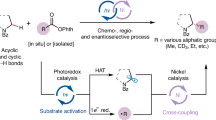
Abstract
While significant progress has been made in developing selective C-H bond cross-couplings in the field of radical chemistry, the site and stereoselectivity remain a long-standing challenge. Here, we present the successful development of stereodivergent allylic C(sp3)-H bond arylations through a systematic investigation of the direction and degree of stereoselectivity in the cross-coupling process. In contrast to the signature photosensitized geometrical isomerization of alkenes, the catalytic reaction demonstrates the feasibility of switching the C-C double bond stereoselectivity by means of ligand control as well as steric and electronic effects. Computational studies explain the stereochemical outcome and indicate that excitation of a Ni-allyl complex from singlet to a triplet state results in a spontaneous change of the allyl group coordination and that the subsequent isomerization can be directed by the choice of the ligand to achieve E/Z selectivity.
Similar content being viewed by others
Introduction
Alkenes occur widely in nature and are feedstocks in various fields of the chemical industry, ranging from the vast scale manufacture of plastics and detergents to the synthesis of pharmaceutical intermediates and fragrances1. The selective construction of C–C double bonds in alkenes, either the E or the higher energetic Z isomer, has long been one of the fundamental yet elusive topics in the advancement of organic chemistry. In this regard, classical carbonyl olefination, elimination reaction and alkyne addition reaction are commonly used, albeit with the loss of valuable functionalities2. On the other hand, the ideal and most straightforward way is the direct functionalization of pre-existing alkenes. In the past decades, a variety of catalytic methodologies, including olefin metathesis, Heck-type cross-coupling and alkene isomerization have emerged as attractive approaches for the convergent synthesis of stereo-defined alkenes3,4. Of these approaches, methods based on transition-metal catalyzed alkene positional and geometrical isomerization were developed to selectively access isomers through an energetically net “downhill” process5, whereas photochemical geometrical isomerization has offered a route in the opposite direction but with the limitation only being suitable for chromophoric substrates6,7,8,9. Despite these advances, there are no corresponding transformations that are broadly applicable to the site-selective and stereodivergent preparation of multi-substituted alkenes under mild conditions. The quest for novel and selective methodologies remains a subject of intensive study.
In recent years, visible-light photocatalysis enabled C–H bond functionalization has received widespread attention10,11,12,13,14,15. One of the most recent prominent advances is photoredox nickel dual catalysis enabled C–H bond cross-coupling. Within this subdomain of metallaphotoredox catalysis16,17, two fundamentally distinct pathways have been utilized for the generation of reactive open-shell species18 from native C–H functionality: (1) direct oxidative single-electron transfer (SET) between substrates and excited state of photocatalyst19,20 and (2) hydrogen atom transfer (HAT) of native sp3 C–H bonds by appropriate photo-generated N, O, S, X (Cl, Br) radicals or photoexcited polyoxometalate species21,22,23,24,25,26,27,28,29,30,31. As a result, a very diverse class of substrates such as ethers, amides, alcohols, aldehydes, alkenes, and alkanes, have been successfully employed in the coupling with aryl or alkyl halides to create valuable C(sp2)-C(sp3) or C(sp3)-C(sp3) bonds32,33. Although significant effort has been dedicated to controlling site-selectivity among these developments, very few examples are associated with the controlling of stereochemistry34,35,36. Specifically, methods concerning the stereoselective preparation of multi-substituted alkenes in either E or Z isomeric forms are still not known. Our continuous interest in photoredox-enabled allylic C–H bond functionalization prompted us to develop a general method that can selectively give the alkenes in either E or Z configuration37,38.
Herein, we demonstrate a highly stereoselective allylic C(sp3)-H bond cross-coupling of alkenes through the synergistic combination of photoredox and nickel catalysis (Fig. 1)39.
