Abstract
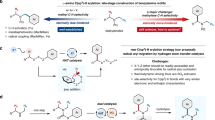
Atropisomers are important organic frameworks in bioactive natural products, drugs as well as chiral catalysts. Meanwhile, silanols display unique properties compared to their alcohol analogs, however, the catalytic synthesis of atropisomers bearing silanol groups is challenging. Here, we show a rhodium-catalyzed torsional strain-promoted asymmetric ring-opening reaction for the synthesis of α-silyl biaryl atropisomers. The reaction features a dynamic kinetic resolution of C(Ar)-Si bond cleavage, whose stereochemistry was controlled by a phosphoramidite ligand derived from (S)-3-methyl-1-((2,4,6-triisopropylphenyl)sulfonyl)piperazine. This work is a demonstration of an aryl-Narasaka acylation, where the C(Ar)-Si bond cleavage is promoted by the torsional strain of α, α’-disubstituted silafluorene.
Similar content being viewed by others
Introduction
Atropisomers are an important class of compounds, which showcased chirality due to the restricted rotation around the single C–C, C–N, or C–O bonds1,2,3. Among them axially chiral biaryls have caught extensive attentions due to their wide existence in bioactive natural products, drugs4,5; furthermore it is one of prior skeletons for chiral ligands or catalysts in asymmetric catalysis.
Despite the importance of biaryl atropisomers, the catalytically asymmetric synthesis was challenging due to the steric hindrance around the axis, which usually required three or four ortho substituents for achieving stable axial chirality6. The aryl–aryl cross-coupling or oxidative coupling is the most straightforward method, which delivered extremely useful axially chiral ligands and catalysts, such as BINOLs7,8,9,10,11,12,13,14,15,16,17,18. In addition, other useful methods, including (dynamic) kinetic resolution19,20,21,22,23,24,25,26, point to axial chirality transfer27,28,29, de novo aryl ring synthesis30,31,32,33,34 have been developed and significantly broadened the diversity of biaryl atropisomers. Recently, organo-catalyzed transformation showed its strong power in organic synthesis and it have been successfully applied to axially chiral molecules construction35,36,37,38,39,40.
In comparison with the classic asymmetric couplings, which proceed via a highly bulky metal intermediate (Fig. 1a), the ring-opening strategy surcumvented this key intermediate and it provided an efficient protocol for construction of biaryl atropisomers. Bringmann pioneered the atropisomer synthesis via ring-opening of lactones, with either stoichiometric or catalytic methods (Fig. 1b)41,42. Recently, Zhang and co-workers realized Ir-catalyzed asymmetric hydrogenation of Bringmann’s lactones43. Under the catalysis of nickel, copper, and palladium, the groups of Hayashi, Gu and others realized the ring-opening of dibenzo[b,d]thiophenes44, diaryliodoniums45,46,47,48, and 9H-fluoren-9-ols (Fig. 1b)49. This ring-opening reactions showed particular advantages in the preparation of sterically hindered ortho tetra-substituted atropisomeric biaryls; furthermore, these ring-opening reactions displayed excellent diversity: hydroxyl, hydroxymethyl, thiol, iodine, and keto groups were efficiently introduced to the position adjacent to the chiral axis.
Silanols display unique applications in pharmaceutical chemistry and organic chemistry50,51. For example, compound 1 displays nuclear receptor modulator activity and has better lipophilicity than the corresponding carbinol52. Silanols 2 and 3 were used as organocatalysts for asymmetric syntheses53,54. Considering the remarkable property of silanol groups, we wonder if ortho silanol substituted biaryl atropiosmers could be accessed via the carbon–silica bond cleavage of silafluorene in a stereoselective manner. Unfortunately, the silanes showed low reactivity in classic cross-couplings, i.e., Hiyama or Hiyama–Denmark Couplings55,56. Pleasingly, the Narasaka acylation57,58, which favored C–SiMe3 bond breaking over the C–SiMe2(OH), provided a potential solution for C–Si bond cleavage of silafluorene (Fig. 2b). However, challenges still remain: (a) only vinyl silanes underwent Narasaka acylation; (b) it is necessary to differentiate up to four C(aryl)–Si bonds in our systems. The releasing of noncyclic aryl ring is the undesired pathway; (c) the classic Narasaka acylation is steric sensitive, and vinyltriphenylsilane displayed extremely low reactivity (Fig. 2c). It is forseeable, the reaction would be more challenging if tetra(arylsilanes) were used. In the previous studies on the ring-opening reactions, we found that the two non-hydrogen groups adjacent to the axis increased the distortion of the molecule. The distorted molecule is the energetic compound, which had relative lower activation energy than the non-distorted one for the ring-opening reaction. Different from the inherent high strain of three- or four-membered rings, we anticipated that the activity of ring-opening reaction can be increased by the torsional strain of α, α′-disubstituted five-membered silafluorenes. Furthermore, the torsional strain also enabled to differentiate one of four C(aryl)–Si bonds to give desired products.
