Abstract
The genus Liriodendron belongs to the family Magnoliaceae, which resides within the magnoliids, an early diverging lineage of the Mesangiospermae. However, the phylogenetic relationship of magnoliids with eudicots and monocots has not been conclusively resolved and thus remains to be determined1,2,3,4,5,6. Liriodendron is a relict lineage from the Tertiary with two distinct species—one East Asian (L. chinense (Hemsley) Sargent) and one eastern North American (L. tulipifera Linn)—identified as a vicariad species pair. However, the genetic divergence and evolutionary trajectories of these species remain to be elucidated at the whole-genome level7. Here, we report the first de novo genome assembly of a plant in the Magnoliaceae, L. chinense. Phylogenetic analyses suggest that magnoliids are sister to the clade consisting of eudicots and monocots, with rapid diversification occurring in the common ancestor of these three lineages. Analyses of population genetic structure indicate that L. chinense has diverged into two lineages—the eastern and western groups—in China. While L. tulipifera in North America is genetically positioned between the two L. chinense groups, it is closer to the eastern group. This result is consistent with phenotypic observations that suggest that the eastern and western groups of China may have diverged long ago, possibly before the intercontinental differentiation between L. chinense and L. tulipifera. Genetic diversity analyses show that L. chinense has tenfold higher genetic diversity than L. tulipifera, suggesting that the complicated regions comprising east–west-orientated mountains and the Yangtze river basin (especially near 30° N latitude) in East Asia offered more successful refugia than the south–north-orientated mountain valleys in eastern North America during the Quaternary glacial period.
Similar content being viewed by others
Main
The Magnoliaceae, a family in the order Magnoliales, is an early diverging lineage of the Mesangiospermae (core angiosperms)8, and thus, it possesses a crucial phylogenetic position for better understanding the evolution of the extant flowering plants. However, the relationships among magnoliids, eudicots, and monocots have not been conclusively resolved despite previous valuable attempts2,5,6. The Liriodendron genus, which belongs to the subfamily Liriodendroideae of the Magnoliaceae, consisted of several species distributed throughout the Northern Hemisphere until the Late Tertiary, but now comprises only of a pair of sister species with a classic intercontinental disjunction distribution: one in East Asia (L. chinense) and the other in eastern North America (L. tulipifera). These two Tertiary relict Liriodendron species have been suggested to have diverged during the middle to late Miocene7,9, a reflection of range restrictions resulting from extinctions in the late Cenozoic10. Moreover, this pair of species is a perfect verification of the second prediction of the geographic speciation theory, which was proposed to explain the origin of species11,12.
Here, we combined three different sequencing technologies (that is, short-read sequencing, long-read sequencing and optical mapping) to de novo assemble the L. chinense genome. First, we achieved ~327.11 gigabases (Gb) of clean Illumina paired-end reads (Supplementary Table 1), ~147.89 Gb of corrected PacBio long reads (length longer than 2 kilobases (kb); Supplementary Table 2) and ~315.41 Gb of Bionano genome map data (Supplementary Table 3). We estimated the genome size to be 1.75 Gb based on Illumina data (Supplementary Fig. 1 and Supplementary Table 4), which was consistent with the estimation of ~1.8 Gb using flow cytometry (Supplementary Note). Then, we assembled the genome of Liriodendron into 4,624 contigs with an N50 length of 1.43 megabases (Mb) using Falcon (Supplementary Table 5). Furthermore, this assembly of long reads was integrated with a Bionano optical map to create a hybrid assembly consisting of 3,711 scaffolds totalling 1.74 Gb with an N50 length of 3.53 Mb (Supplementary Table 5). Finally, we anchored 529 scaffolds totalling ~1.37 Gb to a genetic map with 19 linkage groups, using a total of 1,576 microsatellite markers (Supplementary Fig. 2 and Supplementary Table 6). A high-confidence set of 35,269 gene models was constructed using the genome annotation pipeline MAKER (Supplementary Fig. 3), with 83.59% of genes being assigned putative functional annotations (Supplementary Table 7). To assess the quality of the assembly, we compared ten bacterial artificial chromosomes (BACs), in which potential repeat regions were masked (Supplementary Note), with assembled scaffolds, resulting in an average coverage of 99.75% (Supplementary Fig. 4). Of all 66,934 unigenes (>200 base pairs (bp)) assembled de novo by RNA sequencing (RNA-Seq), more than 90% had a length coverage of greater than 90% within a single scaffold (Supplementary Table 8). In addition, 1,300 (90.28%) genes of the BUSCO plant set were covered by the Liriodendron genome (Supplementary Table 9).
