Abstract
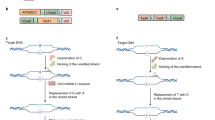
Base editors (BEs) empower the efficient installation of beneficial or corrective point mutations in crop and human genomes. However, conventional BEs can induce unpredictable guide RNA (gRNA)-independent off-target edits in the genome and transcriptome due to spurious activities of BE-enclosing deaminases, and current improvements mostly rely on deaminase-specific mutagenesis or exogenous regulators. Here we developed a split deaminase for safe editing (SAFE) system applicable to BEs containing distinct cytidine or adenosine deaminases, with no need of external regulators. In SAFE, a BE was properly split at a deaminase domain embedded inside a Cas9 nickase, simultaneously fragmenting and deactivating both the deaminase and the Cas9 nickase. The gRNA-conditioned BE reassembly conferred robust on-target editing in plant, human and yeast cells, while minimizing both gRNA-independent and gRNA-dependent off-target DNA/RNA edits. SAFE also substantially increased product purity by eliminating indels. Altogether, SAFE provides a generalizable solution for BEs to suppress off-target editing and improve on-target performance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The amplicon data, WGS data and RNA-seq data reported in this paper can be found at the CNGB Sequence Archive of the China National GeneBank DataBase with the accession number CNP0004043. Amino acid sequences of different SAFE BEs and target amplicon sequences for the gRNAs are provided in the Supplementary Information. Source data are provided with this paper.
References
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Nishida, K. et al. Targeted nucleotide editing using hybrid prokaryotic and vertebrate adaptive immune systems. Science 353, aaf8729 (2016).
Gaudelli, N. M. et al. Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Kang, B. C. et al. Precision genome engineering through adenine base editing in plants. Nat. Plants 4, 427–431 (2018).
Kim, J. S. Precision genome engineering through adenine and cytosine base editing. Nat. Plants 4, 148–151 (2018).
Zhang, H. et al. Genome editing of upstream open reading frames enables translational control in plants. Nat. Biotechnol. 36, 894–898 (2018).
Xue, C., Zhang, H., Lin, Q., Fan, R. & Gao, C. Manipulating mRNA splicing by base editing in plants. Sci. China Life Sci. 61, 1293–1300 (2018).
Chen, K., Wang, Y., Zhang, R., Zhang, H. & Gao, C. CRISPR/Cas genome editing and precision plant breeding in agriculture. Annu. Rev. Plant Biol. 70, 667–697 (2019).
Li, Z., Xiong, X., Wang, F., Liang, J. & Li, J. F. Gene disruption through base editing-induced messenger RNA missplicing in plants. New Phytol. 222, 1139–1148 (2019).
Molla, K. A., Sretenovic, S., Bansal, K. C. & Qi, Y. Precise plant genome editing using base editors and prime editors. Nat. Plants 7, 1166–1187 (2021).
Xue, C. et al. Tuning plant phenotypes by precise, graded downregulation of gene expression. Nat. Biotechnol. https://doi.org/10.1038/s41587-023-01707-w (2023).
Rees, H. A. et al. Improving the DNA specificity and applicability of base editing through protein engineering and protein delivery. Nat. Commun. 8, 15790 (2017).
Kim, D., Kim, D. E., Lee, G., Cho, S. I. & Kim, J. S. Genome-wide target specificity of CRISPR RNA-guided adenine base editors. Nat. Biotechnol. 37, 430–435 (2019).
Liang, Z. et al. Genome editing of bread wheat using biolistic delivery of CRISPR/Cas9 in vitro transcripts or ribonucleoproteins. Nat. Protoc. 13, 413–430 (2018).
Kim, D., Luk, K., Wolfe, S. A. & Kim, J. S. Evaluating and enhancing target specificity of gene-editing nucleases and deaminases. Annu. Rev. Biochem. 88, 191–220 (2019).
Jin, S. et al. Cytosine, but not adenine, base editors induce genome-wide off-target mutations in rice. Science 364, 292–295 (2019).
Zuo, E. et al. Cytosine base editor generates substantial off-target single-nucleotide variants in mouse embryos. Science 364, 289–292 (2019).
Grünewald, J. et al. Transcriptome-wide off-target RNA editing induced by CRISPR-guided DNA base editors. Nature 569, 433–437 (2019).
Zhou, C. et al. Off-target RNA mutation induced by DNA base editing and its elimination by mutagenesis. Nature 571, 275–278 (2019).
