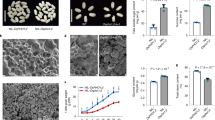
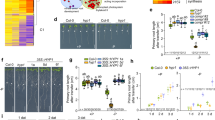
Abstract
Inorganic phosphate (Pi) is an essential component of all life forms. Land plants acquire Pi from the soil through roots and associated symbioses, and it is then transported throughout the plant. When sufficient, excess Pi is stored in vacuoles for remobilization following Pi deficiency. Although Pi release from the vacuoles to the cytoplasm serves as a critical mechanism for plants to adapt to low-Pi stress, the transporters responsible for vacuolar Pi efflux have not been identified. Here, we identified a pair of Oryza sativa vacuolar Pi efflux transporters (OsVPE1 and OsVPE2) that were more abundant in plants grown under Pi-deficient conditions. These OsVPE proteins can transport Pi into yeast cells and Xenopus laevis oocytes. Vacuolar Pi content was higher in the loss-of-function Osvpe1 Osvpe2 double mutant than in wild type, particularly under low-Pi stress. Overexpression of either OsVPE1 or OsVPE2 in transgenic plants reduced vacuolar Pi content, consistent with a role in vacuolar Pi efflux. We demonstrate that these VPE proteins evolved from an ancient plasma membrane glycerol-3-phosphate transporter protein. Together, these data indicate that this transporter was recruited to the vacuolar membrane to catalyse Pi efflux during the course of land plant evolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that supports the findings of this study are available within the article and its Supplementary Information files or from the corresponding authors upon reasonable request.
References
Balzergue, C. et al. Low phosphate activates STOP1–ALMT1 to rapidly inhibit root cell elongation. Nat. Commun. 8, 15300 (2017).
Muller, J. et al. Iron-dependent callose deposition adjusts root meristem maintenance to phosphate availability. Dev. Cell 33, 216–230 (2015).
Kanno, S. et al. A novel role for the root cap in phosphate uptake and homeostasis. eLife 5, e14577 (2016).
Raghothama, K. G. Phosphate acquisition. Annu. Rev. Plant Physiol. Plant Mol. Biol. 50, 665–693 (1999).
Guo, B. et al. Functional analysis of the Arabidopsis PHT4 family of intracellular phosphate transporters. New Phytol. 177, 889–898 (2008).
Versaw, W. K. & Harrison, M. J. A chloroplast phosphate transporter, PHT2;1, influences allocation of phosphate within the plant and phosphate-starvation responses. Plant Cell 14, 1751–1766 (2002).
Picault, N., Hodges, M., Palmieri, L. & Palmieri, F. The growing family of mitochondrial carriers in Arabidopsis. Trends Plant Sci. 9, 138–146 (2004).
Foyer, C. & Spencer, C. The relationship between phosphate status and photosynthesis in leaves: effects on intracellular orthophosphate distribution, photosynthesis and assimilate partitioning. Planta 167, 369–375 (1986).
Liu, J. et al. A vacuolar phosphate transporter essential for phosphate homeostasis in Arabidopsis. Proc. Natl Acad. Sci. USA 112, E6571–E6578 (2015).
Liu, T. Y. et al. Identification of plant vacuolar transporters mediating phosphate storage. Nat. Commun. 7, 11095 (2016).
Wang, C. et al. Rice SPX-major facility superfamily 3, a vacuolar phosphate efflux transporter, is involved in maintaining phosphate homeostasis in rice. Plant Physiol. 169, 2822–2831 (2015).
Elvin, C. M., Hardy, C. M. & Rosenberg, H. Pi exchange mediated by the GlpT-dependent sn-glycerol-3-phosphate transport system in Escherichia coli. J. Bacteriol. 161, 1054–1058 (1985).
Huang, Y., Lemieux, M. J., Song, J., Auer, M. & Wang, D. N. Structure and mechanism of the glycerol-3-phosphate transporter from Escherichia coli. Science 301, 616–620 (2003).
Ramaiah, M., Jain, A., Baldwin, J. C., Karthikeyan, A. S. & Raghothama, K. G. Characterization of the phosphate starvation-induced glycerol-3-phosphate permease gene family in Arabidopsis. Plant Physiol. 157, 279–291 (2011).
Kawai, H. et al. Arabidopsis glycerol-3-phosphate permease 4 is localized in the plastids and involved in the accumulation of seed oil. Plant Biotechnol. 31, 159–165 (2014).
Shen, J. et al. Organelle pH in the Arabidopsis endomembrane system. Mol. Plant 6, 1419–1437 (2013).
Guo, M. et al. Integrative comparison of the role of the PHOSPHATE RESPONSE1 subfamily in phosphate signaling and homeostasis in rice. Plant Physiol. 168, 1762–1776 (2015).
Popova, Y., Thayumanavan, P., Lonati, E., Agrochao, M. & Thevelein, J. M. Transport and signaling through the phosphate-binding site of the yeast Pho84 phosphate transceptor. Proc. Natl Acad. Sci. USA 107, 2890–2895 (2010).
Law, C. J., Enkavi, G., Wang, D. N. & Tajkhorshid, E. Structural basis of substrate selectivity in the glycerol-3-phosphate: phosphate antiporter GlpT. Biophys. J. 97, 1346–1353 (2009).