The genome size of L. chinense is larger than those of most sequenced angiosperms (Supplementary Fig. 5). We further investigated two pertinent aspects of genome evolution—whole-genome duplication (WGD) events and transposable element bursts—both of which have had profound effects on plant genome evolution13. The fraction of synonymous substitutions per synonymous site (Ks) distributions of paralogues in the Liriodendron genome and transcriptome clearly illustrate the occurrence of a single WGD event experienced by Liriodendron (Fig. 1a,b). It has been firmly established that whole-genome triplication (mechanistically originating as two successive WGDs) occurred in the grape14, and there is no evidence for lineage-specific polyploidy events in Amborella15. By performing a comparative genomic analysis of Vitis with Amborella and Liriodendron, we identified 3:1 and 3:2 syntenic depth ratios in the Vitis–Amborella (Supplementary Fig. 6) and Vitis–Liriodendron (Fig. 1c and Supplementary Fig. 7) comparisons, respectively. Furthermore, we mapped the complete repertoire of 1–2–3 orthologous regions in the Amborella–Liriodendron–Vitis genome comparison (Fig. 1d,e). Thus, from these data, we conclude that a single Liriodendron lineage-specific WGD event occurred, consistent with the results of the fourfold synonymous third-codon transversion position analysis (Supplementary Fig. 8). We speculated that the Liriodendron WGD event occurred approximately 116 million years ago (Ma) with a synonymous substitution rate of 3.02 × 10−9 synonymous substitutions yr−1 (ref. 16). Considering the possibly overestimated synonymous substitution rate16 and the divergence time of 113–128 Ma between the families Magnoliaceae and Lauraceae17, the WGD detected in the Liriodendron genome probably predated the separation of these two families.
a, Ks distributions for the whole paranome identified from the whole genome of Liriodendron (green), grape (blue) and Amborella (orange). WGT, whole-genome triplication. b, Ks distribution for the whole paranome identified from the whole transcriptome of L. chinense. c, Comparison of Liriodendron and grape genomes. Dot plots of orthologues show a 2–3 chromosomal relationship between the Liriodendron genome and grape genome. d, Macrosynteny patterns show that a typical ancestral region in the basal angiosperm Amborella can be tracked to up to two regions in Liriodendron and to up to three regions in the grape. Grey wedges in the background highlight major syntenic blocks spanning more than 30 genes between the genomes (highlighted by one syntenic set shown in colour). e, Microcollinearity patterns between genomic regions from Amborella, Liriodendron and the grape. Rectangles represent predicted gene models, with purple and brown showing relative gene orientations. Grey wedges connect matching gene pairs, with two sets highlighted in red.
Transposable elements account for 61.64% of the Liriodendron genome (Supplementary Tables 10 and 11). Long terminal repeat (LTR) retrotransposons are the most abundant transposable element, representing 56.25% of the assembly (Supplementary Table 11). Among the LTR retrotransposons, Gypsy elements are much more abundant than Copia elements (Supplementary Table 12 and Supplementary Fig. 9). In addition, transposable elements are unevenly distributed across the Liriodendron genome and tend to accumulate in intergenic regions rather than genic regions and regions adjoining genes (Supplementary Fig. 10), probably as a result of natural selection due to the potential detrimental effects of transposable elements on gene expression18. With respect to the genic regions, transposable elements have an unequal distribution between exons and introns, and there is an obvious bias towards transposable element accumulation in introns compared with exons (Supplementary Fig. 11), consistent with the natural selection hypothesis, although introns may play an important role in gene expression19. Furthermore, long interspersed nuclear element-1 has an abnormally high rate of accumulation in genic regions, in contrast with the pattern shown by other transposable elements (Supplementary Fig. 12 and Supplementary Table 13). Moreover, we analysed the divergence time distribution for all LTRs in the Liriodendron genome and found a Ks peak at 0.05 (Supplementary Fig. 13). We assumed an intergenic nucleotide substitution rate of 1.51 × 10−9 that was roughly twice as low as that within the genic regions (Supplementary Note), resulting in an insertion time of ~16 Ma. Overall, these results show that an ancient WGD event that occurred approximately 116 Ma, followed by a more recent burst of transposable element insertion that occurred approximately 16 Ma, have both contributed to the expansion of the Liriodendron genome.
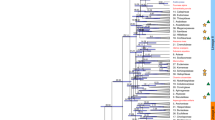
Some features of the Liriodendron phenotype are typical of both monocots and eudicots (Fig. 2a), which is consistent with the obscure phylogenetic relationships among magnoliids, monocots and eudicots. To investigate which of the three previously proposed tree topologies is most likely to be true (that is: (1) ((monocots, (eudicots, magnoliids)), basal angiosperm); (2) ((eudicots, (monocots, magnoliids)), basal angiosperm); or (3) ((magnoliids, (monocots, eudicots)), basal angiosperm) (Supplementary Table 14)), we selected an additional six eudicots, six monocots, three magnoliids and one basal angiosperm, with one gymnosperm being the outgroup (Supplementary Fig. 14), to construct individual orthogroups. In this way, we could use as many gene families as possible to depict a broad picture of the phylogeny. After careful evaluation and selection (Supplementary Note), we finally obtained 502 low-copy orthogroups, with 172 orthogroups (34.26%) supporting topology I, 155 orthogroups (30.88%) supporting topology II and the final 175 orthogroups (34.86%) supporting topology III (Fig. 2b), with no statistically significant difference among the three topologies (χ2 = 1.3904; P = 0.4990). Based on these 502 low-copy orthogroups, quantification of differences in gene-wise log-likelihood scores (ΔGLS) among these three alternative topologies20 showed an equal distribution of phylogenetic signals for each topology at the gene level (Supplementary Fig. 16). Further excluding orthogroups whose ΔGLS values were outliers (Supplementary Note), we obtained 481 low-copy orthogroups, with a lack of statistical significance among the orthogroups supporting each of the three alternative topologies (Fig. 2b; χ2 = 0.2162; P = 0.8975). These results explain why all three possible topologies have been observed in previous studies using different datasets (Supplementary Table 14) and suggest that rapid diversification occurred in the common ancestor of magnoliids, eudicots and monocots, which might be responsible for the phylogenetic incongruence in previous studies.
