Abstract
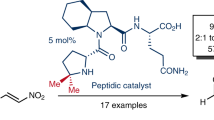
Peptides are important compounds with broad applications in many areas. Asymmetric transamination of α-keto amides can provide an efficient strategy to synthesize peptides, however, the process has not been well developed yet and still remains a great challenge in both enzymatic and catalytic chemistry. For biological transamination, the high activity is attributed to manifold structural and electronic factors of transaminases. Based on the concept of multiple imitation of transaminases, here we report N-quaternized axially chiral pyridoxamines 1 for enantioselective transamination of α-keto amides, to produce various peptides in good yields with excellent enantio- and diastereoselectivities. The reaction is especially attractive for the synthesis of peptides made of unnatural amino acids since it doesn’t need great efforts to make chiral unnatural amino acids before amide bond formation.
Similar content being viewed by others
Introduction
Peptides are one type of the most important compounds with high biological activities, which are widely present in many natural products, pharmaceutically relevant molecules, and biological systems1,2,3. Especially in recent years, there appears a growing interest in therapeutic peptides1,2,3 and more and more peptide drugs have been developed (Fig. 1a)4,5,6. Development of alternative new methods for the synthesis of peptides is always highly desirable and potentially useful7,8.
Enzymatic transamination is an important process to produce chiral amines such as amino acids in biological systems (Fig. 1b)9,10, which is promoted by transaminases (Fig. 1c) with pyridoxal/pyridoxamine 5′-phosphates as the coenzyme9,10,11,12,13. Mimicking the biological process14, i.e., asymmetric biomimetic transamination, affords a highly intriguing method to synthesize NH2-free amines from readily available carbonyl compounds15,16,17. The chemistry has attracted much attention since the 1970s18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36. The studies mainly include stoichiometric chiral pyridoxamine-promoted asymmetric transamination of α-keto acids18,19,20,21, pyridoxal/pyridoxamine-catalyzed asymmetric transamination of α-keto acids22,23,24,25,26, and chiral base/Lewis acid-catalyzed asymmetric transamination of α-keto esters and activated ketones27,28,29,30,31,32,33,34,35,36. Asymmetric transamination of α-keto amides can potentially provide an appealing new strategy to produce peptides. However, to the best of our knowledge, asymmetric transamination of α-keto amides to peptides are barely reported37,38, although the reverse process, transamination of peptides at the N-termini to α-keto amides, has been widely developed and already have been successfully applied to protein modification38,39,40,41,42,43,44,45. In contrast to the transamination of α-keto acids, asymmetric transamination of α-keto amides to peptides remains a challenge for enzymatic catalysis likely due to non-naturally occurring process and it is also a challenge for chemical catalysis probably because the complicated structure of α-keto amides requires more active catalysts to promote transamination.
Previous studies have suggested that asymmetric 1,3-proton shift between the ketimine intermediate and the aldimine is likely a key step for biological transamination (Fig. 1d)11,12,13,46,47. In order to accelerate this step, evolution has elegantly optimized transaminases by incorporating a Lys residue at an appropriate position48,49,50. The ε-NH2 group of the Lys residue serves as an intramolecular base to deprotonate the benzylic C-H of the ketimine (Fig. 1d)48,49,50. In addition, the pKa values of the pyridine N in the coenzyme pyridoxal 5′-phosphate (PLP) and pyridoxamine 5′-phosphate (PMP) are around 8.551,52,53, thus the pyridine N is predominantly protonated in biological systems (near pH 7)53,54,55,56. The strong electron-withdrawing property of the protonated pyridine ring helps to increase the acidity of the benzylic C-H bond (Fig. 1d)54,55,56,57,58. The two effects work together to promote the transformation from the ketimine to the aldimine via 1,3-proton shift, magically accelerating transamination process. Inspired by the controlled protonation of the pyridine N of PLP in biological systems, Rapoport has developed N-methylpyridinium-4-carboxaldehyde benzenesulfonate (Rapoport’s salt) as an effective transamination reagent for conversion of amines to carbonyl compounds59. Francis have found that Rapoport’s salt displays significantly improved efficiency in the transamination of proteins as compared to pyridinium-4-carboxaldehyde, converting the N-termini into the corresponding carbonyl groups44,45. Recently, we have proved that introducing an amine side arm to a chiral pyridoxamine can remarkably increase its activity and enantioselectivity for asymmetric transamination of α-keto acids26 and also have observed that quaternization of the pyridine N of a chiral pyridoxal leads to dramatical improvement of catalytic activity for asymmetric biomimetic Mannich reaction60 and aldol reaction61 of glycinate. On the basis of the structural characteristics of transaminases46,47,48,49,50,53,54,55,56 as well as the previously reported studies26,44,45,59,60,61,62, we has designed N-quaternized biaryl axially chiral pyridoxamines 1 bearing an amine side arm, mimicking transaminases in multiple aspects for catalytic asymmetric transamination of α-keto amides to peptides (Fig. 1c). The quaternization always keeps the pyridine ring with strong electron-withdrawing ability to improve the benzylic C-H acidity of the ketimine intermediate during transamination, no matter under acidic or basic conditions60,61. Like the Lys residue does in biological transamination, the amine side arm can serve as an intramolecular base to facilitate 1,3-proton shift.