Here, we report an aryl-Narasaka acylation reaction catalyzed by the chiral rhodium complex which is in situ formed of [Rh(CO)2Cl]2 and phosphoramidite ligand derived from (S)-3-methyl-1-((2,4,6-triisopropylphenyl)sulfonyl)piperazine. Computational studies show that the storioanl stain of 7,7-diphenyl-7H-dinaphtho[2,1-b:1′,2′-d]silole is about 12.58 kcal mol−1, which efficiently accelerate the C–Si bond cleavage.
Results
Optimization of Rh-catalyzed aryl-Narasaka acylation
Our investigation began with the ring-opening/acylation reaction of dimethyl silafluorene (4a)59,60,61,62 with acetic anhydride (5a) (Table 1). With [Rh(CO)2Cl]2 as the catalyst, primary investigations found that the TADDOL-based phosphoramidite L1 gave satisfied stereo-induction with 49% yield in the presence of 1.0 equiv of Cs2CO3 (Table 1, entry 1). Decreasing the loading of base gradually improve the yield of 6a, until it reached the highest level when 30 mol% of Cs2CO3 was employed (Table 1, entries 2–4). Surprisingly, the reaction without the addition of base did not produce isolable desired product (Table 1, entry 5). Replacing the biphenyl group in the ligand with 4-fluorophenyl group gave a decreased enantioselectivity (Table 1, entry 6). The ligand without the chiral α-methyl group at the piperidine moiety (L3) afforded a reduced stereoselectivity (Table 1, entry 7). Organic base, i.e., Et3N was not suitable for this ring-opening/acylation reaction, while Na2CO3 dramatically improved the yield and maintained high enantioselectivity (Table 1, entries 8–9). The additives, such as KBr or 4 Å molecular sieve did not offer better outcomes (Table 1, entries 10–11).
The synthesis of sterically hindered silanols is challenging63,64. Unfortunately, applying the optimized conditions ([Rh(CO)2Cl]2/L1) to 4a and benzoic anhydride gave conspicuously decreased ee value (54%, 75% ee). Thus, further optimization based on the modification of phosphoramidites were carried out by using Si,Si-diphenylsilafluorene 4b and benzoic anhydride (Table 2). The N-Cbz or N-Boc piperazine derived ligands L4 and L5 gave improved results in comparison with L1 (Table 2, entries 1–3). The 2,4,6-trimethylphenylsulfonamide analog L6 gave 77% ee, while, pleasingly, 2,4,6-tri(isopropyl)phenylsulfonamide derivative L7 afforded the desired product 6b in 92% ee (Table 2, entries 4 and 5). The use of Cs2CO3 dramatically improved the yield (Table 2, entry 6). Decreasing the loading of L7 to 6.25 mol% gave slightly lower yield of 4b (Table 2, entry 7). Increasing the amount of Cs2CO3 is detrimental for the yield (Table 2, entries 8 and 9). Further optimization by using Et3N in lieu of Cs2CO3, or addition of molecular sieve did not produce better results (Table 2, entries 10 and 11).
Substrate scope
Under the optimal conditions, a number of acid anhydrides were tested with 4b as the substrate (Fig. 3). The aliphatic anhydrides also smoothly coupled with 4b to produce the corresponding silonols. The longer alkyl chain slightly decreased the yields and selectivity (6c–6f). 2-Methoxyacetic anhydride steadily underwent this acylation reaction to give 6g in excellent yield and enantioselectivity. The reaction of cyclopropanecarboxylic anhydride afforded the product 6h in 81% yield with 85% ee. The acrylic anhydride derivatives also reacted with 4b uneventfully to furnish the products in excellent ee values (6i–6k). The aromatic anhydrides were also compatible substrates. The p-methylbenzoic anhydride gave a slightly decreased yield, while the electron-withdrawing substituents were advantageous for both yields and stereoselectivity (6l–6o). Subsequently, other substituted aromatic acid anhydrides, including 3,4,5-trimethoxybenzoic anhydride, were submitted to the standard conditions to produce the corresponding silanols 6p–6s with excellent ees. Lastly, the 2-naphthoic or heteroaromatic anhydride were checked, and both of the reactions proceeded smoothly with the ee values were marginally changed (6t and 6u).
