Abstract
Pulmonary endothelial progenitor cells (EPCs) are critical for neonatal lung angiogenesis and represent a subset of general capillary cells (gCAPs). Molecular mechanisms through which EPCs stimulate lung angiogenesis are unknown. Herein, we used single-cell RNA sequencing to identify the BMP9/ACVRL1/SMAD1 pathway signature in pulmonary EPCs. BMP9 receptor, ACVRL1, and its downstream target genes were inhibited in EPCs from Foxf1WT/S52F mutant mice, a model of alveolar capillary dysplasia with misalignment of pulmonary veins (ACDMPV). Expression of ACVRL1 and its targets were reduced in lungs of ACDMPV subjects. Inhibition of FOXF1 transcription factor reduced BMP9/ACVRL1 signaling and decreased angiogenesis in vitro. FOXF1 synergized with ETS transcription factor FLI1 to activate ACVRL1 promoter. Nanoparticle-mediated silencing of ACVRL1 in newborn mice decreased neonatal lung angiogenesis and alveolarization. Treatment with BMP9 restored lung angiogenesis and alveolarization in ACVRL1-deficient and Foxf1WT/S52F mice. Altogether, EPCs promote neonatal lung angiogenesis and alveolarization through FOXF1-mediated activation of BMP9/ACVRL1 signaling.
Similar content being viewed by others
Introduction
Lung morphogenesis is a highly conserved developmental process, which requires specific signaling events and cell-to-cell interactions among a diversity of cells derived from endodermal, ectodermal, and mesodermal progenitors1,2. Formation of alveolus, a gas-exchange unit mediating transport of gases between inhaled air and the blood, occurs prior to and after birth, when respiratory epithelial progenitors differentiate into alveolar type I (AT1) and AT2 cells which are located in close proximity to capillary endothelial cells1,3. Defects in formation of alveoli and pulmonary capillaries are associated with severe pediatric lung diseases, such as Bronchopulmonary Dysplasia (BPD) and Alveolar Capillary Dysplasia with Misalignment of Pulmonary Veins (ACDMPV)4,5,6,7. Recent single cell RNA sequencing of human and mouse neonatal lungs identified two types of alveolar endothelial cells: general capillary cells (gCAPs) and alveolar capillary cells (aCAPs or aerocytes)8. aCAPs express APLN and CAR4, form a majority of alveolar gas-exchange surface area, and require signaling through Vascular Endothelial Growth Factor (VEGF) produced by AT1 cells8,9,10. gCAPs express APLNR and consist of several cell types, including mature gCAPs, cells of the embryonic vascular plexus and other endothelial cells with progenitor properties that are critical for pulmonary vascular development and lung repair after injury8,11. Endothelial progenitor cells (EPCs) expressing FOXF1 transcription factor and cKIT receptor (FOXF1+ EPCs) exhibit high colony-forming capacity and are capable of engraftment into the neonatal lung tissue of ACDMPV mice to form alveolar capillaries11,12. While EPCs hold promise for cell therapies in ACDMPV and other pediatric pulmonary disorders13, molecular mechanisms through which EPCs stimulate neonatal lung angiogenesis are unknown.
FOXF1 is a member of Forkhead Box (FOX) family of transcription factors, which is required for formation of pulmonary capillaries in mice and humans1,3. Heterozygous loss-of-function mutations in FOXF1 are linked to ACDMPV, a severe congenital disorder which is characterized by diminished lung angiogenesis leading to scarcity of the alveolar capillary network4. ACDMPV causes respiratory distress and mortality within the first month of birth in spite of intensive care4,5,14. Global deletion of the Foxf1 gene in mice is embryonic lethal15, whereas haploinsufficiency of Foxf1 (Foxf1+/-) or heterozygous S52F Foxf1 knock-in mutation (Foxf1WT/S52F) disrupts development of alveolar capillaries, recapitulating human ACDMPV14,16. FOXF1 stimulates lung angiogenesis and vascular repair after injury by accelerating endothelial proliferation while maintaining endothelial cell junctions to prevent hemorrhage17,18. FOXF1 regulates expression of genes critical for VEGF, PDGF, NOTCH and TIE2 signaling pathways during lung development and regeneration19,20,21,22. While genetic and histological evidence strongly suggest that endothelial cells play a key role in ACDMPV pathogenesis4,14, endothelial cell diversity and key signaling pathways in ACDMPV remain unclear.
The TGFβ/BMP signaling pathway has been implicated in vascular development. Abnormalities in TGFβ/BMP signaling in endothelium lead to developmental defects in the vasculature23. Hereditary hemorrhagic telangiectasia (HHT) is an autosomal dominant disorder which is associated with severe abnormalities in the microvascular network and linked to mutations in ACVRL1 and ENG genes24,25,26. ACVRL1 forms a receptor complex with BMPR2 and ENG to mediate TGFβ/BMP signaling in endothelial cells through phosphorylation of SMAD1/5/8. BMP9, a high-affinity ligand for the ACVRL1/BMPR2/ENG receptor27, is produced by hepatocytes and delivered by the blood to other organs28. BMP9 promotes proliferation of endothelial cells in vitro and in vivo27, and ameliorates pulmonary arterial hypertension (PAH) through activation of endothelial BMPR2 signaling29. While circulating BMP9 prevents alveolar simplification after neonatal hyperoxia30 and protects pulmonary endothelium after LPS-mediated lung injury31, it is unclear which endothelial cell lineages require BMP9 signaling to promote lung repair and regeneration.
In the present study, we used single cell RNA sequencing to identify a genomic signature of the BMP9/ACVRL1/SMAD1signaling pathway in pulmonary FOXF1+ EPCs. We also used a mouse model of ACDMPV to demonstrate that FOXF1+ EPCs stimulate neonatal lung angiogenesis through FOXF1-mediated activation of BMP9/ACVRL1 signaling.
Results
The S52F Foxf1 mutation alters cellular composition of the mouse lung prior to birth
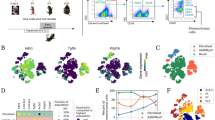
We previously generated Foxf1WT/S52F mice in which the evolutionary conserved serine 52 in the FOXF1 DNA binding domain was replaced with phenylalanine in one of the Foxf1 alleles, recapitulating the S52F FOXF1 mutation found in ACDMPV patients14. Since Foxf1WT/S52F mice developed alveolar capillary dysplasia phenotypically similar to human ACDMPV14, we used single cell RNA sequencing (scRNAseq) of lung tissue to identify molecular mechanisms through which the S52F FOXF1 mutation causes ACDMPV. Single cell transcriptomes from lungs of Foxf1WT/S52F and control WT E18.5 embryos were compared. Since FOXF1 is not expressed in hematopoietic cell lineages1,19, CD45+ cells were depleted from lung cell suspensions using immunomagnetic beads. Efficient depletion of CD45+ cells was confirmed by FACS analysis (Supplementary Fig. 1a, b). Cells from three embryos were combined in each group prior to single cell RNA sequencing. After confirming RNA quality and filtering outlier cells and cell doublets at both cell and feature levels (Supplementary Fig. 1c), 7559 cells from Foxf1WT/S52F and 5911 cells from WT lungs were subjected to bioinformatic analysis using the anchor-based integrative pipeline from Seurat 3.032. Twelve major cell clusters were identified by unsupervised clustering (Fig. 1a, b). These include epithelial AT1, AT2, AT1/AT2, ciliated and club cells, endothelial cells and six mesenchymal cell clusters: fibroblasts 1 (FB-1), FB-2, pericytes, matrix FB-1, matrix FB-2 and myofibroblasts. The integrated cell clustering of E18.5 mouse lungs was performed using cell-specific gene signatures of each cluster (Supplementary Figs. 1d, e and 2a–d) that were identified in published studies1,33,34. Foxf1 mRNA was detected in endothelial cells, FB-1, pericytes and myofibroblasts, but not in Nkx2-1-expressing epithelial cells (Fig. 1c and Supplementary Fig. 2e).
a The integrated projection of CD45-depleted pulmonary cells from WT and Foxf1WT/S52F lungs. Lungs were harvested from E18.5 embryos and enzymatically digested to obtain single cell suspensions. CD45-positive cells were depleted using immunomagnetic beads. Three embryos from each genotype were pooled together prior to the scRNAseq analysis. 12 cell clusters were identified using the Uniform Manifold Approximation and Projection (UMAP) clustering. b A K-nearest neighbor (KNN) graph-based clustering approach was used to identify similar cell clusters in WT (n = 5911 cells) and Foxf1WT/S52F lungs (n = 7559 cells). c Violin plots show the presence of Foxf1 mRNA in endothelial cells, fibroblast-1, pericytes and myofibroblasts based on expression of selective cell markers Cdh5, Pdgfra and Cspg4. d Proportions of cells in each of the 12 clusters are compared in WT and Foxf1WT/S52F lungs. Percentages of endothelial and AT1 cells are decreased in Foxf1WT/S52F lungs. The percentage of matrix FB2 is increased in Foxf1WT/S52F lungs compared to lungs of WT littermates. e Linear regression analysis shows changes in distribution of endothelial, AT1 and matrix FB2 clusters between WT and Foxf1WT/S52F lungs.
While all twelve major cell clusters were present in Foxf1WT/S52F lungs, cellular composition of the mutant lung was changed compared to WT controls (Fig. 1d). Percentages of endothelial and AT1 cells were decreased, whereas the percentage of matrix FB-2 was increased in Foxf1WT/S52F lungs compared to lungs of WT littermates (Fig. 1d). These findings were supported by the linear regression analysis of cell distribution across the clusters (Fig. 1e), and by immunostaining of lung tissue sections using antibodies specific to ERG, HOPX and FN1 (Supplementary Fig. 3). Thus, the S52F Foxf1 mutation alters cellular composition in the mouse lung prior to birth. Since neither AT1 nor matrix FB-2 cells express Foxf1 (Fig. 1c) and gene expression signatures in these cell types were similar between Foxf1WT/S52F and WT lungs (Supplementary Fig. 4a–d), we focused our next studies on pulmonary endothelial cells.