Here we show that asymmetric biomimetic transamination of α-keto amides 2 can be achieved by using chiral pyridoxamines 1 as the catalyst, to produce various peptides 3 with excellent enantiopurities (Fig. 1e).
Results
Catalyst synthesis
The synthesis of chiral pyridoxamines 1 started with reductive amination of compound 526 to introduce the amine side chain. Protecting the two amine groups with di-tert-butyl dicarbonate gave intermediates 6 (Fig. 2). Treatment of 6 with methyl iodide and subsequent deprotection with hydrochloric acid afforded N-methyl pyridoxamines 1a-e in good yields.
Condition optimization
With diphenylglycine (4) as the amine source25,26,63, catalyst chiral pyridoxamine 1b was first tested for the transamination of glycinyl α-keto amide 2a (Fig. 3, entry 1). The originally-formed NH2-free transamination product was treated with di-tert-butyl dicarbonate to avoid the cyclization to piperazinedione during the isolation64, to give the corresponding N-Boc-protected dipeptide 3a in 20% yield with 76% ee. Additives have significant impacts on the reaction in terms of enantioselectivity and activity. Increased yield and enantioselectivity were obtained for transaminations performed in MeOH/H2O or TFEA/H2O with HOAc/KOAc or HOAc/Na2HPO4 as the additives (Fig. 3, entries 2 and 10 vs 3–9). Chiral pyridoxamine 1b exhibited the best performance among the catalysts 1a-e examined (Fig. 3, entries 10–14).
TFEA = 2,2,2-trifluoroethanol. aReaction conditions: 2a (0.10 mmol), 4 (0.11 mmol), 1 (0.0050 mmol), HOAc (0.40 mmol), base (0.20 mmol) in solvent (0.48 mL) and H2O (0.12 mL) at 20 °C for 48 h unless otherwise stated. The reaction mixtures were then treated with di-tert-butyl dicarbonate (0.30 mmol) at rt for 3 h. bIsolated yields based on α-keto amide 2a. cThe ee values were determined by HPLC analysis. dReaction time was 72 h.
Substrate scope
Under the optimal conditions, various glycinyl α-keto amides containing alkyl (for 3b-e), aromatic (for 3a and 3f-i), or heteroatomic alkyl (for 3j-k) groups were all smoothly transaminated to give the corresponding N-Boc-protected glycinyl dipeptides 3a-k in 70-94% yields with up to 98% ee (Fig. 4). Chiral glycinyl α-keto amide (for 3 l) displayed excellent diastereoselectivity (98:2 dr). Transamination of α-keto phenylbutanamides of chiral amino acid esters produced various N-Boc-protected dipeptides 3m-y in 56–93% yields with up to 99:1 diastereoselectivity. Peptidyl α-keto amides were also effective for the asymmetric transamination, to form tripeptides 3z-ab and tetrapeptides 3ac-ad in 60–87% yields with excellent diastereoselectivities under very mild conditions. Various functional groups such as C-C double bond (for 3c, 3 l and 3 y), NH2-sensitive bromide (for 3j), silyl group (for 3k), OH group of Tyr (for 3 s), NH group of Trp (for 3t), amide CONH2 of Asn (for 3 v), Boc-protected Lys residue (for 3w), Boc-protected guanidine (for 3x), and basic NH2 group of Lys (for 3ab) were all well tolerated by the transamination likely due to the mild reaction conditions.
