Abstract
Ductal carcinoma in situ (DCIS) of the breast precedes the development of invasive breast cancer and reflects the genomic changes and protein expression profile of invasive disease. AKT1low cancer cells (QCC) are a rare, drug-tolerant, epigenetically plastic, and quiescent cancer cell subset that we previously identified at a frequency of 0.5–1% in primary breast tumors using the marker profile: AKTlow/H3K9me2low/HES1high. Here we used quantitative immunofluorescence microscopy with computational image analysis to show that AKT1low QCCs are present in DCIS from patients with and without co-existing invasive breast cancer. These data suggest that a drug-resistant, quiescent cancer cell state is present in premalignant breast lesions prior to the development of invasive disease. These findings warrant further study of whether AKT1low QCCs contribute to invasive tumor development and recurrence, similar to their role in more advanced malignancy.
Similar content being viewed by others
Introduction
Ductal carcinoma in situ (DCIS) of the breast is a premalignant lesion that will develop into invasive breast cancer in about 20–50% of cases.1 DCIS reflects the genomic changes (e.g., copy number changes, point mutations) and protein expression profile (e.g., estrogen receptor (ER), HER2 receptor) of subsequent invasive disease.2,3 However, little is known about whether quiescent cancer cell states exist in preinvasive lesions like DCIS and might contribute to tumorigenesis.1 The AKT1low quiescent cancer cell (QCC) is a rare, drug-tolerant, plastic, quiescent (Ki67low/MCM2low) cancer cell state that contributes to tumor growth in xenograft models.4,5,6,7 We previously showed that QCCs exist in clusters at a frequency of 0.5–1% in primary breast tumors and metastases using the marker profile: AKTlow/H3K9me2low/HES1high.7,8 We also found that AKT1low QCCs persist after neoadjuvant chemotherapy in patients with triple negative breast cancer at primary and metastatic sites and, thus, may contribute to breast cancer growth and recurrence.7,8
Here we used quantitative immunofluorescent microscopy with computational image analysis to show that AKT1low QCCs are present in DCIS from patients with and without co-existing invasive breast cancer.
Results
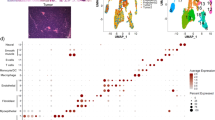
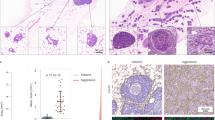
Twenty-two cases of DCIS were analyzed for AKT1low QCCs. 21 cases (96%) were ER+ HER2− and 1 case was ER+ HER2 +. QCCs existed in heterogeneously distributed clusters, as we had found in invasive disease8 (Fig. 1a–c). QCCs were identified in all DCIS samples (22/22) at a mean percentage (QCC-P) of 0.95% ± 0.93 per patient sample (one section), similar to the frequency in invasive breast cancer (Fig. 2a).7,8 Given the small number of samples, it was not possible to test an association between ER or HER2 receptor status and QCC-P. QCC-P did not significantly differ by DCIS grade (1, 2 or 3; p = 0.56, Fig. 2b), patient menopausal status (pre- vs. post-; p = 0.75, Fig. 2c) or presence of concurrent ipsilateral invasive carcinoma (n = 18 vs. 4; p = 0.51, Fig. 2d). Given the short follow up time, no patient had developed recurrent invasive carcinoma.
AKT1low QCCs are found within DCIS. a Hematoxylin and eosin stained section showing a representative area of ductal carcinoma in situ; b Multiplex immunofluorescence of DCIS tissue shown in a with AKT1low QCC cluster marked by a white arrow and the marker profile: AKTlow (green), H3K9me2low (yellow) and HES1high (red); inset image is enlarged to show QCC marker detail. c Digital tumor map of DCIS tissue shown in a and b with AKT1low QCCs shown in red and proliferating cancer cells in blue. Images were taken at ×20
AKT1low QCCs are rare and QCC-P in DCIS does not differ by grade, patient menopausal status or presence of invasive carcinoma. a QCCs were identified in all DCIS samples (22/22) at a mean percentage of 0.95% ± 0.93 per patient sample (one section), similar to the frequency in invasive breast cancer. b QCC-P did not significantly differ by DCIS grade (1, 2 or 3, p = 0.56); patient menopausal status (pre- vs. post-, p = 0.75) or c presence of concurrent ipsilateral invasive carcinoma (n = 18 vs. 4; p = 0.51) (d). Error bars represent mean ± SD
Discussion
We identified AKT1low QCCs in patients with premalignant DCIS at a similar frequency to that found in invasive breast cancer.8 In addition, the frequency of QCCs was not different between the clinical or pathologic groups we analyzed. This suggests that, similar to genetic changes,2 quiescent cancer cell states previously identified in invasive disease may also be found in DCIS. A population of CD44+/ALDH1+/Ki67− cancer ‘stem cells’ has been previously described in patients with DCIS at a frequency of 0.36–15% using triple immunohistochemistry.9 Using flow cytometry, the same group found the frequency of CD44+/CD24−/CK+/CD45− cells in DCIS ranges from 2.6–18%.10 However, Ki67low, AKT1low QCCs do not clearly express the marker profile ascribed to cancer ‘stem cells’ e.g., CD44high/CD24low 4. This suggests that there are likely multiple populations of quiescent cancer cells in DCIS but the morphologic and functional overlap between them remains to be elucidated. These findings warrant further study of how AKT1low QCCs contribute to invasive tumor development and recurrence, particularly in the setting of pre-existing DCIS.
