Abstract
Pepper (Capsicum spp.) is one of the earliest cultivated crops and includes five domesticated species, C. annuum var. annuum, C. chinense, C. frutescens, C. baccatum var. pendulum and C. pubescens. Here, we report a pepper graph pan-genome and a genome variation map of 500 accessions from the five domesticated Capsicum species and close wild relatives. We identify highly differentiated genomic regions among the domesticated peppers that underlie their natural variations in flowering time, characteristic flavors, and unique resistances to biotic and abiotic stresses. Domestication sweeps detected in C. annuum var. annuum and C. baccatum var. pendulum are mostly different, and the common domestication traits, including fruit size, shape and pungency, are achieved mainly through the selection of distinct genomic regions between these two cultivated species. Introgressions from C. baccatum into C. chinense and C. frutescens are detected, including those providing genetic sources for various biotic and abiotic stress tolerances.
Similar content being viewed by others
Introduction
The genus Capsicum, a member of the nightshade family Solanaceae and commonly known as pepper or paprika, originated in South and Central America and includes around 35 species1. In 2021, the global cultivation of peppers exceeded 3.68 Mha, with a total production of 36.29 and 4.84 million tons of green and dry peppers, respectively (FAOSTAT; http://faostat.fao.org/). C. annuum var. annuum is the most commonly planted Capsicum species worldwide and contains both pungent and non-pungent (sweet) pepper fruits. Pungent pepper fruits are not only used as food and nutraceutical resources but also widely used in traditional medicines as pain relief and cancer inhibitors, reflecting the presence of a diverse array of bioactive compounds2.
Pepper has been domesticated in Central and/or South America since around 7000 BC, thus it represents one of the most ancient domesticated crops3. The five cultivated Capsicum species are C. annuum var. annuum domesticated from C. annuum var. glabriusculum, C. baccatum var. pendulum domesticated from C. baccatum var. baccatum, C. chinense, C. frutescens and C. pubescens. They belong to three distinct clades: C. annuum var. annuum, C. chinense and C. frutescens in the Annuum clade, C. baccatum var. pendulum in the Baccatum clade and C. pubescens in the Pubescens clade3. C. pubescens has flowers with purple corollas and dark brown or blackish seeds, while C. baccatum var. pendulum has flowers with white corollas and yellow or golden spots and beige- or tan-colored seeds, and C. baccatum var. baccatum has flowers with white to off-white corollas and corolla spots. Flowers of the cultivated C. frutescens and C. chinense have corollas that are typically off-white but more often are pale green4, while the corollas of C. annuum var. annuum are white (rarely violet). C. chinense is diversified in terms of fruit shape, size and color, while C. frutescens fruits are usually small and elongated. Fruits of C. chinense are typically quite pungent, with a strong fruit-like flavor5 and a distinct aroma. Fruits of C. frutescens are also pungent. Within the Annuum clade, there is a wide range of overlapping morphological characteristics6. For instance, C. frutescens shows some characteristics such as fruit shape, color and fruit position similar to those of C. chinense.
The domestication of peppers has been suggested to have occurred in multiple centers of origin7,8. However, previous large-scale genetic and genomic studies of pepper mainly focused on one single domesticated species, C. annuum var. annuum9,10,11. Therefore, knowledge of phylogenetic relationships, genetic diversity, population differentiation, convergent and divergent selections, and introgression among the various domesticated and wild Capsicum species remains limited. Recent rapid advances in sequencing technologies and abundant available germplasm resources of Capsicum have made it feasible to perform large-scale genome analyses of diverse Capsicum species to evaluate their genetic diversity and decipher genomic regions and genes associated with the evolution and domestication of peppers.
In this study, we first assemble high-quality genomes of three pepper accessions from the Annuum, Baccatum and Pubescens clades, using PacBio long reads and high-throughput chromosome conformation capture (Hi-C) maps, and construct a graph pan-genome from these three genome assemblies. We then perform genome resequencing of 500 accessions that cover all five domesticated species and the important wild progenitors and relatives and construct a single-base resolution variation map of Capsicum using the graph pan-genome as the reference. Our comprehensive population genomic analyses provide insights into population differentiation, convergent and divergent domestication, and introgression of the Capsicum species, and pinpoint candidate genes associated with important agronomic traits such as fruit shape and flavor, flowering time and biotic/abiotic stress responses. This study provides comprehensive and valuable genomic resources for facilitating future functional studies and molecular breeding of the Capsicum species.
Results
Reference genome assembly and graph pan-genome construction
We selected three lines, C. annuum var. annuum Zhangshugang, C. baccatum var. pendulum PI 632928, and C. pubescens Grif 1614, from the Annuum, Baccatum and Pubescens clades, respectively, for sequencing and assembly of pepper reference genomes. Using PacBio long reads, Illumina short reads and Hi-C data (Supplementary Fig. 1 and Supplementary Table 1), we generated chromosome-scale genome assemblies for Zhangshugang, PI 632928, and Grif 1614, with the contig N50 lengths of 35.42 Mb, 27.70 Mb and 46.20 Mb, respectively (Supplementary Fig. 2, Supplementary Data 1 and Supplementary Table 2). Evaluation of base accuracy and completeness of the genome assembly using Merqury12 and BUSCO13 indicated the high quality of these three genome assemblies (Supplementary Data 1 and Supplementary Table 3). The three assemblies showed an overall high collinearity, although some large inversions and genome rearrangements were observed between them (Supplementary Fig. 3).
The percentages of repetitive sequences were 78.14% in the Zhangshugang genome, 87.21% in the PI 632928 genome and 89.25% in the Grif 1614 genome (Supplementary Table 4). Combining evidence from ab initio predictions, transcript mapping and protein homology, a total of 33,688, 32,830 and 33,398 protein-coding genes were predicted in the Zhangshugang, PI 632928 and Grif 1614 genomes, respectively. Gene BUSCO analysis indicated that 94.0–97.7% of the core conserved plant genes were completely captured in the predicted genes of these three genomes, compared with only 77.0–86.7% in the predicted genes of previously published pepper genomes (Supplementary Data 1).
To improve read mapping and variant calling and to provide a more comprehensive reference to access the genetic diversity of the Capsicum species, we constructed a graph pan-genome from these three high-quality chromosome-scale genome assemblies. Using the Zhangshugang genome as the reference, small insertions/deletions (indels; 1–20 bp) and structural variants (SV; >20 bp) were identified in PI 632928 and Grif 1614 based on the whole-genome alignments. These identified variants in PI 632928 and Grif 1614 were integrated into the Zhangshugang genome, resulting in a graph pan-genome that contained a total of 1635.8 Mb sequences not present in the Zhangshugang genome. Of these sequences, 780.9 Mb and 853.5 Mb were uniquely from PI 632928 and Grif 1614, respectively, and 1.38 Mb were from both accessions (Supplementary Data 2).
Phylogeny and population structure of Capsicum
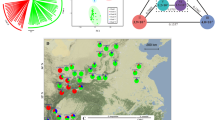
In this study, we first selected 1296 accessions representing nine Capsicum species collected from 94 countries or regions for shallow whole-genome sequencing (~1×) (Supplementary Data 3). Using SNPs called from the shallow whole-genome sequencing data, we constructed a core collection using GenoCore14, which comprised 500 accessions and captured ~97.5% of the total genetic diversity in the 1296 Capsicum accessions. Accessions in the core collection were mainly from Central and South America (Fig. 1a and Supplementary Data 4). These 500 accessions were further sequenced at an average depth of 14.7×, resulting in a total of 22.3 terabases of sequence data (Supplementary Data 4). By mapping the sequencing reads to the graph pan-genome, a total of 100,111,632 high-quality SNPs and 5,306,979 small indels were identified in the core collection. The SNPs included 430,414 that caused non-synonymous changes and 27,251 that resulted in start/stop codon gains or losses (Supplementary Table 5).
a Geographical distribution of the core pepper accessions (after taxonomy correction). The size of the pie corresponds to the total number of accessions from the selected geographical region. For C. galapagoense, only one accession with unknown geographic information was available and is not presented on the map. b Maximum likelihood phylogenetic tree and population structure of the core pepper accessions. c, d Principal component (PC) analysis, PC1 versus PC2 (c) and PC2 versus PC3 (d), of the core pepper accessions.
