Abstract
The seasonal dynamics of the vegetation canopy strongly regulate the surface energy balance and terrestrial carbon fluxes, providing feedbacks to climate change. Whether the seasonal timing of maximum canopy structure was optimized to achieve a maximum photosynthetic carbon uptake is still not clear due to the complex interactions between abiotic and biotic factors. We used two solar-induced chlorophyll fluorescence datasets as proxies for photosynthesis and the normalized difference vegetation index and leaf area index products derived from the moderate resolution imaging spectroradiometer as proxies for canopy structure, to characterize the connection between their seasonal peak timings from 2000 to 2018. We found that the seasonal peak was earlier for photosynthesis than for canopy structure in >87.5% of the northern vegetated area, probably leading to a suboptimal maximum seasonal photosynthesis. This mismatch in peak timing significantly increased during the study period, mainly due to the increasing atmospheric CO2, and its spatial variation was mainly explained by climatic variables (43.7%) and nutrient limitations (29.6%). State-of-the-art ecosystem models overestimated this mismatch in peak timing by simulating a delayed seasonal peak of canopy development. These results highlight the importance of incorporating the mechanisms of vegetation canopy dynamics to accurately predict the maximum potential terrestrial uptake of carbon under global environmental change.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The CSIF dataset is from https://doi.org/10.17605/OSF.IO/8XQY6. The GOME-2 SIF dataset is from https://avdc.gsfc.nasa.gov/pub/data/satellite/MetOp/GOME_F/. The MODIS NDVI dataset is from https://lpdaac.usgs.gov/products/mod13c1v006/. The reprocessed LAI dataset is from http://globalchange.bnu.edu.cn/research/laiv6. The FLUXNET2015 dataset is from https://fluxnet.org/data/fluxnet2015-dataset/. The surface air temperature and Rad datasets are from https://rda.ucar.edu/datasets/ds314.3/. The SWC dataset is from https://disc.gsfc.nasa.gov/datasets/GLDAS_NOAH025_3H_2.1/summary?keywords=GLDAS. The SLA, Nm and Pm datasets are from https://github.com/abhirupdatta/global_maps_of_plant_traits. The canopy height and maximum rooting depth datasets are from https://webmap.ornl.gov/ogc/dataset.jsp?dg_id=10023_1 and https://wci.earth2observe.eu/thredds/catalog/usc/root-depth/catalog.html. The ASR and plant species datasets are from https://ecotope.org/anthromes/biodiversity/plants/data/ and https://databasin.org/datasets/43478f840ac84173979b22631c2ed672/. The tree density dataset is from https://elischolar.library.yale.edu/yale_fes_data/1/.
Code availability
All computer codes for the analysis of the data are available from the corresponding author on reasonable request.
Change history
11 January 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41477-023-01342-y
References
Piao, S., Friedlingstein, P., Ciais, P., Viovy, N. & Demarty, J. Growing season extension and its impact on terrestrial carbon cycle in the Northern Hemisphere over the past 2 decades. Glob. Biogeochem. Cycles 21, GB3018 (2007).
Richardson, A. D. et al. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agric. For. Meteorol. 169, 156–173 (2013).
Xia, J., Niu, S., Ciais, P. & Janssens, I. A. Joint control of terrestrial gross primary productivity by plant phenology and physiology. Proc. Natl Acad. Sci. USA 112, 2788–2793 (2015).
Yang, J. et al. Divergent shifts in peak photosynthesis timing of temperate and alpine grasslands in China. Remote Sens. Environ. 233, 111395 (2019).
Huang, K., Xia, J., Wang, Y. & Ahlstrom, A. Enhanced peak growth of global vegetation and its key mechanisms. Nat. Ecol. Evol. 2, 1897–1905 (2018).
Park, T., Chen, C. & Macias-Fauria, M. Changes in timing of seasonal peak photosynthetic activity in northern ecosystems. Glob. Change Biol. 25, 2382–2395 (2019).
Medlyn, B. E. Physiological basis of the light use efficiency model. Tree Physiol. 18, 167 (1998).
Turner, D. P., Urbanski, S., Bremer, D., Wofsy, S. C. & Gregory, M. A cross-biome comparison of daily light use efficiency for gross primary production. Glob. Change Biol. 9, 383–395 (2003).