The S52F Foxf1 mutation disrupts ACVRL1 signaling in gCAPs
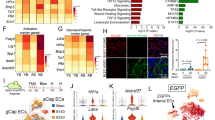
Consistent with recently described gene signatures of pulmonary endothelial cell lineages8,35, 320 endothelial cells from Foxf1WT/S52F and 532 endothelial cells from WT lungs were subdivided into 5 sub-clusters (Fig. 2a, and Supplementary Fig. 5a, b). These included arterial, venous, lymphatic, general capillary cells (gCAPs) and aerocytes (aCAPs). Foxf1 mRNA was mainly expressed in gCAPs and aCAPs (Fig. 2c, d). Foxf1-expressing cells were also detected among arterial and venous clusters but were absent in the lymphatics (Fig. 2c).
a Dimensional reduction plot with UMAP shows five subclusters of pulmonary endothelial cells in E18.5 lungs. Lungs were harvested from E18.5 mouse embryos, enzymatically digested and then incubated with CD45+ immunobeads to deplete hematopoietic cells. Three embryos from each genotype were pooled together prior to the scRNAseq analysis. b Split dimensional reduction plots of pulmonary endothelial cells from WT (n = 532 cells) and Foxf1WT/S52F embryos (n = 320 cells). c, d Violin and scatter plots show an enrichment of Foxf1 mRNA in pulmonary capillary cells. Foxf1 expression is selectively decreased in gCAPs but not aCAPs from Foxf1WT/S52F embryos. T-test (two-tailed) analyses were performed, and pvalue for Foxf1 is 0.01257. *p < 0.05, p > 0.05 is ns. e Heatmap shows representative gCAP genes, expression of which is decreased in Foxf1WT/S52F embryos compared to WT embryos. f Analysis of signaling pathways demonstrates that gCAPs of Foxf1WT/S52F embryos exhibit decreased expression of genes associated with the TGFβ/BMP signaling pathway, sphingolipid metabolism, endocytosis, Rap1 and PPAR pathways. Fisher’s Exact (one-sided) test was used via DAVID bioinformatic resource to measure gene-enrichment p-values. g Violin plots show decreased expression of Acvrl1 in gCAPs but not aCAPs. Expression of Bmpr2 and Eng is unchanged T-test (two-tailed) analyses were performed, and p-value for Acvrl1 is 0.01993. p < 0.05 is *p > 0.05 is ns. h, i Violin plots show decreased expression of downstream target genes of the TGFβ/BMP signaling pathway in pulmonary gCAPs, including Tmem100, Calcrl, Clec14a, Id1 and Id3. T-test (two-tailed) analyses were performed, and pvalue for Tmem100 is 0.01688, for Calcrl is 0.02132, for Clec14a is 0.0213, for Id1 is 0.03623, for Id3 is 0.037811. *p < 0.05, p > 0.05 is ns. The data for box plots in d and g–i were retrieved from single cell normalized counts (WT: n = 532; Foxf1WT/S52F: n = 320) Boxplots: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile. Expression of these genes in pulmonary aCAPs was unchanged. ns is not significant.
Foxf1 mRNA was selectively decreased in Foxf1WT/S52F gCAPs but not in other endothelial cell types (Fig. 2d). Using the likelihood-ratio test, we generated a heatmap and identified gene expression differences between gCAPs from Foxf1WT/S52F and WT lungs. Expression of 93 genes was decreased in Foxf1WT/S52F gCAPs compared to controls (Fig. 2e and Supplementary Table 1), whereas expression of 49 genes was increased (Supplementary Table 2). Bioinformatic analysis identified several functional categories that were impaired in Foxf1WT/S52F gCAPs (Fig. 2f). Expression of genes critical for the TGFβ/BMP signaling pathway was decreased in Foxf1WT/S52F gCAPs compared to control (Fig. 2f and Supplementary Table 3). A systematic profiling of mRNAs encoding TGFβ/BMP receptors among pulmonary cell types showed that Acvrl1 (also known as ALK1) and Bmpr2 are the main TGFβ type I and type II receptors in endothelial cells (Supplementary Fig. 6a, b). Decreased expression of Foxf1 in Foxf1WT/S52F gCAPs (Fig. 2d) was associated with reduced Acvrl1 mRNA (Fig. 2g). Neither Foxf1 nor Acvrl1 mRNAs were changed in aCAPs of Foxf1WT/S52F mice (Fig. 2d–g). We also used scRNAseq to examine expression of downstream target genes of the ACVRL1 signaling pathway, such as Tmem100, Calcrl, Clec14a, Id1 and Id326,36,37. mRNAs of these genes were enriched in pulmonary endothelial cells compared to other cell types (Supplementary Fig. 6a). Expression of Tmem100, Calcrl, Clec14a, Id1 and Id3 was decreased in Foxf1WT/S52F gCAPs compared to gCAPs from WT lungs (Fig. 2h, i), a finding consistent with impaired ACVRL1 signaling. Decreased expression of Acvrl1 and Tmem100 in Foxf1WT/S52F gCAPs was also confirmed by RNAscope (Supplementary Fig. 7). In contrast, expression of ACVRL1 target genes was unchanged in aCAPs (Fig. 2h, i). The Foxf1WT/S52F mutation did not alter Bmpr2 and Eng mRNAs in either gCAPs or aCAPs (Fig. 2g).
Single cell RNAseq and RNAscope identify heterogeneity of pulmonary gCAPs
Based on scRNAseq analysis of murine WT E18.5 lungs, Aplnr-expressing gCAPs consisted of two distinct cell subsets: FOXF1– gCAPs and FOXF1+ gCAPs (Fig. 3a, b). Both gCAP subsets resided in the alveolar region as demonstrated by RNAscope using riboprobes specific to Aplnr and Foxf1 (Fig. 3c). FOXF1+ gCAPs expressed Kit (Fig. 3b) and were similar to recently described FOXF1+cKIT+ lung endothelial progenitor cells (FOXF1+ EPCs)11. The presence of Mki67 in FOXF1+ EPCs (Fig. 3b and Supplementary Table 4) indicates that these cells are highly proliferative. In contrast, FOXF1– gCAPs did not expressed Mki67 but were positive for Adgrg6 and Fbln5 (Fig. 3b and Supplementary Table 5).
a, b Dimensional reduction plot with UMAP was generated using scRNAseq dataset from WT E18.5 lungs and shows gCAP and aCAP clusters of microvascular endothelial cells. Three embryos were pooled together prior to the scRNAseq analysis. gCAPs consist of two distinct subclusters: FOXF1+ gCAPs and FOXF1– gCAPs. c RNAscope of lung sections from WT E18.5 embryos shows APLNR-expressing gCAPs in the alveolar region. High magnification images of area in yellow box are shown on the right. Lung sections were counterstained with DAPI. APLNR co-localizes with FOXF1 in FOXF1+ gCAPs (red arrowheads). FOXF1 is not expressed in FOXF1– gCAPs (yellow arrowheads). The RNAscope assays were performed twice with consistent results. Scale bars are 10 μm.
FOXF1 deficiency disrupts ACVRL1 signaling in mouse and human lungs. We used FACS analysis and fluorescent microscopy to confirm that ACVRL1 and ENG proteins are co-expressed in pulmonary endothelial cells (Supplementary Fig. 8a, b). Using cell sorting, FOXF1+ EPCs (FOXF1+cKIT+CD31+CD45–) were purified from lungs of Foxf1WT-GFP/S52F E18.5 reporter embryos (Supplementary Fig. 9a), in which the first Foxf1 allele contains the S52F Foxf1 mutation and the second Foxf1 allele produces two separate WT-Foxf1 and GFP reporter transcripts11. Expression of Foxf1, Acvrl1 and its downstream target genes was lower in FACS-sorted Foxf1WT-GFP/S52F EPCs compared to EPCs from Foxf1WT-GFP/+ controls (Fig. 4a, b and Supplementary Fig. 9b). In contrast, expression of Bmpr2, Eng and other TGFβ/BMP receptors was unchanged (Supplementary Fig. 9b, c). ACVRL1 staining was decreased in Foxf1WT/S52F lungs (Fig. 4c). Furthermore, Foxf1WT-GFP/S52F EPCs exhibited decreased cell surface expression of ACVRL1 (Fig. 4d, e) and reduced numbers of EPCs co-expressing ACVRL1 and ENG (ACVRL1+ENG+FOXF1+cKIT+CD31+CD45–) (Fig. 4f). Finally, we used publicly available RNAseq datasets from lungs of patients with ACDMPV38 to examine expression of genes critical for the ACVRL1 signaling pathway. mRNAs of ACVRL1 and its target gene CALCRL were decreased in ACDMPV lungs (n = 8) compared to donor lungs (n = 5) (Fig. 4g, h). Consistent with murine Foxf1WT/S52F datasets, mRNAs of BMPR2, ENG and other TGFβ/BMP receptors were unchanged in human ACDMPV lungs (Fig. 4g, h and Supplementary Fig. 10a, b). Altogether, FOXF1 deficiency decreases ACVRL1 and expression of its downstream target genes in human and mouse lungs.