Gehrke, J. M. et al. An APOBEC3A–Cas9 base editor with minimized bystander and off-target activities. Nat. Biotechnol. 36, 977–982 (2018).
Grünewald, J. et al. CRISPR DNA base editors with reduced RNA off-target and self-editing activities. Nat. Biotechnol. 37, 1041–1048 (2019).
Rees, H. A., Wilson, C., Doman, J. L. & Liu, D. R. Analysis and minimization of cellular RNA editing by DNA adenine base editors. Sci. Adv. 5, eaax5717 (2019).
Doman, J. L., Raguram, A., Newby, G. A. & Liu, D. R. Evaluation and minimization of Cas9-independent off-target DNA editing by cytosine base editors. Nat. Biotechnol. 38, 620–628 (2020).
Jin, S. et al. Rationally designed APOBEC3B cytosine base editors with improved specificity. Mol. Cell 79, 728–740 (2020).
Richter, M. F. et al. Phage-assisted evolution of an adenine base editor with improved Cas domain compatibility and activity. Nat. Biotechnol. 38, 883–891 (2020).
Yu, Y. et al. Cytosine base editors with minimized unguided DNA and RNA off-target events and high on-target activity. Nat. Commun. 11, 2052 (2020).
Neugebauer, M. E. et al. Evolution of an adenine base editor into a small, efficient cytosine base editor with low off-target activity. Nat. Biotechnol. 41, 673–685 (2023).
Chen, L. et al. Re-engineering the adenine deaminase TadA-8e for efficient and specific CRISPR-based cytosine base editing. Nat. Biotechnol. 41, 663–672 (2023).
Wang, L. et al. Eliminating base-editor-induced genome-wide and transcriptome-wide off-target mutations. Nat. Cell Biol. 23, 552–563 (2021).
Berríos, K. N. et al. Controllable genome editing with split-engineered base editors. Nat. Chem. Biol. 17, 1262–1270 (2021).
Liu, Y. et al. A Cas-embedding strategy for minimizing off-target effects of DNA base editors. Nat. Commun. 11, 6073 (2020).
Li, S. et al. Docking sites inside Cas9 for adenine base editing diversification and RNA off-target elimination. Nat. Commun. 11, 5827 (2020).
Nguyen Tran, M. T. et al. Engineering domain-inlaid SaCas9 adenine base editors with reduced RNA off-targets and increased on-target DNA editing. Nat. Commun. 11, 4871 (2020).
Liu, K. et al. Mapping single-cell-resolution cell phylogeny reveals cell population dynamics during organ development. Nat. Methods 18, 1506–1514 (2021).
Wang, Y., Zhou, L., Liu, N. & Yao, S. BE-PIGS: a base-editing tool with deaminases inlaid into Cas9 PI domain significantly expanded the editing scope. Signal Transduct. Target. Ther. 4, 36 (2019).
Xiong, X. et al. A cytosine base editor toolkit with varying activity windows and target scopes for versatile gene manipulation in plants. Nucleic Acids Res. 50, 3565–3580 (2022).
Pham, P. et al. Structural analysis of the activation-induced deoxycytidine deaminase required in immunoglobulin diversification. DNA Repair 43, 48–56 (2016).
Zong, Y. et al. Precise base editing in rice, wheat and maize with a Cas9–cytidine deaminase fusion. Nat. Biotechnol. 35, 438–440 (2017).
Wang, Z. P. et al. Egg cell-specific promoter-controlled CRISPR/Cas9 efficiently generates homozygous mutants for multiple target genes in Arabidopsis in a single generation. Genome Biol. 16, 144 (2015).
Chen, Y. et al. CRISPR/Cas9-mediated base-editing system efficiently generates gain-of-function mutations in Arabidopsis. Sci. China Life Sci. 60, 520–523 (2017).
Powles, S. B. & Yu, Q. Evolution in action: plants resistant to herbicides. Annu. Rev. Plant Biol. 61, 317–347 (2010).
Jin, S., Gao, Q. & Gao, C. An unbiased method for evaluating the genome-wide specificity of base editors in rice. Nat. Protoc. 16, 431–457 (2021).
DiCarlo, J. E. et al. Genome engineering in Saccharomyces cerevisiae using CRISPR–Cas systems. Nucleic Acids Res. 41, 4336–4343 (2013).
Long, H. et al. Antibiotic treatment enhances the genome-wide mutation rate of target cells. Proc. Natl Acad. Sci. USA 113, E2498–E2505 (2016).
