Premature ovarian insufficiency was averted among the majority patients ≤ 40 years of age in the Ovarian Protection Trial in Oestrogen Non-responsive Premenopausal Breast Cancer Patients Receiving Adjuvant or Neo-adjuvant Chemotherapy (OPTION) study6,7. Successful post-treatment pregnancies have been reported in the Prevention of Menopause Induced by Chemotherapy: a Study in Early Breast Cancer Patients—Gruppo Italiano Mammella 6 (PROMISE-GIM6) and Prevention of Early Menopause Study (POEMS) studies, with no detrimental effects on survival8,9. The Pregnancy Outcome and Safety of Interrupting Therapy for Women with Endocrine Responsive Breast Cancer (POSITIVE) trial confirmed that patients attempting pregnancy by temporarily interrupting their endocrine therapy did not increase their risk of breast cancer recurrence in the short-term10.
Results from the Suppression of Ovarian Function Trial (SOFT) and Tamoxifen and Exemestane Trial (TEXT) trials, and subsequently the Addition of Ovarian Suppression to Tamoxifen in Young Women With Hormone-Sensitive Breast Cancer Who Remain Premenopausal or Regain Vaginal Bleeding After Chemotherapy (ASTRRA) trial, have indicated that the addition of ovarian function suppression to tamoxifen or an aromatase inhibitor (AI) improves disease-free survival, freedom from distant recurrence, and overall survival11,12,13. A recent meta-analysis of 25 randomized trials using individual patient data from almost 15,000 women confirmed reduction of their 15-year risk of recurrence and death from breast cancer14. Consistent 10-year results were found previously in a systematic literature review of 15 studies representing over 11,000 pre-menopausal women with breast cancer15. This is particularly true among high-risk patients who remain pre-menopausal after chemotherapy, or patients 40 years and younger who have a higher risk of recurrence16. The most substantial benefits were observed when ovarian function suppression is used in combination with an AI, although ovarian function suppression with tamoxifen remains an option, and either of these options conveys a greater benefit than tamoxifen alone in patients with high-risk tumors16.
Ovarian function suppression can be achieved with gonadotropin-releasing hormone (GnRH) agonists, which suppress the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) from the pituitary gland by downregulating the GnRH receptors17. This reduces the primary method of circulating estradiol production from the ovaries, essentially inducing reversible medical ablation (Fig. 1)17. Once GnRH agonist therapy is discontinued and estradiol, FSH, and LH return to normal levels, fertility returns as follicular maturation resumes17. The reversibility of medical ovarian function suppression is an advantage over the irreversibility of surgical or radiation ablation, particularly among pre-menopausal women who have the potential for many remaining childbearing years or a desire for future ovarian function17. The question arises when designing new studies evaluating GnRH agonists as to which biomarker is the most meaningful surrogate for ovarian function suppression among patients with pre-menopausal estrogen receptor-positive (ER+) breast cancer?
(adapted from Lu et al. 2). Upon initiation of GnRH agonist therapy, it mimics endogenous LHRH and stimulates the production of FSH and LH from the pituitary, resulting in an initial surge of E2 from the ovaries. With long-term administration of GnRH agonists, the LHRH receptors are downregulated and desensitized, resulting in reduced ovarian hormone production. Abbreviations: GnRH gonadotropin-releasing hormone, LHRH luteinizing hormone-releasing hormone, FSH follicle-stimulating hormone, LH luteinizing hormone, E2 estradiol, ER estrogen receptor. Graphics used with permission under a Creative Commons Attribution 3.0 unported license from Servier Medical Art by Servier, available at smart.servier.com.