TBDPS = tert-butyldiphenylsilyl. aReaction conditions: 2 (0.10 mmol), 4 (0.11 mmol), 1b (0.0050 mmol), HOAc (0.40 mmol), Na2HPO4 or KOAc (0.20 mmol) in CF3CH2OH or MeOH (0.48 mL) and H2O (0.12 mL) for 48 or 72 h unless otherwise stated (See SI). For 3m-y, the reactions were carried out in a double scale. Isolated yields were based on α-keto amides 2. The ee and dr values were determined by HPLC analysis. bThe crude reaction mixture was treated with di-tert-butyl dicarbonate (0.30 mmol) at room temperature for 3 h. cThe reaction was carried out in a double scale with 7 (0.040 mmol, 20 mol %) at 50 °C for 72 h.
In order to investigate the impacts of catalyst and substrates on diastereomeric induction, several representative α-keto amides (for 3m-n, 3r, 3v-y, and 3aa-ab) were examined respectively using (S)-1b (5 mol%), (R)-1b (5 mol%), and achiral pyridoxamine 7 (20 mol%) as the catalyst. The corresponding peptides were formed with S configurations of the newly generated chiral centers from catalyst (R)-1b and R configurations from (S)-1b. The chiral pyridoxamine catalyst dominated the stereoselectivity, while the chiral groups on the amino acid residues of α-keto amides throwed little influence on the diastereomeric induction probably due to being far away from the reaction centers as well as the flexibility of the skeletons of the α-keto amides. No matter which configuration of the catalyst 1b was applied, excellent diastereoselectivities were always obtained, even for α-keto amides (for 3n and 3aa) with a nearby bulky chiral amino acid residue and for those that displayed obvious substrate-induction on diastereoselectivity in 7-catalyzed non-asymmetric transamination (5:95 dr for 3r and 19:81 dr for 3x). For α-keto amide 2 y bearing two nearby chiral centers, a pair of diastereomers (R,R,S)-3y and (R,S,S)-3y were respectively obtained in good yields with high enantiopurities by using (S)-1b and (R)-1b as the catalyst. The absolute configurations of the newly generated chiral centers of peptides 3 were assigned by analog, based on the X-ray analysis of 3d, 3 m, and 3r (also see Supplementary Figs. 1–3 in SI).
Synthetic applications
Divergent extending an additional amino acid unit from a central peptide is of great interest for peptide drug screening and bioactivity studies. The synthesis would be difficult when the extended unit is a commercially unavailable unnatural amino acid. The transamination process provides an efficient strategy for the amino acid extending. For example, starting from the benzyl ester of protease inhibitor Ubenimex (8)65, condensation with α-keto acids and subsequent asymmetric transamination afforded a variety of enantiopure peptides 3ae-ai with one more amino acid residue extended (Fig. 5a). The unprotected OH group remained untouched during the condensation and transamination.
Based on the “condensation-transamination” process, a new strategy for the synthesis of peptides also can be developed. As illustrated in Fig. 5b, the methyl ester of DPP-IV inhibitor Diprotin A (10)66 underwent condensation with α-keto acid 9a and subsequent asymmetric transamination, forming tetrapeptide 3ad with excellent diastereoselectivity. Repeating the reaction sequence two more times afforded hexapeptide 3ak with high enantiopurity. The chirality of the extended amino acid residues was established along with the transamination process. The protocol is especially attractive for the synthesis of peptides made of unnatural amino acids, since it doesn’t need great efforts to make NH2-protected chiral unnatural amino acids before the amide bond formation.
Reaction mechanism
A plausible mechanism was proposed for the transamination (Fig. 6a)11,12,13,25,26. Pyridoxamine 1b condenses with α-keto amide 2 to form ketimine 11, which undergoes asymmetric 1,3-proton shift to aldimine 13 under the assistance of the amine side arm46,47,48,49,50. Hydrolysis of aldimine 13 releases peptide 3 and generates the pydridoxal, which is in situ converted into iminium 14 via intramolecular condensation. The iminium 14 then undergoes decarboxylative transamination with the amine source diphenylglycine (4) back to pyridoxamine catalyst 1b25,26,63, completing a catalytic cycle.
