Abstract
Leptomeningeal metastases (LM) are a devastating complication of HER2 + metastatic breast cancer (MBC), with no effective treatments. In a case series of 8 patients with heavily pretreated HER2 + MBC and progressing LM, all 8 patients (100%) derived clinical benefit from Trastuzumab deruxtecan (TDXd), and 4 patients (50%) had an objective partial response based on formal neuroradiology MRI reads using the EORTC/RANO-LM Revised-Scorecard. T-DXd warrants further study in LM in HER2 + MBC and solid tumors where T-DXd may be active.
Similar content being viewed by others
Between 5–15% of patients with metastatic breast cancer (MBC) develop leptomeningeal metastases (LM)1,2 with approximately 60% having no parenchymal brain metastases (BrMs)3. The presence of LM has a drastic impact on patients’ function and quality of life, leading to poor survival outcomes, with a median survival of only 4.4 months in human epidermal growth factor receptor 2-positive (HER2+) MBC4. Despite significant advances in the treatment of HER2 + MBC in recent years, there are no U.S. Food and Drug Administration (FDA)-approved therapies specifically to treat LM. Trastuzumab deruxtecan (T-DXd) is a HER2-directed antibody-drug conjugate carrying an exatecan-derivative topoisomerase I inhibitor payload5.
DESTINYBreast03 demonstrated a significant improvement in progression-free survival (PFS) compared with trastuzumab emtansine (T-DM1), and established a new therapeutic option in the second-line setting6. The benefit of T-DXd was observed in the subset of patients with stable BrMs at the time of the enrollment, with an overall response rate (ORR) of 67.4% and BrMs-ORR of 63%7. Notably, patients with LM were not eligible in this trial. Smaller studies have evaluated T-DXd to treat active BrMs, showing promising activity. The DEBBRAH and TUXEDO-1 clinical trials, including patients with active (untreated or progressing) BrMs, demonstrated an intracranial ORR of 46.2% (95% CI, 19.2–74.9)8 and 73.3% (95% CI, 48.1–89.1%) respectively9. Likewise, a recent multi-institution analysis of patients with active HER2 + BrMs showed a brain metastases disease control rate of 75%10. The impressive intracranial and extracranial activity of TDXd has led to widespread uptake in the clinic. However, the efficacy of T-DXd in patients with HER2 + LM have not been previously reported.
The assessment of treatment-response of LM in clinical practice and clinical trials remains challenging. Recently, a joint effort of the European Organisation for Research and Treatment of Cancer (EORTC) Brain Tumor Group and Response Assessment in Neuro-Oncology (RANO) prospectively validated a revised magnetic resonance imaging (MRI)-scorecard for response assessment in LM, showing a substantial interobserver agreement and associated prognostic significance3.
In this retrospective study inspired by our collective clinical observations, we report the activity of T-DXd in patients with HER2 + MBC and active LM, based on the standardized EORTC/RANO MRI-scorecards. The objective of our study was to assess the activity of T-DXd on LM in patients with HER2 + MBC and progressing LM.
Eight patients met the inclusion criteria at the data cut-off (Table 1). Three patients had baseline, positive cerebrospinal fluid (CSF) cytology, three had negative CSF cytology and two patients were not tested. Median age was 42.5 years; 75% of patients had estrogen receptor-positive MBC. Seven patients (87.5%) had prior whole-brain radiation therapy (WBRT), with a median time of 100 days from the most recent RT to the first infusion of T-DXd (range: 5–363 days). Patients were very heavily pretreated with a median of 4.5 prior therapies (Fig. 1). All had received prior HER2-targeted tyrosine kinase inhibitors (TKI): seven patients had prior tucatinib and one neratinib; two patients had received intra-thecal trastuzumab (Fig. 1). Patients had been alive with active LMD for a median of 101 days prior to T-DXd.
LM leptomeningeal disease, M months. TCHP trastuzumab, pertuzumab, carboplatin and docetaxel, THC trastuzumab, pertuzumab and docetaxel. HP pertuzumab and trastuzumab. Atezo-HP Atezolizumab, pertuzumab, and high-dose trastuzumab. T-DM1 trastuzumab emtansine. GDC0084 paxalisib. Cape capecitabine. WBSRT whole brain and spine radiotherapy. SRS stereotactic radiation therapy. WBRT whole brain radiation therapy. VNR vinorelbine. T-DXd trastuzumab deruxtecan. BrMs brain metastasis. ‡bRT brachytherapy with radioactive seed implantation. † new biopsy showed HER2 in situ hybridization positive, UNK§, patient was given recommendation for further treatment, lost to follow-up; alive at data cutoff.