aReaction conditions: silafluorene 4b (0.20 mmol) and acid anhydrides (0.30 mmol, 1.5 equiv), [Rh(CO)2Cl]2 (0.005 mmol, 2.5 mol%), L7 (0.0125 mmol, 6.25 mol%), and base (30 mol%, Na2CO3 for aliphatic anhydride, Cs2CO3 for aromatic anhydride) in 1,4-dioxane (4.0 mL) at 70 °C for 24 h. bL1 was used as the ligand. cThe reaction time is 48 h.
Silafluorenes bearing different substituents were further investigated (Fig. 4). Ligand L1 was found more efficient for substrate 4a, which reacted with propionic anhydride and butyric anhydride to give 6v and 6w in 85% and 90% ee, respectively. The substituted benzene rings attached to the silicon atom have marginal effect on the selectivity, all the silanols were formed in excellent stereoselectivity (6x–6bb). The silafluorenes with biphenyl skeleton gave relatively lower stereoselectivity (6cc–6ee). The diastereochemistry of compound 6cc was determined to be R by single crystal X-ray diffraction analysis (CCDC 1968743). The starting material α,α′,β,β′-tetramethyl biphenylsilafluorene showed slight poor stability, as a result, the corresponding product 6ff was isolated in only 45% yield with 87% ee.
aReaction conditions: silafluorene 4 (0.20 mmol) and acid anhydride 5 (0.30 mmol, 1.5 equiv), [Rh(CO)2Cl]2 (0.005 mmol, 2.5 mol%), L7 (0.021 mmol, 10.5 mol%) and base (30 mol%, Na2CO3 for aliphatic anhydride, Cs2CO3 for aromatic anhydride) in 1,4-dioxane (4.0 mL) at 70 °C for 24 h. bThe reaction was performed with L1 as the ligand at 50 °C for 48 h.
Control experiments
For the unsymmetrical silafluorene 4j, the reaction gave 1:1 diastereomers, although both 6gg (CCDC 2013379) and 6gg′ were obtained in high enantioselectivity (Fig. 5a, eq (1)). The reaction between 4b and benzoic 4-fluorobenzoic anhydride afforded 6m and 6b in a ratio of 1:1.3 (Fig. 5a, eq (2)). Notably, with acetic benzoic anhydride as the reagent, the reaction produced methyl aryl ketone 6c as the major product (6c:6b = 6.7:1) (Fig. 5a, eq (3)). Finally, a control reaction by mixing 4a and 4b (1:1) was performed with acetic anhydride as limiting reagent. It afforded a 1.5:1 mixture of 6a and 6c, indicating 4a was slightly more active than 4b under our conditions (Fig. 5b, eq (4)).
Synthetic application
Treatment of 6b with phenylmagnesium chloride at 0 °C afforded hydroxysilanol 7 in excellent yield (Fig. 5c, eq (5)). Compound 7 was an anologue of corresponding carbols, which was potentially useful in asymmetric catalysis, for example, asymmetric hetero-Diels–Alder reactions65. In addition, vinylation of the ketone, followed by bromocyclization gave cyclic compound 9 (CCDC 1987455) in excellent diastereoselectivity (Fig. 5c, eq (6), (7)) (For reaction conditions optimization, see Supplementary Table 1). The Hiyama coupling by employing these silanols was not successful under various conditions, which was possibly due to the steric hindrance of the silanols. However, silanol 6b was readily transferred to arylbromide 10, which underwent classic cross-coupling to give 11 without losing the enantiopurity (Fig. 5c, eq (8)).