Ren, Q. et al. Improved plant cytosine base editors with high editing activity, purity, and specificity. Plant Biotechnol. J. 19, 2052–2068 (2021).
Randall, L. B. et al. Genome- and transcriptome-wide off-target analyses of an improved cytosine base editor. Plant Physiol. 187, 73–87 (2021).
Li, S., Liu, L., Sun, W., Zhou, X. & Zhou, H. A large-scale genome and transcriptome sequencing analysis reveals the mutation landscapes induced by high-activity adenine base editors in plants. Genome Biol. 23, 51 (2022).
Zong, Y. et al. Efficient C-to-T base editing in plants using a fusion of nCas9 and human APOBEC3A. Nat. Biotechnol. 36, 950–953 (2018).
Lapinaite, A. et al. DNA capture by a CRISPR–Cas9-guided adenine base editor. Science 369, 566–571 (2020).
Chen, L. et al. Engineering a precise adenine base editor with minimal bystander editing. Nat. Chem. Biol. 19, 101–110 (2023).
Liang, P. et al. Genome-wide profiling of adenine base editor specificity by EndoV-seq. Nat. Commun. 10, 67 (2019).
Jiang, F., Zhou, K., Ma, L., Gressel, S. & Doudna, J. A. A Cas9–guide RNA complex preorganized for target DNA recognition. Science 348, 1477–1481 (2015).
Jiang, F. et al. Structures of a CRISPR–Cas9 R-loop complex primed for DNA cleavage. Science 351, 867–871 (2016).
Kummerfeld, S. K. & Teichmann, S. A. Relative rates of gene fusion and fission in multi-domain proteins. Trends Genet. 21, 25–30 (2005).
Chee, W. K. D., Yeoh, J. W., Dao, V. L. & Poh, C. L. Highly reversible tunable thermal-repressible split-T7 RNA polymerases (Thermal-T7RNAPs) for dynamic gene regulation. ACS Synth. Biol. 11, 921–937 (2022).
Komor, A. C. et al. Improved base excision repair inhibition and bacteriophage Mu Gam protein yields C:G-to-T:A base editors with higher efficiency and product purity. Sci. Adv. 3, eaao4774 (2017).
Wang, L. et al. Enhanced base editing by co-expression of free uracil DNA glycosylase inhibitor. Cell Res. 27, 1289–1292 (2017).
Kim, H. S., Jeong, Y. K., Hur, J. K., Kim, J. S. & Bae, S. Adenine base editors catalyze cytosine conversions in human cells. Nat. Biotechnol. 37, 1145–1148 (2019).
Wu, Y. et al. Genome-wide analyses of PAM-relaxed Cas9 genome editors reveal substantial off-target effects by ABE8e in rice. Plant Biotechnol. J. 20, 1670–1682 (2022).
Li, Z. et al. A potent Cas9-derived gene activator for plant and mammalian cells. Nat. Plants 3, 930–936 (2017).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Nishimura, A., Aichi, I. & Matsuoka, M. A protocol for Agrobacterium-mediated transformation in rice. Nat. Protoc. 1, 2796–2802 (2006).
Liu, Q. et al. Hi-TOM: a platform for high-throughput tracking of mutations induced by CRISPR/Cas systems. Sci. China Life Sci. 62, 1–7 (2019).
Gietz, R. D. & Schiestl, R. H. Quick and easy yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat. Protoc. 2, 35–37 (2007).
Lõoke, M., Kristjuhan, K. & Kristjuhan, A. Extraction of genomic DNA from yeasts for PCR-based applications. BioTechniques 50, 325–328 (2011).
Hwang, G. H. et al. Web-based design and analysis tools for CRISPR base editing. BMC Bioinform. 19, 542 (2018).
Clement, K. et al. CRISPResso2 provides accurate and rapid genome editing sequence analysis. Nat. Biotechnol. 37, 224–226 (2019).
Xie, X. et al. CRISPR-GE: a convenient software toolkit for CRISPR-based genome editing. Mol. Plant 10, 1246–1249 (2017).
Bae, S., Park, J. & Kim, J. S. Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30, 1473–1475 (2014).
Wu, X. et al. Genome-wide binding of the CRISPR endonuclease Cas9 in mammalian cells. Nat. Biotechnol. 32, 670–676 (2014).