Species misidentifications and ambiguity among accessions are not uncommon in herbaria15 and Capsicum germplasm collections16. It is well known that C. annuum, C. chinense and C. frutescens show evidence of parallel evolution for various plant and fruit characteristics17. The overlap in morphological characteristics of these three species has led to the recognition of a C. annuum complex18. In most instances, these taxa are effectively identified using a botanical key4 utilizing species-specific characteristics such as those illustrated in Heiser and Smith19. While these characteristics have proven useful in most instances, they are subject to some degree of quantitative variation as affected by environmental and/or genetic influences. Hence, molecular evidence has become increasingly useful in clarifying questionable taxonomic classifications16. Of the 500 accessions in the core collection, 62 were originally unidentified as to species (C. spp.), and 9 were unidentified as to variety (C. baccatum var. baccatum or C. baccatum var. pendulum) (Supplementary Data 4). Based on the phylogenetic relationship and population structure inferred from 28,980 linkage disequilibrium (LD)-pruned SNPs at the fourfold degenerate (4DTv) sites (Supplementary Fig. 4), combined with the manual review of data provided in the U.S. National Plant Germplasm System (NPGS) and images of the fruit of these accessions grown in Changsha, Hunan province, China, all 62 previously unclassified accessions were unambiguously assigned to a specific species and/or variety group within a species, and nine C. baccatum accessions without variety classification were assigned to an appropriate variety group (Supplementary Data 4). An additional 80 accessions were found to have been incorrectly classified and were subsequently reassigned to the correct species (n = 47) or variety group (n = 33). It is notable that 10 accessions, all from Mexico, previously classified as wild C. annuum var. glabriusculum (9) or unclassified (1), were clustered within and shared a similar genetic structure with the cultivated C. annuum var. annuum accessions, suggesting that these accessions are possibly feral forms (Fig. 2b and Supplementary Data 4).
a Fixation index (FST) between different pepper populations. For each comparison, all FST values generated from 100 permutations were lower than the empirical FST value. b Total size of highly differentiated genome regions (top 1% FST) among the five domesticated Capsicum species. c Number of genes within the highly differentiated regions. d UpSet plot of common and unique genes within the differentiated regions among the five domesticated Capsicum species. e History of the effective population size of different pepper groups. f Linkage disequilibrium (LD) decay pattern of different pepper groups. g Boxplots of nucleotide diversity (π) for different pepper groups. For each box plot, the lower and upper bounds of the box indicate the first and third quartiles, respectively, and the center line indicates the median. The numbers of windows (n) for PUB, CHA, BAB, BAP, GLA, ANN, FRU and CHN are 28,893, 28,672, 28,736, 29,061, 29,007, 29,141, 29,242, 29,189, respectively. Note, π values could be overestimated due to the sub-sampling using GenoCore. PUB, C. pubescens; CHA, C. chacoense; BAB, C. baccatum var. baccatum; BAP, C. baccatum var. pendulum; FRU, C. frutescens; CHN, C. chinense; GLA, C. annuum var. glabriusculum; ANN, C. annuum var. annuum. Detailed gene information is provided in Supplementary Data 5.
After taxonomy correction, the 500 accessions were classified into seven Capsicum species, C. pubescens (n = 38), C. chacoense (n = 17), C. baccatum (n = 118), C. annuum (n = 112), C. galapagoense (n = 1), C. frutescens (n = 99) and C. chinense (n = 115). C. annuum was further divided into cultivated (C. annuum var. annuum; n = 90, including 10 possible feral accessions) and wild (C. annuum var. glabriusculum; n = 22) forms. C. baccatum was also divided into cultivated (C. baccatum var. pendulum; n = 109) and wild (C. baccatum var. baccatum; n = 9) groups (Fig. 1a, b). The placement of the seven species in the phylogenetic tree was consistent with the previously reported phylogeny1, with C. pubescens (Pubescens clade) sister to the other six species, followed by C. chacoense, C. baccatum var. baccatum and C. baccatum var. pendulum, forming the Baccatum clade, and C. galapagoense, C. frutescens, C. chinense, C. annuum var. glabriusculum, and C. annuum var. annuum, forming the Annuum clade (Fig. 1b). Population structure analysis revealed seven distinct lineages, which was highly consistent with the phylogenetic tree (Fig. 1b). Principal component analysis (PCA) revealed three primary clusters representing the Pubescens, Baccatum and Annuum clades (Fig. 1c, d), with PC2 clearly separating the four groups, C. frutescens, C. chinense, C. annuum var. annuum, and C. annuum var. glabriusculum, in the Annuum clade (Fig. 1d).
A total of 38 possible hybrids were identified in these pepper accessions and all were from the Annuum clade, including five from C. annuum var. annuum, three from C. annuum var. glabriusculum, 10 from C. chinense and 20 from C. frutescens (Supplementary Data 4). This result is consistent with the cross-compatibility observed between species in the Annuum clade20. These possible hybrids and the 10 possible C. annuum var. annuum feral accessions were excluded from the downstream population analyses.
Population differentiation of Capsicum species
We studied genome-wide population differentiation using the fixation index (FST) among the five domesticated species groups, C. pubescens, C. baccatum var. pendulum, C. frutescens, C. chinense and C. annuum var. annuum, and the three wild groups, C. chacoense, C. baccatum var. baccatum and C. annuum var. glabriusculum. C. baccatum var. baccatum and C. baccatum var. pendulum had the lowest level of genome-wide population differentiation (FST = 0.29) (Fig. 2a). FST values were larger than 0.37 for all other comparisons, supporting strong population differentiation among Capsicum species and independent domestications of the different cultivated species.
The five domesticated species are distributed in specific and unique environments3, have distinct aroma profiles21,22 and exhibit a broad spectrum of biotic and abiotic stress tolerances23. To better understand the genetic basis of these distinct characteristics, we identified highly differentiated genomic regions (top 1% FST windows). The total sizes of highly differentiated genomic regions were similar among different pairwise species comparisons (Fig. 2b), while substantially more genes were found in the differentiated regions between C. pubescens and C. annuum var. annuum (Fig. 2c). Though most of the genes were specific to single comparisons between the five domesticated species, genes involved in biotic/abiotic responses were commonly detected in more than one comparison (Fig. 2d and Supplementary Data 5).
Flowering time is a complex trait that reflects the adaptation of plants to their corresponding growing environments. The timing of flowering differs among the five domesticated Capsicum species. C. pubescens requires the longest time to flower, followed by C. baccatum var. pendulum, C. chinense and C. frutescens, whereas C. annuum var. annuum requires the shortest amount of time. Genes involved in flowering time were found in highly differentiated genomic regions among the domesticated species, such as those homologous to Arabidopsis Suppressor of FRI 4 (SUF4) and FLX Expressor 3 (FLX3) and FY that encodes an RNA 3’ processing protein involved in the regulation of floral transition (Supplementary Data 5). Notably, genes homologous to SUF4, FLX3 and FY were identified in highly differentiated regions in multiple comparisons. These genes provide insights into flowering time variation in the Capsicum species.