a, Liriodendron shows typical features of monocots in its reproductive organs (flower parts in multiples of three and monosulcate pollen grains) and of eudicots in its vegetative organs (two cotyledons, a taproot system, a eudicot-like stem cross-section and netted venation). These experiments were repeated independently at least ten times with similar results. Scale bar, 200 µm. b, Three topologies that coincided with three alternative phylogenetic hypotheses are plotted, and the results of a chi-squared test of the orthogroup numbers supporting each topology are shown below, revealing no statistically significant difference in topology preference. c, The eudicot- and monocot-specific gene families present in Liriodendron are statistically similar to those present in Amborella, whereas Spirodela polyrhiza has a bias towards monocot-specific gene families, and Macleaya cordata has a bias towards eudicot-specific gene families when compared with Amborella. d, Dated phylogeny for 11 plant species with Picea abies as an outgroup. A time scale is shown at the bottom, and red points in some nodes indicate fossil calibration points.
To further confirm the Liriodendron phylogeny, a coalescent-based species tree was constructed using the 502-orthogroup dataset, and this tree supported topology III with low bootstrap support (Supplementary Fig. 17a). Additionally, we performed coalescent-based species tree construction based on the 481-orthogroup dataset, yielding a topology identical to topology III with a bootstrap value increasing from 50 to 54% (Supplementary Fig. 17b). Furthermore, we performed a phylogenetic analysis on the basis of a concatenated sequence alignment of 78 chloroplast genes, yielding a topology consistent with topology III with strong bootstrap support (Supplementary Fig. 18). To continue our investigation, we identified both eudicot- and monocot-specific gene families present in the Liriodendron genome based on the PLAZA 3.0 Monocots database (Supplementary Fig. 19). The gene families from either clade were not significantly over-represented in Liriodendron compared with Amborella (χ2 = 0.1166; P = 0.7328), whereas a monocot plant and a eudicot plant both showed significant biases towards their respective gene families (Fig. 2c). Overall, considering our results, including the mosaic phenotypic characterization, individual and multiple gene tree reconstructions, and lineage-specific gene family identification, we suggest a topology in which eudicots and monocots form a clade that is sister to magnoliids, represented by Liriodendron, with the basal angiosperm Amborella being the next group (Fig. 2d); that is, magnoliids arose before the divergence of eudicots and monocots. Thus, the phylogenetic analysis incorporating the Liriodendron genome provides additional insights into the systematic position and evolution of magnoliids.
At present, the Liriodendron genus contains only two species in regions with a humid subtropical climate, and has partially expanded to the southern margin of the warm temperate climate zone of the Northern Hemisphere21,22 (Fig. 3a and Supplementary Fig. 20). However, a number of extinct Liriodendron species were once widely distributed in relatively high-latitude regions of the Northern Hemisphere before a general cooling of the climate occurred during in the Late Tertiary23, based on fossil records of seeds and leaves (Fig. 3a and Supplementary Fig. 21). To explore the historical demographic fluctuations and present-day genetic diversity within these two Liriodendron species, we resequenced 20 Liriodendron accessions, including 14 L. chinense individuals and six L. tulipifera individuals (Fig. 3a, Supplementary Fig. 22 and Supplementary Table 15).
a, Geographic distribution of Liriodendron accessions. Brown triangles represent the fossil distribution of Liriodendron plants in high-latitude regions of the Northern Hemisphere. Fringe patterns show two principal refugia where Tertiary relict floras occurred: southern East Asia and eastern North America. The natural distributions of L. chinense and L. tulipifera are plotted, with coloured dots representing individual Liriodendron accessions. b, Neighbour-joining tree of all accessions constructed from whole-genome SNPs. Accessions coming from the same geographic areas are grouped together and coloured corresponding to the colours used in a. LY, Liu Yang; SZ, Sang Zhi; EX, E Xi; ST, Song Tao; LP, Li Ping; ML, Meng La; XY, Xu Yong; SN, Sui Ning; DBS, DaBie Shan; HS, Huang Shan; LS1, Lushan_1; SY, Song Yang; LS2, Lushan_2; WYS, WuYi Shan; ON, Ontario; LA, Louisiana; GA, Georgia; TN, Tennessee; NC, North Carolina; MO, Missouri. c, Principal component analysis plots of the first two components for all 20 accessions, with dots coloured corresponding to their provenances. d, Nucleotide diversity (π) and population divergence (FST) across the three groups. The value in each circle represents a measure of nucleotide diversity for this group, and the value on each line indicates the population divergence between the two groups.