Utility of biomarkers in pre-menopausal ER+ breast cancer
Approximately 21% to 71% of young women with breast cancer are at risk for early menopause due to chemotherapy18. The ovarian function of half of these patients resume within 6 months of completing their chemotherapy19. The ovarian recovery can be slower in one of every four patients, with functionality returning 6–24 months after completing their chemotherapy19. Eligibility for GnRH agonist therapy in the ASTRRA trial was based on detection of ovarian recovery during 2 years of tamoxifen therapy19. The Breast Cancer in Young Women (BCY5) guidelines also support the addition of a GnRH agonist to tamoxifen if ovarian function resumes within 2 years of chemotherapy treatment20. Among patients who develop treatment-induced amenorrhea, it has been suggested that biomarkers such as estradiol (E2), follicle-stimulating hormone (FSH), and luteinizing hormone (LH) be monitored serially to predict return of ovarian function21,22. They can potentially be used to determine whether ovarian function has resumed or if there is a need to initiate ovarian suppression therapy21,22. Below, we discuss the merits of each biomarker.
Estradiol
Estradiol (E2) levels ≥ 40 pg/mL are commonly used to establish pre-menopausal status in the absence of regular menstrual cycles for GnRH agonist trial eligibility23. Measurements of E2 are also useful as a marker for endocrine sensitivity, as well as to monitor compliance/adherence and refine treatment regimens24,25. Finding E2 levels within the pre-menopausal range in women receiving GnRH agonist treatment is indicative of incomplete ovarian function suppression per the American Society of Clinical Oncology (ASCO) guidelines26,27. Typically, an E2 level ≤ 10 pg/mL is consistent with menopausal status with ovarian function suppression, and an appropriate level at which an AI can be utilized28. Inadequate ovarian function suppression may reduce the impact of AI28.
It should be noted that E2 monitoring guidelines are not currently available, so the optimal degree of ovarian suppression is undetermined29. Although individual clinical laboratories that assay E2 levels have established acceptable reference ranges, these may differ between laboratories and may not be consistent between assay methods30. The gold standard gas chromatography/mass spectrometry measurement of extracted E2 is more sensitive and specific, but is more expensive and tedious to perform than the immunoassay which may cross-react with other steroids since estradiol is not extracted nor purified22,30,31,32. Accuracy and reliability of E2 assays are known to falter particularly at the low concentration ranges29,32,33. Timing of blood sampling may also affect the results of E2 assays, since E2 exhibits a diurnal cycle such that early morning peaks may differ from a random sample during the day34,35. Although there are no standards regarding timing for estradiol assessment in the real-world setting, the SOFT Estrogen Substudy measured E2 at 3, 6, and 12 months21,36.
Follicle-stimulating hormone
Follicle-stimulating hormone is not usually measured in pre-menopausal patients with breast cancer except at baseline to confirm pre-menopausal status and need for ovarian function suppression37,38,39. Certain clinical trials have included FSH as one of the parameters to determine pre-menopausal status, randomization stratification, and/or confirm ovarian ablation19,40,41. During GnRH agonist therapy, the secretion of FSH is initially downregulated but progressively returns to baseline levels after 1 month28. This is attributed to feedback through inhibin when complete ovarian function suppression is achieved28. An exception perhaps is in cases of incomplete ovarian function suppression as in the presence of rising E2 levels, which have been reported among women with fibroids28. The National Comprehensive Cancer Network® (NCCN®) indicates that both E2 and FSH levels are used to support the diagnosis of menopause, however clear criteria to guide interpretation of FSH and E2 in this population is lacking42. Threshold serum E2 levels ≥ 40 pg/mL and FSH levels < 30 mIU/mL have been used to detect the resumption of ovarian function in the absence of menses23. There is otherwise typically no clinical role of FSH as a stand-alone biomarker in treatment decision-making.