Monteith, J. L. Solar radiation and productivity in tropical ecosystems. Appl. Ecol. 9, 747–766 (1972).
Wang, H. et al. Towards a universal model for carbon dioxide uptake by plants. Nat. Plants 3, 734–741 (2017).
Zhang, Y., Joiner, J., Alemohammad, S. H., Zhou, S. & Gentine, P. A global spatially contiguous solar-induced fluorescence (CSIF) dataset using neural networks. Biogeosciences 15, 5779–5800 (2018).
Frankenberg, C. et al. New global observations of the terrestrial carbon cycle from GOSAT: patterns of plant fluorescence with gross primary productivity. Geophys. Res. Lett. 38, L17706 (2011).
Yuan, H., Dai, Y., Xiao, Z., Ji, D. & Shangguan, W. Reprocessing the MODIS Leaf Area Index products for land surface and climate modelling. Remote Sens. Environ. 115, 1171–1187 (2011).
Elith, J., Leathwick, J. R. & Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 77, 802–813 (2008).
Wang, X. et al. Globally consistent patterns of asynchrony in vegetation phenology derived from optical, microwave, and fluorescence satellite data. J. Geophys. Res. Biogeosci. 125, e2020JG005732 (2020).
Poorter, H. et al. Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol. 193, 30–50 (2012).
Zhang, Y., Commane, R., Zhou, S., Williams, A. P. & Gentine, P. Light limitation regulates the response of autumn terrestrial carbon uptake to warming. Nat. Clim. Change 10, 739–743 (2020).
Yuan, W. et al. Global comparison of light use efficiency models for simulating terrestrial vegetation gross primary production based on the LaThuile database. Agric. For. Meteorol. 192-193, 108–120 (2014).
Reich, P. B. et al. Temperature drives global patterns in forest biomass distribution in leaves, stems, and roots. Proc. Natl Acad. Sci. USA 111, 13721–13726 (2014).
Wright, I. J., Reich, P. B. & Westoby, M. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004).
Reich, P. B., Oleksyn, J. & Wright, I. J. Leaf phosphorus influences the photosynthesis–nitrogen relation: a cross-biome analysis of 314 species. Oecologia 160, 207–212 (2009).
Chen, Y., Han, W., Tang, L., Tang, Z. & Fang, J. Leaf nitrogen and phosphorus concentrations of woody plants differ in responses to climate, soil and plant growth form. Ecography 36, 178–184 (2013).
Jiang, M., Caldararu, S., Zaehle, S., Ellsworth, D. S. & Medlyn, B. E. Towards a more physiological representation of vegetation phosphorus processes in land surface models. New Phytol. 222, 1223–1229 (2019).
Kergoat, L., Lafont, S., Arneth, A., Le Dantec, V. & Saugier, B. Nitrogen controls plant canopy light-use efficiency in temperate and boreal ecosystems. J. Geophys. Res. Biogeosci. 113, G04017 (2008).
Du, E. et al. Global patterns of terrestrial nitrogen and phosphorus limitation. Nat. Geosci. 13, 221–226 (2020).
Cleveland, C. C. et al. Patterns of new versus recycled primary production in the terrestrial biosphere. Proc. Natl Acad. Sci. USA 110, 12733–12737 (2013).
Veneklaas, E. J. et al. Opportunities for improving phosphorus-use efficiency in crop plants. New Phytol. 195, 306–320 (2012).
Janssens, I. A. & Luyssaert, S. Nitrogen’s carbon bonus. Nat. Geosci. 2, 318–319 (2009).
Luo, X. et al. Global variation in the fraction of leaf nitrogen allocated to photosynthesis. Nat. Commun. 12, 4866 (2021).
Lambers, H., Iii, F. & Pons, T. L. Plant Physiological Ecology (Springer, 2008).
Vose, J. M. et al. Factors influencing the amount and distribution of leaf area of pine stands. Ecol. Bull. 43, 102−114 (1994).
Carter, S. K., Saenz, D. & Rudolf, V. H. W. Shifts in phenological distributions reshape interaction potential in natural communities. Ecol. Lett. 21, 1143–1151 (2018).
Sitch, S. et al. Recent trends and drivers of regional sources and sinks of carbon dioxide. Biogeosciences 12, 653–679 (2015).
Farquhar, G. D., von Caemmerer, S. & Berry, J. A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149, 78–90 (1980).