Results
Rational design and optimization
We initially tried to identify suitable reaction conditions that are effective for allylic C(sp3)-H cross-couplings of silyl enol ethers. We also like to highlight an important recent study by Studer and co-workers, who accomplished an efficient C(sp3)-H functionalization of enol ethers40. The investigation began with the coupling of silyl enol ether 1 and methyl 4-bromobenzoate (2). After considerable efforts, we were delighted to see that with readily available reagents, including photocatalyst Ir[dF(CF3)ppy]2(dtbbpy)PF6 (1 mol%), NiCl2•DME (10 mol%), 4,4’-di-tert-butylbipyridine (dtbbpy) (15 mol%) and 2,6-lutidine (1.5 equiv.) we were able to obtain the desired product in excellent yield (83%) with good E selectivity (88%). The reactions were carried out using blue light-emitting diodes (LEDs) at room temperature (Table 1). Control experiments showed that no detectable product was formed in the absence of light irradiation, ligand, base, nickel catalyst, or photocatalyst, showcasing the essential role of all components in this synergistic catalysis. Seeking to improve on the stereoselectivity, we next evaluated several classes of ligands and found that dtbbpy was the best choice of ligand for E selectivity. In the meantime, we were excited to find that a moderate Z selectivity (74%) was realized when 6,6’-dimethyl-2,2’-bipyridine (6,6’-dmbpy) was employed, albeit with low conversion. This observation of ligand effects prompted us to further examine the reaction conditions. After extensive evaluation of different reaction parameters (see Supporting Information), we successfully established two sets of reaction conditions allowing for a stereoselectivity switch: the use of dtbbpy (L1) led to E isomer 3 (93%) in 77% yield (condition A) and employing 6,6’-dmbpy (L2) gave Z isomer 4 in 66% yield (condition B) after isolation. To the best of our knowledge, the use of ligands to tune E/Z stereoselectivity in photoredox nickel dual catalysis has never been reported.
Synthetic scope
With these reaction conditions in hand, we first turned our attention to evaluating the electrophilic coupling partners for the scope of E isomer with condition A. As shown in Fig. 2, a wide range of electron-deficient functional groups (ketone, ester, trifluoromethyl, nitrile, sulfone, chloro, sulfonamide) at the para position on the aryl bromides were well-tolerated using this synergistic strategy and providing the corresponding products 3–11 in good yields (58-83%) and excellent E selectivities (89% to only E isomer). In addition, comparable good results could be obtained for both electron-neutral and electron-rich bromoarenes in contrast to their intrinsic difficulty in oxidative addition (12–17, 58-66% yield, 95% to only E isomer). Moreover, the installation of meta-monosubstituted or disubstituted functional groups on the aryl halide turned out to be successful (18–21, 55–71% yield), excellent levels of stereocontrol were archived regardless of their electronic nature (97% to only E isomer). However, it is noteworthy that the reaction failed with substrates bearing ortho substituents under current reaction conditions. Nearly quantitatively hydro-dehalogenated products were obtained instead. This can be attributed to steric bulkiness. Polycyclic bromoarenes derived from naphthalene, phthalide, and phthalimide performed very well in the reaction (22–24, 61–73% yield, 93% to only E isomer). Additionally, pharmaceutically important heteroaryl bromides bearing indole, azaindole, quinoline, and pyridine moieties proved to be competent coupling partners with this reaction manifold (25–28, 51–71% yield, 88–96% E isomer).
Unless otherwise specified, the reactions were performed on a 0.2 mmol scale under the following reaction conditions: 1 (3 equiv.), aryl halide (1 equiv.), 2,6-lutidine (1.5 equiv.), PC-1 (1 mol%), NiBr2 (10 mol%), dtbbpy (15 mol%), benzene/THF 1:1 (2 mL), rt, 48-72 h, yields and E/Z after purification by chromatography (numbers in parentheses refer to E/Z before purification). Ac, acetyl group; TBS, tert-butyldimethylsilyl.
Having demonstrated the generality of this protocol for relatively simple substrates, we next set out to examine the synthetic potential for the late-stage cross-coupling of structurally diverse bioactive compounds. Aryl bromide electrophiles from indomethacin, sulfadimethoxine, celecoxib, diacetone-D-galactose, and pregnenolone were smoothly converted into their corresponding silyl enol ether (29–33, 52-72% yield, 93% to only E isomer). These results further highlight the robust functional group tolerance of the current development. Notably, only a trace amount of THF arylation adduct resulting from competitive solvent C(sp3)-H coupling was observed.
We next surveyed the scope of alkenes (Fig. 3) by increasing the steric bulkiness of the silyl group from TBS to TIPS, leading to comparable yield with higher level of E selectivity (34, 73% yield, 96% E isomer). High levels of stereoselectivity (93% to only E isomer) and good isolated yields (57–71%) were observed for a variety of enol ethers, as demonstrated by either cyclopentyl (35), phenyl (36), ethyl (38), benzyl (40) substituted or bulky ones from (-)-borneol (37) and (-)-menthol (39). If the size of the additional substituent in the α-position of silyl enol ether is increased, the stereoselectivity decreases, with lower E selectivity obtained for 41 (76%) and 42 (77%). The steric influence is high, and Z selectivity was achieved with substrates carrying tert-butyl and 2-methylphenyl substituents providing the corresponding products in good to excellent Z/E ratio (43–45, 84-98%). Similarly, excellent selectivity was observed in the case of steric-demanding alkene (46). Importantly, further investigation revealed that this method is not restricted to acyclic alkenes. Cyclic substrates have also been efficiently functionalized (47 and 48), showcasing the broad applicability of the current protocol. The cross-coupling can be further extended to styrenes, followed by a visible-light-induced Z to E isomerization. The corresponding styrene products were obtained in moderate yields and excellent E selectivity (49 and 50).