On the basis of phylogenetic analysis of a whole-genome single nucleotide polymorphism (SNP) analysis, we found that these Liriodendron accessions formed three distinct phylogenetic groups (Fig. 3b and Supplementary Fig. 23). This was further supported by a principal component analysis (Fig. 3c) and structure analysis (Supplementary Fig. 24). All L. chinense individuals from western China (CW) clustered together, and the rest of the L. chinense, collected from eastern China (CE), clustered into the second group. The third group comprised all L. tulipifera individuals collected from North America (NA). It is evident that the NA group is phylogenetically positioned between the two L. chinense groups and more closely related to the CE group, suggesting that the earliest divergence occurred between the populations in eastern China and those in western China, followed by differentiation between the eastern Chinese populations and North American populations. This pattern is supported by the phenotypic analysis, which shows that all three groups share one leaf morphological feature, while the CE and NA groups have their own unique leaf morphological feature (Supplementary Fig. 25). Fossil records indicate that similar leaf morphological features to those in the western and eastern China groups had already emerged in two extinct Liriodendron species24,25, again suggesting that these two L. chinense groups may have diverged a very long time ago, possibly preceding the intercontinental differentiation between L. chinense and L. tulipifera (Supplementary Fig. 25).
Nucleotide diversity (π) analysis shows that the CW group has the highest genetic diversity, followed by the CE group, and that the genetic diversity of the NA group is tenfold lower than that of the CW group (Fig. 3d). An analysis of demographic history using the pairwise sequentially Markovian coalescent (PSMC) model26 shows that the two groups from China both had population size peaks at approximately 0.4 Ma and declined afterwards, whereas the NA group population size peak occurred much earlier and continuously declined since approximately 2.3 Ma (Fig. 4), indicating that the populations in eastern China and those in western China underwent a similar demographic history different from that in North American populations. We also calculated genetic differentiation statistics (fixation index; FST) among the three Liriodendron groups, indicating that the genetic differentiation (FST = 0.2055) between the NA group and the CW group was slightly lower than that (FST = 0.2707) between the NA group and the CE group (Fig. 3d). In addition, we also found that the CW group had the highest level of individual differences compared with the other two geographical groups (Supplementary Fig. 26).
a–c, Plots of PSMC results for 20 individuals (7 from western China (a); 7 from eastern China (b); and 6 from North America (c)), as indicated in each legend. The grey lines represent the mass accumulation rate (MAR) of the Chinese Loess Plateau in a and b, and the atmospheric surface air temperature relative to the present in c.
The natural distribution areas of these two Liriodendron species on their respective continents are highly consistent with the two principal areas where Tertiary relict floras occur23 (Fig. 3a). Although Liriodendron species were once distributed over the high-latitude regions of Europe (Fig. 3a), the east–west-orientated mountains are thought to have blocked their southward migration during global cooling in the Late Tertiary and subsequent Quaternary glaciations27, finally leading to the extinction of Liriodendron in Europe7. With respect to the Liriodendron that survived in East Asia and eastern North America, the higher genetic diversity of L. chinense compared with L. tulipifera is consistent with the greater number of suitable refugia in East Asia28,29. In this study, we observed a sustained population decrease during the whole Quaternary glaciation in all L. tulipifera accessions and a population recovery approximately 0.3–0.4 Ma in all L. chinense accessions (Fig. 4), which may have contributed considerably to the severe loss of genetic diversity in L. tulipifera and the relatively high retention of genetic diversity in L. chinense (Fig. 3d and Supplementary Fig. 27), respectively. The population recovery observed in all L. chinense accessions occurred in the interglacial stage between the Guxiang Glaciation (0.3–0.13 Ma) and Naynayxungla Glaciation (0.72–0.5 Ma)30. Considering that the Naynayxungla Glaciation was the most extensive glaciation, including large ice caps and massive valley glaciers, and the following Guxiang Glaciation was characterized by valley glaciers only30, we speculate that the temperature recovery and deglaciation during this interglacial stage provided a foundation for L. chinense population recovery within East Asian refugia. Consequently, in addition to the higher habitat diversity within East Asian refugia29, a suitable living environment during the interglacial stage between the Naynayxungla and Guxiang glaciations may have contributed to the retention, restoration and augmentation of L. chinense genetic diversity.
Methods
Plant materials and sequencing
For genome sequencing, we collected fresh leaves from an adult plant of L. chinense grown in Lushan, which is located in the Jiangxi province of China. For Illumina sequencing, four series of paired-end sequencing libraries with insert sizes of 170, 250, 500 and 800 bp were constructed and subsequently sequenced on the Illumina HiSeq 2000 platform, ultimately resulting in 327.11 Gb clean reads. For PacBio single-molecule real-time sequencing, sequencing libraries with 20-kb DNA inserts were constructed and subsequently sequenced on the Pacific Biosciences RSII instrument, ultimately resulting in a total of 150.18 Gb subread with an N50 length of 15.96 kb for the genome assembly. In addition, purified DNA was labelled at Nt.BspQI sites using the IrysPrep kit, and a 315.41 Gb optical map of the sample was produced from the BioNano Irys system. In addition, abundances of 17-nucleotide k-mers from 170- and 250-bp Illumina sequencing libraries were used to estimate the genome size.
