Abstract
Bacterial bloodstream infections are a major cause of morbidity and mortality among patients undergoing hematopoietic cell transplantation (HCT). Although previous research has demonstrated that pathogens may translocate from the gut microbiome into the bloodstream to cause infections, the mechanisms by which HCT patients acquire pathogens in their microbiome have not yet been described. Here, we use linked-read and short-read metagenomic sequencing to analyze 401 stool samples collected from 149 adults undergoing HCT and hospitalized in the same unit over three years, many of whom were roommates. We use metagenomic assembly and strain-specific comparison methods to search for high-identity bacterial strains, which may indicate transmission between the gut microbiomes of patients. Overall, the microbiomes of patients who share time and space in the hospital do not converge in taxonomic composition. However, we do observe six pairs of patients who harbor identical or nearly identical strains of the pathogen Enterococcus faecium, or the gut commensals Akkermansia muciniphila and Hungatella hathewayi. These shared strains may result from direct transmission between patients who shared a room and bathroom, acquisition from a common hospital source, or transmission from an unsampled intermediate. We also identify multiple patients with identical strains of species commonly found in commercial probiotics, including Lactobacillus rhamnosus and Streptococcus thermophilus. In summary, our findings indicate that sharing of identical pathogens between the gut microbiomes of multiple patients is a rare phenomenon. Furthermore, the observed potential transmission of commensal, immunomodulatory microbes suggests that exposure to other humans may contribute to microbiome reassembly post-HCT.
Similar content being viewed by others
Introduction
Patients undergoing hematopoietic cell transplantation (HCT), a potentially curative treatment for a range of hematologic malignancies and disorders, are at increased risk for bloodstream infections (BSIs) and associated morbidity and mortality1. While the bacterial pathogens that cause BSIs in HCT patients are well understood, their routes of transmission are often unclear. Determining these transmission pathways involves identifying two critical elements: the source of the infection, i.e., how the pathogen was introduced into the patient’s bloodstream, and the origins of the particular pathogen causing the BSI.
The most common pathways by which bacterial pathogens can be introduced into an HCT patient’s bloodstream include contaminated central intravenous lines and translocation of intestinal microbiota across a damaged epithelium2. Indeed, research from our group and others has shown that strains of bacteria isolated from the blood of HCT patients with BSIs may be indistinguishable from the strains in the intestinal microbiota of these patients prior to infection3,4,5. In addition, HCT patients with a microbiome dominated by a single bacterial taxon, such as Enterococcus or Streptococcus, are at increased risk for not only BSI6,7, but also graft-versus-host disease8,9 and death10,11,12,13.
Identifying the source of the BSI is only the first step. To fully understand the transmission pathways of bacterial pathogens in hospital settings, it is also essential to determine the origin of the pathogen that caused the BSI. For gut-based pathogens, there are three possibilities. First, they may exist in the HCT patient’s microbiome upon admission to the hospital. Second, hospital environments and equipment may serve as unintentional reservoirs of pathogens, thereby infecting multiple patients through exposure14. Lastly, a pathogen could originate from the microbiome of other patients, healthcare workers or hospital visitors and be transmitted via person-to-person contact. In cases where traditional epidemiological links cannot be found, this patient–patient transmission of gut microbes may be the “missing link” that explains the persistence of BSIs in hospital environments15.
Transmission of gut bacteria and phages between individuals is known to occur in specific cases, such as from mothers to developing infants16,17,18. By contrast, adults have a microbiome that is relatively resistant to colonization with new organisms even after perturbation by antibiotics19,20,21. While adults living in the same household or in close-knit communities may have more similar microbes than those outside the group22, to our knowledge, direct transmission of gut microbes between adults has not been observed with high-resolution metagenomic methods, with the notable exception of fecal microbiota transplantation23,24,25, a drastic reshaping of the gut microbiota often used in response to Clostridioides difficile infection. Transmission of gut microbiota is thought to occur by a fecal–oral route, which could happen in the hospital environment by exposure to contaminated surfaces or equipment, sharing a room or bathroom, contaminated hands of healthcare workers, or other sources. The perturbed microbiomes of HCT patients, often lacking key species to provide colonization resistance, may be primed to acquire new species from these sources.
Previous studies of the microbiome in HCT patients have often used 16S rRNA sequencing10,26,27,28, which is sufficient for taxonomic classification but may not be sensitive enough to differentiate strains with similar genomes. By contrast, short-read shotgun metagenomic sequencing can capture information from all bacterial, archaeal, eukaryotic, and phage DNA in a stool sample. While short-read sequencing data is accurate on a per-base level, it is often insufficient to assemble complete bacterial genomes due to the presence of repeated genetic elements. Linked-read sequencing captures additional long-range information by introducing molecular barcodes in the library preparation step. This technology allows for significant increases in assembly contiguity29,30 while retaining high per-base accuracy. Both of these technologies also capture information about strain diversity, genetic variation within the population of a species31,32, which is critical for measuring transmission between microbiomes.
Here, we use a collection of short-read and linked-read metagenomic sequencing datasets from 401 stool samples to analyze bacterial transmission between HCT patient microbiomes at a single, high-volume hospital. We apply strain-resolved comparison methods to show that transmission of bacteria between adults hospitalized in the same unit at the same time is likely a rare event, usually occurring when recipients have extremely perturbed microbiomes, such as after exposure to broad-spectrum antibiotics. Bacterial strains shared between individuals include both pathogenic and commensal organisms, demonstrating that transmission may depend more on niche availability than pathogenicity or antibiotic resistance capacity. We find that pathogens colonizing HCT patient microbiomes are present in the first sample in a time course roughly 60–70% of the time in our cohort. This suggests that in most cases, prior colonization, rather than direct transmission from other patients or the hospital environment, is responsible for pathogenic organisms in the gut microbiomes of this patient population. Even though patients were frequently placed into double occupancy hospital rooms with a shared bathroom, we observe relatively few putative transmission events. This implies that sharing a room with another patient may not place a patient recovering from HCT at a greatly increased risk of acquiring pathogens in their gut microbiome.
Results
Sample characteristics and patient geography
We collected weekly stool samples (see the “Methods” section) from adult patients undergoing HCT at Stanford University Medical Center from 2015 to 2019. At the time of the study, our biobank contained over 2000 stool samples from over 900 patients. Samples from October 2015 to November 2018 were considered for this study. Relevant patient health, medication, demographic, hospital admission, and room occupancy data were extracted from electronic health records (Table 1, Supplementary Data 1). All patients stayed in a single ward of the hospital during treatment, which contained 14 single-occupancy and four double-occupancy rooms, the latter of which included shared bathrooms (Fig. S1a). Patients spent a median of 18 days on the ward and were frequently moved between rooms: 42% of patients spent at least one day in three or more rooms during treatment (Table 2, Fig. S1c). 73% of patients shared a room with a roommate for ≥24 h. Over the course of their hospital stays, many patients had several roommates, though never more than one at a time (Fig. S1d). Patients with multi-drug resistant Gram-negative infections were always placed into single rooms with contact precautions. We use the term “hospital overlap” to refer to patients who are in the hospital at the same time. “Roommate overlap” refers to patients who are in the same room at the same time and is a subset of hospital overlap. When not specified otherwise, a minimum time of 24 h was considered for both hospital and roommate overlap.
To understand how roommate overlap may influence transmission of gut microbes, we created a network from patient–roommate interactions lasting at least 24 h (Fig. S1b). 535 patients (77% of patients with at least one roommate, 56% of all patients) fell into the largest connected component of the network. Although the largest component was not densely connected (mean degree 2.2 ± 1.6 standard deviation (SD)), it links together patients over 3 years and may represent a risk for infection transmission. We used the network to select samples for further analysis with metagenomic sequencing, as described in the “Methods” section.
Metagenomic sequencing, assembly and binning
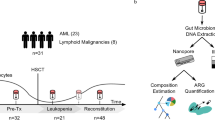
For an overview of the steps used in the generation and processing of sequence data (see Fig. 1a). 328 stool samples from 94 HCT patients were subjected to short-read metagenomic sequencing as part of previous projects (for references and SRA IDs of these samples, see Supplementary Data 2). 96 additional samples from 62 patients were selected for linked-read sequencing to span periods of roommate overlap between patients. Samples were subjected to bead beating-based DNA extraction and bead-based DNA size selection for fragments ≥2 kb (see the “Methods” section). We prepared linked-read sequencing libraries with the 10× Genomics Chromium platform from 89 samples with sufficient DNA concentration. Samples were sequenced to a median of 116 million (M) (±37M SD) read pairs on an Illumina HiSeq4000. In total, 401 stool samples from 149 patients were sequenced (Table 3), with a median of 2 and maximum of 13 samples per patient (Fig. 1b).
a Overview of the experimental and computational workflow used to generate sequencing datasets, bin metagenome-assembled genomes (MAGs), and compare strains between patients. b Number of stool samples sequenced per patient. c Percentage of MAGs meeting each quality level40, stratified by sequencing method. d Of patients who have the given organism detected (≥50% coverage breadth) in a time course sample, percentage of patients where the organism was below the detection threshold (<50% coverage breadth) in the first sample. e Percentage of patients with at least one sample positive with (≥1% relative abundance) or dominated by (≥30% relative abundance) hospital-acquired infection (HAI) organisms, as identified by Kraken2- and Bracken-based classification. f Percentage of bloodstream infections (BSIs) identified with each organism or group in HCT patients and hospital-wide. Source data for this figure are provided in Supplementary data 2–4 and Supplementary Table 1.
We processed all existing short-read data and newly generated linked-read data by first trimming and then removing low-quality reads, PCR duplicates (short-read data only) and reads that aligned against the human genome (see the “Methods” section). After quality control, newly sequenced linked-read samples had a median 104 M (±40 M SD) read pairs, while short-read data had a median 7.6 M (±4.4 M SD) read pairs. Metagenomic assembly was conducted using metaSPAdes33 for short-read data, and MEGAHIT34 followed by Athena29 for linked-read data. Short-read assemblies had a median N50 of 17.2 ± 24.8 kb, while linked-read assemblies had a median N50 of 147.6 ± 165.8 kb. We binned metagenome-assembled genomes (MAGs) using Metabat235, Maxbin36 and CONCOCT37 and aggregated across results from each tool using DASTool38. MAG completeness and contamination was evaluated using CheckM39 and MAG quality was determined by previously established standards40. The vast majority of short-read and linked-read MAGs were at least medium quality, and 27% of linked-read MAGs contained the 5S, 16S, and 23S rRNA genes and at least 18 tRNAs to be considered high-quality (Fig. 1c, Supplementary Data 3). Linked-read MAGs had higher quality than the 4644 species-level genomes in the Unified Human Gastrointestinal Genome collection41, where 573 genomes (12.3%) are high-quality, and only 38 (6.6%) of those came from metagenomes rather than isolates. Sequencing dataset type (short-read vs. linked-read) was not significantly associated with MAG length (student’s t-test, p > 0.9); the increase in quality was mainly due to the inclusion of ribosomal and transfer RNA genes in the linked-read MAGs, which often do not assemble well with short-read sequencing data alone. To understand the diversity of strains present in the microbiomes of our patients, we clustered all medium-quality and high-quality MAGs at 95% and 99% identity thresholds (roughly “species” and “strain” level, see the “Methods” section) using dRep42, yielding 1615 unique genomes representative of the microbial diversity in this sample set.