a Foxf1 and Acvrl1 mRNAs are decreased in FACS-sorted gCAPs of Foxf1WT-GFP/S52F embryos. FOXF1+ gCAPs (FOXF1+cKIT+CD31+CD45–) were purified from enzymatically digested E18.5 mouse lungs (n = 4 embryos in each group). FOXF1-positive gCAPs were identified using the Foxf1-GFP transgene, which contains GFP knocked into the endogenous Foxf1 gene locus. Foxf1WT-GFP/+ embryos were used as controls. Data were shown as mean ± SD. Nonparametric Mann-Whitney U test were performed, and p value for Foxf1 is 0.00022, for Acvrl1 is 0.00015. b Expression of ACVRL1 downstream target genes is decreased in FACS-sorted gCAPs of Foxf1WT-GFP/S52F embryos (n = 4 embryos in each group). Data were shown as mean ± SD, and nonparametric Mann-Whitney U test were performed were performed, and p value for Tmem100 is 0.00009, for Calcrl is 0.00098, for Clec14a is 0.00011. c Immunostaining of E18.5 lungs shows reduced expression of ACVRL1 protein in Foxf1WT/S52F lungs compared to WT controls (n = 6). Scale bars are 10 μm. Data were presented as mean ± SD, and T-test (two-tailed) were performed, p value = 0.01293. d The percentage of ACVRL1+ENG+ cells among pulmonary FOXF1+ gCAPs is decreased in Foxf1WT-GFP/S52F E18.5 embryos. e Measurements of mean fluorescence intensity (MFI) by FACS analysis show decreased ACVRL1 cell surface expression in FOXF1+ gCAPs of Foxf1WT-GFP/S52F embryos compared to Foxf1WT-GFP/+ controls. (n = 6 mice in each group) Boxplots: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile, p value = 0.00298 f Percentages of ACVRL1+ENG+ gCAPs are lower in Foxf1WT-GFP/S52F mice at E18.5 and P7 (n = 6 mice in each group). Data were presented as mean ± SD. T-test (two-tailed) were performed, p value for E18.5 is 6.20943E-07, and p value for P7 is 3.38604E-06. Source data are provided as a Source Data file. g, h, Microarray analysis shows decreased expression of FOXF1, ACVRL1 and CALCRL mRNAs in human ACDMPV lungs (n = 8) compared to donor lungs (n = 5). BMPR2 and ENG mRNAs were unaltered. Boxplots: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile, and student’s T-test (two-tailed) were performed. **p < 0.01, *p < 0.05 is, ns is not significant.
FOXF1 transcriptionally stimulates Acvrl1 through an evolutionarily conserved ACE80 promoter region
Next, we determined whether Acvrl1 is a direct transcriptional target of FOXF1. Based on publicly available FOXF1 ChIPseq datasets from murine E18.5 lungs39, FOXF1 directly bound to DNA in the Acvrl1 promoter region (Supplementary Fig. 11a, b). Using a comparative genomic analysis of eutherian mammals, we identified an evolutionarily conserved 400 bp DNA region located in the Acvrl1 gene (ACE400; −675/−275 bp) (Fig. 5a, b and Supplementary Fig. 11a, b). The −675/−275 bp Acvrl1 promoter region was amplified from mouse genomic DNA and cloned into the luciferase (LUC) reporter plasmid. In co-transfection experiments with fetal lung endothelial MFLM-91U cells, CMV-FOXF1 expression vector increased LUC activity driven by the ACE400 promoter region (Fig. 5c). Thus, FOXF1 directly activates the ACE400 Acvrl1 promoter region in endothelial cells. Interestingly, the ACE400 region contains two FOXF1-binding sites within the 80 bp sub-fragment (ACE80; −538/−458 bp) (Supplementary Fig. 11c). The ACE80 DNA region of the Acvrl1 promoter is evolutionarily conserved between mammals and birds (Fig. 5d, e and Supplementary Fig. 12). CMV-FOXF1 expression vector increased LUC activity driven by the ACE80 promoter region, whereas CMV vector with S52F FOXF1 mutant was transcriptionally inactive (Fig. 5f). Site-directed mutagenesis was used to inactivate both FOXF1-binding sites in the ACE80 region and generate the ACE80 mutant LUC construct (Fig. 4g). Inactivation of FOXF1-binding sites decreased the ability of CMV-FOXF1 vector to stimulate transcriptional activity of the ACE80 promoter region (Fig. 5f). Taken together, FOXF1 transcriptionally activates the Acvrl1 gene through FOXF1-binding sites located in the evolutionarily conserved –538/−458 bp Acvrl1 promoter region.
a, b Schematic diagram of the mouse Acvrl1 gene locus which contains the evolutionarily conserved −675/−275 promoter region (ACE400). Light blue boxes show untranslated DNA regions. Dark blue boxes indicate exons in the Acvrl1 gene. Two FOXF1-binding sites are indicated by green boxes. c Dual luciferase assay shows that CMV-FOXF1 expression plasmid stimulates the ACE400 activity in co-transfection experiments in MFLM-91U cells (n = 6). Data were shown as mean ± SD. T-test (two-tailed) were performed, pvalue for ACE is 0.00000158. p > 0.05 is not significant. CMV-empty expression plasmid (pCMV) and an empty LUC plasmid (vector) were used as controls. d Schematic shows the evolutionary conservation of ACE80 promoter region in amniote vertebrates. Amniotes were classified using UCSC genome browser into five groups: primates (orange), Afrotheria mammals (green), Laurasiatheria mammals (purple), Euarchontoglires mammals (blue) and birds (grey). e Schematic shows sequences of two FOXF1-binding sites in ACE80 region in several species. f Dual luciferase assay shows that CMV-FOXF1 expression vector stimulates the ACE80 activity in co-transfection experiments in MFLM-91U cells (n = 3). Data were shown as mean ± SD. The data were analyzed by one-way ANOVA followed by Tukey’s test (two-tailed). CMV-empty vector (CMV) and transcriptionally inactive S52F FOXF1 mutant plasmid (CMV- S52F-FOXF1) are used as controls. LUC activity is decreased after co-transfection of CMV-FOXF1 plasmid with ACE80mt LUC reporter. **p < 0.01, *p < 0.05, ns is not significant. g Location of FOXF1-binding sites in ACE80 region is indicated by green boxes. FOXF1-binding sites in ACE80mt construct were inactivated by disrupting the FOX core motif.
FOXF1 synergizes with ETS transcription factor FLI1 to stimulate Acvrl1 promoter activity
Since both aCAPs and gCAPs express FOXF1 but Acvrl1 mRNA was only reduced in Foxf1WT/S52F gCAPs (Fig. 2c–g), we tested the possibility that regulation of Acvrl1 promoter requires other transcription factors in addition to FOXF1. Comparative genomic analysis identified two conserved ETS binding sites near FOXF1 sites in the −538/−458 bp Acvrl1 promoter region (Supplementary Fig. 13a, c). Based on scRNAseq data from WT E18.5 lungs, endothelial cells expressed several ETS transcription factors, including Fli1, Erg and Ets1 (Fig. 6a). Fli1 mRNA was enriched in gCAPs compared to aCAPs (Fig. 6b). Both FOXF1– and FOXF1+ subsets of gCAPs expressed high levels of Fli1 (Supplementary Fig. 13d). Erg was enriched in aCAPs (Fig. 6b) and FOXF1– gCAPs (Supplementary Fig. 13d), whereas Ets1 expression was high in both aCAPs and gCAPs (Fig. 6b). Next, we performed co-transfection experiments, testing whether FOXF1 synergizes with FLI1, ERG and ETS1 to regulate Acvrl1 promoter activity in fetal lung endothelial MFLM-91U cells. ETS transcription factor SPI1, which is not expressed in lung endothelial cells (Fig. 6a, b and Supplementary Fig. 13d) was also used in these experiments. FOXF1 synergized with FLI1 but not with other ETS transcription factors to stimulate Acvrl1 promoter activity in vitro (Fig. 6c). Endogenous FLI1 and FOXF1 proteins directly bound to DNA in the −538/−458 bp Acvrl1 promoter region as demonstrated by ChIPseq (Fig. 6d). The S52F FOXF1 mutant, which is deficient in DNA binding14, did not synergize with FLI1 to activate the Acvrl1 promoter (Fig. 6c). Thus, FOXF1 synergizes with ETS transcription factor FLI1 to stimulate Acvrl1 promoter activity in FOXF1+ EPCs. Interestingly, FOXF1 and ETS binding sites were also found in the −1170/−1118 bp Foxf1 promoter region (FARE350), which is conserved in mammals (Supplementary Fig. 14a–c). The −1170/−1118 bp Foxf1 promoter region was amplified from mouse genomic DNA and cloned into the LUC reporter plasmid. Similar to the Acvrl1 promoter, FOXF1 synergized with FLI1 to stimulate its own promoter activity, whereas the S52F FOXF1 mutant had no effect (Supplementary Fig. 15).
a Violin plots were generated using a combined pool of cells from WT and Foxf1WT/S52F E18.5 lungs and show endothelial-enriched expression of ETS transcription factors Fli1, Erg and Ets1. ETS transcription factor Spi1 is not expressed in endothelial, epithelial, and mesenchymal cell lineages in the lung tissue. b Violin plots show that Fli1 is enriched in gCAPs, whereas Erg is enriched in aCAPs. Ets1 expression is similar in aCAPs and gCAPs. The data for box plots were from single cell normalized counts (WT: n = 532; Foxf1WT/S52F: n = 320). Boxplots: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile. T-test (two-tailed) were performed, pvalue for Fli1 is 0.01677, and for Erg is 0.02791. c Dual luciferase (LUC) assay shows transcriptional synergy between CMV-FOXF1 and CMV-FLI1 expression vectors to activate the ACE400 LUC reporter. Co-transfection experiments were performed using fetal lung MFLM-91U cells (n = 4). Data were shown as mean ± SD, and one-way ANOVA followed by Tukey’s test (two-tailed) were performed. **p < 0.01, *p < 0.05, ns is not significant. d ChIPseq shows that FOXF1 and FLI1 proteins bind to the ACE400 region of Acvrl1 gene (red box).