Unless otherwise specified, the reactions were performed on a 0.2 mmol scale under the following reaction conditions: alkene (3 equiv.), aryl bromide (1 equiv.), 2,6-lutidine (1.5 equiv.), PC-1 (1 mol%), NiBr2 (10 mol%), dtbbpy (15 mol%), benzene/THF 1:1 (2 mL), rt, 48-72 h, yields and E/Z after purification by chromatography (numbers in parentheses refer to E/Z before purification). + tert-butyl acrylate (1 equiv.) was used as additive. TIPS, triisopropylsilyl. # Formal β-arylation of ketone was achieved after deprotection. *E/Z before isomerization, 49 (37:63), 50 (33:67), see Supporting Information for reaction condition details.
We subsequently pursued to examine the generality of condition B that could promote Z selective arylation of the allylic C(sp3)-H bonds. As outlined in Fig. 4, a broad range of electron-rich and electron-poor aryl bromides served as effective coupling partners. In all cases, the arylation reactions proceeded with excellent stereoselectivity under the standard conditions (4, 51–57, 91% to only Z isomer). To achieve good efficiency for electron-rich substrates, the use of LiBr as an external bromine additive is crucial (58 to 61, 40–57% yield, only Z isomer). In contrast, only moderate Z selectivity can be obtained for heterocycle derived from pyridine (63, 74% Z). The superior reactivity of C2-substituted bidentate ligand in alkene isomerization41,42,43,44 prompted us to question whether the current protocol could be used to initiate the double bond positional isomerization, allowing for the subsequent regio- and stereoselective arylation reaction at the terminal position of the allylic system. With our optimized condition B, we were pleased to find that the synergistic catalytic mode allows the allylic silyl ether to smoothly participate in the desired isomerization/cross-coupling sequence while achieving high levels of stereocontrol (52, 61% yield, only Z). Furthermore, a commercially available allyl benzene derivative could also be employed, generating the corresponding arylation adduct 64 with a high degree of Z selectivity (only Z). The scope was further extended to an N-allyl-carbazole derivative, affording the desired product 65. Lastly, the synthetic potential of this method was showcased in the stereospecific Simmon-Smith cyclopropanation of silyl enol ether (see Supporting Information).
Unless otherwise specified, the reactions were performed on a 0.2 mmol scale under the following reaction conditions: alkene (3 equiv.), aryl halide (1 equiv.), 2,4,6-collidine (3 or 1.5 equiv.), PC-1 (1 mol%), NiBr2 (20 mol%), 6,6’-dmbpy (20 mol%), dioxane/THF 1:1 (2 mL), rt, 48-72 h, yields and Z/E after purification by chromatography (numbers in parentheses refer to Z/E before purification). + two parallel runs in 0.1 mmol were carried out with LiBr (1 equiv.) as additive.
Mechanistic investigations
To investigate a plausible mechanism for the stereodivergent cross-coupling, we initially looked for direct evidence of allylic radical formation by introducing a well-known carbon radical scavenger TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy). The reaction efficiency diminished dramatically in the presence of the radical inhibitor; the yield dropped to only 19% when 2 equiv. TEMPO was used. The TEMPO adduct 67 was observed by 1H NMR of the crude reaction mixture and further confirmed by HRMS analysis. We next sought to understand the elementary steps that are closely associated with the allylic radical generation. Although (silyl) enol ethers are known to be electron-rich alkenes, cyclic voltammetry studies showed that the direct SET oxidation by the photoexcited PC–1 (*E1/2 = 1.21 V vs. SCE in MeCN) should be difficult. The steady-state Stern-Volmer quenching study also revealed that the silyl enol ether does not quench the excited state of Ir photocatalyst (see Supporting Information). These findings preclude the generation of allylic radicals by the SET event. Moreover, aryl iodide 68 failed to be a competent coupling partner because of the weak hydrogen abstraction ability (Fig. 5b, H-I BDE = 71 kcal/mol)45. We therefore hypothesized a HAT process by bromine radical might be operative. Consistent with this hypothesis, we found that several representative olefins with very high oxidation potentials, cyclopentene (47), cyclohexene (48) and enol acetate (70) were suitable candidates for the cross-coupling.