Classification of abundant healthcare-associated infection organisms
We performed taxonomic classification of sequencing reads with Kraken243 and abundance estimation with Bracken44 using a custom database of bacterial, fungal, archaeal and viral genomes in NCBI Genbank (see the “Methods” section) (Supplementary Data 4 and 5). A median of 33 ± 15% SD reads were classified to the species level with Kraken2 (72 ± 15% SD at the genus level), which was improved to 96 ± 7% SD using Bracken (97 ± 8% SD at the genus level). Organisms that cause healthcare-associated infections (HAI) were identified from the CDC list of pathogens45. Here, we report organisms as present if they achieve 1% relative abundance, but acknowledge that many microbes typically exist at lower concentrations, which may be more difficult to detect with metagenomic sequencing.
Many HAI organisms were prevalent in the microbiomes of the studied HCT patients. 152 samples (38%) from 79 patients (53%) had at least one HAI organism identified at 1% relative abundance or above (Fig. 1e). Escherichia coli was the most common HAI organism (present at ≥1% in 42/149 patients, 28.2%; ≥0.1% = 80/149, 53.7%), followed by Klebsiella pneumoniae (39/149 patients, 26.2%; ≥0.1% = 70/149, 47%) and Enterococcus faecium (26/149 patients, 17.4%; ≥0.1% = 73/149, 49%). Rates of colonization with HAI organisms were much higher than in stool samples from healthy individuals in the Human Microbiome Project46 where E. coli reaches 1% relative abundance in 2.1% of samples, and K. pneumoniae and E. faecium are never found at >1% (present at ≥0.1% in 18.4%, 1.4%, and 0.7% of samples, respectively).
HCT patient microbiomes can become dominated by HAI organisms, often as a result of antibiotic usage. 24 patients (16%) have at least one sample with a dominant HAI organism (≥30% relative abundance), which may place them at increased risk for BSIs6. BSI in this cohort of HCT patients is most frequently caused by E. coli, viridans group Streptococci and E. faecium; these organisms less frequently cause BSI among the entire inpatient population at our hospital (Fig. 1f, Supplementary Table 1). We focused further analysis on E. coli and E. faecium, as these species are both frequently detected in stool and frequently cause BSIs. While viridans group Streptococci frequently cause BSIs in HCT patients, these species are typically more prevalent and abundant in the oral cavity2,47 compared to the gut microbiome (individual species in the group only reach 1% relative abundance in 8/149 patients, 5%).
Detection of E. coli and E. faecium becomes more common during a patient’s hospital stay
We investigated the detection of E. coli and E. faecium in patients with time course samples (82/149 patients, 55%) as a proxy for understanding if these organisms were acquired or became more abundant during the observed hospital stay. Of the 1615 de-replicated MAGs, nine were identified as E. coli and five were identified as E. faecium. We mapped sequencing reads from all samples to these MAGs and evaluated the maximum coverage breadth, the fraction of the reference genome covered with at least one sequencing read. We used a breadth cutoff of 50% to determine “detection” or “absence” in a sample. This threshold is likely specific (it is difficult to achieve 50% breadth by read mis-mapping or homology with a different organism) but not extremely sensitive (it will likely miss very lowly abundant organisms that are truly present).
64/82 (78%) patients with time-course samples have E. coli present at 50% breadth in at least one sample. Of these, 10/64 (16%) have E. coli below 50% breadth in the first sample. 60/82 (73%) patients with time-course samples have E. faecium present at 50% breadth in at least one sample. Of these, 17/60 (28%) have E. faecium below 50% breadth in the first sample (Fig. 1d). In these patients, E. coli or E. faecium may have been newly acquired into the gut microbiome during the hospital stay. Alternatively, the organism could have also been present at low abundance and below our limit of detection in the first sample. It is possible that antibiotic use in the weeks after HCT kills off many of natural microbiome colonizers, allowing antibiotic-resistant HAI organisms to increase in relative abundance above our limit of detection. Overall, the relative abundance of E. coli and E. faecium was not significantly different in first samples compared to later samples (Wilcoxon rank-sum test). While more patients have newly detectable E. faecium than E. coli, the difference was not statistically significant (p = 0.09, binomial likelihood ratio test).
Antibiotic use and its effect on HCT patient microbiomes
HCT patients are frequently prescribed antibiotic, antiviral and antifungal drugs, especially in the days immediately after transplant. These drugs can have a significant impact on the microbial populations in the gut and contribute to the loss of microbial diversity frequently observed following HCT6. Antibiotic use likely impacts the dynamics of bacterial transmission in this patient population, as both natural colonizers, which may provide resistance to newly invading species, and potentially transmitted species can be killed by the drugs. We gathered electronic health record data to understand the characteristics of antibiotic prescription in our patient cohort and its potential impact on the gut microbiome composition (Supplementary Data 6).
Patients were prescribed a median of five different antibiotics (range 1–10) and had a median of 90 cumulative antibiotic-days (range 14–416). The most common antibiotics prescribed were ciprofloxacin (98% of patients prescribed for at least one day), intravenous (IV) vancomycin (80%), cefepime (66%), and piperacillin-tazobactam (57%). Prescription of most antibiotics peaked in the 14 days following HCT, while administration of antifungal drugs like posaconazole and antiviral drugs like ganciclovir were higher up to 50 days following HCT (Fig. 2a). We found that antibiotic usage in the prior seven days before a stool sample was collected was strongly negatively associated with bacterial diversity (linear regression on cumulative antibiotic-days and Shannon diversity, R2 = 0.18, 0.14, p = 8.9e−11, 2.6e−8 for species and genus level, respectively). Samples with a single species dominant at 30% relative abundance or higher also typically came from patients who had higher antibiotic usage in the past seven days prior to that sample (p = 0.001, Wilcoxon rank-sum Test) (Fig. 2b–d).
a Aggregated prescription history of 20 of the most frequently prescribed antibiotic, antifungal and antiviral drugs. Each panel shows the percentage of patients who were prescribed a drug at the given day, relative to the date of HCT. Shannon diversity at the species (b) or genus (c) level compared to total antibiotic-days in the 7 days prior to sample collection. (d) Samples with or without a single species dominant (≥30%), compared with total antibiotic-days in the prior seven days. Boxes extend to the first and third quartile, whiskers extend to the upper and lower value within 1.5*interquartile range (IQR) from the box. Outliers are shown as points. A two-sided Wilcoxon rank-sum test was used to compute the p-value. n = 133 samples from 71 patients in FALSE category; n = 81 samples from 60 patients in TRUE category. Taxonomic similarity at the species level (1−Bray–Curtis dissimilarity) between samples from different patients, evaluated against days of hospital overlap (e) or hours of roommate overlap (f) prior to the earlier sample. Maximum inStrain popANI achieved by comparing all strains in all samples from two patients, evaluated against hours of roommate (g) or days of hospital overlap (h) prior to the earlier sample. In all panels, trend lines are calculated as the best-fit linear regression between the X and Y variables. R and p-values are the Pearson correlation coefficient and correlation p-value, respectively. Pip-Tazo Piperacillin-Tazobactam, TMP-SMX trimethoprim-sulfamethoxazole, PJP Pneumocystis jirovecii pneumonia. Source Data for this figure are provided in Supplementary Data 4–9.
Patients who share time and space in the hospital do not converge in microbiome composition or frequently share strains
To understand the overall impact of hospital and roommate overlap on patient microbiomes, we first studied the taxonomic (Bray–Curtis) similarity between samples from different patients. For each sample pair, we calculated the maximum time the two patients had overlapped in the hospital or as roommates, determined by the earlier sample time (Supplementary Data 7 and 8). There was not a significant linear association between time of overlap and taxonomic similarity (linear regression, p = 0.42 for roommate, p = 0.068 for hospital, Fig. 2e, f), indicating that the broad taxonomic composition of patient microbiomes may not converge under hospital and roommate overlap. However, there may be isolated strains that are shared between patients. We used the strain diversity-aware, SNP-based method inStrain48 to conduct a sensitive analysis of bacterial strains shared between patients. InStrain compares alignments of short reads from multiple samples to the same reference genome and reports two metrics: Consensus ANI (conANI) and PopulationANI (popANI). ConANI counts a SNP when two samples differ in the consensus allele at a position in the reference genome, similar to many conventional SNP calling methods. PopANI counts a SNP only if both samples share no alleles. For example, if A/T alleles were found at frequencies of 90/10% and 10/90% in two samples, a consensus SNP would be called because the consensus base is different. A population SNP would not be called because both samples share an A and T allele.
We mapped sequencing reads from all samples against the collection of 1615 unique MAGs and compared strains in samples from different patients with >50% coverage breadth at a depth of five reads (see methods, Supplementary Data 9). This coverage breadth threshold ensures ANI is calculated across the majority of the strains being compared and is recommended by the authors of inStrain. The maximum log-scaled popANI value was taken as representative of the maximal strain sharing between all pairs of patients. Length of hospital overlap was significantly associated with having a more similar microbiome strain (linear regression on log-scaled popANI values, p = 0.0017, Fig. 2g). Time of roommate overlap was not significantly associated (Fig. 2h), but the limited number of patients who were roommates in our dataset may preclude us from detecting an association. Given that the taxonomic composition of patient microbiomes does not converge during periods of roommate or hospital overlap (Fig. 2e, f) and that the majority of compared strains are less similar than the 99.999% popANI strain sharing threshold discussed below, this correlation likely does not indicate frequent patient–patient or hospital–patient transmission. It may represent a subset of similar strains that commonly colonize patient microbiomes upon extended time spent in the hospital. We followed up on the few cases of high-identity strains as evidence for possible transmission events.