Mechanistic studies
For comparison, compound 4c was also synthesized, and notably silanol 4c showed poor reactivity under standard conditions. To get some structural information of these silafluorenes, single crystals of both 4a (CCDC 1968747) and 4c (CCDC 1968985) were obtained and analyzed by X-ray diffraction. It clearly shows the biphenyl structure in 4c is almost planar, while the increased size of compound 4a makes the naphthyl rings no longer coplanar. It is a twisted molecule with a distortional angle being 31.8° of the binaphthyl skeleton (Fig. 6a). We calculated the rotational barrier [ΔGg (298.15 K, 1 atm)] of 4a, which is around 20.4 kcal/mol. Thus, the calculated half-life of 4a is around 94 s at room temperature (Fig. 6b). In order to learn more about the torsional strain energy of 4a, we calculated the hydrogenation energies of 4a and 4c, respectively (Fig. 6c). Considering the structural differences between 4a and 4c, we further calculated the hydrogenation energy of noncyclic compounds 4a′ and 4c′ (for details see Supplementary Table 2). Thus, the torsional strain energy of 4a is calculated as below:
Markedly, the treatment of [Rh(CO)2Cl]2 with one molar ratio of phosphoramidite L7 in dichloromethane formed a yellowish dimer [Rh(CO)Cl(L7)]2 by releasing two molecules of CO. Recrystallization of this complex in ethyl acetate/hexanes gave a reddish orange crystal that was suitable for X-ray crystallography analysis (CCDC 1969002) (Fig. 6d). In this crystal structure, the (2,4,6-triisopropylphenyl)sulfonylpiperazine moiety worked as a large group by shielding one face of the rhodium center.
Based on the above results and previous studies on Narasaka acylation66, a brief catalytic cycle was tentitatively proposed (Fig. 7). The coordination of L7 to the pre-catalyst formed monomer Rh(I) complex. The oxidative addition of Rh(I) with silafluorene 4b cleavaged C(Ar)–Si bond to form 10, which gave optically active biaryl intermediate 11 via reductive elimination forming a Si–O bond. Subsequently, the second oxidation between 11 with acid anhydride 5 delivered Rh(III) complex 12. Reductive elimination of 12 furnished acylation product 13, which would give the final product 6 after hydrolysis of tri(aryl)silyl benzoate moiety.
The oxidative addition of in situ formed Rh(I) catalyst with 4b gives 10. Subsequent reductive elimination forms optically active biaryl intermediate 11. The second oxidation addition of 11 with acid anhydride 5 gives 12, which regenerates Rh(I) catalyst and compound 6 via reductive elimination, followed by hydrolysis.
Discussion
In conclusion, we reported a Rh-catalyzed asymmetric ring-opening acylation reaction for the synthesis of α-silyl biaryl atropisomers, a class of chiral bulky silanols. The torsional strain of five-membered silafluorenes enabled the success of selective cleavage of C(Ar)–Si bond, thus accomplishing an aryl-Narasaka acylation variant. Additional notable merit of this work is the developed sulfonylpiperazine derived phosphoramidite ligands, which showed high stereo-induction in C(Ar)–Si cleavage/ring-opening reaction.
Methods
Typical procedure for the Rh-catalyzed aryl-Narasaka acylation
Under nitrogen atmosphere, to a Schleck tube was added [Rh(CO)2Cl]2 (2.0 mg, 0.005 mmol, 2.5 mol%), L1 (24.5 mg, 0.021 mmol, 10.5 mol%) and dioxane (2 mL) at room temperature and was stirred for 30 min. The solution was transferred via cannula carefully to another Schlenk tube charged with 4a (62 mg, 0.200 mmol, 1.0 equiv), acetic anhydride (28 μL, 0.300 mmol, 1.5 equiv), Na2CO3 (6.4 mg, 0.060 mmol, 30 mol%), and dioxane (2 mL). The tube was capped with a screw cap and stirred at 70 °C for 24 h. After being cooled to room temperature, the mixture was filtered through Celite and the filtrate was concentrated in vacuum and purified by flash column chromatography (PE/EtOAc 90:10) on silica gel to afford 6a (73 mg, 98%, 90% ee). \([{\upalpha}]_D^{20}\)−2.25 (c 1.13, CH2Cl2). HPLC conditions: Chiralcel AD-H, isopropanol/hexane = 10:90, flow: 1.0 mL/min, λ = 254 nm. 1H NMR (500 MHz, CDCl3) δ 8.04 (d, J = 9.0 Hz, 1H), 7.99 (d, J = 8.0 Hz, 1H), 7.94 (d, J = 8.5 Hz, 1H), 7.92 (d, J = 8.5 Hz, 1H), 7.87 (d, J = 8.0 Hz, 1H), 7.80 (d, J = 9.0 Hz, 1H), 7.55–7.50 (m, 1H), 7.49–7.44 (m, 1H), 7.31–7.26 (m, 1H), 7.26–7.22 (m, 1H), 7.17 (d, J = 8.5 Hz, 1H), 7.13 (d, J = 8.5 Hz, 1H), 2.70 (s, 1H), 1.94 (s, 3H), 0.13 (s, 3H), −0.47 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 205.2, 141.5, 138.3, 137.9, 137.4, 134.2, 133.7, 133.5, 132.7, 130.4, 128.7, 128.2, 128.0, 127.6, 127.5, 127.0, 126.6, 126.5, 126.2, 123.9, 30.5, 0.6. HRMS (ESI) calcd for C24H22O2SiNa [M + Na]+ 393.1287, found 393.1288.