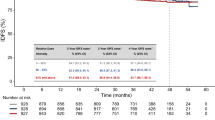
Median time on T-DXd was 7.6 months (range:127–514 days) with a median of 10 (5–23) cycles. Five patients remained on T-DXd at the data cut-off (Fig. 2). One half of patients (n = 4/8) achieved a partial response (PR) on MRI as best response based on the EORTC/RANO-LM Revised-Scorecard, while 4 patients had stable disease (SD) (Fig. 3). Clinical benefit response (CBR) was 100%. Of note, two patients with RANO-LM defined as SD did show radiographic improvement of leptomeningeal linear enhancement, which is not considered evaluable for response. No patient experienced BrMs progression while on T-DXd. No patient had available post-baseline CSF cytology analysis. The median overall survival (OS) from the first cycle of T-DXd was 10.4 months (158–514 days) with 6 patients still alive at the data cut-off. Both deceased patients died due to neurological deterioration. The median OS from the diagnosis of LM was 15.5 months (189–626 days).
In our case series, T-DXd demonstrated encouraging activity with an LM ORR of 50% and CBR of 100%. The median time on treatment was 7.6 months with 5 patients ongoing at the data cutoff. The median OS was 10.4 months since the start of T-DXd with 6 patients remaining alive at the data cutoff. Of note, the median OS from the diagnosis of LM was 15.5 months, suggesting that patients have derived most of the benefit from T-DXd, since the diagnosis of LM. Patients were heavily pretreated, all having received prior RT and a median of 4.5 prior lines of systemic therapy in the metastatic setting, including tucatinib. Limited therapeutic options exist for such patients. In general, LM spread represents a critical event for patients with MBC, with significant prognostic implications, mostly related to the lack of high-efficacy treatments demonstrating durable responses. The major challenge pertains to the CSF-widespread nature of the LM seeding, and the need to treat the whole disease burden. Traditionally, patients with LM have been managed with RT for bulky symptomatic disease with emerging evidence for cranio-spinal RT followed by systemic and/or intrathecal therapies. Attempts for effective intra-CSF drug delivery have been provided through high-dose systemic chemotherapy regimens, like methotrexate, or directly infused in the CSF via lumbar puncture or ventricular reservoir, most commonly methotrexate, liposomal cytarabine and thiotepa, resulting in 30–50% ORR but limited survival rates of approximately 3–5 months11,12. Intrathecal administration of therapies comes with substantial risk and is clinically challenging.
Only select anti-HER2 therapies have reported activity in HER2 + LM. In particular, neratinib plus capecitabine showed preliminary activity in LM, with median LM-PFS of 4 months and OS of 10 months13. In addition, the use of intrathecal trastuzumab has demonstrated ORR of 19.2%, SD in 50%, with a CBR of 70%, and median-OS of 10.5 months14. Given the favorable CSF pharmacokinetic profile and proven intra-cranial activity, tucatinib has been identified as a potential therapeutic for patients with LM, and is under investigation in the TBCRC049 trial (NCT03501979)15. Until now, antibody-drug conjugates held little promise for intracranial efficacy. The body of evidence for TDXd efficacy in stable and active HER2 + BrMs has challenged this paradigm. A potential advantage of T-DXd is the possibility of activity in HER2-low LM in patients with breast cancer, and even potential for activity in LM from other solid tumors, given the broad extracranial efficacy profile reported to date.
Major limitations of our study include its retrospective nature and small sample size, both of which carry intrinsic risks of bias. Also, baseline CSF cytology was rarely available and not performed serially, and LM diagnosis was mostly based on MRI. The centralized neuroradiology assessment of the response based on validated methodology is an important strength of this analysis. Prospective clinical trials are rarely funded in patients with LM and new ways of obtaining clinical data such as prospective registries and multi-institutional case series are needed.
In conclusion, we report activity in a case series of heavily pre-treated progressing HER2 + LM patients treated with TDXd,. Prospective clinical trials are warranted.
Methods
Patient Characteristics
Using institutional electronic medical records, our doctors identified patients with metastatic HER2+ breast cancer at Duke Cancer Institute (Duke) and Dana-Farber Cancer Institute (DFCI) who had brain and/or spine MRI-confirmed LM (+/− positive cytology) and who had received at least one dose of T-DxD between January 1, 2020 and December 31, 2020 as part of their usual clinical care. Data on demographics, clinical, pathologic, and treatment related to their primary and MBC diagnoses were also abstracted based on medical record review.
58 patients treated with T-DXd at Duke and DFCI were identified in the timeframe with biopsy-proven HER2 + metastatic breast cancer. (30 patients at Duke – and 28 patients at DFCI). Amongst those patients, 8 had evidence of progressing LM by clinical assessment, MRI imaging+/− cytology. We utilized the European Association of Neuro-Oncology/European Society for Medical Oncology (EANO/ESMO) proposed classification of LM from solid cancers based on clinical, MRI, and CSF cytology presentation in identification of LMD16. Patients with Type I LM defined by positive CSF cytology (confirmed LM) and type II LM defined by typical clinical and MRI signs (probable or possible LM) were included. As is the case in clinical practice, not all patients underwent CSF testing.