Capsaicinoids contribute greatly to the distinct spicy flavor of fruits of different Capsicum species22,24. Genes involved in the biosynthesis of capsaicinoids were identified in the highly differentiated regions in comparisons among the five domesticated species, including one encoding the cinnamoyl-CoA reductase (CCR; Caz01g35570) between C. frutescens and C. pubescens and between C. chinense and C. pubescens (Fig. 2d and Supplementary Data 5). Volatiles are important flavor-related compounds that impact consumer preferences25. Compounds such as alcohols and aldehydes are commonly found in fruits of C. annuum var. annuum, C. chinense and C. frutescens, but their profiles vary among different species26. Genes regulating alcohol and aldehyde biosynthesis, such as those encoding alcohol dehydrogenases, were found in the identified differentiated genomic regions. Non-volatile compounds such as sugars were also highly correlated with flavor in sweet pepper27 and showed intra- and inter-species variation in Capsicum28. Genes encoding sucrose synthases and sugar transporters were identified in the differentiated genomic regions (Fig. 2d and Supplementary Data 5). These flavor-related genes showed dramatic differences in expression at different fruit development stages (Supplementary Fig. 5). Further functional studies of these genes could improve our understanding of flavor development and variation among domesticated peppers and conceivably lead to the development of more flavorful fruit.
A number of genes involved in biotic/abiotic stress responses were identified in differentiated regions among the domesticated species (Supplementary Data 5). Notably, two tandemly duplicated disease resistance genes, Caz09g00020 and Caz09g00030, were detected in five pairwise comparisons among the five domesticated species. These genes may underlie the resistances found in different pepper groups and could serve as potential targets in pepper resistance breeding.
Population history of Capsicum species
Dynamics of the effective population size (Ne) can provide insights into the impacts of past environmental factors and human domestication events on the demographic history of crops29. We found that Ne for all Capsicum species and variety groups decreased dramatically after the Last Glacial Maximum (LGM) period (33 to 19 ka), which profoundly affected Earth’s climate and environment30. The Ne then rapidly recovered to a plateau starting approximately 4000 years ago when Earth entered the Meghalayan age (4.2 ka to present)31 (Fig. 2e), possibly due to environmental changes and domestication events of the Capsicum species. The Ne of C. annuum var. annuum was the lowest among all the groups until recently, when it became the largest. The increase in the C. annuum var. annuum population size began around 1000 years ago and increased rapidly after Columbus’ discovery of the Americas which led to further migration and the secondary global diversification of pepper3. The intense domestication and selection of C. annuum var. annuum might have led to its slowest genome-wide linkage disequilibrium (LD) decay compared to other groups (Fig. 2f) and a significantly lower level (adjusted P < 0.05) of nucleotide diversity (π = 1.19 × 10−3) compared to the other two domesticated species in the Annuum clade, C. frutescens (π = 2.02 × 10−3) and C. chinense (π = 1.96 × 10−3) (Fig. 2g).
Convergent and divergent domestication of Capsicum species
The principal breeding targets of domesticated Capsicum species include fruit yield and quality (such as shape and pungency), plant architecture and resistance to biotic and abiotic stresses23. Fruits of the two domesticated peppers, C. annuum var. annuum and C. baccatum var. pendulum, were significantly longer and wider and had a significantly higher level of morphological diversity than those of their corresponding wild progenitors (Fig. 3a, b), suggesting possible convergent domestication in C. annuum var. annuum and C. baccatum var. pendulum leading to larger fruit sizes and more variable fruit shapes.
a Fruits of representative wild and domesticated C. annuum and C. baccatum accessions with diverse shapes. Vertical bar represents 1 cm. b Fruit length, fruit width and fruit shape index in wild and domesticated C. annuum and C. baccatum accessions. Significant difference between groups was assessed using the two-tailed Student’s test. c, d Selective sweeps in C. annuum var. annuum (c) and C. baccatum var. pendulum (d). Fruit weight, shape, and pungency-related genes (in bold) and QTLs are presented in pink, orange, and cyan, respectively. Dashed horizontal lines indicate the top 5% Composite likelihood ratio (CLR) scores. Red dots correspond to selective sweeps identified in both C. annuum var. annuum and C. baccatum var. pendulum, and orange, dark green, dark orange and purple dots indicate CLR scores on different chromosomes. e Common and specific genome regions and genes under selection during C. annuum var. annuum and C. baccatum var. pendulum domestication. f, g Manhattan plots of genome-wide association studies of fruit shape of C. annuum (f) and C. baccatum (g) accessions. Red and blue horizontal lines indicate the Bonferroni-corrected significance thresholds of GWAS at α = 0.05 and α = 1, respectively. h Nucleotide diversity (π) and allele conservation of C. baccatum var. baccatum and C. baccatum var. pendulum accessions with fruit shape index >4 (the elongated dedo-de-moça type) and those with fruit shape index <1.2 (the cambuci type). GLA, C. annuum var. glabriusculum; ANN, C. annuum var. annuum; BAB, C. baccatum var. baccatum; BAP, C. baccatum var. pendulum. Detailed information on genes and QTLs is provided in Supplementary Data 6. Source data are provided as a Source Data file.
We searched for genome-wide domestication signals by comparing the domesticated species, C. annuum var. annuum and C. baccatum var. pendulum, with their wild progenitors, C. annuum var. glabriusculum and C. baccatum var. baccatum, respectively. A total of 529 and 615 selective sweeps were identified for C. annuum var. annuum (Fig. 3c and Supplementary Data 6) and C. baccatum var. pendulum (Fig. 3d and Supplementary Data 7), respectively, with a cumulative size of 176.51 Mb (5.84% of the pepper genome) and 181.95 Mb (6.02%). Notably, only 11.17 Mb of genomic regions (containing 68 genes) were found to be under selection in both C. annuum var. annuum and C. baccatum var. pendulum (Fig. 3e), while 165.34 Mb (1553 genes) and 170.78 Mb (890 genes) were specifically selected in C. annuum var. annuum and C. baccatum var. pendulum, respectively.
Fruit size/weight is among the most important traits targeted during pepper domestication. Genes and QTLs associated with fruit size and weight detected in selective sweeps of C. annuum var. annuum were largely different from those detected in C. baccatum var. pendulum (Fig. 3c, d and Supplementary Data 8). One of the strongest domestication signals detected in C. annuum var. annuum was on chromosome 3 that contained fruit weight QTL Han3.132, while the strongest signals in C. baccatum var. pendulum included one on chromosome 2 that overlapped with Han2.132 and one on chromosome 9 that overlapped with fruit size/weight QTLs Fwd/Fwg33. Our results suggest that domestications of C. annuum var. annuum and C. baccatum var. pendulum leading to increased fruit size/weight were likely achieved through the selection of unique genomic regions.
Fruit shape QTLs such as FT4.1 and FT6.1 (ref. 34) were found under selection during the domestication of both C. annuum var. annuum and C. baccatum var. pendulum, whereas a number of other QTLs were uniquely selected, e.g., fs3.1 and FT3.1 only in C. annuum var. annuum and FT1.1, FT2.1, FT9.1 and FT12.1 only in C. baccatum var. pendulum (Fig. 3c, d). Two genes (Caz06g07310 and Caz09g04240) encoding OVATE family proteins, which have been reported in tomato to regulate fruit shape35, were found in the selected regions in C. annuum var. annuum but not in C. baccatum var. pendulum. These results indicate that, like fruit weight, the domestication of fruit shape in C. annuum var. annuum and C. baccatum var. pendulum was also mainly achieved through the selection of different genomic regions. Our core Capsicum collection displayed diverse fruit shapes. The longest and the most elongated pepper accession, Zhudachang, was found in C. annuum var. annuum (Fig. 3a). C. baccatum var. pendulum has two popular types, the bell-shaped cambuci mainly cultivated and consumed in the southwest of Brazil, and the elongated dedo-de-moça in the southern and southeastern regions of Brazil36 (Fig. 3a). Genome-wide association studies (GWAS) of fruit length and width in C. annuum and C. baccatum did not detect significant association signals (Supplementary Fig. 6). However, GWAS of fruit shape index in C. annuum identified a significant association signal on chromosome 2 between 160,915,869 bp and 161,006,859 bp, harboring five genes (Fig. 3f; Supplementary Table 6). GWAS analysis of fruit shape index in C. baccatum identified two highly significant signals, one on chromosome 2 between 172,704,945 bp and 172,706,325 bp, and the other on chromosome 10 between 120,586,110 bp and 122,585,250 bp (Fig. 3g). No genes were annotated in the associated region on chromosome 2, while the region on chromosome 10 contained four genes, including one (Caz10g08850) encoding an OVATE family protein (Supplementary Table 6). The nucleotide diversity of the Caz10g08850 genomic region was much lower in both C. baccatum var. pendulum groups with accessions having a fruit shape index >4 (the elongated dedo-de-moça type) and those having a fruit shape index <1.2 (the cambuci type) compared to C. baccatum var. baccatum (Fig. 3h), suggesting the fixation of different alleles of Caz10g08850 in different pepper types during pepper breeding to meet consumers’ preferences.