Data availability
Additional data supporting the findings described in this paper are available in the Supplementary Information. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Center (CCDC), under deposition numbers CCDC 1968747 (4a), CCDC 19689851 (4c), CCDC 1968743 (6cc), CCDC 2013379 (6gg), CCDC 1969002 (Rh-1), and CCDC 1987455 (9). These data can be obtained free of charge from the Cambridge Crystallographic Data Center via www.ccdc.cam.ac.uk/data_request/cif.
References
Kumarasamy, E., Raghunathan, R., Sibi, M. & Sivaguru, J. Nonbiaryl and heterobiaryl atropisomers: molecular templates with promise for atropselective chemical transformations. Chem. Rev. 115, 11239–11300 (2015).
Bringmann, G. et al. Atroposelective synthesis of axially chiral biaryl compounds. Angew. Chem. Int. Ed. 44, 5384–5427 (2005).
Smyth, J. E., Butler, N. M. & Keller, P. A. A twist of nature-the significance of atropisomers in biological systems. Nat. Prod. Rep. 32, 1562–1583 (2015).
Clayden, J., Moran, W. J., Edwards, P. J. & LaPlante, S. R. The challenge of atropisomerism in drug discovery. Angew. Chem. Int. Ed. 48, 6398–6401 (2009).
Kozlowski, M. C., Morgan, B. J. & Linton, E. C. Total synthesis of chiral biaryl natural products by asymmetric biaryl coupling. Chem. Soc. Rev. 38, 3193–3207 (2009).
Wencel-Delord, J., Panossian, A., Leroux, F. R. & Colobert, F. Recent advances and new concepts for the synthesis of axially stereoenriched biaryls. Chem. Soc. Rev. 44, 3418–3430 (2015).
Li, X., Yang, J. & Kozlowski, M. C. Enantioselective oxidative biaryl coupling reactions catalyzed by 1,5-diazadecalin metal complexes. Org. Lett. 3, 1137–1140 (2001).
Luo, Z. et al. The rational design of novel chiral Oxovanadium(IV) complexes for highly enantioselective oxidative coupling of 2-naphthols. Chem. Commun. 38, 914–915 (2002).
Egami, H. & Katsuki, T. Iron-catalyzed asymmetric aerobic oxidation: oxidative coupling of 2-naphthols. J. Am. Chem. Soc. 131, 6082–6083 (2009).
Hazra, C. K., Dherbassy, Q., Wencel-Delord, J. & Colobert, F. Synthesis of axially chiral biaryls through sulfoxide-directed asymmetric mild C-H activation and dynamic kinetic resolution. Angew. Chem. Int. Ed. 53, 13871–13875 (2014).
Hayashi, T., Hayashizaki, K., Kiyoi, T. & Ito, Y. Asymmetric synthesis catalyzed by chiral Ferrocenylphosphine-transition-metal complexes. 6. practical asymmetric synthesis of 1,1′-binaphthyls via asymmetric cross-coupling with a chiral [(alkoxyalkyl)ferrocenyl]monophosphine/Nickel catalyst. J. Am. Chem. Soc. 110, 8153–8156 (1988).
Genov, M., Almorín, A. & Espinet, P. Efficient synthesis of chiral 1,1′-binaphthalenes by the asymmetric Suzuki-Miyaura reaction: dramatic synthetic improvement by simple purification of naphthylboronic acids. Chem. Eur. J. 12, 9346–9352 (2006).