FOXF1 stimulates BMP9/ACVRL1 signaling in vitro
BMP9 is the main ligand of the ACVRL1/BMPR2/ENG receptor complex in endothelial cells which mediates downstream signaling through phosphorylation of SMAD1/5/827,28. Consistent with published studies, BMP9 increased phosphorylation of SMAD1 in fetal lung endothelial MFLM-91U cells as demonstrated by immunostaining for pSMAD1 (Fig. 7a) and Western blot (Fig. 7b). Furthermore, BMP9 increased ACVRL1 protein levels but did not change the expression of FOXF1 (Fig. 7b). To examine the effect of FOXF1 on BMP9/ACVRL1 signaling in vitro, FOXF1 was inhibited by siRNA prior to BMP9 treatment of MFLM-91U cells (Supplementary Fig. 16a). Inhibition of FOXF1 decreased ACVRL1 and pSMAD1 in BMP9-treated cells but did not affect total SMAD1 (Fig. 7b). There were no changes in pSMAD2 or total SMAD2 (Supplementary Fig. 16b). To examine the consequences of FOXF1 inhibition on transcriptional activity of pSMAD1, we used the BRE-LUC reporter plasmid, which contains a synthetic enhancer element capable of binding to activated pSMAD140. Treatment with BMP9 increased activity of the BRE-LUC reporter (Fig. 7c), consistent with transcriptional activation of pSMAD1. Inhibition of either FOXF1, ACVRL1 or the ACVRL1 co-receptor ENG decreased transcriptional activity of pSMAD1 in BMP9-treated cells (Fig. 7c and Supplementary Fig. 16c). Thus, FOXF1 is required for BMP9/ACVRL1/pSMAD1 signaling in fetal lung endothelial cells in vitro.
a Immunostaining shows an increase in phosphorylated SMAD1 (pSMAD1) after treatment of endothelial MFLM-91U cells with BMP9. Nuclear pSMAD1 staining (red) is indicated by arrows. Endothelial cells are counterstained with DAPI (blue) and ENG (green). The experiment was repeated 3 times with similar results. Scale bars are 10 μm. b Western blot shows that BMP9 treatment increases pSMAD1 and ACVRL1 protein levels but does not affect FOXF1 or total SMAD1 in MFLM-91U cells (n = 3). Inhibition of FOXF1 by siRNA decreases pSMAD1 and ACVRL1 proteins in BMP9-treated cells. c Dual luciferase assay shows that BMP9 increases activity of the BRE-LUC reporter plasmid in MFLM-91U cells (n = 3). siRNA-mediated inhibition of FOXF1, ACVRL1 or ENG decreases BRE-LUC reporter activity in BMP9-treated cells. b, c Data were shown as mean ± SD, and one-way ANOVA followed by Tukey’s test (two-tailed) were performed. **p < 0.01, *p < 0.05 ns is not significant. d–f In vitro angiogenesis assay shows that inhibition of FOXF1 by siRNA decreases the formation of endothelial sprouts in MFLM-91U cells. BMP9 treatment partially restores the in vitro angiogenesis in FOXF1-deficient endothelial cells. Treatment of cells with ALK1-Fc chimeric protein inhibits the effect of BMP9. Scale bars are 20 μm. The complexity of the vascular network in Matrigel is quantitated by measurements of sprout length e and counts of the sprout junctions f (n = 8 per each group). e, f Data were shown as mean ± SD, and one-way ANOVA (two-tailed) followed by Tukey’s multiple comparison test was performed. **p < 0.01, *p < 0.05, ns is not significant.
Next, we used the in vitro angiogenesis assay to determine whether BMP9 can increase angiogenesis in FOXF1-deficient endothelial cells. Consistent with published studies19, siRNA-mediated depletion of FOXF1 inhibited angiogenesis in Matrigel as shown by measurements of the length of endothelial sprouts (Fig. 7d, e) and the number of branching points in the endothelial network (Fig. 7f). Treatment with BMP9 partially restored the ability of FOXF1-deficient endothelial cells to form sprouts in Matrigel (Fig. 7d–f). The effect of BMP9 was completely inhibited by pre-treatment of endothelial cells with ALK1-Fc (Fig. 7d–f), a soluble chimeric protein containing the ACVRL1 ligand-binding domain which functions as a ligand trap for BMP928. Thus, BMP9 increases angiogenesis in FOXF1-deficient lung endothelial cells through the ACVRL1 receptor.
Nanoparticle-mediated inhibition of ACVRL1 in endothelial cells disrupts angiogenesis and alveolarization in the neonatal lung. To determine whether BMP9/ACVRL1 signaling is important for development of pulmonary vasculature after birth, we used Dylight 650-labeled PEI600-MA5/PEG-OA/Cho nanoparticles (Fig. 8a), which were developed by our group to deliver non-integrating DNA expression plasmids, siRNAs and stabilized mRNAs into the pulmonary microvasculature in vivo41,42,43,44. Delivery of the nanoparticles containing either Acvrl1-specific (siAcvrl1) or control siRNA was performed intravenously in WT mice at postnatal day 2 (P2). Forty-eight hours later, the nanoparticles were detected by FACS analysis in approximately 65% of pulmonary endothelial cells (CD31+CD45−) and 30% of CD140+ fibroblasts (CD140+CD31–CD45–CD326–) but not in epithelial (CD326+CD31–CD45–CD140–) or hematopoietic cells (CD45+CD31–) (Supplementary Fig. 17a–c). Compared to CD140+ fibroblasts, the nanoparticle uptake was significantly higher in pulmonary endothelial cells as demonstrated by the mean fluorescence intensity for Dylight 650 (Supplementary Fig. 17d). Forty-eight hours after nanoparticle delivery of Acvrl1 siRNA, endogenous Acvrl1 mRNA was decreased only in endothelial cells but not in other cell types as shown by qRT-PCR analysis of FACS-sorted cells (Fig. 8b). The decrease in endothelial Acvrl1 mRNA in nanoparticle-treated WT mice was approximately similar to that of Acvrl1 mRNA levels in endothelial cells of Foxf1WT/S52F mutant mice (Fig. 4a–c), which exhibit impaired lung angiogenesis and ACDMPV14. Phosphorylation of SMAD1 was decreased in lungs of mice treated with Acvrl1 siRNA (Supplementary Fig. 18a), supporting reduced ACVRL1 signaling. Two weeks after the nanoparticle delivery of Acvrl1 siRNA, arterial oxygenation was decreased (Fig. 8c). Reduced arterial oxygenation in Acvrl1-deficient mice was associated with decreased capillary density (Fig. 8d, e and Supplementary Fig. 19a, b) and increased sizes of alveoli (Supplementary Fig. 19c, d), findings consistent with impaired lung angiogenesis and alveolarization. Interestingly, intravenous administration of BMP9 ligand to siAcvrl1-treated WT mice (Supplementary Fig. 18b) improved arterial oxygenation, increased capillary density, and partially decreased alveolar simplification (Fig. 8c–e and Supplementary Fig. 19a–d). Furthermore, BMP9 treatment restored expression of Acvrl1 and its downstream target gene Tmem100 in purified pulmonary endothelial cells from siAcvrl1-treated mice (Supplementary Fig. 18c). BPM9 treatment did not change Foxf1 mRNA in lung endothelial cells (Supplementary Fig. 18c). Thus, BMP9 increases ACVRL1 expression and stimulates endothelial ACVRL1 signaling to promote neonatal lung angiogenesis and alveolarization.
a Schematic diagram shows structure of PEI600-MA5/PEG-OA/Cho nanoparticles containing duplex siRNA, cholesterol, PEI600-MA5 and PEG2000-OA. b Real-time PCR analysis shows decreased amounts of Acvrl1 mRNA in FACS-sorted pulmonary endothelial cells (CD31+CD45–) from mice treated with nanoparticles containing Acvrl1-specific siRNA (siAcvrl1) (n = 3). Nanoparticles with scrambled siRNA were used as a control (siControl). Nanoparticles were delivered i.v. to P2 mice. Lungs were harvested at P4. Acvrl1 mRNA is not changed in FACS-sorted epithelial cells (CD326+CD31–CD45–), fibroblasts (CD140a+CD31–CD45–) and hematopoietic cells (CD45+CD31–). c Arterial oxygen saturation (SpO2) is decreased in mice treated with nanoparticles containing Acvrl1 siRNA compared to control siRNA (n = 6). Intravenous BMP9 delivery increases arterial oxygenation in siAcvrl1-treated mice (n = 6). Nanoparticles were delivered at P2, BMP9 treatment was performed at P4, arterial oxygenation was measured at P18. b, c Data were presented as mean ± SD. d, e Alveolar capillary density is decreased after nanoparticle delivery of Acvrl1 siRNA but restored after administration of BMP9. Immunostaining lung sections for endothelial marker endomucin (green) and confocal 3D imaging of cleared lung lobes were performed using P18 mouse lungs (n = 6). Boxplots: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile. b–d Data were shown as mean ± SD, and one-way ANOVA (two-tailed) followed by Tukey’s multiple comparison test was performed. **p < 0.01, *p < 0.05, ns is not significant. Scale bars are 50 μm.
BMP9 improves neonatal lung angiogenesis and alveolarization in mouse ACDMPV model. Since BMP9 increases angiogenesis in FOXF1-deficient endothelial cells in vitro (Fig. 7d–f), we tested the efficacy of BMP9 treatment on neonatal lung angiogenesis in Foxf1WT/S52F mouse model of ACDMPV, which is characterized by congenital defects in lung angiogenesis and alveolarization14. Since Foxf1WT/S52F mutant mice with the most severe phenotype die at birth14, we used surviving Foxf1WT/S52F mice with non-lethal congenital defects for BMP9 rescue experiments (Supplementary Fig. 20a, b and41). Foxf1WT/S52F and WT littermates were treated with BMP9 ligand or saline at P3 (Fig. 9a). Two weeks later, the mice were used for measurements of arterial oxygenation, histological evaluation, and RNA analysis of the lung tissue. To visualize perfused alveolar capillaries, we performed intravascular labeling with isolectin B4 before the mouse harvest. Compared to either untreated or saline-treated Foxf1WT/S52F mice, treatment with BMP9 effectively restored the capillary density (Fig. 9b, c), decreased alveolar simplification (Fig. 9d, e), increased arterial oxygenation (Fig. 9f), increased Acvrl1 expression (Supplementary Fig. 21a), and improved survival in the ACDMPV mouse model (Supplementary Table 6). Treatment with BMP9 had no effect on neonatal lung angiogenesis and alveolarization in WT controls (Fig. 9b–f). BMP9 did not change Foxf1 mRNA in either WT or Foxf1WT/S52F lungs (Supplementary Fig. 21b). Finally, BMP9 treatment did not cause abnormal ossification or ectopic bone formation in the developing mice (Supplementary Fig. 21c). Altogether, administration of BMP9 improves survival and alleviates histological and vascular impairments in lungs of ACDMPV mice.
a Schematic shows BMP9 treatment in WT and Foxf1WT/S52F littermates. BMP9 was delivered i.v. at P3. Mice treated with saline were used as controls. Lungs were examined at P18. b, c Delivery of BMP9 improves the capillary density in Foxf1WT/S52F lungs but have no effect in WT controls. Capillary density was measured using the in vivo labeling with isolectin B4 (red) which binds to the luminal surface of perfused blood vessels. Scale bars are 50 μm. Capillary density was quantified using high–resolution confocal microscopy. Ten random images were quantified using n = 10 mice in each group. d–e H&E staining shows that BMP9 treatment protects Foxf1WT/S52F mice from alveolar simplification. Scale bars are 50 μm. Ten random images were quantified using n = 10 mice in each group. f BMP9 treatment increases arterial oxygen saturation (SpO2) in Foxf1WT/S52F mice compared to WT littermates. Arterial oxygenation was measured at P18 (n = 8 mice per group). Boxplots in c, e, and f: center line, median; box boundary, first and third quartiles; whiskers denote 5th–95th percentile. Data were shown as mean ± SD, and one–way ANOVA (two–tailed) followed by Tukey’s multiple comparison test was performed. *p < 0.05,**p < 0.01 ns is not significant.
Discussion
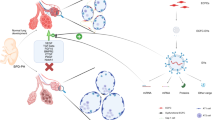
Recent advances in single cell RNA sequencing identified heterogeneity of endothelial cells that form the alveolar microvasculature8. These include aCAPs and gCAPs, the latter of which include EPCs critical for neonatal lung angiogenesis and lung regeneration after injury8,11. EPCs are dependent upon expression of FOXF1 transcription factor in these cells11,12. Inactivating mutations in the FOXF1 gene locus are linked to ACDMPV5,14, a severe congenital disorder associated with paucity of alveolar capillaries4, indicating that FOXF1 is required for development of the alveolar microvasculature. While endothelial cells play a key role in ACDMPV pathogenesis14, endothelial cell diversity and key signaling pathways in ACDMPV remain uncharacterized. In this study, we used Foxf1WT/S52F mice which recapitulate both genetic and pathological findings in ACDMPV patients14, to identify the impairment of BMP9/ACVRL1 signaling in Foxf1WT/S52F EPCs. FOXF1 regulates BMP9/ACVRL1 signaling in EPCs through activation of ACVRL1 gene expression as evidenced by direct binding of the FOXF1 protein to ACVRL1 promoter DNA and by transcriptional activation of the ACVRL1 promoter by CMV–FOXF1 expression vector. While ACVRL1 is an evolutionarily conserved transcriptional target of FOXF1 in higher vertebrates, the activation of ACVRL1 promoter is dependent on transcriptional synergy between FOXF1 and ETS transcription factor FLI1. FOXF1 also synergizes with FLI1 to activate its own promoter region. The presence of functional FOXF1 and FLI1 binding sites in evolutionarily conserved regions of FOXF1 and ACVRL1 promoters is consistent with published studies that identified FOX–ETS DNA–binding motifs as a transcriptional signature of endothelial–specific gene expression45. Since FLI1 is enriched in FOXF1+ EPCs, it is possible that the loss of FOXF1/FLI1 transcriptional synergy (due to the S52F Foxf1 mutation) is more detrimental for the maintenance of proper FOXF1 and ACVRL1 gene expression in FOXF1+ EPCs compared to either aCAPs or FOXF1– gCAPs. Our studies suggest a model in which FOXF1 synergizes with FLI1 to increase its own expression and activate the ACVRL1 promoter, stimulating proangiogenic BMP9/ACVRL1 signaling in pulmonary EPCs (Supplementary Fig. 22). Since decreased expression of ACVRL1 is found in human ACDMPV, impaired BMP9/ACVRL1 signaling in pulmonary EPCs may contribute to diminished angiogenesis in ACDMPV lungs. Our findings that FOXF1 regulates its own expression through a transcriptional auto–regulatory loop are consistent with published studies demonstrating that FOXF1 functions as a “pioneer” transcription factor in gastrointestinal and myogenic tumor cells46,47.
Hereditary hemorrhagic telangiectasia (HHT) is a genetic disorder characterized by mucocutaneous telangiectases, visceral arteriovenous malformations and hemorrhage26. HHT is caused by abnormal TGFβ/BMP9 signaling in endothelial cells through ACVRL1 and its co–receptor ENG24,25. Vascular abnormalities and hemorrhage in HHT type 1 result from inactivating mutations in ENG, whereas vascular defects in HHT type 2 are caused by mutations in ACVRL124,25. ACVRL1 binds to ENG and BMPR2 to form a receptor complex, which promotes TGFβ/BMP signaling through phosphorylation of SMAD1, SMAD5 or SMAD8 that translocate into the nucleus to regulate gene expression27. Heterozygous germline mutations in BMPR2 are linked to a heritable form of pulmonary arterial hypertension (PAH)48. Since ACDMPV is associated with PAH and lung hemorrhage4, it is not surprising that BMP9/ACVRL1/ENG signaling is impaired in ACDMPV. However, obvious differences in pathological and clinical findings between HHT, PAH and ACDMPV suggest that mutations in human ACVRL1, BMPR2, ENG and FOXF1 have unique molecular mechanisms, and perhaps, affect different cell types in addition to regulating BMP9/ACVRL1/ENG signaling in mature endothelial cells and EPCs. Identification of common and unique molecular mechanisms for each mutation will require single cell genomic, transcriptomic, and proteomic approaches using human lung biopsies. Our scRNAseq analysis of murine embryonic lungs demonstrates that Acvrl1, Bmpr2, Eng and Foxf1 are expressed in multiple pulmonary cell types, supporting the potential involvement of these genes in many cellular processes and cell–to–cell interactions.
An important contribution of our study is that i.v. administration of BMP9 ligand stimulates neonatal lung angiogenesis and alveolarization in ACDMPV mice, suggesting that BMP9 agonists may be considered for treatment of human ACDMPV. BMP9 overcomes FOXF1-mediated defects in angiogenesis in Foxf1WT/S52F mutant mice and in vitro by increasing ACVRL1 expression and activating BMP9/ACVRL1/SMAD1 signaling in pulmonary EPCs. Our results are consistent with published studies demonstrating that administration of BMP9 protects the lung from alveolar simplification caused by neonatal hyperoxic injury30, and alleviates pulmonary hypertension in Bmpr2-mutant mice29. Interestingly, BMP9 treatment did not cause aberrant angiogenesis in WT lungs, findings consistent with published studies in which administration of BMP9 had no adverse effects in adult WT mice and rats29. It is possible that short duration of BMP9 stimulation is insufficient to cause aberrant angiogenesis during normal lung development. It is also possible that Foxf1WT/S52F mice are more sensitive to BMP9 stimulation in EPCs due to the presence of a “vascular niche” created by microvascular deficiency in FOXF1-deficient lungs.
In summary, the S52F Foxf1 mutation disrupts BMP9/ACVRL1 signaling in pulmonary EPCs, causing decreased neonatal lung angiogenesis and alveolarization in Foxf1WT/S52F mice. BMP9/ACVRL1 signaling is decreased in human ACDMPV lungs. FOXF1 synergizes with FLI1 to stimulate ACVRL1 gene transcription through an evolutionarily conserved −538/−458 bp promoter region. Administration of BMP9 ligand to either ACVRL1-deficient or Foxf1WT/S52F mice restores neonatal lung angiogenesis and alveolarization. Our studies demonstrate that pulmonary EPCs stimulate neonatal lung angiogenesis and alveolarization through FOXF1-mediated activation of BMP9/ACVRL1 signaling.
Methods
Ethics Statement
The data, analytic methods, and study materials will be made available upon request from the corresponding author of this manuscript to other researchers for purposes of reproducing the results or replicating the procedures. All animal studies were approved by the Institutional Animal Care and Use Committee of the Cincinnati Children’s Hospital. Newborn mouse pups delivered on the same day were randomized at day of birth at P0 and divided to equal–sized litters of 4. Pups were euthanized by injection of pentobarbital sodium at 10 μl/kg. All mice were kept under SPF (specific-pathogen free) conditions in 12/12 light/dark cycle, 18–23 oC and 40–60% humidity. The NIH guidelines for laboratory animal care were strictly followed.
Integrative analysis of single cell RNA sequencing of the mouse lung
Single cell RNAseq libraries were generated from Foxf1WT/S52F and WT E18.5 lungs using the GemCode Single-Cell Instrument and Single Cell 3’ Library & Gel Bead Kit v3 and Chip Kit (10x Genomics) according to manufacturer’s protocol. The lungs were finely minced into small pieces on ice in RPMI-1640 medium containing 0.2 mg/ml Liberase TM (Roche) and 100U/ml Deoxyribonuclease I (Sigma). Lung tissues were digested for 30 min at 37 oC. After digestion, cell suspensions were passed through a 70 μm cell strainer (Corning). Red blood cells were lysed in Ammonium-Chloride-Potassium (ACK) lysis buffer (Thermofisher). Hematopoietic cells were depleted from the cell suspensions using mouse CD45 magnetic beads in MS columns. Cell viability was determined in hemocytometer chambers using trypan blue stain. Equal numbers of cells from three Foxf1WT/S52F and WT E18.5 lungs were combined to prepare sequencing libraries. Approximately 11,000 cells were loaded for each sample with a targeted cell recovery estimate of 7,000 cells. The libraries were sequenced with Novaseq 6000 at a sequencing depth of ~50,000 reads per cell. Starting with BCL files obtained from Illumina sequencing, we used cellranger mkfastq to extract sequence reads in FASTQ format. The cellranger count with mm10 reference genome (as provided by 10X) and the Cell Ranger software (v3.0.0, 10X Genomics) were used to generate gene-count matrices from the FASTQ files. Reads were processed by counting Unique Molecular Identifiers (UMI). Cells with unique feature counts less than 500 and more than 8000 were excluded from the analysis. We also excluded cells that had more than 20% mitochondrial counts. Filtering, normalization criteria and principal component analysis of scRNAseq datasets are described in12,32.
For integrative analysis of WT and Foxf1WT/S52F cells, the combined dataset was analyzed by the Seurat software (version 3.0 in R3.5 environment). For both WT and Foxf1WT/S52F cells, the gene-feature matrices were pre-processed using the same procedures and criteria. The data were log-normalized with a scale factor of 104. The outlier cells were regressed out based on the percentage of mitochondrial reads and the number of UMIs in a negative binomial model. Variable features were determined by the FindVariableGenes function using the Seurat package. FindIntegrationAnchors and IntegrateData functions were used to integrate WT and Foxf1WT/S52F datasets. Next, highly variable features from the integrated matrix were used as input for linear dimensionality reduction via principal component analysis (PCA). Principal components (PCs) were examined using functions ElbowPlot and DimHeatmap to determine the number of PCs for downstream analysis. PCs with standard deviation > 3.5 were used as input for unsupervised clustering with a resolution set to 0.6, using the nonlinear UMAP down dimensional algorithm.
For differential analysis of gene expression in gCAPs, WT and Foxf1WT/S52F gCAPs were defined using the Idents function in the Seurat package. Next, the FindMarkers function was used with the logfc threshold set to 0.4 to identify genes downregulated or upregulated between WT and Foxf1WT/S52F gCAPs. For regression analysis, cells from the cluster AT1 and Matrix. FB -2 were selected and separated based on the orig.ident function from the meta.data. The heatmaps were generated using the function DoHeatmap in the same package. The gene expression signatures were separately produced for WT and Foxf1WT/S52F datasets using the AverageExrpression function and normalized using the log1p function. The linear regression plot was generated using the ggplot2 package. For molecular pathway analysis, the functional annotation clustering was performed using DAVID Bioinformatics Resources Ver.6.8. The KEGG (Kyoto Encyclopedia of Genes and Genomes) pathway enrichment analysis was used to produce the annotation data. Functional clusters with Benjamini-corrected P value < 0.05 were considered statistically significant. Heatmap were generated using the pheatmap package.
FACS analysis and cell sorting
Lungs were perfused through the right ventricle with 5 ml of PBS to remove blood. The lung tissue was enzymatically digested using 0.2 mg/ml Liberase TM (Roche) and 100U/ml Deoxyribonuclease I (Sigma, DN25) for 45 mins, and then passed through nylon mesh (70 µm pores) to obtain a single cell suspension as described42,49,50. Red blood cells were lysed in ACK lysis buffer. To remove cell debris, single cell suspensions from the lung tissue were passed through cell strainer snap cap (Corning life sciences). Single cell suspensions were stained with fixable viability dye (Biolegend) followed by the incubation with TruStain FcX (Biolegend). To identify cell surface antigens, cells were stained with a mixture of fluorochrome-conjugated antibodies (Supplementary Table 7). Data were acquired using BD LSR II cytometer and FACSDiva 8.0 software. Compensation and data analyses were performed using FlowJo software (TreeStar version 10.5.1). The Foxf1-GFP reporter line containing GFP knocked into the endogenous Foxf1 gene locus was used to purify FOXF1+ gCAPs. Stained cells were separated using the five-laser FACS Aria II cell sorter (BD Biosciences). Sorted cells were first collected into EGM-2 medium (Lonza), and then washed once with the medium after centrifugation at 300 g for 5 mins.
RNA preparation and quantitative real-time RT-PCR
RNeasy micro plus kit (Qiagen) was used to prepare total RNA from endothelial cells (CD31+CD45–) that were purified through either FACS sorting or immunomagnetic beads. For the immunomagnetic method, the purity of CD31+CD45– cell enrichment was ~85% as determined by flow cytometry. Quantitative real-time RT-PCR (qRT-PCR) was performed using a StepOnePlus Real-Time PCR system (Thermofisher) as described previously51,52. Samples were amplified using inventoried TaqMan primers (Supplementary Table 8). Reactions were analyzed in triplicates, and expression levels were normalized to β-actin mRNA.
DNA constructs and dual luciferase assay
ACE400 and ACE80 Acvrl1 promoter regions were generated by PCR from mouse genomic DNA and cloned into the pGL4.25 luciferase (LUC) reporter plasmid. PCR primers are provided in Supplementary Table 9. The 400 bp fragment of the Acvrl1 promoter region (ACE400) was generated using a 5′ primer containing a KpnI site, and a 3′ primer containing a NheI site. The amplified ACE400 DNA was purified and cloned into the KpnI-NheI linearized pGL4.25 LUC reporter plasmid containing a minimal promoter. The ACE80 LUC reporter was produced by directly annealing two oligos: 5′ oligo containing a KpnI site, and a 3′ oligo containing a NheI site. The quickchange site-directed mutagenesis was used to disrupt FOXF1-binding sites, and the primers used were listed in Supplementary Table 9. All constructs were verified by DNA sequencing. CMV-FOXF1, CMV-S52F FOXF1 and CMV-empty plasmids were generated previously14,53,54. Expression constructs for ETS transcription factors were obtained from Addgene. Dual LUC assay was performed using the standard protocol from Promega in mouse fetal lung endothelial MFLM-91U cells as described47,55.
RNAscope assay
Mouse lungs were fixed in 4% paraformaldehyde for 24 hr at 4 oC. Tissue sections were cut at 12 μm and the staining was performed using the RNAscope Multiplex Fluorescent Reagent Kit (v.2) and Opal Fluorophore reagents (Akoya Biosciences, 1:750 dilution for Opal 570 and 1:500 dilution for 690 dyes). Sections were counterstained with DAPI and mounted in the Prolong Gold antifade reagent (Thermofisher). The following riboprobes from Advanced Cell Diagnostics were used: Mm-Foxf1 (473051), Mm-Acvrl1(319031), Mm-Tmem100 (435201) and Mm-Aplnr (436171-C2).
Cell culture and immunostaining
The mouse fetal lung mesenchymal MFLM-91U cell line was obtained from Seven Hill Bioreagents. MFLM-91U cells were transfected with siRNAs or treated with recombinant BMP9 (25 ng/ml, R&D) and harvested 48 h after the treatment. After removal of culture medium by aspiration, the cells were washed with PBS and then fixed in 4% paraformaldehyde (PFA) for 15 mins. Cells were permeabilized with 0.1% Triton-X100 and incubated in blocking buffer (10% fetal bovine serum in PBS). The cells were stained with primary antibodies overnight at 4 °C, followed by 1-h incubation with Alexa-488 or Alexa-594-conjugated donkey anti-rabbit IgG (1:1000, Invitrogen, A-21207) and donkey anti-rat IgG (1:1000, Invitrogen A-21208). Cell nuclei were counterstained with Hoechst 33258 (Thermofisher). Images were acquired using Nikon Eclipse Ti2 inverted microscope. Lung cryosections were either stained with Hematoxylin and Eosin (H&E) or used for immunostaining as described55,56,57,58. Antibodies for immunostaining are listed in Supplementary Table 7 and the staining conditions are described in59,60,61,62. Morphometrical measurements of alveolar sizes and intra-vascular labeling with isolectin B4 were described in published studies14,63,64. Co-localization studies and confocal imaging of lung tissue sections were performed as described65,66,67.
In vitro angiogenesis in Matrigel
Fetal lung endothelial MFLM-91U cell line was maintained in serum free medium. For angiogenesis assay, 150 µl of growth factor-reduced Matrigel was placed in each well of 8-well cell culture chamber slides (Corning), pre-incubated on ice, and then incubated for 1 h at 37 oC. For each well, 40,000 cells were seeded and incubated in preconditioned media supplemented with recombinant BMP9 protein (25 ng/ml, R&D). Alk1-Fc protein (R&D, 770-MA) was used to inhibit the effect of BMP9/ACVRL1 signaling. To visualize endothelial sprouts, cells were stained with calceinAM fluorescent viability dye at a final concentration of 2 µg/ml for 30 min. The slides were examined and analyzed using Nikon Eclipse Ti2 inverted microscope and NIS Elements AR software.
Nanoparticle-mediated inhibition of Acvrl1 in the mouse neonatal lung
C57BL/6 mice were purchased from Jackson laboratory. Postnatal day 2 (P2) mice were intravenously (i.v.) injected with PEI600-MA5/PEG-OA/Cho nanoparticles carrying Acvrl1-specific siRNA or control siRNA (5 μg in a total volume of 25 μl) through the facial vein. Chemical synthesis of PEI600-MA5/PEG-OA/Cho nanoparticles is described in14,42,43. BMP9 ligand (R&D) was injected i.v. at P4 via the facial vein (50 ng per mouse in 25 μl of saline).
PEI600-MA5/PEG-OA/Cho nanoparticles were used to encapsulate siRNA duplexes. Briefly, PEI600 was conjugated with myristic acid to form PEI600-MA5. PEG2k was conjugated with oleic acid to form PEG2k-OA. To form NHS esters, carboxylate groups were activated by EDC/NHS for 15 min at 40 oC in 95% ethanol buffered with 25 mM MES (pH 6.0). PEI or PEG-NH2 were quickly added following NHS esters. The mixture was incubated overnight at 40 oC to form PEI600-MA5 or PEG-OA. Residual chemicals in PEI600-MA5 or PEG-OA solutions were removed by dialysis against 90% ethanol or water, respectively. PEI600-MA5, PEG-OA and cholesterol were mixed in ethanol at a mass ratio of 90:10:10. The residual solvent was removed by rotary evaporation to produce a lipid mixture. To label the nanoparticles, the DyLight 650-NHS ester fluorescence dye was coupled to the amine groups on the nanoparticles (1:100 dye to nanoparticle mass ratio). The lipid mixture was resuspended in 25 mM HEPES buffer (pH 7.4). The siRNA stock solution (100 μM) was used to prepare siRNA-encapsulated lipid nanocomplexes at a polymer/siRNA ratio of 20:1 (wt/wt). Nanoparticle complexes with 5 nmol of encapsulated siRNA were diluted in 25 μl carrier solution (25 mM HEPES buffer and 5% glucose) and injected into P2 mice via the facial vein. For BMP9 treatment in vivo, the dose 50 ng/mouse of recombinant BMP9 (5566-BP/CF, R&D) was injected i.v. to nanoparticle-treated P4 mice. Recombinant BMP9 was prepared as 100 μg/ml stock solution. The stock solution was diluted and kept on ice immediately prior to the BMP9 administration.