Wild peppers are usually pungent, while various pungency levels have been selected for during pepper domestication and modern breeding for different culinary uses. A total of 36 and 14 genes involved in capsaicin biosynthesis were found in selective sweeps of C. annuum var. annuum and C. baccatum var. pendulum, respectively (Fig. 3c, d and Supplementary Data 8), with only two (Caz03g22890 and Caz07g10470) of them overlapping. These results indicated that various pungency levels in C. annuum var. annuum and C. baccatum var. pendulum were mainly achieved through the selection of different genes in the capsaicin biosynthetic pathway during their independent domestications.
Introgressions from the Baccatum clade to the Annuum clade
Introgressing resistance (R) genes from closely related wild or cultivated species into modern commercial cultivars is an important and frequently used breeding strategy. For instance, Mi-1, conferring resistance to root-knot nematode in tomato, was introgressed from its distantly related wild species Solanum peruvianum37,38. Among the Capsicum species, one significant gene flow event from the Baccatum clade to a subgroup of the Annuum clade, including C. frutescens and C. chinense was detected using TreeMix39 (Fig. 4a and Supplementary Fig. 7). The significance of this gene flow event was further supported by the ABBA-BABA test40 (D = 0.21; P-value = 2.55 × 10−6) (Fig. 4b). The genome-wide average degree of introgression indicated by fd was 1.71% and the proportion of genome introgression (PGI) was 1.51%. The strongest introgression signals were detected on chromosomes 6, 7 and 12 (Fig. 4c). The region at the end of chromosome 6 (Chr06:242,250,001-246,150,000; top 1% fd as the cutoff, corresponding to fd = 11.38%) contained 72 genes (Supplementary Data 9), including 17 R genes, of which 15 formed two clusters with 6 and 9 genes, respectively (Fig. 4d). One gene in the first cluster, Caz06g28230, homologous to tomato Mi-1 (ref. 41), is known to confer nematode resistance in pepper (denoted as CaRKNR)42. Further scanning of all accessions in the Annuum clade for genomic regions similar to those in the Baccatum clade revealed the presence of a Baccatum introgression in C. chinense and C. frutescens, which was rarely found in C. annuum (Fig. 4e). This introgression led to increased nucleotide diversity levels (Fig. 4d) and stronger LD (Fig. 4f) in this region, in C. chinense and C. frutescens compared to C. annuum var. annuum, possibly due to lack of recombination. Genes known to contribute to biotic and abiotic stress tolerance were also identified in the introgressed regions on chromosomes 7 and 12 (Supplementary Fig. 8 and Supplementary Data 9). These results together suggest that the introgression from the Baccatum clade to C. chinense and C. frutescens could be a consequence of the introduction of resistance traits.
a Phylogenetic structure and gene flow detected between the Baccatum and the Annuum clades. The red arrow line indicates the direction of gene flow. b Four-taxon topology ((P1,P2),P3,O) used in the ABBA-BABA model to detect the degree of introgression (fd) and proportion of introgression across the whole genome (PGI) between C. baccatum (var. baccatum and var. pendulum) and the subgroup of the Annuum clade including C. frutescens and C. chinense. c Degree of introgression (fd) from C. baccatum to C. chinense and C. frutescens across the pepper genome. The top five strongest introgression signals are indicated by black arrows. Dashed horizontal lines indicate the top 1% fd. d Nucleotide diversity (π) of the genome region at the end of chromosome 6 containing the strongest introgression signal. e Introgression from C. baccatum to C. chinense and C. frutescens at the end of chromosome 6. The dark red color represents alleles prevalent in C. baccatum accessions (allele frequency ≥ 0.8). f LD decay patterns of the introgressed region shown in (e). PUB, C. pubescens; BAB, C. baccatum var. baccatum; BAP, C. baccatum var. pendulum; FRU, C. frutescens; CHN, C. chinense; ANN, C. annuum var. annuum.
Discussion
The high-quality chromosome-scale reference genomes and the graph pan-genome of three domesticated pepper species, and the single-base resolution genome variation map of various cultivated and wild pepper species developed in the present study represent a great complement to the recently released pepper reference genomes, pan-genomes and the variation map that mainly focused on one species, C. annuum9,10,11,43, providing additional valuable resources for pepper breeding, and evolutionary and comparative studies within Capsicum and beyond. Large-scale pepper germplasm sequencing reported here serves as an important complementary approach to morphological checking for accurately assigning taxonomy information to plant materials in Capsicum germplasm collections, an accomplishment also demonstrated in a previous study16. Deep resequencing of 500 Capsicum accessions in the core collection captures genetic variation among the five domesticated and three closely related wild species. Phylogenetic and population structure analyses support the independent domestications of the five cultivated Capsicum species. Examination of the genetic basis of population differentiation provides insight into key variations of flowering time, fruit aroma and stress tolerance among the five domesticated pepper species (Fig. 5), which helps understand their adaptations to different environments and the impacts of convergent/divergent domestication in shaping the diversity within the Capsicum genus in response to consumers’ preferences and needs.
Representative fruits are presented for each species/group. Circles and squares represent the domesticated and wild types, respectively. Dark red, light brown and brown represent Pubescens, Baccatum and Annuum clades, respectively. Genes related to population differentiation (dashed white arrows) and introgression (purple arrow) are indicated in white and purple colors, respectively. Selected genes (in bold) and QTLs related to fruit shape, fruit size/weight and pungency during pepper domestication (green arrows) are highlighted in pink, green and cyan, respectively. PUB, C. pubescens; CHA, C. chacoense; BAB, C. baccatum var. baccatum; BAP, C. baccatum var. pendulum; FRU, C. frutescens; CHN, C. chinense; GLA, C. annuum var. glabriusculum; ANN, C. annuum var. annuum. Genes/QTLs are the same as those shown in Figs. 2 and 3 and Supplementary Fig. 8.
Fruit size and shape are among the most dramatic domestication traits of Capsicum species. Selective sweeps identified in C. annuum var. annuum and C. baccatum var. pendulum, two different pepper domesticated species, indicate that shared selection goals such as for enlarged fruit size and elongated fruit shape were mainly achieved by selection of different genomic regions, despite that a few genomic regions overlapping with known fruit shape QTLs have been selected in both cultivated pepper species (Fig. 5). Combined with GWAS, an OVATE family protein gene, Caz10g08850, was identified to be highly associated with fruit shape and has been under selection in different types of C. baccatum var. pendulum with contrasting fruit shapes. OVATE family proteins have been functionally demonstrated to control fruit shape in plants35. Alleles of Caz10g08850 could be utilized in breeding or gene editing programs to create novel shapes to meet consumers’ preferences. Capsaicinoids are the specialized metabolites in Capsicum species that determine the pungency. Different genes in the capsaicinoid biosynthesis pathway have been selected in C. annuum var. annuum and C. baccatum var. pendulum. In summary, the present study demonstrates that genomic regions selected in different Capsicum species for the most important breeding targets, e.g., fruit size, shape and pungency, are largely different, consistent with their independent domestications. Further large-scale phenotypic analysis of the core collection would provide additional insights into the evolution and divergence of these important domestication traits in different pepper species.