Ma, X. et al. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol. Plant 8, 1274–1284 (2015).
Hou, X. L. et al. Regulation of the expression of OsIPS1 and OsIPS2 in rice via systemic and local Pi signalling and hormones. Plant Cell Environ. 28, 353–364 (2005).
Ai, P. et al. Two rice phosphate transporters, OsPht1;2 and OsPht1;6, have different functions and kinetic properties in uptake and translocation. Plant J. 57, 798–809 (2008).
Wang, C. et al. Involvement of OsSPX1 in phosphate homeostasis in rice. Plant J. 57, 895–904 (2009).
Bustos, R. et al. A central regulatory system largely controls transcriptional activation and repression responses to phosphate starvation in Arabidopsis. PLoS Genet. 6, e1001102 (2010).
Wang, X. et al. Trans-Golgi network-located AP1 gamma adaptins mediate dileucine motif-directed vacuolar targeting in Arabidopsis. Plant Cell 26, 4102–4118 (2014).
Rausch, C. & Bucher, M. Molecular mechanisms of phosphate transport in plants. Planta 216, 23–37 (2002).
Tohge, T. et al. Toward the storage metabolome: profiling the barley vacuole. Plant Physiol. 157, 1469–1482 (2011).
Pant, B.-D. et al. Identification of primary and secondary metabolites with phosphorus status-dependent abundance in Arabidopsis, and of the transcription factor PHR1 as a major regulator of metabolic changes during phosphorus limitation. Plant Cell Environ. 38, 172–187 (2015).
Moradi, M., Enkavi, G. & Tajkhorshid, E. Atomic-level characterization of transport cycle thermodynamics in the glycerol-3-phosphate:phosphate antiporter. Nat. Commun. 6, 8393 (2015).
Wang, C. et al. Functional characterization of the rice SPX-MFS family reveals a key role of OsSPX-MFS1 in controlling phosphate homeostasis in leaves. New Phytol. 196, 139–148 (2012).
Deng, M. et al. OsCYCP1;1, a PHO80 homologous protein, negatively regulates phosphate starvation signaling in the roots of rice (Oryza sativa L.). Plant Mol. Biol. 86, 655–669 (2014).
Li, Y. et al. Chalk5 encodes a vacuolar H+-translocating pyrophosphatase influencing grain chalkiness in rice. Nat. Genet. 46, 398–404 (2014).
Wu, F. H. et al. Tape-Arabidopsis Sandwich—a simpler Arabidopsis protoplast isolation method. Plant Methods 5, 16 (2009).
Zhang, Y. et al. A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7, 30 (2011).
Nelson, B. K., Cai, X. & Nebenfuhr, A. A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J. 51, 1126–1136 (2007).
Nour-Eldin, H. H., Hansen, B. G., Norholm, M. H., Jensen, J. K. & Halkier, B. A. Advancing uracil-excision based cloning towards an ideal technique for cloning PCR fragments. Nucleic Acids Res. 34, e122 (2006).
De Angeli, A., Zhang, J., Meyer, S. & Martinoia, E. AtALMT9 is a malate-activated vacuolar chloride channel required for stomatal opening in Arabidopsis. Nat. Commun. 4, 1804–1804 (2013).
Acknowledgements
The authors thank B. Menand for valuable suggestions, J. M. Thevelein for the yeast strains, and L. Jiang and G. Miesenböck for the PEpHluorin and PRpHluorin. This work was supported by the National Key Research and Development Program of China (2017YFD0200204) and the National Natural Science Foundation (31772386, 31801924 and 31670267). K.Y. was supported by the National Program for the Support of Top-notch Young Professionals and the Innovation Program of Chinese Academy of Agricultural Sciences. L.D. is funded by a European Research Council Advanced Grant (EVO-500; contract number 25028). S.L. is funded by a grant from the National Science Foundation.
Author information
Authors and Affiliations
Contributions
K.Y. conceived and supervised the project. L.X., H.Z. and K.Y. designed the research. L.X., H.Z., R.W., Y.L., Z.X., W.T., W.R., F.W., M.D. and J.W. performed the experiments. L.X., H.Z., R.W., L.D., S.L., S.X. and K.Y. analysed the data. L.X., H.Z. and K.Y. wrote the paper with contributions from all the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–13 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Xu, L., Zhao, H., Wan, R. et al. Identification of vacuolar phosphate efflux transporters in land plants. Nature Plants 5, 84–94 (2019). https://doi.org/10.1038/s41477-018-0334-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0334-3
This article is cited by
-
Agrobacterium rhizogenes-mediated marker-free transformation and gene editing system revealed that AeCBL3 mediates the formation of calcium oxalate crystal in kiwifruit
Molecular Horticulture (2024)
-
Modulation of plant immunity and biotic interactions under phosphate deficiency
Journal of Plant Research (2024)
-
Telomere-to-telomere genome of the allotetraploid legume Sesbania cannabina reveals transposon-driven subgenome divergence and mechanisms of alkaline stress tolerance
Science China Life Sciences (2024)
-
Multi-omics strategies uncover the molecular mechanisms of nitrogen, phosphorus and potassium deficiency responses in Brassica napus
Cellular & Molecular Biology Letters (2023)
-
Calcium signaling-mediated transcriptional reprogramming during abiotic stress response in plants
Theoretical and Applied Genetics (2023)