Uozumi, Y., Matsuura, Y., Arakawa, T. & Yamada, Y. M. A. Asymmetric Suzuki-Miyaura coupling in water with a chiral palladium catalyst supported on an amphiphilic resin. Angew. Chem. Int. Ed. 48, 2708–2710 (2009).
Bermejo, A., Ros, A., Fernandez, R. & Lassaletta, J. M. C2-symmetric bis-hydrazones as ligands in the asymmetric Suzuki-Miyaura cross-coupling. J. Am. Chem. Soc. 130, 15798–15799 (2008).
Shen, X., Jones, G. O., Watson, D. A., Bhayana, B. & Buchwald, S. L. Enantioselective synthesis of axially chiral biaryls by the Pd-catalyzed Suzuki-Miyaura reaction: substrate scope and quantum mechanical investigations. J. Am. Chem. Soc. 132, 11278–11287 (2010).
Xu, G., Fu, W., Liu, G., Senanayake, C. H. & Tang, W. Efficient syntheses of Korupensamines A, B and Michellamine B by asymmetric Suzuki-Miyaura coupling reactions. J. Am. Chem. Soc. 136, 570–573 (2014).
Jia, Z. J. et al. General enantioselective C-H activation with efficiently tunable cyclopentadienyl ligands. Angew. Chem. Int. Ed. 56, 2429–2434 (2018).
Shen, D., Xu, Y. & Shi, S.-L. A bulky chiral N-heterocyclic carbene palladium catalyst enables highly enantioselective Suzuki–Miyaura cross-coupling reactions for the synthesis of biaryl atropisomers. J. Am. Chem. Soc. 141, 14938–14945 (2019).
Ros, A. et al. Dynamic kinetic cross-coupling strategy for the asymmetric synthesis of axially chiral heterobiaryls. J. Am. Chem. Soc. 135, 15730–15733 (2013).
Bhat, V., Wang, S., Stoltz, B. M. & Virgil, S. C. Asymmetric synthesis of QUINAP via dynamic kinetic resolution. J. Am. Chem. Soc. 135, 16829–16832 (2013).
Zheng, J. & You, S. L. Construction of axial chirality by rhodium-catalyzed asymmetric dehydrogenative Heck coupling of biaryl compounds with alkenes. Angew. Chem. Int. Ed. 53, 13244–13247 (2014).
Yao, Q. J., Zhang, S., Zhan, B. B. & Shi, B. F. Atroposelective synthesis of axially chiral biaryls by Palladium-catalyzed asymmetric C–H olefination enabled by a transient chiral auxiliary. Angew. Chem. Int. Ed. 56, 6617–6621 (2017).
Jolliffe, J. D., Armstrong, R. J. & Smith, M. D. Catalytic enantioselective synthesis of atropisomeric biaryls by a cation-directed O-alkylation. Nat. Chem. 9, 558–562 (2017).
Moustafa, G. A., Oki, Y. & Akai, S. Lipase-catalyzed dynamic kinetic resolution of C1-and C2-symmetric racemic axially chiral 2, 2′-dihydroxy-1, 1′-biaryls. Angew. Chem. Int. Ed. 57, 10278–10282 (2018).
Wang, Q., Cai, Z.-J., Liu, C.-X., Gu, Q. & You, S.-L. Rhodium-catalyzed atroposelective C–H arylation: efficient synthesis of axially chiral heterobiaryls. J. Am. Chem. Soc. 141, 9504–9510 (2019).
Li, H. et al. Enantioselective synthesis of C–N axially chiral N-aryloxindoles by asymmetric rhodium-catalyzed dual C–H activation. Angew. Chem. Int. Ed. 58, 6732–6736 (2019).
Nishii, Y., Wakasugi, K., Koga, K. & Tanabe, Y. Chirality exchange from sp3 central chirality to axial chirality: benzannulation of optically active diaryl-2,2-dichlorocyclopropylmethanols to axially chiral α-arylnaphthalenes. J. Am. Chem. Soc. 126, 5358–5359 (2004).
Guo, F., Konkol, L. C. & Thomson, R. J. Enantioselective synthesis of biphenols from 1,4-diketones by traceless central-to-axial chirality exchange. J. Am. Chem. Soc. 133, 18–20 (2011).