HAI organisms that colonize HCT patient microbiomes are part of known, antibiotic-resistant and globally disseminated clades
E. coli and E. faecium are common commensal colonizers of human microbiomes49,50,51. These species can also be pathogenic and contribute to inflammation, dysbiosis, and infection in the host52,53. The specific strain of these species is key in determining the balance between a healthy and diseased state in the microbiome. We compared patient-derived E. coli and E. faecium MAGs with several reference genomes (Supplementary Table 2, Supplementary Data 10) to identify the closest strains or sequence types.
Escherichia coli
The 95% identity, or species-level, cluster of E. coli MAGs contained 47 genomes from 26 patients (Fig. 3). Within this alignment average nucleotide identity (ANI) based tree, we observed two clades of genomes where MAGs from multiple patients had >99.9% ANI. These clades were investigated further as they may represent common sequence types. The first clade contained 15 MAGs from 7 patients; these MAGs had >99.9% ANI to pathogenic E. coli sequence type (ST) 131 clade C2 genomes, including EC95854 and JJ188655. ST131 is an extraintestinal, pathogenic, multidrug-resistant E. coli strain which frequently causes urinary tract infections56. E. coli ST131 often carries extended-spectrum β-lactamase (ESBL) genes which convey a wide range of antibiotic resistance. This sequence type is believed to colonize the intestinal tract even in healthy individuals without antibiotic exposure57 and there are reports of this pathogen causing urinary tract infections in multiple individuals within a household58.
MAGs identified as E. coli, medium quality or above and at least 75% the mean length of the reference genomes are included. Several reference genomes are included and labeled with an asterisk. Clusters at the 99% ANI level corresponding to ST131 (purple) and ST648 (orange) are highlighted. Source Data for this figure are provided in Supplementary Data 10.
The second clade contained 12 MAGs from 5 patients with >99.8% ANI to the pathogenic ST648 representative IMT1631659. ST648 is also an ESBL-producing E. coli strain, but it is not as widespread as ST131. Both STs have been isolated from wastewater60 and ST648 has been isolated from the gut of humans61 and other mammals62. Our finding that E. coli ST648 is also prevalent in HCT patient microbiomes suggests that it may become a pathogen of interest in this patient population in the future.
To understand the antibiotic resistance capabilities of the E. coli strains colonizing these HCT patients, we searched for β-lactamase genes in the E. coli MAGs (see the “Methods” section). The most commonly detected genes were ampH and ampC, which are part of the core E. coli genome and likely do not contribute to antibiotic resistance63 (Fig. S2A). CMY-132 was detected exclusively in MAGs in the ST131 clade, and mutations conveying resistance in gyrA were detected in MAGs in multiple clades. CTX-M-type β-lactamases were detected in several samples, but often within the metagenome rather than within the E. coli MAG, indicating they may be on plasmids or mobile genetic elements that did not bin with the rest of the E. coli genomes.
Enterococcus faecium
The species-level cluster of E. faecium MAGs contained 30 genomes from 20 patients (Fig. 4a). All MAGs were ≥99% identical, suggesting a single ST is present in most patients50. These genomes matched closest to E. faecium ST117, a well-described vancomycin-resistant strain that frequently causes BSIs64. Notably, five other MAGs had ~94% ANI (below the 95% clustering threshold, and therefore not shown in Fig. 3a) to the ST117 clade and >99% ANI to commensal E. faecium strains, including strains Com15 and Com1250. To understand the vancomycin resistance capabilities of these strains, we searched for the seven genes in the vanA operon65. Only 2/30 samples had the full operon present within the E. faecium MAG (Fig. S2b). When we looked in the entire metagenome, 22/30 samples had the full operon present, and the vanA genes were usually detected on a contig that was not assigned to any MAG. Van genes are often carried on mobile genetic elements or plasmids in E. faecium, which frequently do not bin well.
a Alignment average nucleotide Identity (ANI) based tree of E. faecium MAGs. MAGs identified as E. faecium, medium quality or above and at least 75% the mean length of the reference genomes are included. Several reference genomes are included and labeled with an asterisk. Two clades containing samples from multiple patients are highlighted for further comparison. Alignment values used to construct this tree can be found in Supplementary Data 10. b, c Heatmaps showing pairwise popANI values calculated with inStrain for clades B and C, respectively. Color scale ranges from 99.99–100% popANI and is in log space to highlight the samples with high popANI. Cells in the heatmap above the transmission threshold of 99.999% popANI are labeled. Four groups containing samples from multiple patients with popANI values above the transmission threshold are highlighted on the top of the heatmaps. Source Data for this figure are provided in Supplementary Data 9 and 10.
Interestingly, no van genes are present in sample 11342_02, but the full operon is present in the E. faecium MAG in sample 11342_03, collected 14 days later from the same patient. In sample 11342_03, the vanA operon appears on a contig 8.7 kb long with 143× coverage, much lower than the ~2000× coverage of the rest of the E. faecium MAG. Mapping reads against this contig revealed scattered coverage in 11342_02, leading us to believe the operon is present, but not at high enough coverage to be assembled in the earlier sample. We attempted to culture vancomycin-resistant Enterococcus (VRE) from these samples (see the “Methods” section), and positively identified E. faecium by MALDI coupled to time-of-flight mass spectrometry in samples 11342_02 and 11342_03. Taken together, these results suggest that there were at least two strains of E. faecium in the gut microbiota of patient 11342. The low relative coverage of the vanA operon indicates that the VRE strain may have been a small fraction of the total E. faecium pop ulation. Patient 11342 was never prescribed vancomycin (Fig. S7a), so the VRE strain may not have had an advantage in this environment.
VanA genes were also not detected in sample 11349_01, where we observed a nearly identical E. faecium strain to patient 11342 after the patients shared a room for 11 days. When we attempted to culture VRE from 11349_01, a bacterium grew poorly on plates containing vancomycin and was identified as Klebsiella pneumoniae. Therefore, we believe the E. faecium strain in the microbiome of this patient was vancomycin sensitive. If transmission from patient 11342 was responsible for colonization of patient 11349, the vancomycin-sensitive strain may have been transmitted.
Nearly identical strains indicative of putative patient–patient Enterococcus faecium, but not Escherichia coli transmission
We used the results from the inStrain comparison to search for nearly identical bacterial strains, which may be indicative of transmission between patients. To determine a threshold for putative transmission, we examined comparisons in several “positive control” datasets where we expect to find identical strains, either as the result of persistence or transmission: time-course samples from the same HCT patient, stool samples from mother–infant pairs18 and samples from fecal microbiota transplantation donors and recipients23. We often observed 100% popANI in these “positive control” comparisons, indicating that there were no SNPs that could differentiate the strain populations in the two samples (Figs. S3 and S4). Due to expected noise and errors in sequencing data, we set the lower bound for transmission in our HCT cohort at 99.999% popANI, equivalent to 30 population SNPs in a 3 megabase (Mb) genome. The same threshold was used to identify identical strains by the authors of inStrain48.
Escherichia coli
While E. coli genomes in samples collected from the same patient over time were always more similar than the putative transmission threshold, in no case did we observe a pair of samples from different patients with ≥99.999% popANI (Supplementary Data 9). This result suggests that all E. coli strains observed are patient-specific and argues that there are not common strains circulating in the hospital environment or passing between patients. Alternatively, patient–patient transmission or acquisition of common environmental strains is either notably rare or rapid evolution after a patient acquires a new strain is reducing popANI levels below the threshold. Deeper metagenomic sequencing or isolation and sequencing of E. coli strains may allow us to detect transmission in previously missed cases.
Enterococcus faecium
We performed the same analysis in E. faecium and observed four examples where two patients shared a strain with ≥99.999% popANI (Fig. 4b, c). In one case, the two patients were roommates and direct transmission appears to be the most likely route. In the other three cases, epidemiological links were less clear, suggesting the patients may have acquired a similar strain from the hospital environment or through unsampled intermediates. In the following descriptions, samples are referred to by the day of collection, relative to the first sample from patients in the comparison.
Case 1
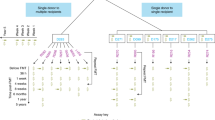
Patients 11342 and 11349 overlapped in the hospital for 21 days and were roommates for 11 days (Fig. 5a). Patient 11342 had a gut microbiome that was dominated by E. faecium; the two samples from this patient have 60% and 87% E. faecium relative abundance. A single sample from patient 11349 was obtained 14 days after starting to share a room with patient 11342. This sample is dominated by Klebsiella pneumoniae, and E. faecium is at 0.4% relative abundance. InStrain comparisons between the E. faecium strains in 11342 (the presumed “donor”) and 11349 (the presumed “recipient”) of the strain revealed 0-2 population SNPs (popANI 100–99.9999%) with 87% of the reference MAG (2.24 Mb) covered ≥5× in both samples. MAGs from each patient were also structurally concordant (representative dotplots in Fig. S6a). These genomes were the most similar out of all E. faecium genomes compared from different patients. Samples from these patients were extracted in different batches and sequenced on different lanes, minimizing the chance that sample contamination or “barcode swapping”66 (see Supplementary Methods) could be responsible for this result. No other strains were shared between these two patients.
Each panel shows the composition of two patients over time. The height of each bar represents the proportion of classified sequence data assigned to each taxon. Samples are labeled relative to the date of the first sample in each set. Bars above each plot represent the approximate time patients spent in the same room (black bars) or in the hospital (gray bars). Red symbols indicate approximate dates of bloodstream infection with the specified organism. Postulated direction of transmission progresses from the top to the bottom patient. Fractions of the bar with >99.999% popANI strains in each panel are indicated with solid colors, and different strains are indicated with hashed colors. All taxa except E. faecium are shown at the genus level for clarity. a Case 1: Putative transmission from patient 11342 to 11349. b Case 2: Putative transmission from patient 11575 to 11568. c Case 3: Putative transmission from patient 11605 to 11673. Source Data for this figure are provided in Supplementary Data 7–9.