Luteinizing hormone
In women with breast cancer, LH is sometimes measured at baseline to confirm the patient’s pre-menopausal status and need for ovarian function suppression37. Luteinizing hormone is otherwise not followed routinely by oncologists during GnRH agonist therapy as it has no clinical role in treatment decision-making. None of the leading clinical practice guidelines recommend monitoring LH levels in patients with breast cancer, such as ASCO26,27, or the International Consensus Symposium for Breast Cancer in Young Women (BCY5) from the European School of Oncology and European Society of Medical Oncology (ESO-ESMO)20. Certain trials have included LH assessment as a criteria for eligibility, including the Austrian Breast and Colorectal Cancer Study Group (ABCSG-12) and German Adjuvant Breast Cancer Study Group (GABG) IV-B-93 when LH > 50 pg/mL was used as one of the acceptable parameters to confirm menopause in the absence of regular menstrual cycles38,39.
Other considerations
For a patient with estrogen receptor-positive breast cancer, the goal is estrogen suppression, and therefore it is critical to measure E2. However, inadequate estrogen suppression may be due in part to body habitus and elevated body mass index (BMI). Since the primary source of serum estrogen is from adipose tissue, overweight patients are more likely to have larger reserves of estrogen precursors to be metabolized by the aromatase enzyme43. Even patients who have had an oophorectomy may have E2 levels above post-menopausal range due to contributions from adipose tissue as an exogenous source of estrogen. Patients with high BMI and intact ovaries may be at higher risk of incomplete ovarian function suppression43. A subanalysis of the SOFT study indicated that patients with higher BMI (median 27 kg/m2) whose E2 levels rose above the strict 2.72 pg/mL suboptimal threshold also had lower baseline FSH (median 8 IU/L) and LH (median 7 IU/L) levels21,24. Regular E2 monitoring has been suggested as the primary biomarker to detect any potential for breakthrough resumption of ovarian function24. The threshold for ovarian function suppression is recognized to be E2 < 40 pg/mL, although threshold levels as low as 10 pg/mL have been documented2,28.
There is less of a concern regarding any potential confounding effects of concomitant therapy such as AI or tamoxifen on E2 levels. Since AI primarily inhibits the peripheral conversion of circulating androgens into estrogens (from androstenedione to E1, with a minor pathway from testosterone to E2), AI will not suppress estrogen to subphysiologic levels if the patient’s ovaries are not being suppressed by other means such as GnRH agonist therapy44. Similarly, the mixed agonist/antagonist effect of tamoxifen is thought to be counteracted by the estrogen-depleting effects of goserelin45.
Evaluating GnRH agonist efficacy
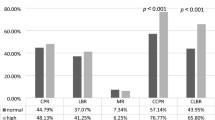
The ASCO guidelines on ovarian suppression are based on data from trials involving the 1-month formulation of GnRH agonists27. In previous trials designed to evaluate the efficacy of a GnRH agonist, E2 < 30 pg/mL was consistently reached indicating ovarian function suppression among the majority of pre-menopausal women40,46,47,48. Estradiol levels were consistent between agents (leuprolide and goserelin) regardless of age, chemotherapy history, taxane regimen, and tumor characteristics46. The other biomarkers were less consistent, as differences in FSH levels were observed with patient age (P = 0.02), cERB-B2(+) tumors (P = 0.05), and lack of taxane in chemotherapy regimen (P = 0.05)46. Likewise, differences in LH levels were observed between GnRH agonists (P = 0.03) and tumor stage (T1 + T2 vs T3 + T4, P = 0.03)46.
In the prostate cancer setting, neither FSH or LH are measured during GnRH agonist therapy49. Testosterone is the biomarker of choice to document castrate levels during androgen deprivation therapy for prostate cancer49,50,51. This correlates to measuring E2 in breast cancer. Authorities have released guidance for appropriate clinical endpoints to be used to assess efficacy of GnRH analogs in 2022 for advanced prostate cancer52. In this setting, plasma testosterone is suggested as the primary endpoint, with levels of <50 ng/dL defined as the threshold for establishing castrate level52. Extrapolating from the prostate cancer setting, it would not be unreasonable to utilize E2 as the primary biomarker for GnRH agonist efficacy in breast cancer.