Introducing resistance genes from related species into modern commercial cultivars is an important and frequently used strategy for enhancing resistance characteristics. Our results provide evidence of introgression from C. baccatum into C. chinense and C. frutescens, carrying genes conferring various biotic and abiotic stress resistances (Fig. 5). The introgressed alleles from C. baccatum were not found in C. annuum var. annuum, which might reflect prezygotic and postzygotic barriers preventing direct crosses between C. annuum var. annuum and C. baccatum44,45. Readily crossable with C. annuum var. annuum, the C. chinense and C. frutescens accessions carrying the beneficial C. baccatum alleles could be used to introduce the disease resistance traits into C. annuum var. annuum.
Methods
Plant materials and genome and transcriptome sequencing
A total of 1296 accessions from nine different Capsicum species (prior to taxonomy reclassification) were used in this study, of which 1210 were obtained from the U.S. National Plant Germplasm System and the remaining 86 were obtained from the Pepper Germplasm Bank at Hunan Academy of Agricultural Science, China. Genomic DNA was extracted from young fresh leaves of these accessions using the Plant Genomic DNA kit (Tiangen, China) following the manufacturer’s instructions. A total of 1.5 µg DNA per sample was used to construct Illumina DNA libraries with insert sizes of ~350 bp, using the Truseq Nano DNA HT Sample Preparation Kit (Illumina, USA) following the manufacturer’s protocol. These libraries were sequenced on the Illumina NovaSeq 6000 platform, and paired-end reads of 150 bp in length were generated.
For genome assemblies, seedlings of C. annuum var. annuum Zhangshugang, C. baccatum var. pendulum PI 632928 and C. pubescens Grif 1614 were grown in a greenhouse and transferred to a dark room for 24 h before collection of young fresh leaves. High molecular weight DNA was extracted from young fresh leaves using the cetyltrimethylammonium bromide method46 and used for the construction of the PacBio SMRT library following the standard SMRT bell construction protocol (PacBio, USA). The SMRT library was sequenced on a PacBio Sequel platform to generate CLR reads for Zhangshugang and PI 632928, and on a PacBio Sequel II platform to generate HiFi reads for Grif 1614. In addition, Illumina paired-end libraries were constructed using the Truseq Nano DNA HT Sample Preparation Kit (Illumina, USA), and Hi-C libraries were prepared following the proximo Hi-C plant protocol (Phase Genomics, USA) for the three pepper lines. Both Illumina and Hi-C libraries were sequenced on the Illumina NovaSeq 6000 platform with the paired-end, 150-bp mode.
To assist protein-coding gene prediction, transcriptome sequencing was performed using tissues from leaf, flower and fruit. Total RNA was extracted using the TRIzol reagent (Invitrogen). Strand-specific RNA-Seq libraries were constructed using the Illumina TruSeq RNA Sample Prep Kit and sequenced on the NovaSeq 6000 platform. In addition, a pool of RNA mixed from all samples was used to construct one PacBio Iso-Seq library with the SMRTbell Express Template Prep kit 2.0 and sequenced on the PacBio Sequel II platform.
Plant growth conditions and phenotyping
Seeds of pepper accessions were sterilized with 5% sodium hypochlorite solution for 15 min, washed with water and then sown in a 200-hole seedling tray filled with vermiculite. The seedlings were grown in greenhouses at 25/18 °C day/night temperature and with a 16/8-h light/dark cycle, relative humidity of 60–70%, and light intensity of 15000 Lux at Changsha (N28°15′10″, E113°5’9″), Hunan province, China. Three ripe fruits were harvested per plant, and each fruit was photographed, and the lateral and longitudinal lengths of the fruit were measured with three replicates.
De novo genome assembly
For Zhangshugang and PI 632928, PacBio CLR reads were error-corrected and de novo assembled into contigs using MECAT2 (ref. 47) with default parameters. By aligning the PacBio reads to the assembled contigs, errors in the assembled contigs were corrected using the Arrow pipeline from the SMRT link 4 toolkit (https://www.pacb.com/products-and-services/analytical-software/smrt-analysis/). The assembled contigs were further polished with the Illumina short reads using Pilon48 (v1.22). For Grif 1614, PacBio HiFi reads were assembled into contigs using Hifiasm49 (v0.16.0) with default parameters. Purge Haplotigs50 (v1.1.2) was then used to remove redundancies in the assembled contigs with default parameters. Assembled contigs were also compared against the NCBI non‐redundant nucleotide database to remove possible contaminated sequences from organelle and microorganism genomes. To build pseudomolecules from the assembled contigs, Hi-C reads were cleaned to remove adapters and low-quality sequences using Trimmomatic51 (v0.39), and then aligned to the contigs using BWA52 (v0.7.17) with default parameters. The contigs were then clustered into pseudomolecules using the agglomerative hierarchical clustering method implemented in LACHESIS53. Base accuracy and completeness of the genome assemblies were evaluated using Merqury12 with default settings and BUSCO13 using the embryophyta odb10 database, respectively.
Repetitive sequence annotation and protein-coding gene prediction
Repetitive sequences in the three genomes were identified by integrating homology-based and de novo predictions. For homology-based prediction, transposable elements (TEs) in Repbase54 were used to scan the genomes with RepeatMasker55 (v4.0.9). The de novo prediction was performed using RepeatModeler (http://www.repeatmasker.org/RepeatModeler/), which is based on de novo repeat detection programs, RECON56 (v1.08) and RepeatScout57 (v1.0.5). Furthermore, a de novo search for long terminal repeat (LTR) retrotransposons in the genomes was performed using LTR_FINDER58 (v1.0.7).
Protein-coding genes were predicted from the repeat-masked genomes by integrating evidence from three different approaches: ab initio, homology-based and transcriptome-based gene predictions. First, RNA-Seq reads from different tissues and developing stages were aligned to the genome assemblies using HISAT2 (ref. 59) (v2.0.4) and then assembled into transcripts using StringTie60 (v2.0). These transcripts, combined with the open reading frames predicted from the Iso-Seq transcripts using PASA61 (v2.0.1), were used as transcript evidence for gene prediction. AUGUSTUS62 (v3.3.1) and GENESCAN63 were used to perform ab initio gene prediction. A set of high-quality full-length cDNA sequences derived from the transcriptome sequences were used to train the ab initio gene predictors. Exonerate64 (v2.2.0) was used for homology-based gene prediction with protein sequences from the four Capsicum genomes, C. annuum var. annuum CM334, C. baccatum PBC81, C. chinense PI 159236 and C. annuum var. annuum Zunla-1 (refs. 7,8). Finally, prediction results from the three approaches were integrated by MAKER65 (v3.00) to derive a set of consensus gene models for each of the three genome assemblies. The predicted protein-coding genes were functionally annotated by comparing their protein sequences against the NCBI non-redundant (nr), TrEMBL/SwissProt66 and InterPro domain67 databases. Gene ontology (GO) terms were assigned to predicted genes using Blast2GO68.
Graph pan-genome construction
A graph pan-genome was constructed using the Zhangshugang genome as the backbone. Genome sequences of PI 632928 and Grif 1614 were aligned to the Zhangshugang genome using minimap269 (v2.23) with the parameter ‘-asm20’. Small indels and large SVs were then identified using the paftools in the minimap2 package with the parameter ‘-L 10000’. In addition, genome sequences of PI 632928 and Grif 1614 were also aligned to the Zhangshugang genome using MUMmer70 (v4.0.0) with the parameter ‘--maxmatch’, and based on the alignments SVs were identified using Assemblytics71 (v1.2.1) with unique anchor length set to 5000 bp, minimum and maximum SV lengths set to 20 bp and 2 Mb, respectively. SVs identified from paftools and Assemblytics were then merged using bcftools72 (v1.14), and inconsistent SVs called from the two programs were removed. The Zhangshugang reference genome and the identified small indels and SVs were used to build the graph pan-genome using the vg toolkit73 (v1.40.0).