Case 2
Patients 11575 and 11568 overlapped in the hospital for 36 days but were never roommates (Fig. 5b). Samples from patient 11575 span 97 days, during which this patient experienced a BSI with Klebsiella pneumoniae and treatment with intravenous (IV) vancomycin, ciprofloxacin, meropenem (Fig. S7c). Antibiotic treatment likely resulted in a reduction in microbiome diversity and domination by E. faecium in samples collected on days 16 and 28. Two samples were collected from patient 11568 on days 28 and 119. The first sample from 11568 was also dominated by E. faecium, but strains from the two patients were distinct (99.95% popANI). 91 days later, the second sample from 11568 has a lower relative abundance of E. faecium but a nearly identical strain to patient 11575. Five population SNPs (99.9997% popANI) were detected with 88% of the reference MAG covered ≥5× in both samples (representative dotplot in Fig. S6b). This suggests that the E. faecium strain in 11568 was replaced by a different strain with high identity to the strain in 11575. Patient 11568 was discharged from the HCT ward during the period between the two samples. The shared strain may represent an acquisition from a common environmental source or transmission from unobserved patients, rather than a direct transmission event between these two patients. While we observed different E. faecium strains in the two samples collected from patient 11568, an E. faecalis strain remained identical between the two time points (99.9997% popANI).
Case 3
Patients 11605 and 11673 did not overlap in the hospital (Fig. 5c). Two samples were collected from 11605 on days 0 and 14. This patient experienced a BSI with E. faecium and treatment with meropenem (Fig. S7e) prior to a sample dominated by the same species on day 14. Patient 11673 experienced a BSI with E. coli and treatment with vancomycin, meropenem, and cefepime (Fig. S7f) prior to the single sample we collected from this patient. Comparing E. faecium strains between the two patients revealed 2 population SNPs (99.9998% popANI) with 48% of the reference MAG covered ≥5× in both samples (representative dotplot in Fig. S6c). Although slightly below the 50% coverage threshold, the high degree of similarity caused us to consider this result. While E. faecium strains in the two patients were nearly identical, the samples were collected 161 days apart and the patients had no overlap in the hospital. This suggests both patients may have acquired the strain from the hospital environment, through transmission from unsampled patients, or another source such as healthcare workers.
Case 4
Patients 11360 and 11789 did not overlap in the hospital. E. faecium remained at relatively low abundance in all samples. Comparing E. faecium strains between patients revealed 5–10 population SNPs (99.9993–99.9996% popANI) with 50–57% genome coverage. Neither patient had a BSI during the sampling period. As these samples were collected at least 428 days apart, a shared source again may be the most likely explanation.
Comparisons with E. faecium and E. coli in published data
The E. faecium and E. coli strains we observe in our patients may be unique to this patient population and hospital environment. Alternatively, they may be hospital acquired strains that are present in other settings around the globe. We searched through several published datasets to differentiate between these possibilities. Our comparison dataset included metagenomic shotgun sequence data from 189 stool samples from adult HCT patients67, 113 stool samples from pediatric HCT patients3,68,69, 732 stool samples from hospitalized infants70 and 58 vancomycin-resistant E. faecium isolates71. Sequence data were downloaded from SRA and processed in the same manner as other short-read data. Each sample was aligned against the E. faecium and E. coli MAGs used in the inStrain analysis above, profiled for SNPs, and compared against samples collected from our HCT patients. Comparisons within our data and comparisons within individual external datasets frequently achieved popANI values of ≥99.999%, typically from comparisons of samples from the same patient over time. Meanwhile, comparisons between our samples and external samples had lower popANI values (Fig. S5).
Comparisons of E. faecium strains in samples from patient 11346 in our dataset and patient 688 in the HCT microbiome dataset collected at Memorial Sloan Kettering Cancer Center67 demonstrated a maximum of 99.9993% popANI (16 population SNPs detected in 2.3 Mb compared). While direct transmission is likely not involved here, this observation does align with the nearly identical E. faecium strains we observed in patients with no hospital or roommate overlap (cases 3 and 4) and speaks to the global dissemination of vancomycin-resistant E. faecium ST 117. Comparing E. coli to external datasets revealed a maximum similarity of 99.996% popANI (200 population SNPs detected in 5.0 Mb compared).
Putative transmission of commensal bacteria
Next, we extended the inStrain analysis to compare all species that were present in multiple patients. We found nearly identical genomes of commensal organisms that may be the result of transmission between patients, as well as several species shared between patients without clear explanations.
Hungatella hathewayi
Patients 11639 and 11662 overlapped in the hospital for 34 days and were roommates for a single day, after which 11639 was discharged (Fig. 6a). Hungatella hathewayi was at 5–10% relative abundance in the two samples from 11639. Patient 11662 developed Streptococcus mitis BSI on day 10 and was treated with IV vancomycin and cefepime (Fig. S8b). The microbiome of this patient recovered with markedly different composition, including an abundant H. hathewayi strain reaching 54% and 17% relative abundance on days 58 and 100, respectively. Comparing H. hathewayi genomes between these two patients revealed 0–1 population SNPs (100–99.99998% popANI) with 94–98% coverage ≥5× (6.9–7.1 Mb sequence covered in both samples). This was the single highest ANI comparison among all strains shared between patients. H. hathewayi MAGs from these patients were also structurally concordant and had few structural variations (Fig. S6d). No other strains were shared between these patients.
Each panel shows the composition of two patients over time. The height of each bar represents the proportion of classified sequence data assigned to each taxon. Samples are labeled relative to the date of the first sample in each set. Bars above each plot represent the approximate time patients spent in the same room (black bars) or in the hospital (gray bars). Red symbols indicate approximate dates of bloodstream infection with the specified organism. Hypothesized direction of transmission progresses from the top to the bottom patient. Fractions of the bar with >99.999% popANI strains in each panel are indicated with solid colors, and different strains are indicated with hashed colors. All taxa except H. hathewayi or A. muciniphila are shown at the genus level for clarity. a Putative case of H. hathewayi transmission from 11639 to 11662. b Putative case of A. muciniphila transmission from 11742 and 11647. Source Data for this figure are provided in Supplementary Data 7–9.
Patient 11662 had H. hathewayi in the first two samples at 1.2% and 0.3% relative abundance, respectively. Although we were limited by coverage, comparing early to late samples with inStrain revealed 472 population SNPs in 3% of the genome that was covered at least 5×, implying 11662 was initially colonized by a different H. hathewayi strain, which was eliminated and subsequently replaced by the strain present in 11639. Given that samples were collected weekly, determining the direction of transmission is challenging. However, 11639 to 11662 appears to be the most likely direction, given the sampling times and perturbation 11662 experienced. However, it is possible that transmission occurred in the opposite direction or from a common source. Interestingly, 11662 was also re-colonized with Flavonifractor plautii in later samples. This strain was different from the strain in earlier samples from this patient, as well as all other Flavonifractor strains in our sample collection.
H. hathewayi is known to form spores and is able to persist outside a host for days72. Although these patients were only roommates for a single day, 11662 remained in the same room for 4 days after 11639 was discharged, increasing the chance that a H. hathewayi spore could be transmitted from a surface in the shared room or bathroom. The question remains as to why transmission of H. hathewayi is not more common, given it is found at ≥1% relative abundance in 31 patients. Perhaps the earlier colonization of the microbiome of 11662 with a different H. hathewayi strain was key—the microbiome in this patient was “primed” to receive a new strain of the same species, despite the significant perturbation this patient experienced.
Notably, H. hathewayi was recently reclassified from Clostridium hathewayi73, and was previously shown to induce regulatory T-cells and suppress inflammation74. Although the interaction of this microbe with the HCT process is not known, it may be interesting to investigate further given that the microbe may be transmitted between individuals and may contribute to inflammation suppression that may be relevant in diseases such as graft-vs.-host disease. However, H. hathewayi may not be entirely beneficial or harmless and has been reported to cause BSI and sepsis in rare cases75,76.
Akkermansia muciniphila
Patients 11742 and 11647 overlapped in the hospital for 11 days and were roommates for nine days (Fig. 6b). Patient 11647 experienced a BSI with Klebsiella pneumoniae (perhaps related to previous K. pneumoniae domination of the microbiome) and was treated with piperacillin-tazobactam and cefepime (Fig. S8d). The final sample from 11647 has Akkermansia muciniphila at 9.4% relative abundance, while the single sample from 11742 was dominated by A. muciniphila (85% relative abundance). Comparing these genomes revealed 0 population SNPs and 7 consensus SNPs with 90% coverage, as well as concordant MAGs from each sample (Fig. S6e). No other strains were shared between these two patients.
In contrast to H. hathewayi, A. muciniphila is not known to form spores, which may reduce the chance of this microbe being transmitted. However, it is an aerotolerant anaerobe that may survive in oxygen for short periods of time77. The microbiome domination of 11742 with A. muciniphila and the relatively long overlap period of 9 days in the same room may provide a greater “infectious dose” (abundance*exposure time) to the recipient patient.
Widespread strain sharing of commercially available probiotic organisms
Several organisms were found with identical or nearly identical genomes across multiple patient microbiomes without clear epidemiological links. The largest clade was found for Lactobacillus rhamnosus, which included 11 samples collected from eight patients over a span of 2.5 years (Fig. 7a, d). All 11 samples in this clade had pairwise popANI of ≥99.999%, and in a subset of eight samples from seven patients, all pairs were identical from a popANI perspective (100%). Of the eight patients in this clade, only two pairs were roommates or overlapped in the hospital (patients 11537/11547 and 11647/11662, roommates for three days and one day, hospital overlap for 20 and 44 days, respectively). All 11 samples in this clade were collected after HCT (median time from HCT to first sample 43 days, range 12–93 days), and L. rhamnosus always had <0.1% relative abundance in pre-HCT samples, when samples were available. Five of eight patients were discharged from the hospital after HCT and prior to acquiring L. rhamnosus, which we observed in a sample collected during a subsequent admission. We also observe L. rhamnosus falling below 0.1% relative abundance in a subsequent sample in five patients, suggesting that this strain may be a transient colonizer of the microbiome (Fig. 7e). We evaluated antibiotic prescriptions in these patients and found that acquisition and loss of L. rhamnosus typically occur independently of antibiotic use. For example, in patient 11537, L. rhamnosus is first detected and expands to 23.4% relative abundance while the patient is prescribed ciprofloxacin and cefepime. L. rhamnosus declines in relative abundance after the antibiotic prescription ends (Fig. S9). Similar clades of high-identity genomes from different patients were found for Lactobacillus gasseri (Fig. 7b) and Streptococcus thermophilus (Fig. 7c).
Population ANI-based tree of (a) Lactobacillus rhamnosus, (b) Lactobacillus gasseri, (c) Streptococcus thermophilus strains present in patient samples. Clades containing samples from different patients with ≥99.999% popANI are highlighted with a gray background. Clades with 100% popANI between all pairs are additionally bolded and italicized. d Timeline of approximate date of samples containing a L. rhamnosus strain in the transmission cluster in (a). Patients who were discharged from the hospital after HCT and prior to acquiring L. rhamnosus are bolded and italicized. e Microbiome composition of patient 11537. L. rhamnosus abundance at each time point is indicated above the bar. This patient received HCT on relative day 3. Source Data for this figure are provided in Supplementary Data 7–10.