Considerations for future trials
Based on the above discussion, we recommend that the primary endpoint of ovarian function suppression be measured by E2 levels in breast cancer trials. Estradiol should be used since it most closely reflects ovarian function and is the most relevant hormone activating estrogen receptors in the breast (Fig. 1)2. The ASCO guidelines indicate that detection of premenopausal E2 levels in women while on GnRH agonist therapy is a sign of incomplete ovarian suppression27. Measurement of FSH and/or LH levels could be secondary endpoints. Resumption of menses regardless of hormone levels would indicate failure of therapy. The primary concern when evaluating any formulation is the potential for E2 escape if the GnRH agonist formulation wears off at the end of the dosing period, before the next dose is scheduled. We suggest trials also checking E2 levels towards the end of the dosing interval to confirm it is still suppressed. While in clinical practice it would be important to evaluate for the resumption of menses and/or cyclical fluctuations in climacteric symptoms27, whether clinical trial procedures would include such assessments needs to be considered.
Data availability
Source data for all figures and tables are provided in the paper. No new data sets have been generated or analyzed for this article.
References
Giaquinto, A. N. et al. Breast cancer statistics, 2022. CA Cancer J. Clin. 72, 524–541 (2022).
Lu, Y.-S., Wong, A. & Kim, H.-J. Ovarian function suppression with luteinizing hormone-releasing hormone agonists for the treatment of hormone receptor-positive early breast cancer in premenopausal women. Front. Oncol. 11, 700722 (2021).
Lambertini, M. et al. Ovarian suppression with triptorelin during adjuvant breast cancer chemotherapy and long-term ovarian function, pregnancies, and disease-free survival. A randomized clinical trial. JAMA 314, 2632–2640 (2015).
Moore, H. C. F. et al. Goserelin for ovarian protection during breast-cancer adjuvant chemotherapy. N. Engl. J. Med. 372, 923–932 (2015).
Zong, X. et al. Effects of gonadotropin-releasing hormone analogs on ovarian function against chemotherapy-induced gonadotoxic effects in premenopausal women with breast cancer in China: A randomized clinical trial. JAMA Oncol. 8, 252–258 (2022).
Leonard, R. C. F. et al. GnRH agonist for protection against ovarian toxicity during chemotherapy for early breast cancer: the Anglo Celtic Group OPTION trial. Ann. Oncol. 28, 1811–1816 (2017).
Lambertini, M. et al. Gonadotropin-releasing hormone agonists during chemotherapy for preservation of ovarian function and fertility in premenopausal patients with early breast cancer: a systematic review and meta-analysis of individual patient-level data. J. Clin. Oncol. 36, 1981–1990 (2018).
Lambertini, M. et al. Long-term outcomes with pharmacological ovarian suppression during chemotherapy in premenopausal early breast cancer patients. J. Natl. Cancer Inst. 114, 400–408 (2022).
Moore, H. C. F. et al. Final analysis of the prevention of early menopause study (POEMS)/SWOG Intergroup S0230. JNCI J. Natl. Cancer Inst. 111, 210–213 (2019).
Partridge, A. H. et al. Interrupting endocrine therapy to attempt pregnancy after breast cancer. N. Engl. J. Med. 388, 1645–1656 (2023).
Pagani, O. et al. Adjuvant exemestane with ovarian suppression in premenopausal breast cancer: long-term follow-up of the combined TEXT and SOFT trials. J. Clin. Oncol. 41, 1376–1382 (2023).
Francis, P. A. et al. Adjuvant endocrine therapy in premenopausal breast cancer: 12-year results from SOFT. J. Clin. Oncol. 41, 1370–1375 (2023).
Baek, S. Y. et al. Adding ovarian suppression to tamoxifen for premenopausal women with hormone receptor-positive breast cancer after chemotherapy: an 8-year follow-up of the ASTRRA Trial. J. Clin. Oncol. 41, 4864–4871 (2023).
Gray, R. G. et al. Effects of ovarian ablation or suppression on breast cancer recurrence and survival: Patient-level meta-analysis of 14,993 pre-menopausal women in 25 randomized trials. J. Clin. Oncol. 41, 503 (2023).