As expected, N-quaternization of the pyridine ring of the chiral pyridoxamines resulted in higher catalytic activity and better enantioselectivity for the asymmetric transamination (Fig. 6b, 1b vs 1g). The stronger electron-withdrawing property makes the benzylic C-H of ketimine 11 more acidic and also stabilizes the corresponding delocalized carbanion 12 better54,55,56,57,58,67,68,69, thus favoring the 1,3-proton shift and accelerating the transamination process. The control experiment confirmed the amazing effect of the amine side arm again (Fig. 6b, 1b vs 1f). Introducing an acetyl group onto the nitrogen to eliminate the basicity of the amine on the side arm led to marked decreases in activity and enantioselectivity. The amine side arm not only promotes the 1,3-proton shift by acting as an intramolecular base to deprotonate the benzylic C-H of ketimine 11 (Fig. 6a) but also helps to orient the α-keto amide by hydrogen bonding with the carbonyl oxygen of the amide group (Fig. 6c), resulting in improved activity and stereoselectivity48,49,50. Protonation of the delocalized carbanion 12 occurs at α−position of the amide group from the up side of the pyridine ring away from the amine side arm26,46,47, to form the newly generated chiral center with S configuration from catalyst (R)-1b.
Discussion
In summary, based on the concept of multiple imitation of transaminases, we have developed N-quaternized axially chiral pyridoxamines 1 containing an amine side arm. With pyridoxamine 1b as the catalyst, challenging substrates α-keto amides were successfully transaminated to peptides in good yields with excellent enantio- and diastereoselectivities. The catalyst dominated the diastereoselective control for the transamination of chiral α-keto amides. Thus, a pair of diastereomeric peptides could be respectively obtained with high enantiopurities by switching the configuration of the pyridoxamine catalyst. The strong electron-withdrawing property of the N-quaternized pyridine ring together with the cooperative catalysis of the amine side arm account for the increased catalytic activity and selectivity of the pyridoxamine 1b in the transamination. The reaction can provide an efficient strategy for divergent and successive extension of peptides via condensation-transamination reaction sequence, which is especially attractive for the synthesis of peptides made of unnatural amino acids.
Methods
General procedure for the asymmetric biomimetic transamination Reaction (Fig. 4)
A mixture of α-keto amide 2 (0.10 mmol), chiral pyridoxamine 1b (0.0050 mmol), 2,2-diphenylglycine (4) (0.11 mmol), HOAc (0.40 mmol), Na2HPO4 or KOAc (0.20 mmol), CF3CH2OH or MeOH (0.48 mL), and H2O (0.12 mL) was stirred at 16–25 °C for the specified time. For glycinyl α-keto amides (for 3a-l) and α-keto phenylbutanamides of amino acid esters (for 3m-y), the crude reaction mixtures were treated with di-tert-butyl dicarbonate (0.3 mmol) at room temperature for 3 h after the transamination was completed, then concentrated via rotary evaporator to remove most of the solvent and isolated by column chromatography on silica gel with a mixed solvent ethyl acetate and petroleum ether as the eluant to give the products N-Boc-protected dipeptides 3a-y. For Peptidyl α-keto amides (for 3z-ad), the reaction mixtures were submitted to concentration via rotary evaporator to remove most of the solvent and then isolated by column chromatography on silica gel with a mixed solvent of dichloromethane, methanol and ammonia solution in ethanol (2.9 M) as the eluant to give the transamination products tripeptides 3z-aa and tetrapeptides 3ab-ad without NH2-protection. The ee and dr values of 3a-ac were determined by HPLC analysis.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and Supplementary Information file, or from the corresponding author upon reasonable request. For the experimental procedures, characterization data, and NMR spectra along with HPLC chromatograms, see Supplementary Information. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers of CCDC 2036531 (3d), CCDC 2036532 (the cyclized derivative of 3 m), and CCDC 2036529 (3r). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via https://www.ccdc. cam.ac.uk/structures/.