Given that we did not observe hospital or roommate overlap between most patients in the L. rhamnosus cluster, the most likely explanation is that patients acquired this strain from a common source. L. rhamnosus is a component of several commercially available probiotic supplements, is present in certain live active-culture foods such as yogurt, and is among the most commonly prescribed probiotic species in US hospitals78. However, HCT recipients were not allowed to take probiotics or consume high-bacteria dairy products, such as probiotic yogurt or soft cheese, while inpatients on the HCT ward. We also verified that no prescriptions were written for probiotics by examining electronic health records. A majority of patients were discharged from the hospital between HCT and acquiring the L. rhamnosus strain, which may have provided them with the opportunity to consume a probiotic supplement or dairy product. Contact with a family member or other individual who had the strain in their microbiome could also be responsible for colonization of the HCT patient.
If this L. rhamnosus strain is a commonly used probiotic supplement or is found in commonly consumed dairy products, it may be found in other gut microbiome sequencing datasets. Comparing MAGs from this cluster against all Genbank genomes revealed a maximum alignment-based ANI of 99.95% to L. rhamnosus ATCC 853079. Instrain-based comparisons against this reference had a maximum popANI of 99.98%, below the putative transmission threshold. We then searched against all genomes in the Unified Human Gastrointestinal Genome collection41 and identified two genomes that were nearly identical to the strain found in HCT patients. These genomes were originally from the Human Gastrointestinal Bacteria Culture Collection80 (accessions ERR2221226 and ERR1203919, belonging to the same isolate per a personal communication with the authors). Assembled isolate and patient-derived genomes had ≥99.99% ANI; inStrain-based SNP comparisons had ≥99.999% popANI. This suggests that a L. rhamnosus strain that is nearly identical to the genomes in our HCT patients has been isolated from human stool in the past.
Discussion
Our investigation using high-resolution metagenomic sequencing attempts to quantify if and when patient-patient microbiome transmission is involved in the spread of pathogenic organisms. We first found that hospitalized HCT patients frequently harbor HAI organisms in their gut microbiome, validating previous studies which used culture-based approaches or 16S rRNA sequencing6,26,27. MAGs created from patient samples had a high identity to several globally disseminated and antibiotic-resistant sequence types, including Escherichia coli ST131 and ST648. Interestingly, whereas ST131 is a well-recognized multi-drug resistant pathogen, ST648 is nearly as prevalent as ST131 in our sample collection, and may thus be an emerging pathogen in this patient population.
Despite the high degree of hospital and roommate overlap between patients in our study, we found no association between these factors and the taxonomic similarity of patient microbiomes, and only a weak association between hospital overlap and maximum strain identity. These findings suggest that patient-patient transmission is not driving microbiome composition, but individual strains may still be shared between patients. We did not identify any pairs of patients harboring E. coli strains with popANI values above the 99.999% popANI transmission threshold. Taken together with the observation that E. coli is commonly detected in the patient’s microbiome upon admission, this finding argues that patients usually enter the hospital with an “individual-specific” E. coli strain and do not frequently transmit it to others. An exclusion principle may be at play, where an E. coli niche can only be filled by a single strain or a small number of strains, and new strains are unlikely to engraft when the niche is already occupied. In contrast to E. coli, we observed four pairs of patients with E. faecium strains that were more similar than 99.999% popANI. In one case, the two patients spent 11 days sharing a room and bathroom prior to observing the shared strain. Direct links between patients were less clear or non-existent in the other three cases, and transmission through unsampled intermediates or acquisition from an environmental source may have been responsible. We also found evidence for an E. faecium strain in a published dataset from HCT patient microbiomes67 that had above 99.999% popANI to a strain in our sample collection. While this finding is likely not the result of patient-patient transmission, it does indicate that very similar strains may exist within patients in different geographic locations.
We then expanded the transmission analysis to examine all species that were present in the microbiome of multiple patients. Identical Hungatella hathewayi strains were found in two patients who were in the hospital together for 34 days and roommates for a single day. Earlier samples from patient 11662 had a significantly different H. hathewayi strain than the strain present in later samples. It is possible that the earlier colonization with the same species exhibited a priority effect81 and primed this individual to be re-colonized. In another set of patients who overlapped in the hospital for 11 days and were roommates for nine days, we identified identical Akkermansia muciniphila strains. In both examples, the likely “recipient” patient experienced BSI prior to the putative transmission event. The subsequent antibiotic treatment initiated for the treatment of BSI resulted in vast microbiome modification and simplification, which may have opened a niche for the new organism to engraft into. Both H. hathewayi and A. muciniphila can survive outside the host for periods of time, but H. hathewayi can form spores that enable it to live in aerobic conditions for days72. In the case of H. hathewayi transmission, patient 11662 remained in the same room with a shared bathroom after the single day of overlap with patient 11639. Spores surviving on surfaces may be responsible for transmission, given the relatively short period of overlap. In these cases, patient-patient transmission may help in the recovery of microbiome diversity following BSI and may play a role in ameliorating post-HCT inflammatory processes, such as acute graft-vs-host disease.
Finally, we observed identical probiotic species in multiple patients without clear geographic or temporal links, including Lactobacillus rhamnosus, Lactobacillus gasseri, and Streptococcus thermophilus. Acquisition from a commercially available probiotic or live-active culture food appears to be the most likely explanation. While patients hospitalized for HCT were not allowed to consume probiotics or high-bacterial dairy foods, a majority of patients were discharged after HCT and prior to a subsequent admission, upon which L. rhamnosus was detected. Many patients lost the strain in later samples, independent of antibiotic prescription, suggesting that L. rhamnosus was a transient colonizer. This matches the observation that abundance of probiotic species in the gut often declines after supplementation ends20.
The healthy adult gut microbiome is relatively resistant to perturbation and colonization with new strains or species19. By contrast, mother-to-infant transmission of bacteria and phages is common and well-described16,17,18. Patients in our study often shared spaces, were exposed to dramatic “niche clearing” therapies and were often immunosuppressed. We frequently observed patients acquiring new organisms into their gut microbiome during their hospital stay, especially following BSI. Still, we found that patient-patient transmission of gut microbes is relatively rare. This suggests that other factors such as age, rather than perturbation or microbial exposure, may play the largest role in microbiome transmission. There are also several alternative explanations for the relative lack of transmission between patients. First, the adult gut microbiome may remain densely colonized even when dramatically perturbed by antibiotics and chemotherapy, and thus may be resistant to invasion with new strains. Strains we observed in later samples may have existed at low very levels in earlier samples from the individual, therefore evading detection. Second, it is possible that the microbiome of healthcare workers, hospital visitors, or other staff serves as the source of newly colonizing strains. Third, it is possible that the built environment, equipment used in the care of these individuals, and other environmental sources such as food and personal items harbored the microbes that were later transmitted. As we did not sample these other potential sources, it is difficult to know the extent to which they contributed to the collective reservoir of potentially transmitted organisms. Fourth, adaptive evolution may rapidly change the genomes of newly acquired microbes, as has been described for E. faecium in the human gut82 and E. coli in the mouse gut83,84,85. Rapid evolution would move the genomes of transmitted microbes below the popANI transmission threshold. Finally, transmitted organisms may be killed by antibiotics before they can establish a community within the host and thus be detected with metagenomic sequencing.
Our findings have important implications for hospital management and infection prevention. 55/149 patients (37%) in our study experienced BSIs, which is comparable to the rate of BSI in other transplant centers86. Our findings suggest that microbiome transmission does not play a large role in spreading infections among HCT patients, and that established contact precautions and procedures for patient isolation were working as intended. Recently, the HCT ward at our hospital moved to a new location with exclusively single rooms, which may further reduce the opportunity for transmission.
Our analysis of transmission of microbes between HCT patients does have several limitations. We analyzed hundreds of samples collected over many years, but most sampling was done on a weekly basis. We did not explicitly collect samples on the day of admission or discharge. Our sample collection also ignores previous hospital stays, either in a different ward in our hospital, or other hospitals entirely, that may be responsible for the acquisition of HAI organisms. Our study was focused on collecting fewer samples from a larger number of individuals, potentially limiting our ability analyze microbiome changes over time. By contrast, the second largest study using shotgun metagenomics to study the microbiome of HCT patients had much more dense sampling, analyzing an average of 8 samples from each of the 49 patients in the study67. We also did not perform any sampling of the hospital environment, healthcare workers or visitors, which would allow us to track transmission patterns in detail and more conclusively state where newly acquired microbes originated87. Our work is also entirely based on metagenomic sequencing data, which has its own challenges and sources of bias, including “barcode swapping”66, which could contribute to false positive transmission findings. To address this, we measured the impact of barcode swapping in linked-read data, and eliminated linked-read and short-read comparisons where a finding of identical strains could be the result of barcode swapping (see Supplementary Methods). Additionally, metagenomic sequencing may fail to detect lowly-abundant colonizers, especially when samples are contaminated with host DNA88, which is often the case in stool samples from HCT patients. These challenges undoubtedly affect our sensitivity and specificity in measuring acquisition and transmission of both pathogenic and commensal microbes. While our comparison methods were sensitive to strain populations in the gut microbiome, we did not attempt to phase strain haplotypes. Haplotype phasing with long-read sequencing technology like Nanopore or PacBio89,90,91 could help determine whether sets of SNPs occurred in the same or different strains.
Our study leaves several questions unanswered that we hope future work on microbiome transmission will attempt to answer. First, our findings need to be validated in an external cohort in a different hospital. Collecting stool samples during an infection outbreak may lead to more transmission events being identified and may implicate microbiome transmission in perpetuating the outbreak. Similar experiments in a pediatric patient population may reveal more gut-to-gut transmission, as young children have microbiomes that are still developing and more susceptible to colonization with new species. Our work did not investigate any possible sources of microbial transmission other than the gut microbiota of HCT patients. A more granular study where samples are collected from the hospital environment, as well as hand swabs and stool samples from healthcare workers, visitors, and family members, is clearly indicated by these early results. As more transmission events are observed with high-resolution genomic methods, we will start to uncover the general principles governing community assembly in the human microbiome. These new insights may help prevent infections and other illnesses in this vulnerable patient population in the future.