De novo assembly
The Liriodendron genome was de novo assembled using FALCON (https://github.com/PacificBiosciences/FALCON) based on PacBio long reads (only reads longer than 10 kb were used in the assembly). Errors in the PacBio reads were corrected within the FALCON pipeline. Contigs was first polished based on raw PacBio data and finally corrected using Illumina short reads with Pilon31. A hybrid assembly was created based on contigs and optical maps using the Bionano Solve Pipeline (https://bionanogenomics.com/support-page/bionano-access/). Then, the corrected PacBio long reads were used for superscaffold gap filling using PBJelly32. We constructed a reference genetic map of L. chinense based on an F1 population of 150 plants from a cross between L. chinense and L. tulipifera using JoinMap 4.0 (ref. 33). Markers with inconsistent placement were manually screened and the collinearity of common markers was inspected using MapChart 2.2 (ref. 33). Markers in common were used as anchor points. Possible chimeric scaffolds were identified as those containing sequences of markers mapped to different locations in the same linkage group or different linkage groups, and these scaffolds were manually inspected. This process generated 19 Liriodendron pseudomolecules.
Genome assessment
We assessed the coverage of the genome assembly by mapping 89 BACs back to assembly with 97% of these BAC sequences covered without any obvious misassemblies. A comparison of 9 randomly chosen BACs sequenced by 454 sequencing technology indicated a low error rate. In addition, we used the BUSCO34 database to assess the genome assembly. We also validated the assembled genome using 66,934 unigenes (length ≥ 200 bp) from RNA-Seq.
Repeat annotation
We identified tandem repeats and transposable elements separately. Tandem repeats were predicted using Tandem Repeats Finder 4.04 (ref. 35). For transposable element identification, we performed a combination of similarity-based and de novo approaches. First, we used RepeatMasker with the Repbase 16.10 (ref. 36) database of known repeat sequences to search for transposable elements in the genome, and we additionally used RepeatProteinMask, implemented in RepeatMasker, to identify transposable elements by aligning the genome sequence to the transposable element protein database. Then, to apply our de novo approach, we constructed a repeat library generated by RepeatModeler37 with default parameters and ran RepeatMasker on the genome sequences, using the RepeatModeler consensus sequence as a library. Finally, all the repeat sequences identified by the different methods were combined into the final repeat annotation.
Gene prediction
Gene model prediction was conducted by the MAKER pipeline38, integrating ab initio prediction with de novo assembled transcripts from short-read messenger RNA sequencing, isoform-sequencing full-length transcripts, and protein homology data. A high-confidence gene model was constructed by further removing transposons and low-confidence predictions. Gene functional annotation was performed using the Swiss-Prot and TrEMBL databases39, while motifs and domains were annotated using InterProScan40 by searching against publicly available protein databases. Descriptions of gene products (that is, Gene Ontology terms) were retrieved from the corresponding InterPro entries. We also mapped the Liriodendron reference genes to KEGG41 pathway maps.
Transfer RNA genes were predicted based on tRNAscan-SE42. Ribosomal RNA fragments were identified by aligning plant ribosomal RNA sequences43 to the Liriodendron genome by BLASTN44. micro RNA and small nuclear RNA genes were detected by INFERNAL45 software against the Rfam database46 (release 9.1).
Genome synteny
We performed synteny searches to compare the L. chinense genome structure with that of the grape and Amborella genomes using MCscan47, requiring at least five gene pairs per syntenic block. The resulting dot plots were inspected to confirm the paleoploidy level of L. chinense in relation to the other genomes by counting the syntenic depth in each genomic region.
Ks values for homologous gene pairs were calculated as described in Maere et al.48. Fourfold synonymous third-codon transversion position values were calculated for syntenic segments from the concatenated alignments and constructed by dividing the number of transversions at all fourfold degenerate third-codon positions by the number of fourfold degenerate third-codon positions.
Phylogenetic analysis
Orthogroups were constructed with 14 other sequenced plants—6 eudicots (Arabidopsis thaliana, Populus trichocarpa, Vitis vinifera, Coffea canephora, Ipomoea nil and Fraxinus excelsior); 6 monocots (Brachypodium distachyon, Xerophyta viscosa, Asparagus officinalis, Musa acuminata, Ananas comosus and Oryza sativa); 1 basal angiosperm (Amborella trichopoda); and 1 gymnosperm (Gnetum montanum)—and three other magnoliid transcriptome datasets, including two sequenced in this study (Magnolia grandiflora and Michelia alba) and one available in Ibarra-Laclette et al.49 (Persea americana), using the software OrthoFinder50. We selected low-copy orthogroups with the number of putative orthologues less than two in each species, and putative orthologues were found in at least four eudicots, four monocots, three magnoliids, one basal angiosperm and one gymnosperm, resulting in 1,163 orthogroups. Then, each orthogroup was aligned using Clustal Omega51, and all alignments were further trimmed using TrimAl 1.2 (ref. 52). Next, we constructed 1,163 single-gene trees using RAxML53 with the PROTCATWAG mode. Then, we compared these single-gene trees with the species tree and screened them as described in Zeng et al.6. Finally, after careful examination, a total of 502 low-copy orthogroups were selected for further analysis.