Data collection and evaluation
Retrospective data collection included patients’ demographic and tumor clinicopathologic characteristics, previous RT, presence of BrMs, and previous anti-HER2 therapies.
Radiological assessment was based on the EORTC/RANO revised LM scorecard3 and performed by a dedicated neuroradiologist at DFCI (HL) and Duke (CL). The scorecards were completed for each patient at each MRI re-evaluation; BrMs response was also reported (Supplementary Table 1). LM partial response corresponds to a decrease by >50% in the summed product of orthogonal diameters of the LM implants, with no increase of the ventricular size. ORR included patients reporting a PR or complete response (CR). CBR included the proportion of patients experiencing SD, PR, or CR, with stable or improved neurological symptoms. Available reports on CSF analyses were all collected.
Data availability
The datasets of this study are hosted with a protected password known to the lead authors in a data repository and are not for open access, given their sensitive nature. The corresponding author, Sarah Sammons, MD (sarahl_sammons@dfci.harvard.edu) may be contacted for potential collaborations, upon careful screening of the proposals, to share the data grouped and anonymized, provided an IRB authorization.
References
Lee, D. W., Lee, K. H., Kim, J. W. & Keam, B. Molecular targeted therapies for the treatment of leptomeningeal carcinomatosis: current evidence and future directions. Int J. Mol. Sci. 17, 1074 (2016).
Figura, N. B. et al. Breast leptomeningeal disease: a review of current practices and updates on management. Breast Cancer Res Treat. 177, 277–294 (2019).
Le Rhun, E. et al. Prospective validation of a new imaging scorecard to assess leptomeningeal metastasis: A joint EORTC BTG and RANO effort. Neuro Oncol. 24, 1726–1735 (2022).
Abouharb, S. et al. Leptomeningeal disease and breast cancer: the importance of tumor subtype. Breast Cancer Res Treat. 146, 477–486 (2014).
Doi, T. et al. Safety, pharmacokinetics, and antitumour activity of trastuzumab deruxtecan (DS-8201), a HER2-targeting antibody-drug conjugate, in patients with advanced breast and gastric or gastro-oesophageal tumours: a phase 1 dose-escalation study. Lancet Oncol. 18, 1512–1522 (2017).
Cortes, J. et al. Trastuzumab Deruxtecan versus Trastuzumab Emtansine for breast cancer. N. Engl. J. Med 386, 1143–1154 (2022).
Hurvitz, S. A. et al. Trastuzumab deruxtecan versus trastuzumab emtansine in patients with HER2-positive metastatic breast cancer: updated results from DESTINY-Breast03, a randomised, open-label, phase 3 trial. Lancet. 401, 105–117 (2023).
Perez-Garcia, J. M. et al. Trastuzumab Deruxtecan in patients with central nervous system involvement from HER2-positive breast cancer: The DEBBRAH Trial. Neuro Oncol. 25, 157–166 (2022).
Bartsch, R. et al. Trastuzumab deruxtecan in HER2-positive breast cancer with brain metastases: a single-arm, phase 2 trial. Nat. Med. 28, 1840–1847 (2022).
Kabraji, S. et al. Preclinical and clinical efficacy of trastuzumab deruxtecan in breast cancer brain metastases. Clin. Cancer Res. 29, 174–182 (2022).
Carausu, M. et al. Breast cancer patients treated with intrathecal therapy for leptomeningeal metastases in a large real-life database. ESMO Open 6, 100150 (2021).
Kapke, J. T. et al. High-dose intravenous methotrexate in the management of breast cancer with leptomeningeal disease: Case series and review of the literature. Hematol. Oncol. Stem Cell Ther. 12, 189–193 (2019).
Pellerino, A. et al. Neratinib and Capecitabine for the treatment of Leptomeningeal Metastases from HER2-positive breast cancer: a series in the setting of a compassionate program. Cancers 14, 1192 (2022).
Kumthekar, P. U. et al. A Phase I/II Study of Intrathecal Trastuzumab in HER-2 positive cancer with leptomeningeal metastases: safety, efficacy, and cerebrospinal fluid pharmacokinetics. Neuro Oncol. 25, 557–565 (2022).
Li, J. et al. Mechanistic modeling of central nervous system pharmacokinetics and target engagement of HER2 Tyrosine Kinase inhibitors to inform treatment of breast cancer brain metastases. Clin. Cancer Res. 28, 3329–3341 (2022).
Le Rhun, E. et al. EANO-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up of patients with brain metastasis from solid tumours. Ann. Oncol. 32, 1332–1347 (2021).