Bui, K. T., Willson, M. L., Goel, S., Beith, J. & Goodwin, A. Ovarian suppression for adjuvant treatment of hormone receptor-positive early breast cancer. Cochrane Database Syst. Rev. 3, CD013538 (2020).
Francis, P. A. et al. Tailoring adjuvant endocrine therapy for premenopausal breast cancer. N. Engl. J. Med. 379, 122–137 (2018).
Robertson, J. F. & Blamey, R. W. The use of gonadotrophin-releasing hormone (GnRH) agonists in early and advanced breast cancer in pre- and perimenopausal women. Eur. J. Cancer 39, 861–869 (2003).
Fornier, M. N. et al. Incidence of chemotherapy-induced, long-term amenorrhea in patients with breast carcinoma age 40 years and younger after adjuvant anthracycline and taxane. Cancer 104, 1575–1579 (2005).
Kim, H. A. et al. Adding ovarian suppression to tamoxifen for premenopausal breast cancer: a randomized phase III trial. J. Clin. Oncol. 38, 434–443 (2020).
Paluch-Shimon, S. et al. ESO-ESMO fifth international consensus guidelines for breast cancer in young women (BCY5). Ann. Oncol. 33, 1097–1118 (2022).
Bellet, M. et al. Twelve-month estrogen levels in premenopausal women with hormone receptor-positive breast cancer receiving adjuvant triptorelin plus exemestane or tamoxifen in the Suppression of Ovarian Function Trial (SOFT): the SOFT-EST Substudy. J. Clin. Oncol. 34, 1584–1593 (2016).
Smith, I. E. et al. Adjuvant aromatase inhibitors for early breast cancer after chemotherapy-induced amenorrhoea: caution and suggested guidelines. J. Clin. Oncol. 24, 2444–2447 (2006).
Kim, H. J. et al. Five-year changes in ovarian function restoration in premenopausal patients with breast cancer taking tamoxifen after chemotherapy: an ASTRRA study report. Eur. J. Cancer 151, 190–200 (2021).
Arboleda, B. et al. Ovarian function suppression: a deeper consideration of the role in early breast cancer and its potential impact on patient outcomes: a consensus statement from an international expert panel. Oncologist 27, 722–731 (2022).
Folkerd, E. J., Lønning, P. E. & Dowsett, M. Interpreting plasma estrogen levels in breast cancer: caution needed. J. Clin. Oncol. 32, 1396–1400 (2014).
Burstein, H. J. et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: ASCO Clinical Practice Guideline focused update. J. Clin. Oncol. 37, 423–438 (2019).
Burstein, H. J. et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline update on ovarian suppression. J. Clin. Oncol. 34, 1689–1701 (2016).
Dowsett, M., Lønning, P. E. & Davidson, N. E. Incomplete estrogen suppression with gonadotropin-releasing hormone agonists may reduce clinical efficacy in premenopausal women with early breast cancer. J. Clin. Oncol. 34, 1580–1583 (2016).
Fleege, N. M. G. et al. Ovarian function suppression in premenopausal women with concurrent endocrine therapy use. Clin. Breast Cancer 23, 454–460 (2023).
Demers, L. M. et al. Measuring estrogen exposure and metabolism: Workshop recommendations on clinical issues. J. Clin. Endocrinol. Metab. 100, 2165–2170 (2015).
Santen, R. J. et al. Superiority of gas chromatography/tandem mass spectrometry assay (GC/MS/MS) for estradiol for monitoring of aromatase inhibitor therapy. Steroids 72, 666–671 (2007).
Lee, J. S. et al. Comparison of methods to measure low serum estradiol levels in postmenopausal women. J. Clin. Endocrinol. Metab. 91, 3791–3797 (2006).
Rosner, W. et al. Challenges to the measurement of estradiol: an endocrine society position statement. J. Clin. Endocrinol. Metab. 98, 1376–1387 (2013).