Employing an E/Z mixture of silyl enol ether 71 gave similar results as in the case of the Z isomer only, indicating the geometry loss of the double bond during the course of the reaction. Interestingly, we found that the Z-configured starting material in the latter case was converted into an E/Z mixture following a careful inspection of the reaction crude mixture. In a control experiment without nickel catalyst under the standard condition, the Z isomer remained to a high degree (92%) intact (Fig. 6a). These experiments suggest the possibility that the nickel-allyl species was involved in the reaction. We then carried out the stoichiometric experiment of aryl-Ni(II) complex 73 (derived from 4-methylchlorobenzene and dtbbpy) with 1, no product of 13 was found (Fig. 6b). In comparison, the stoichiometric reaction of in-situ generated allylnickel(II) complex 74 produced the corresponding silyl enol ether product in moderate yields. More importantly, the stereoselectivity for product formation by choice of the ligand is consistent with the catalytic reaction, albeit in lower ratio (Fig. 6c). We also observed that the ratio of ligand to nickel could affect the stereoselectivity with dtbbpy ligand, and the nickel-allyl isomerization mechanism can be excluded given that the same E selectivity was obtained when the stoichiometric experiment was performed in the presence of Zn as reductant without light irradiation (see Supporting Information). Based on our mechanistic studies, a plausible mechanism is proposed (Fig. 6d). Upon irradiation with visible light, the Ir photocatalyst generates a long-lived triplet excited state, which should be able to oxidize the bromide anion to the active bromine radical. Subsequent hydrogen atom abstraction from the allylic position of alkene 1 yields the carbon-centered radical 1′. In line with previous reports, this allyl radical is readily trapped by the doublet Ni(I)Br (A) to produce the Ni(II)(allyl)Br intermediate B. Single-electron reduction of B by the reduced state of the photocatalyst leads to the Ni(I)(allyl) complex C, which undergoes oxidative addition of the aryl bromide to produce the corresponding Ni(III)(allyl)(aryl)Br complex D. The catalytic cycle ends with the facile reductive elimination of the desired cross-coupling product and concurrent regeneration of the active catalyst A.
In support of the above control experiments and our proposed reaction mechanism, we performed DFT calculations (see Computational Details in Supporting Information) to define the catalytic mechanism. Possible pathways of photocatalytic cycles are discussed in Supplementary Figs. 15 and 16. Our results show that the oxidative photoredox cycle is favored during the formation of active Ni-complex from the pre-catalyst NiBr2. On the other hand, the reductive cycle is favored in the case of the Ni-catalytic pathway.
We have investigated the Ni-catalytic cycle starting from LNiI-Br as the active catalyst46,47. The free-energy profile of the complete nickel catalytic pathway for the E-product (3) formation in the presence of dtbbpy ligand is shown in Fig. 7a, while the Z-product (4) pathway is shown in the Supplementary Fig. 17. In addition, both E- and Z-product formation pathways with 6,6’-dmbpy ligand are depicted in the Supplementary Figs. 18 and 19. The Ni-catalytic cycle starts with the radical addition RE to the nickel atom of 2A1 (Fig. 7a). The resulting 1A2E undergoes endergonic 1e− reduction by 2IrII to generate 2A3E and Br− (SET1, Fig. 7a). Further 1e− oxidation of Br− coupled with 3IrIII/2IrII reduction (SET2) leads to Br•, which is involved in the HAT step to generate allyl radical from 1. The reaction continues with the oxidative addition of 2 to the 2A3E via transition state 2[A3E-A4E]‡ and the energy barrier of 25.3 kcal/mol, leading to 2A4E. The Ni-cycle is completed by facile reductive elimination of the product from 2A4E via transition state 2[A4E-A1]‡ and an activation barrier of 5.4 kcal/mol, regenerating active catalyst 2A1 with the liberation of the E-product 3. Consistently with the experiments (Fig. 6c), calculations suggest that E–Z isomerization of the allyl moiety is possible at the LNi(II)(allyl)Br complex, 1A2E in Fig. 7a. Therefore, the E-/Z- product selectivity is occurring at this intermediate, depending on the relative feasibility between isomerization step and the corresponding SET.
a Free-energy profile of Ni-catalytic cycle in the presence of dtbbpy ligand for E-product (3) formation. The M06(SMD-THF)/def2-TZVPP//PBE/def2-SVP/def2-TZVP level of theory was used to calculate the reported values. b Free-energy profiles for isomerization of allyl group at LNi(II)(allyl)Br complex for dtbbpy ligand (left) and 6,6’-dmbpy ligand (right). For energy conventions, refer to Fig. 7a.