Acknowledgements
The authors express sincerest gratitude to the Kiki Fund for Breast Cancer Leptomeningeal Metastases Research and Translating Duke Health (CKA) for providing support in the field and helping catalyse the research in an area of high-unmet clinical need, and limited funding. We are also immensely thankful to all the patients who provided consent to be included in this study. All the personnel involved for the administrative support and the incredible value of all the health providers involved in the care of patients is highlighted. Invaluable editorial support was added by Valerie Hope Goldstein from the Dana-Farber Cancer Institute.
Author information
Authors and Affiliations
Contributions
L.A., D.T., S.S., and N.L. conceived of the presented idea. S.T., N.L., S.S., D.T., and C.A. cared for presented patients. C.B. performed the statistical analysis. C.L. and L.H. performed neuroradiology reads. All authors contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
SMT reports a consulting or advisory board role for Novartis, Pfizer, Merck, Eli Lilly, AstraZeneca, Genentech/Roche, Eisai, Sanofi, Bristol Myers Squibb, Seattle Genetics, Odonate Therapeutics, CytomX Therapeutics, Daiichi Sankyo, Athenex, Gilead, Mersana, Certara, Chugai Pharma, Ellipses Pharma, Infinity, 4D Pharma, OncoSec Medical Inc., BeyondSpring Pharmaceuticals, OncXerna, Zymeworks, Zentalis, Blueprint Medicines, Reveal Genomics, ARC Therapeutics, Myovant, Zetagen, Umoja Biopharma, Menarini/Stemline, Aadi Bio and Bayer; and reports institutional research funding from Genentech/Roche, Merck, Exelixis, Pfizer, Eli Lilly, Novartis, Bristol Myers Squibb, Eisai, AstraZeneca, NanoString Technologies, Cyclacel, Nektar, Gilead, Sanofi and Seattle Genetics. MK reports research funding paid to their institution from BMS, AbbVie, Biontech, Astellas, Celldex, Daiichi Sankyo and CNS pharmaceuticals; and honoraria from George Clinical, Voyager Therapeutics, Johnson and Johnson, and the Jax lab for genomic research. CKA reports research funding paid to their institution from PUMA, Lilly, Merck, Seattle Genetics, Nektar, Tesaro, G1-Therapeutics, ZION, Novartis, Pfizer, Astra Zeneca, and Elucida; honoraria from Genentech, Eisai, IPSEN, Seattle Genetics, Astra Zeneca, Novartis, Immunomedics, Elucida, and Athenex; and royalties from Uptodate.com and Jones and Bartlett Publishing Company. NUL reports institutional research funding from Genentech, Merck, Pfizer, Seattle Genetics, AstraZeneca, Zion Pharmaceuticals, and Olema Pharmaceuticals; consultant/advisory board work for Puma, Seattle Genetics, Daiichi Sankyo, AstraZeneca, Prelude Therapeutics, Denali Therapeutics, Olema Pharmaceuticals, Aleta BioPharma, Affinia Therapeutics, Voyager Therapeutics, Janssen and BluePrint Therapeutics; stock and other ownership interests in Artera, Inc. (<$50k and <5% as it relates to consulting activities - options are not currently valued or in-hand); and royalties from UpToDate (book). SS declares research funding to their institution from Astra Zeneca, Abbvie, Bristol Myers Squibb, Eli Lilly, SEAGEN, and Sermonix; and consulting fees from Foundation Medicine, Astra Zeneca, Daichii Sankyo, Eli Lilly, Pfizer, Sermonix and Novartis. The remaining authors declare no conflicts of interest.
Ethics
All relevant ethical regulations, including the Declaration of Helsinki, were observed. Patients at DFCI provided written informed consent to participate in an Institutional Review Boards (IRB)-approved registry study (IRB #20–482), including publication of this case series. Patients at Duke were included by an IRB-approved waiver of consent for this case series (IRB #Pro00106347). Data was extracted from patients’ electronic medical records and entered into a secure, patient deidentified database.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alder, L., Trapani, D., Bradbury, C. et al. Durable responses in patients with HER2+ breast cancer and leptomeningeal metastases treated with trastuzumab deruxtecan. npj Breast Cancer 9, 19 (2023). https://doi.org/10.1038/s41523-023-00519-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-023-00519-0
This article is cited by
-
Prognostic impact of clinical and radiological factors on leptomeningeal metastasis from solid cancers
Journal of Neuro-Oncology (2024)
-
Treatment with trastuzumab deruxtecan in patients with HER2-positive breast cancer and brain metastases and/or leptomeningeal disease (ROSET-BM)
npj Breast Cancer (2023)
-
Incidence of HER2-expressing brain metastases in patients with HER2-null breast cancer: a matched case analysis
npj Breast Cancer (2023)
-
Systemic Treatment for Brain Metastasis and Leptomeningeal Disease in Breast Cancer Patients
Current Oncology Reports (2023)