Methods
Cohort selection
HCT patients were recruited at the Stanford Hospital Blood and Marrow Transplant Unit under an IRB protocol approved by The Stanford University Research Compliance Office (Protocol #8903; Principal Investigator: Dr. David Miklos, co-Investigator: Drs. Ami Bhatt and Tessa Andermann). Informed consent was obtained from all individuals whose samples were collected. Consent included explicit agreement to long-term storage of samples for future research studies and use of health information including but not limited to medication data as well as transplant outcomes. Stool samples were placed at 4 °C immediately upon collection and processed for storage at the same or following day. Stool samples were aliquoted into 2-mL cryovial tubes and homogenized by brief vortexing. The aliquots were stored at −80 °C until extraction.
We identified all samples that had been sequenced previously by our group. Samples were selected for linked-read sequencing to augment this collection. We examined the network of patient roommate overlaps to find cases where we were likely to uncover transmission events, if they were happening. These included patient pairs from whom we ideally had samples before and after the roommate overlap period. 96 samples that provided the best coverage of roommate overlaps were selected for linked-read sequencing.
The following clinical data were extracted from the electronic health record: demographic information, underlying disease, type of transplantation (allogeneic vs. autologous), date and type of bloodstream infection, medication prescriptions, time of admission and discharge and location of patients (rooms) over time. Hospital-wide BSI data were obtained from an electronic report generated by the clinical microbiology laboratory. Medication prescription data was filtered by the following criteria:
-
1.
Only entries for antibiotics, antifungals, antivirals, antibacterials, and Pneumocystis jirovecii pneumonia prophylaxis were retained.
-
2.
Medications with a missing start or end date were excluded.
-
3.
Medications with a frequency of “PRN” (pro re nata, or as needed) or a prescription status of “Canceled” were excluded.
-
4.
Medications with a difference between start and end date of less than one day were excluded.
-
5.
Medications prescribed to be taken by eyes, ears, topical application, or “swish and spit” were excluded.
-
6.
Medication prescriptions occurring outside the window of HCT date ± 100 days were excluded for the aggregated analysis.
We do acknowledge the challenge of working with electronic health record data, and recognize that there is a disconnect between medications prescribed and medications consumed by a patient.
DNA Extraction, library preparation and sequencing
DNA was extracted from stool samples using a mechanical bead-beating approach with the Mini-Beadbeater-16 (BioSpec Products) and 1-mm diameter zirconia/silica beads (BioSpec Products) followed by the QIAamp Fast DNA Stool Mini Kit (Qiagen) according to manufacturer’s instructions. Bead-beating consisted of 7 rounds of alternating 30 s bead-beating bursts followed by 30 s of cooling on ice. For samples subjected to linked-read sequencing, DNA fragments less than ~2 kb were eliminated with a SPRI bead purification approach92 using a custom buffer with minor modifications: beads were added at 0.9×, and eluted DNA was resuspended in 50 μl of water. DNA concentration was quantified using a Qubit fluorometer (Thermo Fisher Scientific). DNA fragment length distributions were quantified using a TapeStation 4200 (Agilent Technologies).
Short-read sequencing libraries were prepared with either the Nextera Flex or Nextera XT kit (Illumina) according to manufacturer’s instructions. Linked-read sequencing libraries were prepared on the 10× Genomics Chromium platform (10× Genomics). Linked-read libraries have a single sample index, and were pooled to minimize the possibility of barcode swapping between samples from patients who were roommates (see Supplementary Methods). Libraries were sequenced on an Illumina HiSeq 4000 (Illumina).
Sequence data processing
TrimGalore version 0.5.093 was used to perform quality and adapter trimming with the flags “–clip_R1 15–clip_R2 15–length 60”. SeqKit version 0.9.194 was used to remove duplicates in short-read data with the command “seqkit rmdup–by-seq”. Due to excessive processing time, this step was skipped for linked-read data. Reads were mapped against the GRCh38 assembly of the human genome using BWA version 0.7.17-r118895 and only unmapped reads were retained. Quality metrics were verified with FastQC version 0.11.896. Bioinformatics workflows were implemented with Snakemake97.
Short-read classification with Kraken2
We classified all short-read data with a Kraken243 database containing all bacteria, viral and fungal genomes in NCBI GenBank assembled to complete genome, chromosome or scaffold quality as of January 2020. Human and mouse reference genomes were also included in the database. A Bracken44 database was also built with a read length of 150 and k-mer length of 35. Kraken2 version 2.0.8-beta and Bracken version 2.0 were run in paired-end mode with default parameters. Bray–Curtis distances were calculated with the R package vegan98 version 2.5–7.
Assembly and binning
Short-read sequencing samples were assembled using SPAdes version 3.14.033 using the ‘–meta’ flag. Linked-read sequencing samples were assembled with Megahit version 1.2.934 to generate seed contigs, which were then assembled with the barcode-aware assembler Athena version 1.329. Metagenome-assembled genomes (MAGs) were binned with Metabat2 version 2.1535, Maxbin version 2.2.736 and CONCOCT version 1.1.037 and aggregated using DASTool version 1.1.138. MAG completeness and contamination was evaluated using CheckM version 1.0.1339 and MAG quality was evaluated by the standards set in40. All assembled contigs were classified with Kraken2 using the database described above. To generate bin identifications, contig classifications were pooled such that contigs making up at least two-thirds the length of the bin were classified as a particular species. If a classification could not be assigned at the species level, the process was repeated at the genus level, and so on.
Genome de-replication and SNP profiling
MAGs were filtered to have minimum completeness 50% and maximum contamination 15% as measured by CheckM, then were de-replicated with dRep version 2.6.242 with default parameters except the primary clustering threshold set to 0.95. In further steps, a single de-replicated genome will be referred to as a cluster. Reads from all samples were mapped against the de-replicated set of genomes with BWA. Clusters that had >1× average coverage in at least two samples were retained for further analysis. Individual bam files were extracted for each sample–cluster pair with at least 1× coverage. Bam files were randomly subsetted to a maximum of 2 million reads for computational efficiency. Alignments were profiled and then compared across samples with inStrain version 1.3.1148 using default parameters.
Building phylogenetic trees
MAG average nucleotide identity (ANI) trees (Figs. 2 and 3) were created using the pairwise alignment values from dRep, which uses the MUMmer program99. MAGs were filtered to be at least 75% the length of the mean length of reference genomes used in the tree. Reference genomes were selected by searching literature for collections of well-described isolates with genomes available. References that were not relevant and clustered in isolated sections of the tree were removed. Pairwise ANI values were transformed into a distance matrix and clustered using the ‘hclust’ function with the ‘average’ method in R version 4.0.3100. Heatmaps were created using pairwise popANI values from inStrain, transformed into a distance matrix, and hierarchically clustered using the ‘ward.D2’ method.
Determining transmission thresholds
To determine the ANI threshold to call a comparison a “putative transmission event” we evaluated the distributions of ANI values for within- and between-patient comparisons for different species (Fig. S3). We often detected zero population SNPs in time course samples from the same patient, including E. faecium in a pair of samples collected from the same patient 323 days apart. Meanwhile, between-patient comparisons typically had lower ANI values. To verify that transmission events would also result in population ANI values near 100%, we examined external datasets where transmission of bacteria in the microbiome is known to occur as a “positive control”. We gathered sequencing data from stool samples of matched mother-infant pairs18 and fecal microbiota transplantation donors and recipients23 and processed them with the same methods. In these datasets, we regularly observed genomes with 100% popANI between matched individuals, and did not find cases of 100% popANI between unmatched individuals (Fig. S4). In the ideal cases, we expect transmission of bacteria between the microbiomes of HCT patients to result in genomes with 100% popANI. However, the measured genomes may not reach this level of identity, due to mutations since the transmission event, sequencing errors, or other factors. Therefore, we set the transmission threshold at 99.999% popANI, equivalent to 30 population SNPs in a 3 megabase (Mb) genome. Although this threshold is stringent, we recognize that it may allow for false positives where two closely related strains exist in different patients solely by chance.
Despite our efforts to minimize the impact of barcode swapping on detecting transmission (see Supplementary Methods), we still identified many comparisons with >99.999% popANI that we believe to be false positives. These were filtered out based on the following criteria: short read samples from different patients that were sequenced on the same lane and shared one index sequence or linked read samples that were sequenced on the same lane and reads mapping to the organism shared >40% of barcodes. We also removed pairs that could be affected by “secondary” swapping, where the two samples were not directly affected, but an interaction between other samples from the two patients could cause false positives. In total, we removed 31 comparisons from the final table with >99.999% popANI.
Pairwise MAG comparison
MAGs were aligned with the mummer program using default settings99 and filtered for 1-1 alignments. Dotplots were visualized with the “Dot” program101 filtering for non-repetitive alignments ≥1 kb.
Antibiotic resistance gene detection
Antibiotic resistance genes (ARGs) were profiled in contigs from all samples using Resistance Gene Identifier version 5.1.1 (RGI) and the Comprehensive Antibiotic Resistance Database (CARD)102 with default parameters. Genes were counted if they met the “strict” or “perfect” threshold from RGI. ARGs were annotated both if they occurred on a contig in the MAG of interest, or anywhere in the metagenomic assembly.
Isolation, culture and identification of VRE organisms
Stool samples from patients 11342 and 11349 were resuspended in glycerol and streaked on SpectraVRE (R01830, Thermo Fischer Scientific) plates and incubated at 35 °C overnight. The following day, four colonies from each of the plates that displayed growth were picked and streaked out on a separate quadrant of fresh SpectraVRE plates. These Round 1 (R1) plates were incubated at 35 °C overnight and checked for growth the following day. For each R1 plate, a colony was picked from each quadrant and streaked out on a new quadrant of a fresh SpectraVRE plate. These Round 2 (R2) plates were incubated at 35 °C overnight and checked for growth the following day. For each R2 plate, a colony from each quadrant was subjected to MALDI-TOF bacterial species identification analysis on a Bruker Biotyper (Burker) per manufacturer instructions
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Raw sequence data for this manuscript, when not previously published, have been uploaded to NCBI SRA under project number PRJNA707487. MAGs generated in this study are available from Zenodo in a tar.gz archive (8.4 Gb) under record number 5768708. Additional information on patient clinical metadata, sequencing datasets generated, statistics of MAGs generated, kraken2 classification results, ANI and inStrain results, days of overlap between patients and antibiotic prescription are available as Supplementary Data files. The kraken2 classification database was built from genomes contained in NCBI Genbank [https://www.ncbi.nlm.nih.gov/genbank/]. The Comprehensive Antibiotic Resistance Database is available online [https://card.mcmaster.ca/].