We also calculated the phylogenetic signal based on three alternative topological hypotheses and quantified the difference in gene-wise log-likelihood scores (ΔGLS) among each of the three topologies using RAxML20,53. To diminish the influence of tiny amounts of data on phylogenetic inference, we further excluded orthogroups with outlier ΔGLS values, defined as described in Shen et al.20. To estimate the species tree, we performed a coalescent-based approach using Astral 5.6.1 (ref. 54). We also performed phylogenetic analyses based on 78 chloroplast genes among 24 land plant species using RAxML53.
To estimate divergence time, we used PAML MCMCTREE55 to perform Bayesian estimation with soft fossil constraints56 based on 235 single-copy orthologous genes that are shared by L. chinense and 10 other species. Markov chain Monte Carlo analysis was run to sample 1,000,000 times with a sampling frequency of 50 and a burn-in of 5,000,000 iterations. We also used CAFE57 to identify gene families that had undergone expansions or contractions across the maximum likelihood tree.
Resequencing and diversity analysis
DNA from 14 L. chinense and 6 L. tulipifera adult plants was extracted, and paired-end libraries with insert sizes of 100–150 bp were sequenced using Illumina technology at BGI. We first called SNPs using BWA58, GATK59 and SAMtools60, then annotated these SNPs using SNPEFF61, ultimately summarizing them by a customized Perl script.
The neighbour-joining phylogenetic tree was constructed using TreeBeST62 based on SNPs. Population structure and ancestry information was inferred using FRAPPE63 with the best K value determined by ADMIXTURE64 based on a cross-validation test. We additionally performed a principal component analysis using the STRATPCA programme from EIGENSOFT 3.2 (ref. 65).
Population genetic parameters, including nucleotide diversity (π)66 and the Watterson estimator (θw)67, were estimated on the basis of the genotypes of each line at the SNP positions using BioPerl.
The PSMC model, which was originally applied to human genomes26 and subsequently also applied to plant genomes15,68, was applied to study the effective population sizes (Ne) of the two Liriodendron species over time.
See the Supplementary Note for additional details.
Reporting Summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Code availability
The custom Perl script used for summarization of SNP annotation is available from the corresponding author upon request.
Data availability
The raw reads and genome assembly have been deposited as a BioProject under accession PRJNA418360. The resequencing data for 20 Liriodendron individuals have been deposited as a BioProject under accession PRJNA418361.
Change history
23 January 2019
In the Supplementary Information file originally published with this Letter, the authors mistakenly omitted Supplementary Table 14; this has now been amended.
References
Soltis, P. S. & Soltis, D. E. The origin and diversification of angiosperms. Am. J. Bot. 91, 1614–1626 (2004).
Qui, Y. L. et al. Phylogenetic analyses of basal angiosperms based on nine plastid, mitochondrial, and nuclear genes. Int. J. Plant Sci. 166, 815–842 (2005).
Moore, M. J. & Soltis, D. E. Phylogenetic analysis of the plastid inverted repeat for 244 species: insights into deeper-level angiosperm relationships from a long, slowly evolving sequence region. Int. J. Plant Sci. 172, 541–558 (2011).
Zhang, N., Zeng, L., Shan, H. & Ma, H. Highly conserved low-copy nuclear genes as effective markers for phylogenetic analyses in angiosperms. New Phytol. 195, 923–937 (2012).
Wickett, N. J. et al. Phylotranscriptomic analysis of the origin and early diversification of land plants. Proc. Natl Acad. Sci. USA 111, E4859–E4868 (2014).
Zeng, L. et al. Resolution of deep angiosperm phylogeny using conserved nuclear genes and estimates of early divergence times. Nat. Commun. 5, 4956 (2014).
Parks, C. R. & Wendel, J. F. Molecular divergence between Asian and North American species of Liriodendron (Magnoliaceae) with implications for interpretation of fossil floras. Am. J. Bot. 77, 1243–1256 (1990).
Azuma, H., Garcia-Franco, J. G., Rico-Gray, V. & Thien, L. B. Molecular phylogeny of the Magnoliaceae: the biogeography of tropical and temperate disjunctions. Am. J. Bot. 88, 2275–2285 (2001).
Nie, Z. L. et al. Phylogenetic and biogeographic complexity of Magnoliaceae in the Northern Hemisphere inferred from three nuclear data sets. Mol. Phylogenet. Evol. 48, 1027–1040 (2008).
Parks, C. R., Miller, N. G., Wendel, J. F. & McDougal, K. M. Genetic divergence within the genus Liriodendron (Magnoliaceae). Ann. MO Bot. Gard. 70, 658–666 (1983).
Darwin, C. R. On the Origin of Species by Means of Natural Selection or the Preservation of Favoured Races in the Struggle for Life (John Murray, London, 1859).