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Shen, M. W. et al. Predictable and precise template-free CRISPR editing of pathogenic variants. Nature 563, 646–651 (2018).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Acknowledgements
This work was supported by the National Key Research and Development Program of China grant no. 2019YFA0906202, the Guangdong Provincial Key Project for Basic and Applied Basic Research (Cornerstone grant) and the National Natural Science Foundation of China (NSFC) grant no. 32125004 to J.-F.L.; NSFC grant no. 32293191 to X.H.; NSFC grant no. 32200494 and the China Postdoctoral Science Foundation grant no. 2022M723662 to K.L.; and NSFC grant no. 32100485 to F.-N.X. We thank Azenta Life Sciences (Suzhou, China) for help in deep sequencing and Y. Chen for assistance in flow cytometry.
Author information
Authors and Affiliations
Contributions
J.-F.L. and K.L. conceived and designed the study. X.X., K.L., Z.L., F.-N.X. and X.-M.R. performed the experiments. K.L. and X.X. analysed the data. J.-F.L. and X.H. supervised the research. J.-F.L. wrote the manuscript. All authors approved the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
X.X., K.L., Z.L., X.H. and J.-F.L. have been granted a China invention patent (ZL202210503831.5) based on some results reported in this paper. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Seiichi Toki and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 N- or C-terminal split fragment of PIGS-AID10 alone is catalytically inactive.
a, PIGS-AID10 exhibits higher editing efficiency than N-AID10. Comparison of C-to-T editing efficiencies between PIGS-AID10 and N-AID10 was conducted at indicated Arabidopsis endogenous genomic loci in protoplasts by deep sequencing of target amplicons. Data are shown as mean values of two biological replicates. Ctrl corresponds to cells transfected with the gRNA only. b, Amino acid sequence of AID10 with seven candidate split sites highlighted in red and the catalytic center shadowed in blue. c,d, None of the N-terminal (c) or C-terminal (d) split fragments of PIGS-AID10 alone can induce CBE reporter activity in the presence of gRNA. The gRNA only was used as a negative control (Ctrl). Relative luciferase (LUC) activity was calculated by setting the LUC activity of the unsplit CBE (PIGS-AID10) as 100%. Data are shown as mean values of two biological replicates.
Extended Data Fig. 2 Split-AID10 can edit the AtALS locus to confer herbicide resistance.
a, The AtALS target locus. Intended C·G to T·A (red) conversion by Split-AID10 would introduce an A-to-T amino acid substitution. The PAM is in green. b, Summary of Split-AID10 editing outcome in transgenic Arabidopsis T1 plants. The editing efficiency was calculated as the percentage of transgenic plants containing indicated mutation type at the target site. WT, Ho, He, Bi, and Chi denote wild type, homozygous, heterozygous, biallelic, and chimeric plants, respectively. c, Sanger sequencing validates homozygous C-to-T mutation introduced by Split-AID10. d, Homozygous C-to-T mutation at the AtALS locus confers the plant with imazethapyr herbicide resistance. Transgenic Arabidopsis T1 plants of 40-day-old were sprayed with 30 mg/l imazethapyr (IMZ) herbicide once and were photographed in one month. Unedited plants died of imazethapyr toxicity.
Extended Data Fig. 3 Split-AID10 enables robust on-target editing in human and yeast cells.
a,c, Split-AID10 exhibits sufficiently high on-target editing efficiency in human HEK293T cells (a) and yeast cells (c). Data are shown as mean values and standard deviation of three biological replicates for six target loci and the highest C-to-T editing efficiency among multiple Cs was used to represent the editing efficiency at a given target locus. b,d, Split-AID10 minimizes C-to-A/G and indel byproducts in human HEK293T cells (b) and yeast cells (d). The inlaid panel in d corresponds to a magnified view. Each dot in the violin plot represents the editing efficiency for the indicated editing product per target site, while the three lines mark quartile positions. e, Domain structures of Split-AID10N6, Split-AID10C6, and PIGS-AID10N6. Note that PIGS-AID10N6 contains a complete nCas9 and an intact deaminase catalytic center. f, None of Split-AID10N6, Split-AID10C6 and PIGS-AID10N6 is catalytically active in yeast cells. Data are shown as mean values and standard deviation of three biological replicates.
Extended Data Fig. 4 Rationale of visible selection of on-target editing in the EASY assay.
The intended nonsense mutation in ScAde1 or ScCan1 by converting a Trp or Gln codon (shadowed in yellow) to a stop codon via C-to-T editing (red) makes edited yeast clones visually distinguishable. The on-target C-to-T editing of ScAde1 generated red colonies in YPD agar plates, while that of ScCan1 produced white colonies in SC-Arg agar plates containing L-Canavanine.