References
Henninot, A., Collins, J. C. & Nuss, J. M. The current state of peptide drug discovery: back to the future? J. Med. Chem. 61, 1382–1414 (2018).
Lau, J. L. & Dunn, M. K. Therapeutic peptides: historical perspectives, current development trends, and future directions. Bioorg. Med. Chem. 26, 2700–2707 (2018).
Vlieghe, P., Lisowski, V., Martinez, J. & Khrestchatisky, M. Synthetic therapeutic peptides: science and market. Drug Discov. Today 15, 40–56 (2010).
Piliero, P. J. Atazanavir: A Novel HIV-1 Protease Inhibitor. Expert Opin. Inv. Drugs 11, 1295–1301 (2002).
Ricci, F. et al. Intermediate dose Ara-C followed by G-CSF: an effective and predictable mobilization regimen in hematological malignancies. Blood 110, 3290–3290 (2007).
BEER, T. M., GARZOTTO, M., EILERS, K. M. & LEMMON, D. Phase II study of abarelix depot for androgen independent prostate cancer progression during gonadotropin-releasing hormone agonist therapy. J. Urol. 169, 1738–1741 (2003).
Humphrey, J. M. & Chamberlin, A. R. Chemical synthesis of natural product peptides: coupling methods for the incorporation of noncoded amino acids into peptides. Chem. Rev. 97, 2243–2266 (1997).
El-Faham, A. & Albericio, F. Peptide coupling reagents, more than a letter soup. Chem. Rev. 111, 6557–6602 (2011).
Taylor, P. P., Pantaleone, D. P., Senkpeil, R. F. & Fotheringham, I. G. Novel biosynthetic approaches to the production of unnatural amino acids using transaminases. Trends Biotechnol. 16, 412–418 (1998).
Fuchs, M., Farnberger, J. E. & Kroutil, W. The industrial age of biocatalytic transamination. Eur. J. Org. Chem. 2015, 6965–6982 (2015).
Metzler, D. E., Ikawa, M. & Snell, E. E. A general mechanism for vitamin B6-catalyzed reactions. J. Am. Chem. Soc. 76, 648–652 (1954).
Ayling, J. & Snell, E. E. Mechanism of action of pyridoxamine pyruvate transaminase. Biochemistry 7, 1616–1625 (1968).
Liao, R.-Z., Ding, W.-J., Yu, J.-G., Fang, W.-H. & Liu, R.-Z. Theoretical studies on pyridoxal 5′-phosphate-dependent transamination of α-amino acids. J. Comput. Chem. 29, 1919–1929 (2008).
Breslow, R. Biomimetic chemistry: biology as an inspiration. J. Biol. Chem. 284, 1337–1342 (2009).
Breslow, R. Biomimetic chemistry and artificial enzymes: catalysis by design. Acc. Chem. Res. 28, 146–153 (1995).
Xie, Y., Pan, H., Liu, M., Xiao, X. & Shi, Y. Progress in asymmetric biomimetic transamination of carbonyl compounds. Chem. Soc. Rev. 44, 1740–1748 (2015).
Chen, J., Liu, Y. E., Gong, X., Shi, L. & Zhao, B. Biomimetic chiral pyridoxal and pyridoxamine catalysts. Chin. J. Chem. 37, 103–112 (2019).
Kuzuhara, H., Komatsu, T. & Emoto, S. Synthesis of a chiral pyridoxamine analog and nonenzymatic stereoselective transamination. Tetrahedron Lett. 19, 3563–3566 (1978).
Breslow, R., Hammond, M. & Lauer, M. Selective transamination and optical induction by a β-cyclodextrin-pyridoxamine artificial enzyme. J. Am. Chem. Soc. 102, 421–422 (1980).
Zimmerman, S. C. & Breslow, R. Asymmetric synthesis of amino acids by pyridoxamine enzyme analogs utilizing general base-acid catalysis. J. Am. Chem. Soc. 106, 1490–1491 (1984).
Wei, S., Wang, J., Venhuizen, S., Skouta, R. & Breslow, R. Dendrimers in solution can have their remote catalytic groups folded back into the core: enantioselective transaminations by dendritic enzyme mimics-II. Bioorg. Med. Chem. Lett. 19, 5543–5546 (2009).