Krinner, G. et al. A dynamic global vegetation model for studies of the coupled atmosphere–biosphere system. Glob. Biogeochem. Cycles 19, GB1015 (2005).
Murray-Tortarolo, G. et al. Evaluation of land surface models in reproducing satellite-derived LAI over the high-latitude Northern Hemisphere. Part I: Uncoupled DGVMs. Remote Sens. 5, 4819–4838 (2013).
Lawrence, D. M. et al. The community land model version 5: description of new features, benchmarking, and impact of forcing uncertainty. J. Adv. Model. Earth Syst. 11, 4245–4287 (2019).
Goll, D. S., Winkler, A. J. & Raddatz, T. Carbon–nitrogen interactions in idealized simulations with JSBACH (version 3.10). Geosci. Model Dev. 10, 2009–2030 (2017).
Goll, D. S., Vuichard, N. & Maignan, F. A representation of the phosphorus cycle for ORCHIDEE (revision 4520). Geosci. Model Dev. 10, 3745–3770 (2017).
Sun, Y., Goll, D. S. & Chang, J. Global evaluation of the nutrient-enabled version of the land surface model ORCHIDEE-CNP v1.2 (r5986). Geosci. Model Dev. 14, 1987–2010 (2021).
Clark, D. B., Mercado, L. M. & Sitch, S. The Joint UK Land Environment Simulator (JULES), model description—Part 2: Carbon fluxes and vegetation dynamics. Geosci. Model Dev. 4, 701–722 (2011).
Terrer, C. et al. Nitrogen and phosphorus constrain the CO2 fertilization of global plant biomass. Nat. Clim. Change 9, 684–689 (2019).
Reyes-Fox, M. et al. Elevated CO2 further lengthens growing season under warming conditions. Nature 510, 259–262 (2014).
Guanter, L. et al. Global and time-resolved monitoring of crop photosynthesis with chlorophyll fluorescence. Proc. Natl Acad. Sci. USA 111, E1327–E1333 (2014).
Sun, Y. et al. OCO-2 advances photosynthesis observation from space via solar-induced chlorophyll fluorescence. Science 358, eaam5747 (2017).
Joiner, J. et al. The seasonal cycle of satellite chlorophyll fluorescence observations and its relationship to vegetation phenology and ecosystem atmosphere carbon exchange. Remote Sens. Environ. 152, 375–391 (2014).
Chu, D. et al. Long time-series NDVI reconstruction in cloud-prone regions via spatio-temporal tensor completion. Remote Sens. Environ. 264, 112632 (2021).
Joiner, J. et al. Global monitoring of terrestrial chlorophyll fluorescence from moderate-spectral-resolution near-infrared satellite measurements: methodology, simulations, and application to GOME-2. Atmos. Meas. Tech. 6, 2803–2823 (2013).
Zhang, Y., Joiner, J., Gentine, P. & Zhou, S. Reduced solar-induced chlorophyll fluorescence from GOME-2 during Amazon drought caused by dataset artifacts. Glob. Change Biol. 24, 2229–2230 (2018).
Rodell, M., Houser, P. R. & Jambor, U. The Global Land Data Assimilation System. Bull. Am. Meteorol. Soc. 85, 381–394 (2004).
Pastorello, G. et al. The FLUXNET2015 dataset and the ONEFlux processing pipeline for eddy covariance data. Sci. Data 7, 225 (2020).
Reichstein, M. et al. On the separation of net ecosystem exchange into assimilation and ecosystem respiration: review and improved algorithm. Glob. Change Biol. 11, 1424–1439 (2005).
LASSLOP, G. et al. Separation of net ecosystem exchange into assimilation and respiration using a light response curve approach: critical issues and global evaluation. Glob. Change Biol. 16, 187–208 (2010).
Vautard, R., Yiou, P. & Ghil, M. Singular-spectrum analysis: a toolkit for short, noisy chaotic signals. Phys. D. 58, 95–126 (1992).
Zhou, S. et al. Dominant role of plant physiology in trend and variability of gross primary productivity in North America. Sci. Rep. 7, 41366 (2017).
Butler, E. E., Datta, A. & Flores-Moreno Mapping local and global variability in plant trait distributions. Proc. Natl Acad. Sci. USA 114, E10937–E10946 (2017).
Simard, M., Pinto, N., Fisher, J. B. & Baccini, A. Mapping forest canopy height globally with spaceborne lidar. J. Geophys. Res. Biogeosci. 116, G04021 (2011).
Ellis, E. C., Antill, E. C. & Kreft, H. All is not loss: plant biodiversity in the anthropocene. PLoS ONE 7, e30535 (2012).
Kier, G., Mutke, J., Dinerstein, E., Ricketts, T. H. & Barthlott, W. Global patterns of plant diversity and floristic knowledge. J. Biogeogr. 32, 1107–1116 (2005).
Boles, S. H. et al. Land cover characterization of temperate East Asia using multi-temporal VEGETATION sensor data. Remote Sens. Environ. 90, 477–489 (2004).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (41988101), the Second Tibetan Plateau Scientific Expedition and Research Program (grant no. 2019QZKK0208) and the Xplorer Prize. Z.Z. was supported by the National Natural Science Foundation of China (41901122) and the Shenzhen Fundamental Research Program (GXWD20201231165807007-20200814213435001). J.P. was supported by the Spanish Government grant PID2019-110521GB-I00, the Fundación Ramón Areces grant CIVP20A6621 and the Catalan Government grants SGR2017-1005 and AGAUR-2020PANDE00117. We thank H. Vallicrosa from CSIC, Global Ecology Unit CREAF-CSIC-UAB for discussion. We also thank I. MacLachlan from Peking University for proofreading. We are grateful for the computational resources provided by the High-performance Computing Platform of Peking University’s supercomputing facility.
Author information
Authors and Affiliations
Contributions
S.P. and Z.Z. designed the study. Q.Z. performed the analysis. Q.Z., S.P. and Z.Z. wrote the initial draft. All authors, including H.Z., R.M., Y.Z. and J.P., contributed to the interpretation of the results and the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks William Smith and Sujong Jeong for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
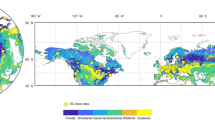
Extended Data Fig. 1 Comparison between the timings of seasonal peak photosynthesis and canopy structure in northern ecosystems based on multiple proxies.
Spatial patterns of the seasonal peak timing difference between photosynthesis and canopy structure represented by δDOYGOME-2 SIF, NDVI (a) and δDOYCSIF, LAI (b).
Extended Data Fig. 2 Mean differences of seasonal peak timings between photosynthesis and canopy structure for different ecosystem types.
Northern ecosystems (n = 2578665), forests (n = 788343), shrublands (n = 765928), and grasslands (n = 344205). Boxplots show the median, maximum, minimum, 25th, and 75th quartiles values (without outliers). The coloured letters represent significant differences (all p values = 9.56 × 10−10, two-sided Tukey’s HSD test) in average δDOYCSIF, NDVI among ecosystems estimated by one-way analysis of variance (ANOVA).
Extended Data Fig. 3 Illustration of the optimal GPPmax conceptual model.
Coloured curves indicate the seasonal cycles of environmental resources (Resource, blue), photosynthesis (GPP, orange), and canopy structure (NDVI, green). The seasonal peak timing of the canopy structure is adjusted to match the highest availability of environmental resources (DOYNDVI = DOYResource), and therefore the optimized maximum seasonal GPP was achieved.
Extended Data Fig. 4 Attribution of the trends in absolute δDOYCSIF, NDVI in northern ecosystems during 2000–2017.
a, Trends in spatially averaged absolute δDOYCSIF, NDVI derived from satellite observation (OBS) and BRT models (Predicted), and attributed respectively to rising CO2 (CO2), climate change (Climate), and other factors (Others). b-e, Spatial patterns of the trends in absolute δDOYCSIF, NDVI corresponding to the columns in a. The satellite observation was resampled to 0.5° to match the spatial resolution of explanatory variables in the BRT model.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7 and Tables 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Q., Zhu, Z., Zeng, H. et al. Seasonal peak photosynthesis is hindered by late canopy development in northern ecosystems. Nat. Plants 8, 1484–1492 (2022). https://doi.org/10.1038/s41477-022-01278-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-022-01278-9
This article is cited by
-
Solar-Induced Chlorophyll Fluorescence (SIF): Towards a Better Understanding of Vegetation Dynamics and Carbon Uptake in Arctic-Boreal Ecosystems
Current Climate Change Reports (2024)