BMP9 treatment of Foxf1 WT/S52F mice and measurements of capillary density using the whole mount lung imaging
Foxf1WT//S52F and Foxf1WT-GFP/+ mouse lines were described previously11,14. Foxf1WT//S52F mice were maintained in C57BL/6 × 129/J genetic background14. Mice were anesthetized for in vivo treatment with recombinant BMP9 protein (R&D). At P2, Foxf1WT/S52F and WT littermates were given i.v. injections of recombinant BMP9 (50 ng/mouse) via the facial vein. Lungs were harvested at P18. Alexa fluor 594-conjugated IsolectinB4 (Thermofisher) was injected i.v. one hour prior to lung harvest to label pulmonary vasculature. Lung was perfused with PBS, inflated with 4% PFA and fixed in 4% PFA overnight. Fixed tissues were washed with PBS three times and incubated in the hydrogel monomer solution A4P0 (4% acrylamide in PBS) supplemented with 0.25% photoinitiator 2,2ʹʹ-Azobis(2-(2-imidazolin-2-yl) propane) dihydrochloride (VA-044, Wako Chemicals, USA) for two days, and then incubated for 6 h at 37 °C to initiate tissue-hydrogel hybridization. After removing excess of hydrogel by washing the tissues with PBS, tissues were cleared with X-clarity system (Logos biosystems) in electrophoretic tissue clearing solution for 6 h. After several washes in PBS for two days, the samples were transferred to RIMS to align the refractive indices of the objective. RIMS consists of 88% w/v Histodenz (D2158, Sigma-Aldrich), pH adjusted to 7.5 with NaOH. Samples were incubated in RIMS overnight and then mounted in fresh RIMS for imaging. The 3D images of pulmonary vasculature were acquired using a NIKON multiphoton confocal microscope. For each treatment, 10 random lung images from 3–5 mice were used for quantification of the microvascular network. The images were captured as Z-series stack containing 11 slices (9 μm thickness). Confocal images were quantified using NIS elements and ImageJ software.
Measurements of arterial oxygen saturation
Arterial oxygenation was measured using MouseOx + pulse oximeter (STARR Life Sciences). The MouseOx + photodiode sensor was placed on the shaved skin of the neck, and the measurements were taken for 10 min during stable heart rate and oxygen saturation readings.
Western blot
MFLM-91U cells were transfected in six-well plates and harvested 48 h after the transfection. After rinsing cells with ice-cold PBS, cells were lysed in cold RIPA buffer (Cell Signaling Technology) supplemented with protease and phosphatase inhibitors. The protein concentrations were determined using Bio-Rad DC assays. Western blot analysis was performed as described68,69,70. Briefly, cell lysates (30 μg of total protein) were separated on 4–12% gradient gels and transferred to PVDF transfer membranes (0.2 μm) (Thermo Scientific) using a wet transfer. After blocking with Odyssey blocking buffer (Li-Cor Biosciences), membranes were incubated with primary antibodies overnight at 4 °C. The membranes were washed 3 times for 10 mins with TBS-T and incubated for 1 h with secondary antibodies from Li-Cor Biosciences. The membranes were washed 3 times for 10 mins with TBS-T and imaged using LI-COR Odyssey CLx.
Statistical analysis
Statistical analyses were performed using R software and Graphpad Prism 8.0. One-way ANOVA and Student’s T-test were used to determine statistical significance. P < 0.05 were considered statistically significant. Multiple means were compared using one-way analysis of variance with the post-hoc Tukey’s test when comparing the groups for which a post hoc analysis of each group was required. Non-parametric Mann-Whitney U test was used for datasets with n < 6 to determine statistical significance. Values were presented as mean ± standard deviation (SD).
Data availability
Single cell RNA sequencing data generated in this study from WT and Foxf1WT/S52F E18.5 lungs were deposited in GEO database (accession number GSE178184). FOXF1 ChIPseq data from mouse lungs (GSE77951; https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE54780) and FLI1 ChIPseq (GSE69101) are also available in a public database (http://cistrome.org/db/). Microarray data from ACDMPV lungs were retrieved from GEO database (GSE54780) using GEOquery 2.5.6 R package. The data were analyzed by the R package limma (3.8.3). Source data are provided with this paper.
Code availability
Code used for the analysis of scRNAseq data is available in a public repository at https://github.com/WGLUN/acdmodel.
References
Whitsett, J. A., Kalin, T. V., Xu, Y. & Kalinichenko, V. V. Building and regenerating the lung cell by cell. Physiological Rev. 99, 513–554 (2019).
Morrisey, E. E. & Hogan, B. L. Preparing for the first breath: genetic and cellular mechanisms in lung development. Developmental Cell 18, 8–23 (2010).
Bolte, C., Whitsett, J. A., Kalin, T. V. & Kalinichenko, V. V. Transcription factors regulating embryonic development of pulmonary vasculature. Adv. Anat. Embryol. Cell Biol. 228, 1–20 (2018).
Bishop, N. B., Stankiewicz, P. & Steinhorn, R. H. Alveolar capillary dysplasia. Am. J. Respiratory Crit. Care Med. 184, 172–179 (2011).
Dharmadhikari, A. V., Szafranski, P., Kalinichenko, V. V. & Stankiewicz, P. Genomic and epigenetic complexity of the FOXF1 locus in 16q24.1: implications for development and disease. Curr. Genomics 16, 107–116 (2015).
Bolte, C., Kalin, T. V. & Kalinichenko, V. V. Molecular, cellular, and bioengineering approaches to stimulate lung regeneration after injury. Semin Cell Dev. Biol. 100, 101–108 (2020).
Pradhan, A., Kalin, T. V. & Kalinichenko, V. V. Genome editing for rare diseases. Curr. Stem Cell Rep. 6, 41–51 (2020).
Gillich, A. et al. Capillary cell-type specialization in the alveolus. Nature 586, 785–789 (2020).
Vila Ellis, L. et al. Epithelial vegfa specifies a distinct endothelial population in the mouse lung. Developmental Cell 52, 617–630 (2020).
Niethamer T. K., et al. Defining the role of pulmonary endothelial cell heterogeneity in the response to acute lung injury. Elife 9, e53072 (2020).
Wang, G. et al. Generation of pulmonary endothelial progenitor cells for cell-based therapy using interspecies Mouse-Rat Chimeras. Am. J. Respiratory Crit. Care Med. 204, 326–338 (2021).
Ren, X. et al. Postnatal alveologenesis depends on FOXF1 signaling in c-KIT(+) endothelial progenitor cells. Am. J. respiratory Crit. Care Med. 200, 1164–1176 (2019).
Kolesnichenko, O. A., Whitsett, J. A., Kalin, T. V. & Kalinichenko, V. V. Therapeutic potential of endothelial progenitor cells in pulmonary diseases. Am. J. Respiratory Cell Mol. Biol. 65, 473–488 (2021).
Pradhan, A. et al. The S52F FOXF1 mutation inhibits stat3 signaling and causes alveolar capillary dysplasia. Am. J. Respiratory Crit. Care Med. 200, 1045–1056 (2019).
Mahlapuu, M., Ormestad, M., Enerback, S. & Carlsson, P. The forkhead transcription factor Foxf1 is required for differentiation of extra-embryonic and lateral plate mesoderm. Development 128, 155–166 (2001).
Lim, L., Kalinichenko, V. V., Whitsett, J. A. & Costa, R. H. Fusion of right lung lobes and pulmonary vessels in mice heterozygous for the Forkhead Box f1 targeted allele. Am. J. Physiol. 282, L1012–L1022 (2002).
Bolte, C. et al. FOXF1 transcription factor promotes lung regeneration after partial pneumonectomy. Sci. Rep. 7, 10690 (2017).
Cai, Y. et al. FOXF1 maintains endothelial barrier function and prevents edema after lung injury. Sci. Signal 9, ra40 (2016).
Ren, X. et al. FOXF1 transcription factor is required for formation of embryonic vasculature by regulating VEGF signaling in endothelial cells. Circulation Res. 115, 709–720 (2014).
Kalinichenko, V. V. et al. Wild type levels of the mouse forkhead box f1 gene are essential for lung repair. Am. J. Physiol. Lung Cell Mol. Physiol. 282, L1253–L1265 (2002).
Kalin, T. V. et al. Pulmonary mastocytosis and enhanced lung inflammation in mice heterozygous null for the Foxf1 gene. Am. J. Respiratory Cell Mol. Biol. 39, 390–399 (2008).
Xu, J. et al. A Shh-Foxf-Fgf18-Shh molecular circuit regulating palate development. PLoS Genet. 12, e1005769 (2016).
Cunha, S. I., Magnusson, P. U., Dejana, E. & Lampugnani, M. G. Deregulated TGF-beta/BMP signaling in vascular malformations. Circulation Res. 121, 981–999 (2017).
Johnson, D. W. et al. Mutations in the activin receptor-like kinase 1 gene in hereditary haemorrhagic telangiectasia type 2. Nat. Genet. 13, 189–195 (1996).
Shovlin, C. L. et al. A gene for hereditary haemorrhagic telangiectasia maps to chromosome 9q3. Nat. Genet. 6, 205–209 (1994).
Fernandez, L. A., Sanz-Rodriguez, F., Blanco, F. J., Bernabeu, C. & Botella, L. M. Hereditary hemorrhagic telangiectasia, a vascular dysplasia affecting the TGF-beta signaling pathway. Clin. Med. Res. 4, 66–78 (2006).
Suzuki, Y. et al. BMP-9 induces proliferation of multiple types of endothelial cells in vitro and in vivo. J. Cell Sci. 123, 1684–1692 (2010).
Bidart, M. et al. BMP9 is produced by hepatocytes and circulates mainly in an active mature form complexed to its prodomain. Cell Mol. Life Sci. 69, 313–324 (2012).
Long, L. et al. Selective enhancement of endothelial BMPR-II with BMP9 reverses pulmonary arterial hypertension. Nat. Med. 21, 777–785 (2015).
Chen, X. et al. Bone Morphogenetic protein 9 protects against neonatal hyperoxia-induced impairment of alveolarization and pulmonary inflammation. Front Physiol. 8, 486 (2017).
Li, W. et al. Circulating BMP9 protects the pulmonary endothelium during inflammation-induced lung injury in mice. Am. J. Respiratory Crit. Care Med. 203, 1419–1430 (2021).
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F. & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
Guo, M. et al. Single cell RNA analysis identifies cellular heterogeneity and adaptive responses of the lung at birth. Nat. Commun. 10, 37 (2019).
Wen, B. et al. In Vivo Generation of Lung and Thyroid Tissues from Embryonic Stem Cells Using Blastocyst Complementation. Am. J. Respiratory Crit. Care Med. 203, 471–483 (2021).
Schupp, J. C. et al. Integrated Single-Cell Atlas of Endothelial Cells of the Human Lung. Circulation 144, 286–302 (2021).
Somekawa, S. et al. Tmem100, an ALK1 receptor signaling-dependent gene essential for arterial endothelium differentiation and vascular morphogenesis. Proc. Natl Acad. Sci. USA 109, 12064–12069 (2012).
Bocci, M. et al. Activin receptor-like kinase 1 is associated with immune cell infiltration and regulates CLEC14A transcription in cancer. Angiogenesis 22, 117–131 (2019).
Sen, P. et al. Comparative analyses of lung transcriptomes in patients with alveolar capillary dysplasia with misalignment of pulmonary veins and in foxf1 heterozygous knockout mice. PloS One 9, e94390 (2014).
Dharmadhikari, A. V. et al. Lethal lung hypoplasia and vascular defects in mice with conditional Foxf1 overexpression. Biol. Open 5, 1595–1606 (2016).
Korchynskyi, O. & ten Dijke, P. Identification and functional characterization of distinct critically important bone morphogenetic protein-specific response elements in the Id1 promoter. J. Biol. Chem. 277, 4883–4891 (2002).
Sun, F. et al. Nanoparticle Delivery of STAT3 Alleviates Pulmonary Hypertension in a Mouse Model of Alveolar Capillary Dysplasia. Circulation 144, 539–555 (2021).
Bolte, C. et al. Nanoparticle delivery of proangiogenic transcription factors into the neonatal circulation inhibits alveolar simplification caused by hyperoxia. Am. J. Respiratory Crit. Care Med. 202, 100–111 (2020).
Dunn, A. W., Kalinichenko, V. V. & Shi, D. Highly efficient in vivo targeting of the pulmonary endothelium using novel modifications of polyethylenimine: an importance of charge. Adv. Health. Mater. 7, e1800876 (2018).
Deng, Z., Kalin, G. T., Shi, D. & Kalinichenko, V. V. Nanoparticle delivery systems with cell-specific targeting for pulmonary diseases. Am. J. Respiratory Cell Mol. Biol. 64, 292–307 (2021).
De Val, S. et al. Combinatorial regulation of endothelial gene expression by ets and forkhead transcription factors. Cell 135, 1053–1064 (2008).
Ran, L. et al. FOXF1 defines the core-regulatory circuitry in gastrointestinal stromal tumor. Cancer Disco. 8, 234–251 (2018).
Milewski, D. et al. FOXF1 is required for the oncogenic properties of PAX3-FOXO1 in rhabdomyosarcoma. Oncogene 40, 2182–2199 (2021).
International, P. P. H. C. et al. Heterozygous germline mutations in BMPR2, encoding a TGF-beta receptor, cause familial primary pulmonary hypertension. Nat. Genet. 26, 81–84 (2000).
Ren, X. et al. Forkhead box M1 transcription factor is required for macrophage recruitment during liver repair. Mol. Cell Biol. 30, 5381–5393 (2010).
Black, M. et al. FOXF1 Inhibits Pulmonary Fibrosis by Preventing CDH2-CDH11 Cadherin Switch in Myofibroblasts. Cell Rep. 23, 442–458 (2018).
Milewski, D. et al. FOXM1 activates AGR2 and causes progression of lung adenomas into invasive mucinous adenocarcinomas. PLoS Genet. 13, e1007097 (2017).
Bolte, C. et al. Expression of Foxm1 transcription factor in cardiomyocytes is required for myocardial development. PloS One 6, e22217 (2011).
Hoggatt, A. M. et al. The transcription factor Foxf1 binds to serum response factor and myocardin to regulate gene transcription in visceral smooth muscle cells. J. Biol. Chem. 288, 28477–28487 (2013).
Bolte, C. et al. Forkhead box F2 regulation of platelet-derived growth factor and myocardin/serum response factor signaling is essential for intestinal development. J. Biol. Chem. 290, 7563–7575 (2015).
Kim, I. M. et al. Functional characterization of evolutionary conserved DNA regions in forkhead box f1 gene locus. J. Biol. Chem. 280, 37908–37916 (2005).
Li, E. et al. Blastocyst complementation reveals that NKX2-1 establishes the proximal-peripheral boundary of the airway epithelium. Dev. Dyn. 250, 1001–1020 (2021).
Kalinichenko, V. V., Gusarova, G. A., Shin, B. & Costa, R. The forkhead box f1 transcription factor is expressed in brain and head mesenchyme during mouse embryonic development. Gene Expr. Patterns 3, 153–158 (2003).
Wang, X. et al. Rapid Hepatocyte Nuclear Translocation of the Forkhead Box M1B (FoxM1B) Transcription factor Causes a Transient Increase in Size of Regenerating Transgenic Hepatocytes. Gene Expr. 11, 149–162 (2003).
Ustiyan, V. et al. FOXF1 transcription factor promotes lung morphogenesis by inducing cellular proliferation in fetal lung mesenchyme. Developmental Biol. 443, 50–63 (2018).
Balli, D. et al. Foxm1 transcription factor is required for macrophage migration during lung inflammation and tumor formation. Oncogene 31, 3875–3888 (2012).
Shukla, S. et al. The FOXM1 inhibitor RCM-1 decreases carcinogenesis and nuclear beta-Catenin. Mol. Cancer Therapeutics 18, 1217–1229 (2019).
Goda, C. et al. Loss of FOXM1 in macrophages promotes pulmonary fibrosis by activating p38 MAPK signaling pathway. PLoS Genet. 16, e1008692 (2020).
Xia, H. et al. Foxm1 regulates resolution of hyperoxic lung injury in newborns. Am. J. Respiratory Cell Mol. Biol. 52, 611–621 (2015).
Sun L. et al. The FOXM1 inhibitor RCM-1 suppresses goblet cell metaplasia and prevents IL-13 and STAT6 signaling in allergen-exposed mice. Sci. Signal 10, eaai8583 (2017).
Ustiyan, V. et al. beta-catenin and Kras/Foxm1 signaling pathway are critical to restrict Sox9 in basal cells during pulmonary branching morphogenesis. Dev. Dyn. 245, 590–604 (2016).
Ustiyan, V. et al. Foxm1 transcription factor is critical for proliferation and differentiation of Clara cells during development of conducting airways. Developmental Biol. 370, 198–212 (2012).
Bolte, C. et al. Postnatal ablation of Foxm1 from cardiomyocytes causes late onset cardiac hypertrophy and fibrosis without exacerbating pressure overload-induced cardiac remodeling. PloS One 7, e48713 (2012).
Pradhan, A., Ustiyan, V., Zhang, Y., Kalin, T. V. & Kalinichenko, V. V. Forkhead transcription factor FoxF1 interacts with Fanconi anemia protein complexes to promote DNA damage response. Oncotarget 7, 1912–1926 (2016).
Wang, I. C. et al. Increased expression of FoxM1 transcription factor in respiratory epithelium inhibits lung sacculation and causes Clara cell hyperplasia. Developmental Biol. 347, 301–314 (2010).
Milewski, D. et al. FoxF1 and FoxF2 transcription factors synergistically promote rhabdomyosarcoma carcinogenesis by repressing transcription of p21Cip1 CDK inhibitor. Oncogene 36, 850–862 (2017).
Acknowledgements
We thank Erika Smith for excellent editorial assistance and Dr. Andreas Damianos for helpful comments.
Funding
This work was supported by NIH Grants HL141174 (to V.V.K.), HL149631 (to V.V.K.) and HL152973 (to V.V.K and T.V.K.).
Author information
Authors and Affiliations
Contributions
G.W. and V.V.K. designed the study; G.W., B.W., Z.D., Y.Z., O.A.K., V.U. and A.P. conducted experiments and analyzed the data; G.W., T.V.K. and V.V.K. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, G., Wen, B., Deng, Z. et al. Endothelial progenitor cells stimulate neonatal lung angiogenesis through FOXF1-mediated activation of BMP9/ACVRL1 signaling. Nat Commun 13, 2080 (2022). https://doi.org/10.1038/s41467-022-29746-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-29746-y
This article is cited by
-
Epigenetic scores derived in saliva are associated with gestational age at birth
Clinical Epigenetics (2024)
-
Macrophage extracellular vesicle-packaged miR-23a-3p impairs maintenance and angiogenic capacity of human endothelial progenitor cells in neonatal hyperoxia-induced lung injury
Stem Cell Research & Therapy (2024)
-
Identification of endothelial and mesenchymal FOXF1 enhancers involved in alveolar capillary dysplasia
Nature Communications (2024)
-
FOXF1 promotes tumor vessel normalization and prevents lung cancer progression through FZD4
EMBO Molecular Medicine (2024)
-
Crosstalk between endothelial progenitor cells and HCC through periostin/CCL2/CD36 supports formation of the pro-metastatic microenvironment in HCC
Oncogene (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.