Kikuchi, J.-i, Zhang, Z.-Y. & Murakami, Y. Enantioselective catalysis by a supramolecular bilayer membrane as an artificial aminotransferase. stereochemical roles of an L-lysine residue and L-phenylalanine at the reaction site. J. Am. Chem. Soc. 117, 5383–5384 (1995).
Kuang, H. & Distefano, M. D. Catalytic enantioselective reductive amination in a host−guest system based on a protein cavity. J. Am. Chem. Soc. 120, 1072–1073 (1998).
Svenson, J., Zheng, N. & Nicholls, I. A. A molecularly imprinted polymer-based synthetic transaminase. J. Am. Chem. Soc. 126, 8554–8560 (2004).
Shi, L. et al. Chiral pyridoxal-catalyzed asymmetric biomimetic transamination of α-keto acids. Org. Lett. 17, 5784–5787 (2015).
Liu, Y. E. et al. Enzyme-inspired axially chiral pyridoxamines armed with a cooperative lateral amine chain for enantioselective biomimetic transamination. J. Am. Chem. Soc. 138, 10730–10733 (2016).
Bernauer, K., Deschenaux, R. & Taura, T. Stereoselectivity in reactions of metal complexes VII. Asymmetric synthesis of amino acids by metal ion-promoted transamination. Helv. Chim. Acta 66, 2049–2058 (1983).
Soloshonok, V. A., Kirilenko, A. G., Galushko, S. V. & Kukhar, V. P. Catalytic asymmetric synthesis of β-fluoroalkyl-β-amino acids via biomimetic [1,3]-proton shift reaction. Tetrahedron Lett. 35, 5063–5064 (1994).
Willems, J. G. H., de Vries, J. G., Nolte, R. J. M. & Zwanenburg, B. Asymmetric imine isomerisation in the enantioselective synthesis of chiral amines from prochiral ketones. Tetrahedron Lett. 36, 3917–3920 (1995).
Hjelmencrantz, A. & Berg, U. New approach to biomimetic transamination using bifunctional [1,3]-proton transfer catalysis in thioxanthenyl dioxide imines. J. Org. Chem. 67, 3585–3594 (2002).
Knudsen, K. R., Bachmann, S. & Jørgensen, K. A. Catalytic enantioselective transaminiation of α-keto esters: an organic approach to enzymatic reactions. Chem. Commun. 20, 2602–2603 (2003).
Xiao, X., Xie, Y., Su, C., Liu, M. & Shi, Y. Organocatalytic asymmetric biomimetic transamination: from α-keto esters to optically active α-amino acid derivatives. J. Am. Chem. Soc. 133, 12914–12917 (2011).
Wu, Y. & Deng, L. Asymmetric synthesis of trifluoromethylated amines via catalytic enantioselective isomerization of imines. J. Am. Chem. Soc. 134, 14334–14337 (2012).
Xie, Y., Pan, H., Xiao, X., Li, S. & Shi, Y. Organocatalytic asymmetric biomimetic transamination of aromatic ketone to optically active amine. Org. Biomol. Chem. 10, 8960–8962 (2012).
Zhou, X., Wu, Y. & Deng, L. Cinchonium betaines as efficient catalysts for asymmetric proton transfer catalysis: the development of a practical enantioselective isomerization of trifluoromethyl imines. J. Am. Chem. Soc. 138, 12297–12302 (2016).
Kang, Q.-K., Selvakumar, S. & Maruoka, K. Asymmetric synthesis of α-amino acids by organocatalytic biomimetic transamination. Org. Lett. 21, 2294–2297 (2019).
Herbst, R. M. & Shemin, D. The synthesis of peptides by transamination. J. Biol. Chem. 147, 541–547 (1943).
Lee, S. H., Kyung, H., Yokota, R., Goto, T. & Oe, T. N-terminal α-ketoamide peptides: formation and transamination. Chem. Res. Toxicol. 27, 637–648 (2014).
Cennamo, C., Carafoli, B. & Bonetti, E. P. Non-enzymatic transamination between peptides and pyridoxal. Isolation of the 2,4-dinitrophenylhydrazones of some ketopeptides. J. Am. Chem. Soc. 78, 3523–3527 (1956).
Dixon, H. Transamination of peptides. Biochem. J. 92, 661–666 (1964).
Papanikos, A., Rademann, J. & Meldal, M. α-ketocarbonyl peptides: a general approach to reactive resin-bound intermediates in the synthesis of peptide isosteres for protease inhibitor screening on solid support. J. Am. Chem. Soc. 123, 2176–2181 (2001).
Gilmore, J. M., Scheck, R. A., Esser-Kahn, A. P., Joshi, N. S. & Francis, M. B. N-terminal protein modification through a biomimetic transamination reaction. Angew. Chem. Int. Ed. 45, 5307–5311 (2006).
Scheck, R. A., Dedeo, M. T., Iavarone, A. T. & Francis, M. B. Optimization of a biomimetic transamination reaction. J. Am. Chem. Soc. 130, 11762–11770 (2008).
Witus, L. S. et al. Site-specific protein transamination using N-methylpyridinium-4-carboxaldehyde. J. Am. Chem. Soc. 135, 17223–17229 (2013).
Palla, K. S., Witus, L. S., Mackenzie, K. J., Netirojjanakul, C. & Francis, M. B. Optimization and expansion of a site-selective N-methylpyridinium-4-carboxaldehyde-mediated transamination for bacterially expressed proteins. J. Am. Chem. Soc. 137, 1123–1129 (2015).
Yoshimura, T., Jhee, K.-H. & Soda, K. Stereospecificity for the hydrogen transfer and molecular evolution of pyridoxal enzymes. Biosci. Biotechnol. Biochem. 60, 181–187 (1996).
Soda, K., Yoshimura, T. & Esaki, N. Stereospecificity for the hydrogen transfer of pyridoxal enzyme reactions. Chem. Rec. 1, 373–384 (2001).
Kirsch, J. F. et al. Mechanism of action of aspartate aminotransferase proposed on the basis of its spatial structure. J. Mol. Biol. 174, 497–525 (1984).
Kochhar, S., Finlayson, W. L., Kirsch, J. F. & Christen, P. The stereospecific labilization of the C-4’ pro-S hydrogen of pyridoxamine 5’-phosphate is abolished in (Lys258—Ala) aspartate aminotransferase. J. Biol. Chem. 262, 11446–11448 (1987).
Malashkevich, V. N. et al. Structural basis for the catalytic activity of aspartate aminotransferase K258H lacking the pyridoxal 5’-phosphate-binding lysine residue. Biochemistry 34, 405–414 (1995).
Adrover, M. et al. Towards a detailed description of pyridoxamine tautomeric species. N. J. Chem. 36, 1751–1761 (2012).
Gansow, O. A. & Holm, R. H. Aqueous solution equilibria of pyridoxamine, pyridoxal, 3-hydroxypyridine-4-aldehyde, and 3-hydroxypyridine-2-aldehyde as studied by proton resonance. Tetrahedron 24, 4477–4487 (1968).
Limbach, H.-H. et al. Critical hydrogen bonds and protonation states of pyridoxal 5′-phosphate revealed by NMR. Biochim. Biophys. Acta 1814, 1426–1437 (2011).
Yano, T., Kuramitsu, S., Tanase, S., Morino, Y. & Kagamiyama, H. Role of Asp222 in the catalytic mechanism of Escherichia coli aspartate aminotransferase: the amino acid residue which enhances the function of the enzyme-bound coenzyme pyridoxal 5’-phosphate. Biochemistry 31, 5878–5887 (1992).
Sharif, S. et al. NMR localization of protons in critical enzyme hydrogen bonds. J. Am. Chem. Soc. 129, 9558–9559 (2007).
Griswold, W. R. & Toney, M. D. Role of the pyridine nitrogen in pyridoxal 5′-phosphate catalysis: activity of three classes of plp enzymes reconstituted with deazapyridoxal 5′-phosphate. J. Am. Chem. Soc. 133, 14823–14830 (2011).
Crugeiras, J., Rios, A., Riveiros, E. & Richard, J. P. Substituent effects on electrophilic catalysis by the carbonyl group: anatomy of the rate acceleration for PLP-catalyzed deprotonation of glycine. J. Am. Chem. Soc. 133, 3173–3183 (2011).
Crugeiras, J., Rios, A., Amyes, T. L. & Richard, J. P. Carbon acidity of the α-pyridinium carbon of a pyridoxamine analog. Org. Biomol. Chem. 3, 2145–2149 (2005).
Buckley, T. F. & Rapoport, H. Mild and simple biomimetic conversion of amines to carbonyl compounds. J. Am. Chem. Soc. 104, 4446–4450 (1982).
Chen, J. et al. Carbonyl catalysis enables a biomimetic asymmetric Mannich reaction. Science 360, 1438–1442 (2018).
Cheng, A. et al. Efficient Asymmetric Biomimetic Aldol Reaction of Glycinates and Trifluoromethyl Ketones by Carbonyl Catalysis. Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.202104031 (2021).
Ma, J. et al. Enantioselective synthesis of pyroglutamic acid esters from glycinate via carbonyl catalysis. Angew. Chem. Int. Ed. 60, 10588–10592 (2021).
Liu, L., Zhou, W., Chruma, J. & Breslow, R. Transamination reactions with multiple turnovers catalyzed by hydrophobic pyridoxamine cofactors in the presence of polyethylenimine polymers. J. Am. Chem. Soc. 126, 8136–8137 (2004).
Yamazaki, Y. et al. Acid catalyzed monodehydro-2,5-diketopiperazine formation from N-α-ketoacyl amino acid amides. Tetrahedron 65, 3688–3694 (2009).
Yoneda, J. et al. Inhibition of tumor invasion and extracellular matrix degradation by ubenimex (bestatin). Clin. Exp. Metastasis 10, 49–59 (1992).
Juillerat-Jeanneret, L. Dipeptidyl peptidase IV and its inhibitors: therapeutics for type 2 diabetes and what else? J. Med. Chem. 57, 2197–2212 (2014).
Crugeiras, J., Rios, A., Riveiros, E., Amyes, T. L. & Richard, J. P. Glycine enolates: the effect of formation of iminium ions to simple ketones on α-amino carbon acidity and a comparison with pyridoxal iminium ions. J. Am. Chem. Soc. 130, 2041–2050 (2008).
Tang, S., Zhang, X., Sun, J., Niu, D. & Chruma, J. J. 2-azaallyl anions, 2-azaallyl cations, 2-azaallyl radicals, and azomethine ylides. Chem. Rev. 118, 10393–10457 (2018).
Chen, W.-W. & Zhao, B. Decarboxylative umpolung synthesis of amines from carbonyl compounds. Synlett 31, 1543–1550 (2020).
Acknowledgements
We are grateful for the generous financial support from the National Natural Science Foundation of China (21672148, 21871181), the Shanghai Municipal Education Commission (2019-01-07-00-02-E00029), the Shanghai Municipal Committee of Science and Technology (18ZR1447600, 20JC1416800), “111” Innovation and Talent Recruitment Base on Photochemical and Energy Materials (D18020), and Shanghai Engineering Research Center of Green Energy Chemical Engineering (18DZ2254200).
Author information
Authors and Affiliations
Contributions
B.Z. conceived and directed the project and wrote the paper. W.C and X.Q. conducted most of the experiments including the synthesis of the chiral pyridoxamine catalysts and the development of the asymmetric biomimetic transamination reaction. H.Z. synthesized some pyridoxamine intermediates and several α-keto amides for the transamination. B.L. performed some experiments for the catalyst development. J.G. and L.Z synthesized several α-keto amides for the transamination. W.C. revised the manuscript and the Supplementary Information.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cai, W., Qiao, X., Zhang, H. et al. Asymmetric biomimetic transamination of α-keto amides to peptides. Nat Commun 12, 5174 (2021). https://doi.org/10.1038/s41467-021-25449-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-021-25449-y
This article is cited by
-
Biomimetic asymmetric catalysis
Science China Chemistry (2023)
-
Prebiotic synthesis of α-amino acids and orotate from α-ketoacids potentiates transition to extant metabolic pathways
Nature Chemistry (2022)
-
Catalytic asymmetric α C(sp3)–H addition of benzylamines to aldehydes
Nature Catalysis (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.