Hillebrand, J. J., Wickenhagen, W. V. & Heijboer, A. C. Improving science by overcoming laboratory pitfals with hormone measurements. J. Clin. Endocrinol. Metab. 106, e1504–e1512 (2021).
Ball, L. J., Palesh, O. & Kriegsfeld, L. J. The pathophysiologic role of disrupted circadian and neuroendocrine rhythms in breast carcinogenesis. Endocr. Rev. 37, 450–466 (2016).
Burns, E. et al. Measuring ovarian escape in premenopausal estrogen receptor-positive breast cancer patients on ovarian suppression therapy. Oncologist 26, e936–e942 (2021).
Eisen, A. et al. Optimal systemic therapy for early breast cancer in women: a clinical practice guideline. Curr. Oncol. 22, S67–S81 (2015).
Gnant, M. et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-month follow-up from the ABCSG-12 randomised trial. Lancet Oncol. 12, 631–641 (2011).
Kaufmann, M. et al. A randomised trial of goserelin versus control after adjuvant, risk-adapted chemotherapy in premenopausal patients with primary breast cancer - GABG-IV B-93. Eur. J. Cancer 43, 2351–2358 (2007).
Kendzierski, D. C., Schneider, B. P. & Kiel, P. J. Efficacy of different leuprolide administration schedules in premenopausal breast cancer: a retrospective review. Clin. Breast Cancer 18, e939–e942 (2018).
LHRH-agonists in Early Breast Cancer Overview group, et al. Use of luteinising-hormone-releasing hormone agonists as adjuvant treatment in premenopausal patients with hormone-receptor-positive breast cancer: a meta-analysis of individual patient data from randomised adjuvant trials. Lancet 369, 1711–1723 (2007).
Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Breast Cancer V.5.2023. © National Comprehensive Cancer Network, Inc 2023. All rights reserved. Accessed December 21, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org. NATIONAL COMPREHENSIVE CANCER NETWORK®, NCCN®, NCCN GUIDELINES®, and all other NCCN Content are trademarks owned by the National Comprehensive Cancer Network, Inc. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. (2023).
Pfeiler, G. et al. Impact of body mass index on the efficacy of endocrine therapy in premenopausal patients with breast cancer: an analysis of the prospective ABCSG-12 trial. J. Clin. Oncol. 29, 2653–2659 (2011).
Lønning, P. E. & Lien, E. A. Mechanisms of action of endocrine treatment in breast cancer. Crit. Rev. Oncol. Hematol. 21, 158–193 (1995).
Johansson, A. et al. Twenty-year benefit from adjuvant goserelin and tamoxifen in premenopausal patients with breast cancer in a controlled randomized clinical trial. J. Clin. Oncol. 40, 4071–4082 (2022).
Aydiner, A. et al. Two different formulations with equivalent effect? Comparison of serum estradiol suppression with monthly goserelin and trimonthly leuprolide in breast cancer patients. Med. Oncol. 30, 354 (2013).
Boccardo, F. et al. Endocrinological and clinical evaluation of two depot formulations of leuprolide acetate in pre- and perimenopausal breast cancer patients. Cancer Chemother. Pharmacol. 43, 461–466 (1999).
Wu, Z. Y. et al. Effectiveness of a 6-month 22.5-mg leuprolide acetate depot formulation with tamoxifen for postoperative premenopausal estrogen suppression in hormone receptor-positive breast cancer. Front. Oncol. 11, 665426 (2021).
Virgo, K. S. et al. Initial management of noncastrate advanced, recurrent, or metastatic prostate cancer: ASCO Guideline update. J. Clin. Oncol. 39, 1274–1305 (2021).
Morote, J. et al. Serum testosterone levels in prostate cancer patients undergoing luteinizing hormone-releasing hormone agonist therapy. Clin. Genitourin. Cancer 16, e491–e496 (2018).
Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.4.2023. © National Comprehensive Cancer Network, Inc 2023. All rights reserved. Accessed October 31, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org. NATIONAL COMPREHENSIVE CANCER NETWORK®, NCCN®, NCCN GUIDELINES®, and all other NCCN Content are trademarks owned by the National Comprehensive Cancer Network, Inc. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. (2023).
Food and Drug Administration: Advanced Prostate Cancer: Developing gonadotropin-releasing hormone analogues, in U.S. Department of Health and Human Services (ed). Guidance for Industry (ed May 2022). https://www.fda.gov/regulatory-information/search-fda-guidance-documents/advanced-prostate-cancer-developing-gonadotropin-releasing-hormone-analogues-guidance-industry (2022).
Acknowledgements
Medical writing and editorial assistance were provided under the direction of the authors by LoAn K. Ho, PharmD, of Forward WE Go, a division of Wesley Enterprise, Inc. with funding support from TerSera Therapeutics.
Author information
Authors and Affiliations
Contributions
All authors collaborated in the conception of the manuscript, contributed to the collection and interpretation of data, critically reviewed and revised all drafts for intellectual content, provided final approval to the manuscript, and agreed to share responsibility and accountability for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
K.E.M. discloses clinical trial research support to her institution from Gilead Sciences, H3 Biomedicine, ZWI Therapeutics, Eli Lilly, Olema Oncology, G1 Therapeutics, AstraZeneca; receives consulting fees from Puma Biotechnology; receives honoraria from OncLive, Best of ASCO, Physicians’ Education Resource, Eli Lilly’s speaker’s bureau and Dava Oncology; receives support for attending meetings from Eli Lilly and Dava Oncology; and participates on the advisory board for Daiichi Sankyo, Puma Biotechnology, Sermonix and Eli Lilly. T.A.T. reports research support (grants) to her institution from Pfizer, AstraZeneca, Genentech/Roche, Daiichi Sankyo, Ayala Pharmaceuticals and Astellas Pharma; consulting fees from Pfizer, AstraZeneca, Genentech/Roche, Daiichi Sankyo, Merck, Gilead Sciences, Novartis, GlaxoSmithKline, GE Healthcare, Biotheranostics, Hengrui Pharmaceuticals, G1 Therapeutics, TerSera Therapeutics, Stemline Therapeutics and Exact Sciences; and an advisory role with I-SPY trial data safety and monitoring board. M.M.R. reports research support (grants) and/or consulting fees to her institution from AstraZeneca, Bayer, Biotheranostics, Bristol-Myers Squibb, Debiopharm, Ipsen, Novartis, Pfizer, Roche, Tolmar and TerSera Therapeutics; honoraria from Bristol-Myers Squibb; and an advisory role with AstraZeneca, Bristol-Myers Squibb, TerSera Therapeutics and Tolmar. N.V. discloses research funding to her institution from Merck, Dae Hwa, Radius, Novartis, Pfizer; and participation in advisory boards for Gilead Sciences, TerSera Therapeutics, Aadi Bioscience, OncoSec Medical, AbbVie and the steering committee for the Stemline study. V.K. receives royalties from UpToDate and honoraria from Gilead Sciences, AstraZeneca and Seagen; consults for Puma Biotechnology, AstraZeneca, Menarini Group, and Eli Lilly; and participates on an advisory board for Bristol-Myers Squibb.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McCann, K.E., Goldfarb, S.B., Traina, T.A. et al. Selection of appropriate biomarkers to monitor effectiveness of ovarian function suppression in pre-menopausal patients with ER+ breast cancer. npj Breast Cancer 10, 8 (2024). https://doi.org/10.1038/s41523-024-00614-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-024-00614-w
This article is cited by
-
Estrogen levels in young women with hormone receptor-positive breast cancer on ovarian function suppression therapy
npj Breast Cancer (2024)
-
De novo design of a nanoregulator for the dynamic restoration of ovarian tissue in cryopreservation and transplantation
Journal of Nanobiotechnology (2024)