The allyl isomerization is expected to occur via a classic η3 → η1 → η3 mechanism involving C−C bond rotation after the formation of the η1 Ni–C–C=C species and dissociation of the localized C=C bond from Ni. However, in the singlet state this isomerization is highly disfavored for 1A2E and 1B2E, with free-energy barriers of 48.4 and 50.8 kcal/mol, respectively (Supplementary Figs. 21, 22). Importantly, under irradiation of light, promoting excitation of the complexes from singlet to a triplet state (1A2E/1B2E → 3A2E/3B2E), the coordination mode of the allyl group spontaneously changes from ɳ3 to promote one electron from a filled d-orbital of Ni to the empty Ni d-orbital involved in accepting the 4e of the ɳ3 coordinated allyl. The ɳ1-isomers of 3A2E and 3B2E undergo C−C bond rotation to generate the respective Z-isomer 3A2Z and 3B2Z at the triplet state (Fig. 7b). In the case of the dtbbpy ligand, the most stable ɳ1 isomer has the unsubstituted carbon of the allyl group σ-bonded to the Ni atom, which requires Z-isomerization occurring through rotation around the partial C−C(OTBS) double bond, with an energy barrier (ΔG‡ = 20.2 kcal/mol) higher than that of the 1e− reduction step, (ΔGMH‡ = 8.6 kcal/mol, SET1 in Fig. 7a). This indicates that formation of the Z-product is unfeasible. Differently, in the case of the 6,6′-dmbpy ligand, the most stable ɳ1 isomer has a well-defined Ni–C(OTBS) σ-bond. Consequently, E/Z isomerization occurs via rotation around a C–C single bond with a low free-energy barrier (ΔG‡ = 10.8 kcal/mol), and the corresponding Z-isomer (3B2Z) is favored energetically. This suggests a higher population of the Z-isomer over the E one prior to the 1e− reduction step (ΔGMH‡ = 20.5 kcal/mol, SET4 in Supplementary Fig. 18). Alternative mechanisms were explored for the E/Z isomerization with dtbbpy ligand. However, those are unfavored due to very high energy pathways (see Supplementary Fig. 19).
Comparison of the 3A2E and 3B2E geometries explains the different impact of the two ligands on the isomerization step (Fig. 7b). The reduced steric hindrance in 3A2E allows partial π coordination to nickel of the formally ɳ1 allyl group, with a relatively short C−C(OTBS) bond (1.379 Å), allowing rotation with the high energy barrier of Fig. 7b. Differently, due to steric substitutes at 6,6’ position in 3B2E, only one C atom of the ɳ1 allyl is interacting with nickel, allowing for low energy rotation (see Fig. 7b) around a C−C bond having a strong single bond character (1.477 Å, Fig. 8).
Although the cross-coupling of substrates bearing geminal dimethyl moiety has occurred with high levels of regio- and stereocontrol, we observed a small amount of product (2%) from diarylation. With this in mind, we next examined the feasibility of the direct use of a monoarylated product as the coupling partner. To our delight, the sequential diarylation is successfully applied with both electron-rich and electron-deficient aryl bromides to prepare diarylated silyl enol ethers (Fig. 9). Interestingly, our standard reaction conditions can achieve stereocontrol by discriminating substituent electronics. For instance, if the aryl ring of the substrate has an electron-donating substituent, introducing another one bearing an electron-withdrawing para substituent could give 76 in 51% yield and 85% E selectivity using L1. By switching to conditions with L2, the inverse configuration of the double bond can be obtained (77, 57% yield and 82% Z selectivity). This shows the potential for stereoselective alkene synthesis by exploiting the electronic property of the substrates.
a Condition A: the reactions were performed on a 0.2 mmol scale under the following reaction conditions: alkene (3 equiv.), aryl bromide (1 equiv.), 2,6-lutidine (1.5 equiv.), PC-1 (1 mol%), NiBr2 (10 mol%), dtbbpy (15 mol%), benzene (2 mL), rt, 60 h. b Condition B: the reaction was performed on a 0.2 mmol scale under the following reaction conditions: alkene (3 equiv.), aryl bromide (1 equiv.), 2,4,6-collidine (3 equiv.), PC-1 (1 mol%), NiBr2 (20 mol%), 6,6’-dmbpy (20 mol%), dioxane (2 mL), rt, 60 h. Yields and E/Z after purification by chromatography.