Extended Data Fig. 5 Split-AID10 minimizes off-target DNA edits in human cells.
a,b, R-loop assay reveals that Split-AID10 eliminates unguided DNA edits relative to other tested CBEs in HEK293T cells. In a, data are shown as mean values and standard deviation of three biological replicates. In b, each dot in the violin plot represents the C-to-T editing efficiency per target site, while the three lines mark quartile positions. ns, not significant, two-sided Mann-Whitney U test. c, Split-AID10 induces substantially reduced edits at predicted off-target sites for the HEK-1 or HEK-5 targeting gRNA in HEK293T cells. Data are shown as mean values and standard deviation of three biological replicates.
Extended Data Fig. 6 Split-AID10 eliminates off-target RNA edits in human cells.
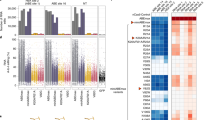
a,b, Gating strategies of flow cytometry for sorting base editor-expressing HEK293T cells and control cells. In a, cells expressing base editors were flow-sorted for the top 5% of gated cells (% parent) with the highest GFP (488 nm) and mScarlet (561 nm) signal. In b, the nCas9-UGI control was sorted for the top 5% of gated cells with the highest signal and for a population of cells with a mean fluorescence intensity matching the top 5% of BE3-transfected cells collected on the same day. c, On-target DNA editing validation for all base editor groups in HEK293T cell-based RNA off-target assay. Data are shown for three biological replicates (Rep.1-Rep.3). The lowercase number indicates the position of individual Cs in the protospacer (counting PAM as 21-23). d, Representative Manhattan plot (Rep.3) for transcriptome-wide off-target C-to-U edits in individual chromosomes in each group. n, total number of C-to-U edits. Dashed lines indicate a C-to-U editing level of 5%.
Extended Data Fig. 7 Split-BE3 eliminates off-target RNA edits in human cells.
Representative Manhattan plot (Rep.2) for transcriptome-wide off-target C-to-U edits in individual chromosomes in each group. n, total number of C-to-U edits. Dashed lines indicate a C-to-U editing level of 5%.
Extended Data Fig. 8 Split-ABE8e minimizes off-target RNA/DNA edits.
a,b, None of the N-terminal (a) or C-terminal (b) split fragments of PIGS-ABE8e alone can induce ABE reporter activity in protoplasts in the presence of gRNA. The gRNA only was used as a negative control (Ctrl). Relative luciferase (LUC) activity was calculated by setting the LUC activity of the unsplit ABE (PIGS-ABE8e) as 100%. Data are shown as mean values of two biological replicates. c, Representative Manhattan plot (Rep.3) for transcriptome-wide off-target A-to-I edits in individual chromosomes in each group in human HEK293T cell-based RNA off-target assay. n, total number of A-to-I edits. Dashed lines indicate a A-to-I editing level of 5%. d, Split-ABE8e induces substantially reduced edits at predicted off-target sites for the HEK-8 targeting gRNA in HEK293T cells. Data are shown as mean values and standard deviation of three biological replicates.
Extended Data Fig. 9 Split fragments of a BE are expressed at higher levels than the unsplit BE.
a, Split-AID10N6 and Split-AID10C6 are expressed at higher levels than PIGS-AID10 in both rice and Arabidopsis protoplasts. b, Split-ABE8eN6 and Split-ABE8eC6 are expressed at higher levels than PIGS-ABE8e in both rice and Arabidopsis protoplasts. In (a) and (b), 200 µL rice or Arabidopsis protoplasts were transfected with equal amounts of plasmid(s) encoding PIGS-BE alone or Split-BE-N plus Split-BE-C. Since Split-BE-N and Split-BE-C carry the same protein tag (2×HA tag for both PIGS-AID10 split fragments and 2×FLAG tag for both PIGS-ABE8e split fragments), Split-BE-N or Split-BE-C was also expressed alone to help distinguish individual products. A plasmid encoding GFP was co-transfected as an internal transfection control. The experiments were conducted twice with similar results. The white, orange, and blue arrowheads mark PIGS-BE, Split-BE-N, and Split-BE-C, respectively. Rubisco staining indicates equal protein loading.
Supplementary information
Supplementary Information
Supplementary Tables 1–5 and Sequences.
Source data
Source Data Extended Data Fig. 9
Unprocessed western blots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiong, X., Liu, K., Li, Z. et al. Split complementation of base editors to minimize off-target edits. Nat. Plants 9, 1832–1847 (2023). https://doi.org/10.1038/s41477-023-01540-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-023-01540-8