Methods
Patient cohort
We reviewed medical records for 80 women diagnosed with DCIS between 2015 and 2016 at Massachusetts General Hospital, under an IRB-approved discarded tissue protocol (2009P002302), for which informed consent from participants was not required. Twenty-two patients had sufficient tissue available for analysis. DCIS was confirmed by pathologic review. 4 um thick formalin-fixed paraffin embedded sections (one per patient) underwent analysis for QCCs using quantitative immunofluorescence microscopy, as previously described.8
Semi-automated immunofluorescence staining and imaging
Briefly, tissue sections underwent semi-automated sequential labelling for pan-AKT at 1:3000 (CST, 4691S), HES1 at 1:1000 (EMD Millipore, AB5702) and H3K9me2 at 1:150 (Abcam, ab1220) using tyramide-signal amplifying immunofluorescence (Perkin Elmer) on an automated staining platform (Bond, Leica). Images were acquired on a semi-automated confocal microscope (Vectra, Perkin Elmer) and tissue and cell masks were applied to identify areas of DCIS, using InForm software (Perkin Elmer). AKT1low QCCs were determined using prespecified fluorescence intensity thresholds, as previously described.8 Specifically, a cancer cell was classified as an AKT1low QCC if the HES1 fluorescence intensity for that cell fell in the top 75% percentile of cancer cells for that section and the AKT and H3K9me2 fluorescence intensities for that cell fell in the bottom 25% percentile of cancer cells for that section. QCC percentage for each patient tissue section was determined and digital tumor maps were generated using R software (R Foundation).
Determining image sample size
To be 99% confident of detecting QCCs in each section at a prevalence (P) of 1% of cancer cells, we calculated that (assuming precision of 0.00625 and Z of 2.58) we would need to image at least 1688 cancer cells per patient section.11 We imaged 718 ± 549 (10–3162) cancer cells per x20 field and an average of 29 (4–120) x20 fields were imaged per patient section. Thus, on average, we imaged 20,822 cancer cells per patient section and are 99% confident of detecting QCCs in each tissue section with a precision of 0.625%. Values are shown as mean ± standard deviation. Range shown is maximum and minimum. Differences in groups were determined by unpaired t-test or one-way ANOVA and p < 0.05 was considered significant using Graphpad Prism (v. 7).
Code availability
Code used for calculating QCC-P and generating digital tumor maps is available on request.
Data Availability
All images and raw QCC counts available on request. All other data are available from the authors.
References
Yeong, J., Thike, A. A., Tan, P. H. & Iqbal, J. Identifying progression predictors of breast ductal carcinoma in situ. J. Clin. Pathol. 70, 102–108 (2017).
Casasent, A. K. et al. Multiclonal Invasion in Breast Tumors Identified by Topographic Single Cell Sequencing. Cell 172, 205–217 e212 (2018).
Abba, M. C. et al. A Molecular Portrait of High-Grade Ductal Carcinoma In Situ. Cancer Res. 75, 3980–3990 (2015).
Dey-Guha, I. et al. Asymmetric cancer cell division regulated by AKT. Proc. Natl. Acad. Sci. USA 108, 12845–12850 (2011).
Dey-Guha, I. et al. A mechanism for asymmetric cell division resulting in proliferative asynchronicity. Mol. Cancer Res.: MCR 13, 223–230 (2015).
Facompre, N. D. et al. JARID1B enables transit between distinct states of the stem-like cell population in oral cancers. Cancer Res. 76, 5538–5549 (2016).
Alves, C. P. et al. AKT1(low) quiescent cancer cells promote solid tumor growth. Mol. Cancer Ther. 17, 254–263 (2018).
Kabraji, S. et al. AKT1(low) quiescent cancer cells persist after neoadjuvant chemotherapy in triple negative breast cancer. Breast Cancer Res. 19, 88 (2017).
A, D. A. C. P. et al. Characterization of CD44+ALDH1+Ki-67− cells in non-malignant and neoplastic lesions of the breast. Anticancer Res. 36, 4629–4638 (2016).
Da Cruz Paula, A. et al. Molecular characterization of CD44(+)/CD24(−)/Ck(+)/CD45(−) cells in benign and malignant breast lesions. Virchows. Arch. 470, 311–322 (2017).
Biostatistics: a foundation for analysis in the health sciences. 7th edn, (John Wiley @ Sons, 1999).
Acknowledgements
We thank the MGH/DFHCC Histopathology Core and MGH Breast Pathology Department for technical support. This work was supported by awards from the National Cancer Institute (R01 CA185086, C06 CA05926), Susan G. Komen for the Cure (IIR 12223648), and Harvard-Ludwig Center for Cancer Research (to S. Ramaswamy). S. Ramaswamy was also supported by a Stand Up to Cancer Innovative Research Grant (Grant number SU2C-AACR-IRG0911). Stand Up To Cancer is a program of the Entertainment Industry Foundation administered by the American Association for Cancer Research. X. Sole was supported by a “Bolsa de Ampliacion de Estudios, Instituto de Salud Carlos III, Ministerio de Economía y Competitividad (BA12/00021)”—Spanish postdoctoral fellowship award. D. Sgroi is supported in part by a grant from the Breast Cancer Research Foundation and the Avon Foundation.
Author information
Authors and Affiliations
Contributions
S.K., X.S. and S.R. designed the experiments, analyzed the data and wrote the first draft of the manuscript. D.S. and Y.H. served as study pathologists. Y.H. and C.B. performed automated staining and acquired the microscopy images. All authors reviewed and contributed to the final draft of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kabraji, S., Sole, X., Huang, Y. et al. AKT1low quiescent cancer cells in ductal carcinoma in situ of the breast. npj Breast Cancer 5, 10 (2019). https://doi.org/10.1038/s41523-019-0105-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-019-0105-y
This article is cited by
-
Diversity and molecular network patterns of symptom phenotypes
npj Systems Biology and Applications (2021)