Coyne, J. A. Why Evolution Is True (Univ. Press, Oxford, 2009).
Wendel, J. F., Jackson, S. A., Meyers, B. C. & Wing, R. A. Evolution of plant genome architecture. Genome Biol. 17, 37 (2016).
Jaillon, O. et al. The grapevine genome sequence suggests ancestral hexaploidization in major angiosperm phyla. Nature 449, 463–467 (2007).
Amborella Genome, P. The Amborella genome and the evolution of flowering plants. Science 342, 1241089 (2013).
Cui, L. et al. Widespread genome duplications throughout the history of flowering plants. Genome Res. 16, 738–749 (2006).
Kumar, S., Stecher, G., Suleski, M. & Hedges, S. B. TimeTree: a resource for timelines, timetrees, and divergence times. Mol. Biol. Evol. 34, 1812–1819 (2017).
Hollister, J. D. & Gaut, B. S. Epigenetic silencing of transposable elements: a trade-off between reduced transposition and deleterious effects on neighboring gene expression. Genome Res. 19, 1419–1428 (2009).
Rose, A. B. & Beliakoff, J. A. Intron-mediated enhancement of gene expression independent of unique intron sequences and splicing. Plant Physiol. 122, 535–542 (2000).
Shen, X. X., Hittinger, C. T. & Rokas, A. Contentious relationships in phylogenomic studies can be driven by a handful of genes. Nat. Ecol. Evol. 1, 126 (2017).
Little, E. L. Atlas of United States Trees. Volume 1: Conifers and Important Hardwoods (US Department of Agriculture, 1971).
Hao, R. M. Geographical distribution of Liriodendron chinense in China and its significance. J. Plant Resour. Environ. (China) 4, 1–6 (1995).
Milne, R. I. Northern Hemisphere plant disjunctions: a window on tertiary land bridges and climate change? Ann. Bot. 98, 465–472 (2006).
Baghai, N. L. Liriodendron (Magnoliaceae) from the Miocene Clarkia flora of Idaho. Am. J. Bot. 75, 451–464 (1988).
Bell, W. A. Upper Cretaceous floras of the Dunvegan, Bad Heart, and Milk River Formations of Western Canada. Geol. Surv. Canada Bull. 94, 1–76 (1962).
Li, H. & Durbin, R. Inference of human population history from individual whole-genome sequences. Nature 475, 493–496 (2011).
Tiffney, B. H. The Eocene North Atlantic land bridge: its importance in Tertiary and modern phytogeography of the Northern Hemisphere. J. Arnold Arbor. 66, 243–273 (1985).
Wen, J. Evolution of eastern Asian and eastern North American disjunct distributions in flowering plants. Annu. Rev. Ecol. Syst. 30, 421–455 (1999).
Qian, H. & Ricklefs, R. E. Large-scale processes and the Asian bias in species diversity of temperate plants. Nature 407, 180–182 (2000).
Sun, Y. B. & An, Z. S. Late Pliocene–Pleistocene changes in mass accumulation rates of eolian deposits on the central Chinese Loess Plateau. J. Geophys. Res. Atmos. 110, D23101 (2005).
Walker, B. J. et al. Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE 9, e112963 (2014).
English, A. C. et al. Mind the gap: upgrading genomes with Pacific Biosciences RS long-read sequencing technology. PLoS ONE 7, e47768 (2012).
Van Ooijen, J. W. JoinMap 4: Software for the Calculation of Genetic Linkage Maps in Experimental Populations (Kyazma B.V., Wageningen, 2006).
Simao, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Benson, G. Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res. 27, 573–580 (1999).
Jurka, J. et al. Repbase Update, a database of eukaryotic repetitive elements. Cytogenet. Genome Res. 110, 462–467 (2005).
Price, A. L., Jones, N. C. & Pevzner, P. A. De novo identification of repeat families in large genomes. Bioinformatics 21, i351–i358 (2005).
Cantarel, B. L. et al. Maker: an easy-to-use annotation pipeline designed for emerging model organism genomes. Genome Res. 18, 188–196 (2008).
Bairoch, A. & Apweiler, R. The Swiss-Prot protein sequence database and its supplement trembl in 2000. Nucleic Acids Res. 28, 45–48 (2000).
Zdobnov, E. M. & Apweiler, R. InterProScan—an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17, 847–848 (2001).
Kanehisa, M. & Goto, S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Lowe, T. M. & Eddy, S. R. tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 25, 955–964 (1997).
Garcia, S., Galvez, F., Gras, A., Kovarik, A. & Garnatje, T. Plant rDNA database: update and new features. Database 2014, 1–7 (2014).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Nawrocki, E. P., Kolbe, D. L. & Eddy, S. R. Infernal 1.0: inference of RNA alignments. Bioinformatics 25, 1335–1337 (2009).
Griffithsjones, S. et al. Rfam: annotating non-coding RNAs in complete genomes. Nucleic Acids Res. 33, 121–124 (2005).
Wang, Y. et al. MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 40, e49 (2012).