Quinonero, O. et al. Combining organocatalysis with central-to-axial chirality conversion: atroposelective Hantzsch-type synthesis of 4-arylpyridines. Angew. Chem. Int. Ed. 55, 1401–1405 (2016).
Gutnov, A. et al. Cobalt(I)-catalyzed asymmetric [2+2+2] cycloaddition of alkynes and nitriles: synthesis of enantiomerically enriched atropoisomers of 2-arylpyridines. Angew. Chem. Int. Ed. 43, 3795–3797 (2004).
Tanaka, K., Nishida, G., Wada, A. & Noguchi, K. Enantioselective synthesis of axially chiral phthalides through cationic [RhI(H8-binap)]-catalyzed cross alkyne cyclotrimerization. Angew. Chem. Int. Ed. 43, 6510–6512 (2004).
Shibata, T., Fujimoto, T., Yokota, K. & Takagi, K. Iridium complex-catalyzed highly enantio- and diastereoselective [2+2+2] cycloaddition for the synthesis of axially chiral teraryl compounds. J. Am. Chem. Soc. 126, 8382–8383 (2004).
Xue, F. & Hayashi, T. Asymmetric synthesis of axially chiral 2-aminobiaryls by Rhodium-catalyzed benzannulation of 1-arylalkynes with 2-(cyanomethyl) phenylboronates. Angew. Chem. Int. Ed. 57, 10368–10372 (2018).
Zheng, S. C., Wang, Q. & Zhu, J. Catalytic atropenantioselective heteroannulation between isocyanoacetates and alkynyl ketones: synthesis of enantioenriched axially chiral 3-arylpyrroles. Angew. Chem. Int. Ed. 58, 1494–1498 (2019).
Wang, Y.-B. & Tan, B. Construction of axially chiral compounds via asymmetric organocatalysis. Acc. Chem. Res. 51, 534–547 (2018).
Li, G.-Q., Gao, H., Keene, C., Devonas, M., Ess, D. H. & Kürti, L. Organocatalytic aryl-aryl bond formation: an atroposelective [3,3]-rearrangement approach to BINAM derivatives. J. Am. Chem. Soc. 135, 7414–7417 (2013).
Link, A. & Sparr, C. Organocatalytic atroposelective Aldol condensation: synthesis of axially chiral biaryls by arene formation. Angew. Chem. Int. Ed. 53, 5458–5461 (2014).
Chen, Y.-H. et al. Atroposelective synthesis of axially chiral biaryldiols via organocatalytic arylation of 2-naphthols. J. Am. Chem. Soc. 137, 15062–15065 (2015).
Yu, C., Huang, H., Li, X., Zhang, Y. & Wang, W. Dynamic kinetic resolution of biaryl lactones via a chiral bifunctional amine thiourea-catalyzed highly atropo-enantioselective transesterification. J. Am. Chem. Soc. 138, 6956–6959 (2016).
Lotter, D., Castrogiovanni, A., Neuburger, M. & Sparr, C. Catalyst-controlled stereodivergent synthesis of atropisomeric multiaxis systems. ACS Cent. Sci. 4, 656–660 (2018).
Bringmann, G. & Menche, D. Stereoselective total synthesis of axially chiral natural products via biaryl lactones. Acc. Chem. Res. 34, 615–624 (2001).
Bringmann, G. & Reuscher, H. Atropdiastereoselective ring opening of bridged, “axial-prostereogenic” biaryls: directed synthesis of (+)-ancistrocladisine. Angew. Chem. Int. Ed. Engl. 28, 1672–1673 (1989).
Chen, G.-Q. et al. Design and synthesis of chiral oxa-spirocyclic ligands for Ir-catalyzed direct asymmetric reduction of Bringmann’s Lactones with molecular H2. J. Am. Chem. Soc. 140, 8064–8068 (2018).
Shimada, T., Cho, Y.-H. & Hayashi, T. Nickel-catalyzed asymmetric Grignard cross-coupling of dinaphthothiophene giving axially chiral 1, 1′-Binaphthyls. J. Am. Chem. Soc. 124, 13396–13397 (2002).
Kian, A., Miki, H., Cho, Y.-H. & Hayashi, T. Palladium-catalyzed Heck and carbonyl reactions of a dinaphthaleneiodonium salts forming functionalized 2-iodo-1,1′-binaphthyls. Adv. Synth. