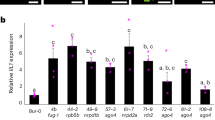
Abstract
Zinc (Zn) is an essential micronutrient for plants and animals owing to its structural and catalytic roles in many proteins1. Zn deficiency affects around 2 billion people, mainly those who live on plant-based diets relying on crops from Zn-deficient soils2,3. Plants maintain adequate Zn levels through tightly regulated Zn homeostasis mechanisms involving Zn uptake, distribution and storage4, but evidence of how they sense Zn status is lacking. Here, we use in vitro and in planta approaches to show that the Arabidopsis thaliana F-group bZIP transcription factors bZIP19 and bZIP23, which are the central regulators of the Zn deficiency response, function as Zn sensors by binding Zn2+ ions to a Zn-sensor motif. Deletions or modifications of this Zn-sensor motif disrupt Zn binding, leading to a constitutive transcriptional Zn deficiency response, which causes a significant increase in plant and seed Zn accumulation. As the Zn-sensor motif is highly conserved in F-group bZIP proteins across land plants, the identification of this plant Zn sensor will promote new strategies to improve the Zn nutritional quality of plant-derived food and feed, and contribute to tackling the global Zn-deficiency health problem.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Accession numbers of genes mentioned in this study are as follows: bZIP19, AT4G35040; bZIP23, AT2G16770; ZIP4, AT1G10970; ZIP5, AT1G05300; NAS2, AT5G56080; ACT2, AT3G18780. All data supporting the findings of this study are available in the main text, Extended Data Figs. 1–6 or in the Supplementary Information. Additional data related to this study are available from the corresponding author on request. All biological materials used in this study are available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Maret, W. & Li, Y. Coordination dynamics of zinc in proteins. Chem. Rev. 109, 4682–4707 (2009).
Welch, R. M. & Graham, R. D. Breeding for micronutrients in staple food crops from a human nutrition perspective. J. Exp. Bot. 55, 353–364 (2004).
Wessells, K. R. & Brown, K. H. Estimating the global prevalence of zinc deficiency: results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 7, e50568 (2012).
Clemens, S. Molecular mechanisms of plant metal tolerance and homeostasis. Planta 212, 475–486 (2001).
Andreini, C., Banci, L., Bertini, I. & Rosato, A. Zinc through the three domains of life. J. Proteome Res. 5, 3173–3178 (2006).
Choi, S. & Bird, A. J. Zinc’ing sensibly: controlling zinc homeostasis at the transcriptional level. Metallomics 6, 1198–1215 (2014).
Colvin, R. A., Holmes, W. R., Fontaine, C. P. & Maret, W. Cytosolic zinc buffering and muffling: their role in intracellular zinc homeostasis. Metallomics 2, 297–356 (2010).
Patzer, S. I. & Hantke, K. The ZnuABC high-affinity zinc uptake system and its regulator Zur in Escherichia coli. Mol. Microbiol. 28, 1199–1210 (1998).
Andrews, G. K. Cellular zinc sensors: MTF-1 regulation of gene expression. Biometals 14, 223–237 (2001).
Eide, D. J. Homeostatic and adaptive responses to zinc deficiency in Saccharomyces cerevisiae. J. Biol. Chem. 284, 18565–18569 (2009).
Assunção, A. G. L. et al. Arabidopsis thaliana transcription factors bZIP19 and bZIP23 regulate the adaptation to zinc deficiency. Proc. Natl Acad. Sci. USA 107, 10296–10301 (2010).
Vinson, C. R., Sigler, P. B. & McKnight, S. L. Scissors-grip model for DNA recognition by a family of leucine zipper proteins. Science 246, 911–916 (1989).
Deppmann, C. D., Alvania, R. S. & Taparowsky, E. J. Cross-species annotation of basic leucine zipper factor interactions: insight into the evolution of closed interaction networks. Mol. Biol. Evol. 23, 1480–1492 (2006).
Castro, P. H. et al. Phylogenetic analysis of F-bZIP transcription factors indicates conservation of the zinc deficiency response across land plants. Sci. Rep. 7, 3806 (2017).
Guerinot, M. & Lou The ZIP family of metal transporters. Biochim. Biophys. Acta 1465, 190–198 (2000).
Clemens, S., Deinlein, U., Ahmadi, H., Höreth, S. & Uraguchi, S. Nicotianamine is a major player in plant Zn homeostasis. BioMetals 26, 623–632 (2013).
Inaba, S. et al. Identification of putative target genes of bZIP19, a transcription factor essential for Arabidopsis adaptation to Zn deficiency in roots. Plant J. 84, 323–334 (2015).
Lilay, G. H., Castro, P. H., Campilho, A. & Assunção, A. G. L. The Arabidopsis bZIP19 and bZIP23 activity requires zinc deficiency—insight on regulation from complementation lines. Front. Plant Sci. 9, 1955 (2019).
Assunção, A. G. L. et al. Model of how plants sense zinc deficiency. Metallomics 5, 1110–1116 (2013).
Persson, D. P., Hansen, T. H., Laursen, K. H., Schjoerring, J. K. & Husted, S. Simultaneous iron, zinc, sulfur and phosphorus speciation analysis of barley grain tissues using SEC-ICP-MS and IP-ICP-MS. Metallomics 1, 418–426 (2009).
Azevedo, H. et al. Transcriptomic profiling of Arabidopsis gene expression in response to varying micronutrient zinc supply. Genom. Data 7, 256–258 (2016).
Maret, W. Zinc Biochemistry: from a single zinc enzyme to a key element of life. Adv. Nutr. 4, 82–91 (2013).
Krężel, A. & Maret, W. The biological inorganic chemistry of zinc ions. Arch. Biochem. Biophys. 611, 3–19 (2016).
Clemens, S. Metal ligands in micronutrient acquisition and homeostasis. Plant Cell Environ. 42, 2902–2912 (2019).
Kashiv, Y. et al. Imaging trace element distributions in single organelles and subcellular features. Sci. Rep. 6, 21437 (2016).
Zhao, H. et al. Regulation of zinc homeostasis in yeast by binding of the ZAP1 transcriptional activator to zinc-responsive promoter elements. J. Biol. Chem. 273, 28713–28720 (1998).
Lyons, T. J. et al. Genome-wide characterization of the Zap1p zinc-responsive regulon in yeast. Proc. Natl Acad. Sci. USA 97, 7957–7962 (2000).
Bird, A. J. et al. Zinc fingers can act as Zn2+ sensors to regulate transcriptional activation domain function. EMBO J. 22, 5137–5146 (2003).
Frey, A. G. et al. Zinc-regulated DNA binding of the yeast Zap1 zinc-responsive activator. PLoS ONE 6, e22535 (2011).
Kobayashi, T. Understanding the complexity of iron sensing and signaling cascades in plants. Plant Cell Physiol. 60, 1440–1446 (2019).
Kim, S. A., LaCroix, I. S., Gerber, S. A. & Guerinot, M. Lou. The iron deficiency response in Arabidopsis thaliana requires the phosphorylated transcription factor URI. Proc. Natl Acad. Sci. USA 116, 24933–24942 (2019).
Castro, P. H., Lilay, G. H. & Assuncao, A. G. L. in Plant Micronutrient Use Efficiency: Molecular and Genomic Perspectives in Crop Plants (eds Hossain M. A. et al.) Ch. 1, 1–10 (Academic Press Books, Elsevier, 2018).
Dubeaux, G., Neveu, J., Zelazny, E. & Vert, G. Metal sensing by the IRT1 transporter-receptor orchestrates its own degradation and plant metal nutrition. Mol. Cell 69, 953–964 (2018).
Cakmak, I. & Kutman, U. B. Agronomic biofortification of cereals with zinc: a review. Eur. J. Soil Sci. 69, 172–180 (2018).
White, P. J. & Broadley, M. R. Biofortification of crops with seven mineral elements often lacking in human diets–iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 182, 49–84 (2009).
Nazri, A. Z., Griffin, J. H. C., Peaston, K. A., Alexander-Webber, D. G. A. & Williams, L. E. F-group bZIPs in barley—a role in Zn deficiency. Plant. Cell Environ. 40, 2754–2770 (2017).
Evens, N. P., Buchner, P., Williams, L. E. & Hawkesford, M. J. The role of ZIP transporters and group F bZIP transcription factors in the Zn-deficiency response of wheat (Triticum aestivum). Plant J. 92, 291–304 (2017).
Lilay, G. H. et al. Rice F-bZIP transcription factors regulate the zinc deficiency response. J. Exp. Bot. 71, 3664–3677 (2020).
Shan, Q. et al. Targeted genome modification of crop plants using a CRISPR–Cas system. Nat. Biotechnol. 31, 686–688 (2013).
Earley, K. W. et al. Gateway-compatible vectors for plant functional genomics and proteomics. Plant J. 45, 616–629 (2006).
Jefferson, R. A., Kavanagh, T. A. & Bevan, M. W. GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6, 3901–3907 (1987).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Olsen, L. I. et al. Mother-plant-mediated pumping of zinc into the developing seed. Nat. Plants 2, 16036 (2016).
Acknowledgements
This work was supported by the Independent Research Fund Denmark, DFF-YDUN-program (4093–00245B); Portuguese Foundation for Science and Technology, FCT-IF program (IF/01641/2014), FCT-MCTES (PTDC/BAA-AGR/31122/2017; POCI-01-0145-FEDER- 031122); Novo Nordisk Foundation, Biotechnology-based Synthesis and Production Research program (NNF18OC0034598; to G.H.L., P.H.C., F.L. and A.G.L.A.); DFF-FTP-program (50544600-1126521001-112652; to D.P.P.) and the Netherlands Genome Initiative (40-41009-98-11084; to R.A. and M.G.M.A.). Element analysis was performed at CHEMI Center, and imaging data were collected at the CAB Center at PLEN, University of Copenhagen.
Author information
Authors and Affiliations
Contributions
G.H.L., D.P.P., P.H.C., F.L., R.D.A. and A.G.L.A. designed and performed experiments. G.H.L. and A.G.L.A. analysed and interpreted data. G.H.L., M.G.M.A. and A.G.L.A. wrote the manuscript. All of the authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Plants thanks Marc Hanikenne and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 SEC-ICP-MS chromatogram of MBP incubated with 67Zn stable isotope.
SEC-ICP-MS chromatogram showing a sample of maltose binding protein (MBP) incubated with 67Zn stable isotope. The MBP eluted after ~590 s and was detected both a, by UV and b, by a weak 48SO signal. The 48SO signal is weak due to the low-amount of S-containing amino acids in the MBP. c, There is no co-elution of 67Zn with the MBP at this retention time, showing that there is no Zn-binding to MBP. Free 67Zn eluted after ~680 s.
Extended Data Fig. 2 Shoot ionome analysis of the motif deletion mutant lines (bZIP19 del1 del2).
Shoot ionome analysis of the motif deletion mutant lines (bZIP19 del1 del2). Plants (aged 6 weeks) of WT (Col), bzip19/23 (-), bzip19/23-bZIP19, bzip19/23-bZIP19 del1 del2 and bzip19/23-pZIP4::GUS-bZIP19 del1 del2 lines grown in hydroponics with control nutrient solution. a, Fe b, Cu c, Mn and d, P concentration, and e, dry weight (DW), of shoot tissue. # represents independently transformed T3 homozygous lines. Bars represent element concentration or DW of shoots as mean ± s.e.m. (n = 4 or n = 5). Different letters indicate significant differences (p < 0.05), determined using one-way analysis of variance followed by Tukey post-hoc test.
Extended Data Fig. 3 Seed ionome analysis of the motif deletion mutant lines (bZIP19 del1 del2).
Seed ionome analysis of the motif deletion mutant lines (bZIP19 del1 del2). Seeds from soil-grown plants of WT (Col), bzip19/23 (-), bzip19/23-bZIP19, bzip19/23-bZIP19 del1 del2 and bzip19/23-pZIP4::GUS-bZIP19 del1 del2 lines were analyzed. a, Fe b, Cu c, Mn and d, P concentration in seeds. # represents independently transformed T3 homozygous lines. Bars represent element concentration in seeds as mean ± s.e.m. (n = 4–7). Different letters indicate significant differences (p < 0.05), determined using one-way analysis of variance followed by Tukey post-hoc test.
Extended Data Fig. 4 Histochemical GUS staining analysis under different micronutrient deficiencies.
Histochemical GUS staining analysis under different micronutrient deficiencies. a, Phenotypic analysis and b, histochemical GUS staining of seedlings (aged 12 d) of pZIP4::GUS and bzip19/23-pZIP4::GUS lines grown on ½MS medium with control or under different micronutrient deficiencies; that is, -Zn, -Fe, -Cu and -Mn. Four to six plates (a), and 3–5 seedlings (b) per treatment and genotype were analyzed.
Extended Data Fig. 5 Deletion mutant lines grown with Zn deficient (-Zn) or sufficient (control) media.
Deletion mutant lines grown with Zn-deficient (-Zn) or -sufficient (control) media. a, Seedlings (aged 14 d) of Arabidopsis WT (Col), bzip19/23 double mutant (-), bzip19/23-bZIP19 and bzip19/23-bZIP19 del1 del2 lines, grown with control or -Zn ½MS medium. b, Plants (aged 6 weeks) of WT (Col), bzip19/23 (-), bzip19/23-bZIP19, bzip19/23-bZIP19 del1 del2 and bzip19/23-pZIP4::GUS-bZIP19 del1 del2 lines grown in hydroponics with control nutrient solution. # represents independently transformed T3 homozygous lines.
Extended Data Fig. 6 Protein gel electrophoresis of purified bZIP19 and bZIP19 del1 del2 proteins.
Protein gel electrophoresis (SDS-PAGE) of purified MBP-bZIP19 and MBP-bZIP19 del1 del2 proteins stained with Coomassie Blue. Forty µg of each purified protein was loaded. The purified proteins show the expected molecular weight (arrow) ca. 68 KDa (28 KDa from bZIP19 + 40 KDa from MBP). The analysis was performed twice with two independent protein extractions. Molecular weight markers (MW) are displayed.
Supplementary information
Supplementary Information
Supplementary Table 1.
Source data
Source Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed gel.
Rights and permissions
About this article
Cite this article
Lilay, G.H., Persson, D.P., Castro, P.H. et al. Arabidopsis bZIP19 and bZIP23 act as zinc sensors to control plant zinc status. Nat. Plants 7, 137–143 (2021). https://doi.org/10.1038/s41477-021-00856-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-021-00856-7
This article is cited by
-
The LRR receptor-like kinase ALR1 is a plant aluminum ion sensor
Cell Research (2024)
-
Comprehensive in silico analysis of the underutilized crop tef (Eragrostis tef (Zucc.) Trotter) genome reveals drought tolerance signatures
BMC Plant Biology (2023)
-
The auxin signaling pathway contributes to phosphorus-mediated zinc homeostasis in maize
BMC Plant Biology (2023)
-
The Role of Membrane Transporters in the Biofortification of Zinc and Iron in Plants
Biological Trace Element Research (2023)
-
Foxtail millet (Setaria italica) as a model system to study and improve the nutrient transport in cereals
Plant Growth Regulation (2023)