SNP and small indel calling for the pepper collection
Raw Illumina DNA reads were processed to remove adapter and low-quality sequences using Trimmomatic51 (v0.39) with parameters “TruSeq3-PE-2.fa:2:30:10:1:TRUE SLIDINGWINDOW:4:20 LEADING:3 TRAILING:3 MINLEN:40”. The cleaned reads were then mapped to the graph pan-genome using the Giraffe mapper74 with default parameters. The resulting aligned gam files were converted to bam files using ‘vg surject’. SNPs and small indels were then called from bam files using the Sentieon software package (https://www.sentieon.com/), which was built based on the GATK variant calling tool75. Briefly, duplicated read pairs in each alignment file were marked, and indel realignment and base quality score recalibration were performed. Variants from each sample were called using the Sentieon Haplotyper function, and joint variant calling was performed using the GVCFtyper function in Sentieon. Raw SNPs were then filtered using GATK75 (v3.8) with parameters ‘QD < 2.0||FS > 60.0||MQ < 40.0||SOR > 3.0 || MQRankSum < −12.5 || ReadPosRankSum < −8.0’ and raw small indels were filtered with parameters ‘QD < 2.0||FS > 200.0 || ReadPosRankSum < −20.0’. Filtered bi-allelic variants with minor allele frequency (MAF) ≥ 0.05 and missing data rate ≤ 0.3 were kept. Variants were annotated using SnpEff76 (v5.0e). SNPs were further LD-pruned with parameters ‘--indep-pairwise 50 10 0.3’ using PLINK (v1.9)77, and the LD-pruned SNPs were used for downstream population genomic analyses unless otherwise indicated.
Phylogenetic and population genomic analyses
A total of 28,980 LD-pruned SNPs at fourfold degenerate (4DTv) sites were used to construct the maximum likelihood (ML) phylogenetic tree and to perform population structure and principal component analysis (PCA). The ML phylogenetic tree was constructed using IQ-TREE78 (v1.6.12) with 1000 bootstrap replicates and the optimal substitution model of TVM+F+R4, which was determined based on the lowest BIC (Bayesian Information Criterion) value using ModelFinder79. Four wild tomato accessions, Solanum pennellii LA1272, S. pennellii LA0716, S. habrochaites LA1777 and S. habrochaites LA1718, were used as the outgroup. Illumina reads of these four accessions were downloaded from NCBI Sequencing Read Archive under accession numbers ERR418106, ERR418107, ERR418103 and ERR418102, and aligned to the pepper graph pan-genome for SNP calling as described above. The population structure of the Capsicum accessions was inferred using fastSTRUCTURE80 (v1.0) with the number of population clusters (K) set from 2 to 20. The optimal K (7) was determined using the chooseK.py script in fastSTRUCTURE. PCA was performed using PLINK77 (v1.9). Accessions with significant genome-wide hybridization were identified using HyDe81 with a P-value cutoff of 0.05 and were excluded from the downstream population genomic analyses (Supplementary Data 4).
Genome-wide LD decay pattern for each of the Capsicum groups was calculated using PopLDdecay82 (v3.41) with the parameter ‘-MaxDist 1000’. FST values between different Capsicum groups were calculated using VCFtools (v0.1.17) with a window size of 100 kb and a step size of 10 kb. To evaluate the significance of population differentiation, for each pair of compared populations, 100 FST values were generated from permutations through randomly reshuffling the accessions and these values were compared to the empirical FST value. Highly differentiated genomic regions between different Capsicum groups were identified as windows with top 1% FST values, and windows were merged if they overlapped. Genome-wide nucleotide diversity (π) was calculated in 100-kb non-overlapping windows using VCFtools83. The significance of the mean π values between Capsicum groups was calculated using the two-tailed t-test with the Bonferroni adjust method using the pairwise.t.test() function in the R package stats (v3.6.2).
Inference of effective population size
The effective population size (Ne) for each of the eight Capsicum groups was inferred using SMC++84 (v1.15.4.dev18+gca077da) with genome-wide SNPs. Ten accessions from each group from different representative origins were selected for the analysis. SNP missing rate was set to less than 0.2, and the runs of homozygosity longer than 5 kb were treated as missing to avoid potential bias. In order to improve the estimation accuracy, we used the composite likelihood approach by setting each of the 10 accessions as the ‘distinguished individual’ and the remaining nine as ‘undistinguished individuals’. The mutation rate and generation time were set at 6.96 × 10−9 (ref. 85) and one year, respectively.
Identification of selective sweeps
Selective sweeps across the pepper genome in the cultivated C. annuum var. annuum population and in the cultivated C. baccatum var. pendulum population were identified using SweeD86 (v4.0.0) with genome-wide SNPs, by incorporating the empirical estimate of the effective population size derived from the SMC++ analyses described above. Composite likelihood ratios (CLR) were calculated in an average of a 10-kb window across the genome by setting grid numbers according to chromosome lengths (number of grid = chromosome length/10000). Those with the top 5% highest CLR values were identified as potential selective sweeps and sweeps with physical distance no larger than 10 kb were merged. Furthermore, π ratios between the wild C. annuum var. glabriusculum and the cultivated C. annuum var. annuum, and between the wild C. baccatum var. baccatum and the cultivated C. baccatum var. pendulum were calculated. Potential selective sweeps identified from SweeD having the top 30% of π ratios between the wild and cultivated species were kept as the final selective sweeps.
Genome-wide association study
Genome-wide SNPs were used for GWAS analysis using EMMAX87. Phenotype data of fruit shape-related traits, including fruit length, fruit width and fruit width/length ratio, were log2 transformed. The top 5 principal components and the Balding-Nichols kinship were used as cofactors in the association model. The effective number of SNPs was calculated using Genetic Type I error Calculator88. The Bonferroni-corrected P-value thresholds were 2.34 × 10−8 at Type I error α = 0.05 (highly significant) and 4.68 × 10−7 at α = 1 (significant), respectively, for C. annuum, corresponding to −log10(P) values of 7.63 and 6.33, and 2.13 × 10−8 (α = 0.05) and 4.25 × 10−7 (α = 1) for C. baccatum, corresponding to −log10(P) values of 7.67 and 6.37. The candidate gene region was determined by LD between the peak SNP and its nearby SNPs with the LD cutoff of 0.351 and 0.366 (half value of the maximum averaged LD) for C. annuum var. annuum and C. baccatum var. pendulum, respectively.
Detection of gene flow and introgressions among the Capsicum species
Gene flow between the eight Capsicum groups was detected using TreeMix39 (v1.13). To obtain the optimal number of migration edges, the number of migrations was set from 0 to 20, each with 10 interactions. The optimal number of migration edges (m = 1) was obtained using the linear modeling estimate in the OptM R package (v0.1.6; https://cran.r-project.org/web/packages/OptM), where negligible variance was further explained with the increase of migration (Supplementary Fig. 7).
Genome-wide introgressions were detected by calculating the fd statistics40 in 500-kb sliding windows with a step size of 50 kb, using genome-wide SNPs. The minimum number of SNPs per window was set to 100, and the minimum proportion of samples genotyped per site was set to 0.5. We further calculated the PGI (proportion of introgression across the whole genome), following the recently proposed formula89:
where PGI refers to the average proportion of introgression across the whole genome; fdi refers to the fd value of the ith window; Gi refers to the ith window size in base pairs; G refers to the total genome size in base pairs.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Genome assemblies, raw genome and transcriptome sequencing reads have been deposited in the National Center for Biotechnology Information BioProject database under the accession nos. PRJNA800056 and PRJNA801499. Genome assemblies and annotated genes, pan-genome graphs (Giraffe indexes), and variant data (VCF format) are also available at the Pepper Genomics Database (http://ted.bti.cornell.edu/pepper). Source data are provided with this paper.
References
Carrizo García, C. et al. Phylogenetic relationships, diversification and expansion of chili peppers (Capsicum, Solanaceae). Ann. Bot. 118, 35–51 (2016).
Hernández-Pérez, T., Gómez-García, M., del, R., Valverde, M. E. & Paredes-López, O. Capsicum annuum (hot pepper): an ancient Latin-American crop with outstanding bioactive compounds and nutraceutical potential. A review. Compr. Rev. Food Sci. Food Saf. 19, 2972–2993 (2020).
Djian-Caporalino, C., Lefebvre, V., Sage-Daubèze, A.-M. & Palloix, A. Capsicum. Genetic Resources Chromosome Engineering & Crop Improvement (ed. Singh, R. J.) 186–232 (CRC Press, 2006).
D’Arcy, W. G. & Eshbaugl, W. H. New world peppers [Capsicum—Solanaceae] North of Colombia: a resume. Baileya 19, 93–105 (1974).
Moses, M. & Umaharan, P. Genetic structure and phylogenetic relationships of Capsicum chinense. J. Am. Soc. Hortic. Sci. 137, 250–262 (2012).
Pereira-Dias, L., Vilanova, S., Fita, A., Prohens, J. & Rodríguez-Burruezo, A. Genetic diversity, population structure, and relationships in a collection of pepper (Capsicum spp.) landraces from the Spanish centre of diversity revealed by genotyping-by-sequencing (GBS). Hortic. Res. 6, 54 (2019).
Kraft, K. H. et al. Multiple lines of evidence for the origin of domesticated chili pepper, Capsicum annuum, in Mexico. Proc. Natl Acad. Sci. USA 111, 6165–6170 (2014).
Perry, L. et al. Starch fossils and the domestication and dispersal of chili peppers (Capsicum spp. L.) in the Americas. Science 315, 986–988 (2007).
Cao, Y. et al. Pepper variome reveals the history and key loci associated with fruit domestication and diversification. Mol. Plant 15, 1744–1758 (2022).
Ou, L. et al. Pan-genome of cultivated pepper (Capsicum) and its use in gene presence-absence variation analyses. N. Phytol. 220, 360–363 (2018).
Lee, J.-H. et al. High-quality chromosome-scale genomes facilitate effective identification of large structural variations in hot and sweet peppers. Hortic. Res. 9, 1–13 (2022).
Rhie, A., Walenz, B. P., Koren, S. & Phillippy, A. M. Merqury: reference-free quality, completeness, and phasing assessment for genome assemblies. Genome Biol. 21, 245 (2020).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Jeong, S. et al. GenoCore: a simple and fast algorithm for core subset selection from large genotype datasets. PLoS ONE 12, e0181420 (2017).
Goodwin, Z. A., Harris, D. J., Filer, D., Wood, J. R. I. & Scotland, R. W. Widespread mistaken identity in tropical plant collections. Curr. Biol. 25, R1066–R1067 (2015).
Tripodi, P. et al. Global range expansion history of pepper (Capsicum spp.) revealed by over 10,000 genebank accessions. Proc. Natl Acad. Sci. USA 118, e2104315118 (2021).
Pickersgill, B. Relationships between weedy and cultivated forms in some species of chili peppers (Genus capsicum). Evolution 25, 683 (1971).
McLeod, M. J., Guttman, S. I. & Eshbaugh, W. H. Early evolution of chili peppers (Capsicum). Econ. Bot. 36, 361–368 (1982).
Heiser, C. B. & Smith, P. G. The cultivated capsicum peppers. Econ. Bot. 7, 214–227 (1953).
Parry, C., Wang, Y. W., Lin, S. W. & Barchenger, D. W. Reproductive compatibility in Capsicum is not necessarily reflected in genetic or phenotypic similarity between species complexes. PLoS ONE 16, e0243689 (2021).
Rodríguez-Burruezo, A., Kollmannsberger, H., González-Mas, M. C., Nitz, S. & Fernando, N. HS-SPME comparative analysis of genotypic diversity in the volatile fraction and aroma-contributing compounds of Capsicum fruits from the annuum-chinense-frutescens complex. J. Agric. Food Chem. 58, 4388–4400 (2010).
Kollmannsberger, H., Rodríguez-Burruezo, A., Nitz, S. & Nuez, F. Volatile and capsaicinoid composition of ají (Capsicum baccatum) and rocoto (Capsicum pubescens), two Andean species of chile peppers. J. Sci. Food Agric. 91, 1598–1611 (2011).
Jaiswal, V., Gahlaut, V., Kumar, N. & Ramchiary, N. Genetics, genomics and breeding of chili pepper Capsicum frutescens L. and other Capsicum species. Advances in Plant Breeding Strategies: Vegetable Crops: Volume 9: Fruits and Young Shoots (eds Al-Khayri, J. M. et al.) 59–86 (Springer, 2021).
Wahyuni, Y. et al. Metabolomics and molecular marker analysis to explore pepper (Capsicum sp.) biodiversity. Metabolomics 9, 130–144 (2013).
Tieman, D. et al. A chemical genetic roadmap to improved tomato flavor. Science 355, 391–394 (2017).
Sarpras, M. et al. Comparative analysis of fruit metabolites and pungency candidate genes expression between Bhut jolokia and other Capsicum species. PLoS ONE 11, e0167791 (2016).
Eggink, P. M. et al. A taste of sweet pepper: volatile and non-volatile chemical composition of fresh sweet pepper (Capsicum annuum) in relation to sensory evaluation of taste. Food Chem. 132, 301–310 (2012).
Zamljen, T., Jakopič, J., Hudina, M., Veberič, R. & Slatnar, A. Influence of intra and inter species variation in chilies (Capsicum spp.) on metabolite composition of three fruit segments. Sci. Rep. 11, 1–11 (2021).
Sun, X. et al. Phased diploid genome assemblies and pan-genomes provide insights into the genetic history of apple domestication. Nat. Genet. 52, 1423–1432 (2020).
Clark, P. U. et al. The last glacial maximum. Science 325, 710–714 (2009).
Voosen, P. New geological age comes under fire. Science 361, 537–538 (2018).
Hill, T. A. et al. Regions underlying population structure and the genomics of organ size determination in Capsicum annuum. Plant Genome 10, plantgenome2017.03.0026 (2017).
Lee, H.-Y. et al. Uncovering candidate genes controlling major fruit-related traits in pepper via genotype-by-sequencing based QTL mapping and genome-wide association study. Front. Plant Sci. 11, 1100 (2020).
Chunthawodtiporn, J., Hill, T., Stoffel, K. & Van Deynze, A. Quantitative trait loci controlling fruit size and other horticultural traits in bell pepper (Capsicum annuum). Plant Genome 11, 160125 (2018).
Wu, S. et al. A common genetic mechanism underlies morphological diversity in fruits and other plant organs. Nat. Commun. 9, 4734 (2018).
Gomes, G. P. et al. Diallel analysis of the morphoagronomic, phytochemical, and antioxidant traits in Capsicum baccatum var. pendulum. Hortic. Environ. Biotechnol. 62, 435–446 (2021).
Seah, S., Yaghoobi, J., Rossi, M., Gleason, C. A. & Williamson, V. M. The nematode-resistance gene, Mi-1, is associated with an inverted chromosomal segment in susceptible compared to resistant tomato. Theor. Appl. Genet. 108, 1635–1642 (2004).
Lin, T. et al. Genomic analyses provide insights into the history of tomato breeding. Nat. Genet. 46, 1220–1226 (2014).
Pickrell, J. K. & Pritchard, J. K. Inference of population splits and mixtures from genome-wide allele frequency data. PLoS Genet. 8, e1002967 (2012).
Martin, S. H., Davey, J. W. & Jiggins, C. D. Evaluating the use of ABBA-BABA statistics to locate introgressed loci. Mol. Biol. Evol. 32, 244–257 (2015).
Vos, P. et al. The tomato Mi-1 gene confers resistance to both root-knot nematodes and potato aphids. Nat. Biotechnol. 16, 1365–1369 (1998).
Mao, Z. et al. Cloning and functional analyses of pepper CaRKNR involved in Meloidogyne incognita resistance. Euphytica 205, 903–913 (2015).
Liao, Y. et al. The 3D architecture of the pepper genome and its relationship to function and evolution. Nat. Commun. 13, 3479 (2022).
Manzur, J. P., Fita, A., Prohens, J. & Rodríguez-Burruezo, A. Successful wide hybridization and introgression breeding in a diverse set of common peppers (Capsicum annuum) using different cultivated ají (C. baccatum) accessions as donor parents. PLoS ONE 10, e0144142 (2015).
Martins, K. C., Pereira, T. N. S., Souza, S. A. M., Rodrigues, R. & do Amaral Junior, A. T. Crossability and evaluation of incompatibility barriers in crosses between capsicum species. Crop Breed. Appl. Biotechnol. 15, 139–145 (2015).
Murray, M. G. & Thompson, W. F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 8, 4321–4326 (1980).
Xiao, C. L. et al. MECAT: fast mapping, error correction, and de novo assembly for single-molecule sequencing reads. Nat. Methods 14, 1072–1074 (2017).
Walker, B. J. et al. Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE 9, e112963 (2014).
Cheng, H., Concepcion, G. T., Feng, X., Zhang, H. & Li, H. Haplotype-resolved de novo assembly using phased assembly graphs with hifiasm. Nat. Methods 18, 170–175 (2021).
Roach, M. J., Schmidt, S. & Borneman, A. R. Purge haplotigs: allelic contig reassignment for third-gen diploid genome assemblies. BMC Bioinformatics 19, 460 (2018).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2009).
Burton, J. N. et al. Chromosome-scale scaffolding of de novo genome assemblies based on chromatin interactions. Nat. Biotechnol. 31, 1119–1125 (2013).
Jurka, J. et al. Repbase Update, a database of eukaryotic repetitive elements. Cytogenet. Genome Res. 110, 462–467 (2005).
Chen, N. Using RepeatMasker to identify repetitive elements in genomic sequences. Curr. Protoc. Bioinformatics 4, 4.10 (2004).
Bao, Z. & Eddy, S. R. Automated de novo identification of repeat sequence families in sequenced genomes. Genome Res. 12, 1269–1276 (2002).
Price, A. L., Jones, N. C. & Pevzner, P. A. De novo identification of repeat families in large genomes. Bioinformatics 21, 351–358 (2005).
Xu, Z. & Wang, H. LTR-FINDER: an efficient tool for the prediction of full-length LTR retrotransposons. Nucleic Acids Res. 35, 265–268 (2007).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Pertea, M. et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33, 290–295 (2015).
Haas, B. J. et al. Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nucleic Acids Res. 31, 5654–5666 (2003).
Stanke, M. et al. AUGUSTUS: ab initio prediction of alternative transcripts. Nucleic Acids Res. 34, 435–439 (2006).
Burge, C. & Karlin, S. Prediction of complete gene structures in human genomic DNA. J. Mol. Biol. 268, 78–94 (1997).
Slater, G. S. C. & Birney, E. Automated generation of heuristics for biological sequence comparison. BMC Bioinformatics 6, 31 (2005).
Cantarel, B. L. et al. MAKER: an easy-to-use annotation pipeline designed for emerging model organism genomes. Genome Res. 18, 188–196 (2008).
Boeckmann, B. et al. The SWISS-PROT protein knowledgebase and its supplement TrEMBL in 2003. Nucleic Acids Res. 31, 365–370 (2003).
Mitchell, A. et al. The InterPro protein families database: the classification resource after 15 years. Nucleic Acids Res. 43, D213–D221 (2015).
Conesa, A. & Götz, S. Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int. J. Plant Genomics 2008, 619832 (2008).
Li, H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34, 3094–3100 (2018).
Marçais, G. et al. MUMmer4: a fast and versatile genome alignment system. PLoS Comput. Biol. 14, e1005944 (2018).
Nattestad, M. & Schatz, M. C. Assemblytics: a web analytics tool for the detection of variants from an assembly. Bioinformatics 32, 3021–3023 (2016).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, giab008 (2021).
Garrison, E. et al. Variation graph toolkit improves read mapping by representing genetic variation in the reference. Nat. Biotechnol. 36, 875–881 (2018).
Sirén, J. et al. Pangenomics enables genotyping of known structural variants in 5202 diverse genomes. Science 374, abg8871 (2021).
Mckenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff. Fly 6, 80–92 (2012).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., Von Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587–589 (2017).
Raj, A., Stephens, M. & Pritchard, J. K. FastSTRUCTURE: variational inference of population structure in large SNP data sets. Genetics 197, 573–589 (2014).
Blischak, P. D., Chifman, J., Wolfe, A. D. & Kubatko, L. S. HyDe: a Python Package for Genome-Scale Hybridization Detection. Syst. Biol. 67, 821–829 (2018).
Zhang, C., Dong, S. S., Xu, J. Y., He, W. M. & Yang, T. L. PopLDdecay: a fast and effective tool for linkage disequilibrium decay analysis based on variant call format files. Bioinformatics 35, 1786–1788 (2019).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Terhorst, J., Kamm, J. A. & Song, Y. S. Robust and scalable inference of population history from hundreds of unphased whole genomes. Nat. Genet. 49, 303–309 (2017).
Moniz De Sá, M. & Drouin, G. Phylogeny and substitution rates of angiosperm actin genes. Mol. Biol. Evol. 13, 1198–1212 (1996).
Pavlidis, P., Živković, D., Stamatakis, A. & Alachiotis, N. SweeD: likelihood-based detection of selective sweeps in thousands of genomes. Mol. Biol. Evol. 30, 2224–2234 (2013).
Kang, H. M. et al. Variance component model to account for sample structure in genome-wide association studies. Nat. Genet. 42, 348–354 (2010).
Li, M., Yeung, J. M. Y., Cherny, S. S. & Sham, P. C. Evaluating the effective numbers of independent tests and significant p-value thresholds in commercial genotyping arrays and public imputation reference datasets. Hum. Genet. 131, 747–756 (2012).
Zhou, Y. et al. Triticum population sequencing provides insights into wheat adaptation. Nat. Genet. 52, 1412–1422 (2020).
Acknowledgements
This research was supported by grants from the Laboratory of Lingnan Modern Agriculture Project (NT2021004), the National Natural Science Foundation of China (U19A2028, U21A20230 and 32130097), the Science and Technology Innovation Program of Hunan Province, China (2021NK1006 and 2021JC0007), and the US National Science Foundation (IOS-1855585 to Z.F.).
Author information
Authors and Affiliations
Contributions
X.Z., Z.F. and S.W. designed and managed the project. F.L., C.X., Z.W., J.W., B.T., H.X., B.H., H.Suo, B.Y., L.O., X.L., S.Z., S.Y., Z.L., F.Y., Z.P., Y.M. and X.D. collected samples and performed experiments. J.Z., F.L., H.Sun, X.S., X.W. and R.J. performed data analyses. J.Z. and S.W. wrote the manuscript. Z.F. revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, F., Zhao, J., Sun, H. et al. Genomes of cultivated and wild Capsicum species provide insights into pepper domestication and population differentiation. Nat Commun 14, 5487 (2023). https://doi.org/10.1038/s41467-023-41251-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-41251-4
This article is cited by
-
Genome-wide identification of CaWD40 proteins reveals the involvement of a novel complex (CaAN1-CaDYT1-CaWD40-91) in anthocyanin biosynthesis and genic male sterility in Capsicum annuum
BMC Genomics (2024)
-
Comparative chloroplast genome analyses of cultivated and wild Capsicum species shed light on evolution and phylogeny
BMC Plant Biology (2024)
-
Two telomere-to-telomere gapless genomes reveal insights into Capsicum evolution and capsaicinoid biosynthesis
Nature Communications (2024)
-
Plant pangenomes for crop improvement, biodiversity and evolution
Nature Reviews Genetics (2024)
-
Biodiversity and network complexity of rhizosphere soil microbiomes regulate the differentiation of Capsicum growth strategies
Plant and Soil (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.