In conclusion, we have developed a general method that permits the regio- and stereoselective synthesis of alkenes. This protocol enables catalytic allylic radical generation directly from simple and readily available alkenes and accommodates a wide variety of aryl halide coupling partners. Different from previous mechanistic proposals on C(sp3)-H functionalization with photoredox nickel dual catalysis, our experimental evidence, and detailed computational mechanistic studies support a Ni(I)-allylNi(II)-allylNi(I)-Ni(III) catalytic cycle. Computational studies further explain the stereochemical outcome and indicate that excitation of a Ni(II)-allyl complex from singlet to a triplet state results in a spontaneous change of the allyl group coordination and that the subsequent ligand-dependent isomerization can be directed by choice of the ligand to achieve E/Z selectivity. These unexpected findings led us systematically investigate the stereoselectivity and to develop stereodivergent allylic C(sp3)-H bond arylations in which a ligand switch allows the target synthesis of E and Z isomers of silyl enol ethers.
Methods
General procedure E for the cross-coupling reaction towards E selectivity (condition A)
To a 15 mL vial equipped with a stir bar was added NiBr2 (4.4 mg, 20 µmol, 0.1 equiv.) and 4,4’-di-tert-butyl-2,2’-bipyridine (8.1 mg, 30 µmol, 0.15 equiv), Ir[dF(CF3)ppy]2(dtbbpy)PF6 (2.3 mg, 0.002 mmol, 1 mol%), aryl halide (0.2 mmol, 1 equiv.), 2,6-lutidine (32.1 mg (∼35 µL), 0.3 mmol, 1.5 equiv.) and alkene (0.6 mmol, 3 equiv.). A mixture of benzene (1 mL) and THF (1 mL) was added, then the vial was degassed and under stirring irradiated with the corresponding blue LEDs photoreactor.
General procedure F for the cross-coupling reaction towards Z selectivity (condition B)
To a 15 mL vial equipped with a stir bar was added NiBr2 (8.8 mg, 40 µmol, 0.2 equiv.) and 6,6′-dimethyl-2,2′-bipyridine (7.4 mg, 0.04 mmol, 20 mol%), Ir[dF(CF3)ppy]2(dtbbpy)PF6 (2.3 mg, 0.002 mmol, 1 mol%), aryl halide (0.2 mmol, 1 equiv.), 2,4,6-collidine (72.7 mg (∼80 µL), 0.6 mmol, 3.0 equiv.) and alkene (0.6 mmol, 3 equiv.). A mixture of dioxane (1 mL) and THF (1 mL) was added, then the vial was degassed and under stirring irradiated with the corresponding blue LEDs photoreactor.
Data availability
The authors declare that all data generated in this study are available within the article and the Supplementary Information.
References
Larock, R. C. Comprehensive Organic Transformations: A Guide to Functional Group Preparations. 2 edn, (VCH, 1989).
Wang, J. Stereoselective Alkene Synthesis. (Springer, Berlin, Heidelberg, 2012).
Negishi, E.-I. et al. Recent advances in efficient and selective synthesis of Di-, Tri-, and tetrasubstituted alkenes via Pd-catalyzed alkenylation−carbonyl olefination synergy. Acc. Chem. Res. 41, 1474–1485 (2008).
Dugave, C. & Demange, L. Cis−trans isomerization of organic molecules and biomolecules: implications and applications. Chem. Rev. 103, 2475–2532 (2003).
Massad, I. & Marek, I. Alkene isomerization through allylmetals as a strategic tool in stereoselective synthesis. ACS Catal. 10, 5793–5804 (2020).
Neveselý, T., Wienhold, M., Molloy, J. J. & Gilmour, R. Advances in the E → Z isomerization of alkenes using small molecule photocatalysts. Chem. Rev. 122, 2650–2694 (2022).
Singh, K., Staig, S. J. & Weaver, J. D. Facile synthesis of Z-alkenes via uphill catalysis. J. Am. Chem. Soc. 136, 5275–5278 (2014).
Song, F. et al. Visible-light-enabled stereodivergent synthesis of E- and Z-configured 1,4-dienes by photoredox/nickel dual catalysis. Angew. Chem. Int. Ed. 59, 177–181 (2020).
Molloy, J. J. et al. Boron-enabled geometric isomerization of alkenes via selective energy-transfer catalysis. Science 369, 302–306 (2020).
Hu, X.-Q., Chen, J.-R. & Xiao, W.-J. Controllable remote C−H Bond Functionalization by Visible-light Photocatalysis. Angew. Chem. Int. Ed. 56, 1960–1962 (2017).
Wang, C.-S., Dixneuf, P. H. & Soulé, J.-F. Photoredox catalysis for building c–c bonds from C(sp2)–H bonds. Chem. Rev. 118, 7532–7585 (2018).
Chen, Y., Lu, L.-Q., Yu, D.-G., Zhu, C.-J. & Xiao, W.-J. Visible light-driven organic photochemical synthesis in China. Sci. China Chem. 62, 24–57 (2019).
Shu, C., Noble, A. & Aggarwal, V. K. Metal-free photoinduced C(sp3)–H borylation of alkanes. Nature 586, 714–719 (2020).
Guo, W., Wang, Q. & Zhu, J. Visible light photoredox-catalysed remote C–H functionalisation enabled by 1,5-hydrogen atom transfer (1,5-HAT). Chem. Soc. Rev. 50, 7359–7377 (2021).
Sarkar, S., Cheung, K. P. S. & Gevorgyan, V. C–H functionalization reactions enabled by hydrogen atom transfer to carbon-centered radicals. Chem. Sci. 11, 12974–12993 (2020).
Tellis, J. C. et al. Single-electron transmetalation via photoredox/nickel dual catalysis: unlocking a new paradigm for sp3–sp2 cross-coupling. Acc. Chem. Res. 49, 1429–1439 (2016).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Leifert, D. & Studer, A. The persistent radical effect in organic synthesis. Angew. Chem. Int. Ed. 59, 74–108 (2020).
Zuo, Z. et al. Merging photoredox with nickel catalysis: Coupling of α-carboxyl sp3-carbons with aryl halides. Science 345, 437–440 (2014).
Joe, C. L. & Doyle, A. G. Direct acylation of C(sp3)−H bonds enabled by nickel and photoredox catalysis. Angew. Chem. Int. Ed. 55, 4040–4043 (2016).
Shaw, M. H., Shurtleff, V. W., Terrett, J. A., Cuthbertson, J. D. & MacMillan, D. W. C. Native functionality in triple catalytic cross-coupling: sp3 C–H bonds as latent nucleophiles. Science 352, 1304–1308 (2016).
Heitz, D. R., Tellis, J. C. & Molander, G. A. Photochemical nickel-catalyzed C–H arylation: synthetic scope and mechanistic investigations. J. Am. Chem. Soc. 138, 12715–12718 (2016).
Deng, H.-P., Fan, X.-Z., Chen, Z.-H., Xu, Q.-H. & Wu, J. Photoinduced nickel-catalyzed chemo- and regioselective hydroalkylation of internal alkynes with ether and amide α-hetero C(sp3)–H bonds. J. Am. Chem. Soc. 139, 13579–13584 (2017).
Shen, Y., Gu, Y. & Martin, R. sp3 C–H arylation and alkylation enabled by the synergy of triplet excited ketones and nickel catalysts. J. Am. Chem. Soc. 140, 12200–12209 (2018).
Zhang, L. et al. The combination of benzaldehyde and nickel-catalyzed photoredox C(sp3)−H alkylation/arylation. Angew. Chem. Int. Ed. 58, 1823–1827 (2019).
Kariofillis, S. K. & Doyle, A. G. Synthetic and mechanistic implications of chlorine photoelimination in nickel/photoredox C(sp3)–H cross-coupling. Acc. Chem. Res. 54, 988–1000 (2021).
Perry, I. B. et al. Direct arylation of strong aliphatic C–H bonds. Nature 560, 70–75 (2018).
Huang, H.-M., Bellotti, P., Chen, P.-P., Houk, K. N. & Glorius, F. Allylic C(sp3)–H arylation of olefins via ternary catalysis. Nat. Synth. 1, 59–68 (2022).
Buendia, M. B., Higginson, B., Kegnæs, S., Kramer, S. & Martin, R. Redox-neutral Ni-catalyzed sp3 C–H alkylation of α-olefins with unactivated alkyl bromides. ACS Catal. 12, 3815–3820 (2022).
Kancherla, R. et al. Mechanistic insights into photochemical nickel-catalyzed cross-couplings enabled by energy transfer. Nat. Commun. 13, 2737 (2022).
Maity, B., Scott, T. R., Stroscio, G. D., Gagliardi, L. & Cavallo, L. The role of excited states of LNiII/III(aryl)(halide) complexes in Ni–halide bond homolysis in the arylation of Csp3–H bonds. ACS Catal. 21, 13215–13224 (2022).
Crespi, S. & Fagnoni, M. Generation of alkyl radicals: from the tyranny of tin to the photon democracy. Chem. Rev. 120, 9790–9833 (2020).
Zhu, S., Zhao, X., Li, H. & Chu, L. Catalytic three-component dicarbofunctionalization reactions involving radical capture by nickel. Chem. Soc. Rev. 50, 10836–10856 (2021).
Cheng, X., Lu, H. & Lu, Z. Enantioselective benzylic C–H arylation via photoredox and nickel dual catalysis. Nat. Commun. 10, 3549 (2019).
Shu, X., Huan, L., Huang, Q. & Huo, H. Direct enantioselective C(sp3)–H acylation for the synthesis of α-amino ketones. J. Am. Chem. Soc. 142, 19058–19064 (2020).
Xu, J., Li, Z., Xu, Y., Shu, X. & Huo, H. Stereodivergent synthesis of both Z- and E-alkenes by photoinduced, Ni-catalyzed enantioselective C(sp3)–H alkenylation. ACS Catal. 11, 13567–13574 (2021).
Huang, L. & Rueping, M. Direct cross-coupling of allylic C(sp3)−H bonds with aryl- and vinylbromides by combined nickel and visible-light catalysis. Angew. Chem. Int. Ed. 57, 10333–10337 (2018).
Jia, J., Kancherla, R., Rueping, M. & Huang, L. Allylic C(sp3)–H alkylation via synergistic organo- and photoredox catalyzed radical addition to imines. Chem. Sci. 11, 4954–4959 (2020).
Zhao, C.-G., Xia, S., Wang, C., Wang, W. & Xie, J. Opportunities and challenges of visible-light-driven triple-synergistic catalysis. Chem. Catal. 2, 458–467 (2022).
Liu, K. & Studer, A. Formal β-C−H arylation of aldehydes and ketones by cooperative nickel and photoredox catalysis. Angew. Chem. Int. Ed. 61, e202206533 (2022).
He, Y., Cai, Y. & Zhu, S. Mild and regioselective benzylic C–H functionalization: Ni-catalyzed reductive arylation of remote and proximal olefins. J. Am. Chem. Soc. 139, 1061–1064 (2017).
Peng, L. et al. Ligand-controlled nickel-catalyzed reductive relay cross-coupling of alkyl bromides and aryl bromides. ACS Catal. 8, 310–313 (2018).
Jiao, K.-J. et al. Nickel-catalyzed electrochemical reductive relay cross-coupling of alkyl halides to aryl halides. Angew. Chem. Int. Ed. 59, 6520–6524 (2020).
Kumar, G. S. et al. Nickel-catalyzed chain-walking cross-electrophile coupling of alkyl and aryl halides and olefin hydroarylation enabled by electrochemical reduction. Angew. Chem. Int. Ed. 59, 6513–6519 (2020).
Blanksby, S. J. & Ellison, G. B. Bond dissociation energies of organic molecules. Acc. Chem. Res. 36, 255–263 (2003).
Zhu, C. et al. A multicomponent synthesis of stereodefined olefins via nickel catalysis and single electron/triplet energy transfer. Nat. Catal. 2, 678–687 (2019).
Maity, B. et al. Mechanistic Insight into the photoredox-nickel-HAT triple catalyzed arylation and alkylation of α-amino Csp3–H bonds. J. Am. Chem. Soc. 142, 16942–16952 (2020).
Acknowledgements
B.M. and L.C. acknowledge King Abdullah University of Science and Technology (KAUST) for support and the KAUST Supercomputing Laboratory for providing computational resources of the supercomputer Shaheen II.
Funding
Open Access funding enabled and organized by Projekt DEAL..
Author information
Authors and Affiliations
Contributions
L.H. and M.R. conceived and designed the project. L.H. performed and analyzed the experiments. R.K. and M.S. contributed to the scope and mechanism. B.M. and C.Z. performed the density functional theory calculations. L.H., B.M., and M.R. co-wrote the manuscript with contributions from all authors. M.R. and L.C. directed the whole research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Haohua Huo, and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, L., Szewczyk, M., Kancherla, R. et al. Modulating stereoselectivity in allylic C(sp3)-H bond arylations via nickel and photoredox catalysis. Nat Commun 14, 548 (2023). https://doi.org/10.1038/s41467-023-36103-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-36103-0
This article is cited by
-
Metallaphotoredox catalysis for sp3 C–H functionalizations through single-electron transfer
Nature Catalysis (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.