Code availability
Workflows for data processing, Kraken2 Classification, MAG binning, and inStrain comparisons can be found at https://github.com/bhattlab/bhattlab_workflows103. A workflow to process kraken2 results into taxonomic matrices is available at https://github.com/bhattlab/kraken2_classification104.
References
Young, J.-A. H. et al. Infections after transplantation of bone marrow or peripheral blood stem cells from unrelated donors. Biol. Blood Marrow Transplant. 22, 359–370 (2016).
See, I. et al. Mucosal barrier injury laboratory-confirmed bloodstream infection: results from a field test of a new National Healthcare Safety Network definition. Infect. Control Hosp. Epidemiol. 34, 769–776 (2013).
Kelly, M. S. et al. Gut colonization preceding mucosal barrier injury bloodstream infection in pediatric hematopoietic stem cell transplantation recipients. Biol. Blood Marrow Transplant. 25, 2274–2280 (2019).
Tamburini, F. B. et al. Precision identification of diverse bloodstream pathogens in the gut microbiome. Nat. Med. 1, https://doi.org/10.1038/s41591-018-0202-8 (2018).
Zhai, B. et al. High-resolution mycobiota analysis reveals dynamic intestinal translocation preceding invasive candidiasis. Nat. Med. 26, 59–64 (2020).
Taur, Y. et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin. Infect. Dis. 55, 905–914 (2012).
Ubeda, C. et al. Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J. Clin. Investig. 120, 4332–4341 (2010).
Jenq, R. R. et al. Intestinal blautia is associated with reduced death from graft-versus-host disease. Biol. Blood Marrow Transplant. 21, 1373–1383 (2015).
Mathewson, N. D. et al. Gut microbiome-derived metabolites modulate intestinal epithelial cell damage and mitigate graft-versus-host disease. Nat. Immunol. 17, 505–513 (2016).
Peled, J. U. et al. Microbiota as predictor of mortality in allogeneic hematopoietic-cell transplantation. N. Engl. J. Med. 382, 822–834 (2020).
Shono, Y. et al. Increased GVHD-related mortality with broad-spectrum antibiotic use after allogeneic hematopoietic stem cell transplantation in human patients and mice. Sci. Transl. Med. 8, 339ra71 (2016).
Taur, Y. et al. The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood 124, 1174–1182 (2014).
Weber, D. et al. Microbiota disruption induced by early use of broad-spectrum antibiotics is an independent risk factor of outcome after allogeneic stem cell transplantation. Biol. Blood Marrow Transplant. 23, 845–852 (2017).
Livornese, L. L. Jr et al. Hospital-acquired infection with vancomycin-resistant Enterococcus faecium transmitted by electronic thermometers. Ann. Intern. Med. 117, 112–116 (1992).
Raven, K. E. et al. Complex routes of nosocomial vancomycin-resistant Enterococcus faecium transmission revealed by genome sequencing. Clin. Infect. Dis. 64, 886–893 (2017).
Bäckhed, F. et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 17, 690–703 (2015).
Siranosian, B. A., Tamburini, F. B., Sherlock, G. & Bhatt, A. S. Acquisition, transmission and strain diversity of human gut-colonizing crAss-like phages. Nat. Commun. 11, 1–11 (2020).
Yassour, M. et al. Strain-level analysis of mother-to-child bacterial transmission during the first few months of life. Cell Host Microbe 24, 146–154e4 (2018).
Lozupone, C. A., Stombaugh, J. I., Gordon, J. I., Jansson, J. K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230 (2012).
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174, 1406–1423e16 (2018).
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388–1405e21 (2018).
Brito, I. L. et al. Transmission of human-associated microbiota along family and social networks. Nat. Microbiol. 1, https://doi.org/10.1038/s41564-019-0409-6 (2019).
Smillie, C. S. et al. Strain tracking reveals the determinants of bacterial engraftment in the human gut following fecal microbiota transplantation. Cell Host Microbe 23, 229–240e5 (2018).
Watson, A. R. et al. Adaptive ecological processes and metabolic independence drive microbial colonization and resilience in the human gut. Preprint at bioRxiv https://doi.org/10.1101/2021.03.02.433653 (2021).
Li, S. S. et al. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 352, 586–589 (2016).
Andermann, T. M. et al. The microbiome and hematopoietic cell transplantation: past, present, and future. Biol. Blood Marrow Transplant. 24, 1322–1340 (2018).
Rashidi, A. et al. Pre-transplant recovery of microbiome diversity without recovery of the original microbiome. Bone Marrow Transplant. 1, https://doi.org/10.1038/s41409-018-0414-z (2018).
Shono, Y. & van den Brink, M. R. M. Gut microbiota injury in allogeneic haematopoietic stem cell transplantation. Nat. Rev. Cancer 18, 283–295 (2018).
Bishara, A. et al. High-quality genome sequences of uncultured microbes by assembly of read clouds. Nat. Biotechnol. https://doi.org/10.1038/nbt.4266 (2018).
Kang, J. B. et al. Intestinal microbiota domination under extreme selective pressures characterized by metagenomic read cloud sequencing and assembly. BMC Bioinform. 20, 585 (2019).
Truong, D. T., Tett, A., Pasolli, E., Huttenhower, C. & Segata, N. Microbial strain-level population structure and genetic diversity from metagenomes. Genome Res. 27, 626–638 (2017).
Van Rossum, T., Ferretti, P., Maistrenko, O. M. & Bork, P. Diversity within species: interpreting strains in microbiomes. Nat. Rev. Microbiol. 18, 491–506 (2020).
Nurk, S., Meleshko, D., Korobeynikov, A. & Pevzner, P. A. metaSPAdes: a new versatile metagenomic assembler. Genome Res. 27, 824–834 (2017).
Li, D., Liu, C.-M., Luo, R., Sadakane, K. & Lam, T.-W. MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinforma. Oxf. Engl. 31, 1674–1676 (2015).
Kang, D. D. et al. MetaBAT 2: an adaptive binning algorithm for robust and efficient genome reconstruction from metagenome assemblies. PeerJ 7, e7359 (2019).
Wu, Y.-W., Simmons, B. A. & Singer, S. W. MaxBin 2.0: an automated binning algorithm to recover genomes from multiple metagenomic datasets. Bioinformatics 32, 605–607 (2016).
Alneberg, J. et al. Binning metagenomic contigs by coverage and composition. Nat. Methods 11, 1144–1146 (2014).
Sieber, C. M. K. et al. Recovery of genomes from metagenomes via a dereplication, aggregation and scoring strategy. Nat. Microbiol. 1, https://doi.org/10.1038/s41564-018-0171-1 (2018).
Parks, D. H., Imelfort, M., Skennerton, C. T., Hugenholtz, P. & Tyson, G. W. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 25, 1043–1055 (2015) .
Bowers, R. M. et al. Minimum information about a single amplified genome (MISAG) and a metagenome-assembled genome (MIMAG) of bacteria and archaea. Nat. Biotechnol. 35, 725–731 (2017).
Almeida, A. et al. A unified catalog of 204,938 reference genomes from the human gut microbiome. Nat. Biotechnol. 39, 105–114 (2021).
Olm, M. R., Brown, C. T., Brooks, B. & Banfield, J. F. dRep: a tool for fast and accurate genomic comparisons that enables improved genome recovery from metagenomes through de-replication. ISME J. 11, 2864–2868 (2017).
Wood, D. E., Lu, J. & Langmead, B. Improved metagenomic analysis with Kraken 2. Genome Biol. 20, 1–13 (2019).
Lu, J., Breitwieser, F. P., Thielen, P. & Salzberg, S. L. Bracken: estimating species abundance in metagenomics data. PeerJ Comput. Sci. 3, e104 (2017).
Centers for Disease Control and Prevention (U.S.). Diseases and Organisms in Healthcare Settings | HAI | CDC. https://www.cdc.gov/hai/organisms/organisms.html (2019).
Lloyd-Price, J. et al. Strains, functions and dynamics in the expanded Human Microbiome Project. Nature 550, 61–66 (2017).
Abranches, J. et al. Biology of oral Streptococci. Microbiol. Spectr. 6, (2018).
Olm, M. R. et al. inStrain profiles population microdiversity from metagenomic data and sensitively detects shared microbial strains. Nat. Biotechnol. https://doi.org/10.1038/s41587-020-00797-0 (2021).
Dubin, K. & Pamer, E. G. Enterococci and their interactions with the intestinal microbiome. Microbiology Spectrum 5, (2018).
Palmer, K. L. et al. Comparative genomics of Enterococci: variation in Enterococcus faecalis, clade structure in E. faecium, and defining characteristics of E. gallinarum and E. casseliflavus. mBio 3, (2012).
Tenaillon, O., Skurnik, D., Picard, B. & Denamur, E. The population genetics of commensal Escherichia coli. Nat. Rev. Microbiol. 8, 207–217 (2010).
Kittana, H. et al. Commensal Escherichia coli strains can promote intestinal inflammation via differential interleukin-6 production. Front. Immunol. 9, (2018).
Seishima, J. et al. Gut-derived Enterococcus faecium from ulcerative colitis patients promotes colitis in a genetically susceptible mouse host. Genome Biol. 20, 252 (2019).
Forde, B. M. et al. The complete genome sequence of Escherichia coli EC958: a high quality reference sequence for the globally disseminated multidrug resistant E. coli O25b:H4-ST131 clone. PLoS ONE 9, e104400 (2014).
Andersen, P. S. et al. Complete genome sequence of the epidemic and highly virulent CTX-M-15-producing H30-Rx subclone of Escherichia coli ST131. Genome Announc. 1, (2013).
Gurnee, E. A. et al. Gut colonization of healthy children and their mothers with pathogenic ciprofloxacin-resistant Escherichia coli. J. Infect. Dis. 212, 1862–1868 (2015).
Whitmer, G. R., Moorthy, G. & Arshad, M. The pandemic Escherichia coli sequence type 131 strain is acquired even in the absence of antibiotic exposure. PLoS Pathog. 15, e1008162 (2019).
Madigan, T. et al. Extensive household outbreak of urinary tract infection and intestinal colonization due to extended-spectrum β-lactamase-producing Escherichia coli sequence type 131. Clin. Infect. Dis. 61, e5–e12 (2015).
Schaufler, K. et al. Genomic and functional analysis of emerging virulent and multidrug-resistant Escherichia coli lineage sequence type 648. Antimicrob. Agents Chemother. 63, (2019).
Paulshus, E. et al. Repeated isolation of extended-spectrum-β-lactamase-positive Escherichia coli sequence types 648 and 131 from community wastewater indicates that sewage systems are important sources of emerging clones of antibiotic-resistant bacteria. Antimicrob. Agents Chemother. 63, (2019).
Müller, A., Stephan, R. & Nüesch-Inderbinen, M. Distribution of virulence factors in ESBL-producing Escherichia coli isolated from the environment, livestock, food and humans. Sci. Total Environ. 541, 667–672 (2016).
Ewers, C. et al. CTX-M-15-D-ST648 Escherichia coli from companion animals and horses: another pandemic clone combining multiresistance and extraintestinal virulence? J. Antimicrob. Chemother. 69, 1224–1230 (2014).
Henderson, T. A., Young, K. D., Denome, S. A. & Elf, P. K. AmpC and AmpH, proteins related to the class C beta-lactamases, bind penicillin and contribute to the normal morphology of Escherichia coli. J. Bacteriol 179, 6112–6121 (1997).
Tedim, A. P. et al. Complete genome sequences of isolates of Enterococcus faecium sequence type 117, a globally disseminated multidrug-resistant clone. Genome Announc. 5, (2017).
Ahmed, M. O. & Baptiste, K. E. Vancomycin-resistant enterococci: a review of antimicrobial resistance mechanisms and perspectives of human and animal health. Microb. Drug Resist. 24, 590–606 (2017).
Illumina, Inc. Minimizing Index Hopping. https://www.illumina.com/techniques/sequencing/ngs-library-prep/multiplexing/index-hopping.html (2018).
Schluter, J. et al. The gut microbiota is associated with immune cell dynamics in humans. Nature 588, 303–307 (2020).
D’Amico, F. et al. Gut resistome plasticity in pediatric patients undergoing hematopoietic stem cell transplantation. Sci. Rep. 9, 5649 (2019).
Simms-Waldrip, T. R. et al. Antibiotic-induced depletion of anti-inflammatory clostridia is associated with the development of graft-versus-host disease in pediatric stem cell transplantation patients. Biol. Blood Marrow Transplant. 23, 820–829 (2017).
Olm, M. R. et al. Necrotizing enterocolitis is preceded by increased gut bacterial replication, Klebsiella, and fimbriae-encoding bacteria. Sci. Adv. 5, eaax5727 (2019).
Howden, B. P. et al. Genomic insights to control the emergence of vancomycin-resistant Enterococci. mBio 4, (2013).
Browne, H. P. et al. Culturing of ‘unculturable’ human microbiota reveals novel taxa and extensive sporulation. Nature 533, 543–546 (2016).
Kaur, S., Yawar, M., Kumar, P. A. & Suresh, K. Hungatella effluvii gen. nov., sp. nov., an obligately anaerobic bacterium isolated from an effluent treatment plant, and reclassification of Clostridium hathewayi as Hungatella hathewayi gen. nov., comb. nov. Int. J. Syst. Evol. Microbiol. 64, 710–718 (2014).
Atarashi, K. et al. T reg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature 500, 232–236 (2013).
Linscott, A. J. et al. Fatal septicemia due to Clostridium hathewayi and Campylobacter hominis. Anaerobe 11, 97–98 (2005).
Woo, P. C. Y. et al. Bacteremia due to Clostridium hathewayi in a patient with acute appendicitis. J. Clin. Microbiol. 42, 5947–5949 (2004).
Reunanen, J. et al. Akkermansia muciniphila adheres to enterocytes and strengthens the integrity of the epithelial cell layer. Appl. Environ. Microbiol. 81, 3655–3662 (2015).
Yi, S. H., Jernigan, J. A. & McDonald, L. C. Prevalence of probiotic use among inpatients: a descriptive study of 145 U.S. hospitals. Am. J. Infect. Control 44, 548–553 (2016).
Pittet, V., Ewen, E., Bushell, B. R. & Ziola, B. Genome sequence of Lactobacillus rhamnosus ATCC 8530. J. Bacteriol. 194, 726–726 (2012).
Forster, S. C. et al. A human gut bacterial genome and culture collection for improved metagenomic analyses. Nat. Biotechnol. 37, 186–192 (2019).
Fukami, T. Historical contingency in community assembly: integrating niches, species pools, and priority effects. Annu. Rev. Ecol. Evol. Syst. 46, 1–23 (2015).
Dubin, K. A. et al. Diversification and evolution of vancomycin-resistant Enterococcus faecium during intestinal domination. Infect. Immun. 87, (2019).
Giraud, A. et al. Dissecting the genetic components of adaptation of Escherichia coli to the mouse gut. PLoS Genet. 4, e2 (2008).
Paepe, M. D. et al. Trade-off between bile resistance and nutritional competence drives Escherichia coli diversification in the mouse gut. PLoS Genet. 7, e1002107 (2011).
Barroso-Batista, J. et al. The first steps of adaptation of Escherichia coli to the gut are dominated by soft sweeps. PLoS Genet. 10, e1004182 (2014).
Dandoy, C. E., Ardura, M. I., Papanicolaou, G. A. & Auletta, J. J. Bacterial bloodstream infections in the allogeneic hematopoietic cell transplant patient: new considerations for a persistent nemesis. Bone Marrow Transplant. 52, 1091–1106 (2017).
Brooks, B. et al. Strain-resolved analysis of hospital rooms and infants reveals overlap between the human and room microbiome. Nat. Commun. 8, 1–7 (2017).
Pereira-Marques, J. et al. Impact of host DNA and sequencing depth on the taxonomic resolution of whole metagenome sequencing for microbiome analysis. Front. Microbiol. 10, (2019).
Bertrand, D. et al. Hybrid metagenomic assembly enables high-resolution analysis of resistance determinants and mobile elements in human microbiomes. Nat. Biotechnol. 1, https://doi.org/10.1038/s41587-019-0191-2 (2019).
Moss, E. L., Maghini, D. G. & Bhatt, A. S. Complete, closed bacterial genomes from microbiomes using nanopore sequencing. Nat. Biotechnol. 38, 701–707 (2020).
Tsai, Y.-C. et al. Resolving the complexity of human skin metagenomes using single-molecule sequencing. mBio 7, (2016).
Ramawatar. D. N. A. size selection (>3–4 kb) and purification of DNA using an improved homemade SPRI beads solution. https://doi.org/10.17504/protocols.io.n7hdhj6 (2018).
Andrews, S. Babraham Bioinformatics-Trim Galore! https://www.bioinformatics.babraham.ac.uk/projects/trim_galore/ (2019).
Shen, W., Le, S., Li, Y. & Hu, F. SeqKit: a cross-platform and ultrafast toolkit for FASTA/Q file manipulation. PLoS ONE 11, e0163962 (2016).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Babraham Bioinformatics—FastQC A. Quality Control tool for High Throughput Sequence Data. https://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
Köster, J. & Rahmann, S. Snakemake—a scalable bioinformatics workflow engine. Bioinformatics 28, 2520–2522 (2012).
Oksanen, J. et al. vegan: Community Ecology Package (2020).
Kurtz, S. et al. Versatile and open software for comparing large genomes. Genome Biol. 5, R12 (2004).
R Development Core Team. R: A Language and Environment for Statistical Computing (R Development Core Team, 2012).
Nattestad, M. MariaNattestad/dot (2020).
Alcock, B. P. et al. CARD 2020: antibiotic resistome surveillance with the comprehensive antibiotic resistance database. Nucleic Acids Res. (2019) https://doi.org/10.1093/nar/gkz935.
Siranosian, B. et al. bhattlab/bhattlab_workflows: v1.0.1 (Zenodo, 2021). https://doi.org/10.5281/zenodo.5546646.
Siranosian, B. & Moss, E. bhattlab/kraken2_classification (Zenodo, 2021) https://doi.org/10.5281/zenodo.5219057.
Acknowledgements
We would like to acknowledge the contributions of several members of the Bhatt lab for their assistance in the experimental design, laboratory work and manuscript preparation, including C. Severyn, D. Maghini, S. Vance, A. Natarajan, P. West, A. Behr, M. Chakraborty, and S. Zlitni. We would also like to thank B. Wu at Stanford Health Care for providing information on the geographic layout of the HCT ward, K. Chen for providing information relevant to restrictions on patients consuming probiotics, A. Sidow, and Z. Weng for their instruction and assistance in preparing linked-read sequencing libraries, K. Siranosian for edits to the manuscript, and S. Madison for providing information on HCT patient isolation practices. We would like to thank the Stanford Clinical Microbiology Lab for assistance in identifying isolated and cultured bacterial strains. This work was supported by NIH P01 CA049605 (PI: R. Negrin; Cell Therapy for Leukemia and Lymphoma), and NIH R01 AI143757, CDC BAA 75D30118C02921, Sloan Foundation Fellowship, V Scholar award, and a Damon Runyon Clinical Investigator Award to A.S.B. B.A.S. is supported by a NIST JIMB training grant. Computing costs were supported via a NIH S10 Shared Instrumentation Grant 1S10OD02014101. This work utilized computing resources provided by the Stanford Genetics Bioinformatics Service Center. Finally, we would like to acknowledge the patients and nurses on the Blood and Marrow Transplantation service for their enthusiastic participation in this project.
Author information
Authors and Affiliations
Contributions
B.A.S., A.S.B., H.T., N.B., T.A. designed the study. B.A.S. performed laboratory and computational work, conducted the analysis and wrote the manuscript. E.F.B. performed laboratory work and contributed to writing the manuscript. T.A. performed laboratory work. A.R.R. and T.A. Designed the stool sample collection effort. N.B. assisted with analysis of BSI data and bacterial culture identification. H.T. assisted with computational analysis. A.S.B. oversaw the project and contributed to writing of the manuscript. All authors contributed to editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Marcel van den Brink, Marcus de Goffau and the other, anonymous, reviewer for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Siranosian, B.A., Brooks, E.F., Andermann, T. et al. Rare transmission of commensal and pathogenic bacteria in the gut microbiome of hospitalized adults. Nat Commun 13, 586 (2022). https://doi.org/10.1038/s41467-022-28048-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-28048-7
This article is cited by
-
Oral bacteria relative abundance in faeces increases due to gut microbiota depletion and is linked with patient outcomes
Nature Microbiology (2024)
-
Sequencing-based analysis of microbiomes
Nature Reviews Genetics (2024)
-
Eco-evolutionary feedbacks in the human gut microbiome
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.