Maere, S. et al. Modeling gene and genome duplications in eukaryotes. Proc. Natl Acad. Sci. USA 102, 5454–5459 (2005).
Ibarra-Laclette, E. et al. Deep sequencing of the Mexican avocado transcriptome, an ancient angiosperm with a high content of fatty acids. BMC Genom. 16, 599 (2015).
Emms, D. M. & Kelly, S. OrthoFinder: solving fundamental biases in whole genome comparisons dramatically improves orthogroup inference accuracy. Genome Biol. 16, 157 (2015).
Sievers, F. & Higgins, D. G. Clustal Omega for making accurate alignments of many protein sequences. Protein Sci. 27, 135–145 (2018).
Capella-Gutierrez, S., Silla-Martinez, J. M. & Gabaldon, T. TrimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 25, 1972–1973 (2009).
Stamatakis, A. RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22, 2688–2690 (2006).
Mirarab, S. et al. ASTRAL: genome-scale coalescent-based species tree estimation. Bioinformatics 30, i541–i548 (2014).
Yang, Z. PAML 4: phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 24, 1586–1591 (2007).
Yang, Z. & Rannala, B. Bayesian estimation of species divergence times under a molecular clock using multiple fossil calibrations with soft bounds. Mol. Biol. Evol. 23, 212–226 (2006).
De Bie, T., Cristianini, N., Demuth, J. P. & Hahn, M. W. CAFE: a computational tool for the study of gene family evolution. Bioinformatics 22, 1269–1271 (2006).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
DePristo, M. A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 6, 80–92 (2012).
Vilella, A. J. et al. EnsemblCompara GeneTrees: complete, duplication-aware phylogenetic trees in vertebrates. Genome Res. 19, 327–335 (2009).
Tang, H., Peng, J., Wang, P. & Risch, N. J. Estimation of individual admixture: analytical and study design considerations. Genet. Epidemiol. 28, 289–301 (2005).
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 19, 1655–1664 (2009).
Price, A. L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38, 904–909 (2006).
Nei, M. & Li, W. H. Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc. Natl Acad. Sci. USA 76, 5269–5273 (1979).
Watterson, G. A. On the number of segregating sites in genetical models without recombination. Theor. Popul. Biol. 7, 256–276 (1975).
Ibarra-Laclette, E. et al. Architecture and evolution of a minute plant genome. Nature 498, 94–98 (2013).
Acknowledgements
This work was supported by the National High Technology Research and Development Program of China (863 Program; number 2013AA102705), Key Research and Development Plan of Jiangsu Province (BE2017376), Foundation of Jiangsu Forestry Bureau (LYKJ[2017]42), Specialized National Basic Research Program of China (973 Program; 2012CB114504), National Natural Science Foundation of China (31770715), National High-level Personnel of Special Support Program, Qinglan Project of Jiangsu Province, Talent Project by the Ministry of Science and Technology and Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
J.S. and J.C. were the leading investigators of this research programme. J.S., J.C., S.Y. and N.L. designed the experiments and coordinated the project. Z.H., P.W., Y.S., Y.Z., Z.W., S.L. and T.C. performed field work and collected samples. X.G., C.Z., P.W, L.Y. and H.X. performed the sequencing experiments. X.G., C.Z., L.X, Qihui Z. and M.S. performed the genome assemblies. Z.H., X.G., C.Z. and L.G. constructed the genetic map. L.X, Q.Z., H.X., X.L. and Y.L. provided RNA-Seq data. X.G., C.Z., L.Y., Q.G. and N.L. performed the gene annotation and genome assembly assessment. J.C., Z.H., X.G., J.Z. and S.Y. performed the genome duplication analysis. Z.H., L.X., L.Y., Y.P. and S.Y. analysed the transposable element insertion. J.C., Z.H., X.G., N.L., S.Y. and J.S. performed the phylogenetic analysis. J.C., Z.H., X.G., T.L., S.Y. and J.S. performed the population genetic structure analysis. J.C., Z.H., S.G., S.Y. and J.S. wrote and edited most of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Note, Supplementary Tables 1–15 and Supplementary Figures 1–27.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, J., Hao, Z., Guang, X. et al. Liriodendron genome sheds light on angiosperm phylogeny and species–pair differentiation. Nature Plants 5, 18–25 (2019). https://doi.org/10.1038/s41477-018-0323-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0323-6
This article is cited by
-
Comprehensive deciphering the alternative splicing patterns involved in leaf morphogenesis of Liriodendron chinense
BMC Plant Biology (2024)
-
Characterization, evolution, and abiotic stress responses of leucine-rich repeat receptor-like protein kinases (LRR-RLK) in Liriodendron chinense
BMC Genomics (2024)
-
A high-quality haplotype genome of Michelia alba DC reveals differences in methylation patterns and flower characteristics
Molecular Horticulture (2024)
-
Genomic survey and expression analysis of LcARFs reveal multiple functions to somatic embryogenesis in Liriodendron
BMC Plant Biology (2024)
-
The chromosome-level genomes of the herbal magnoliids Warburgia ugandensis and Saururus chinensis
Scientific Data (2024)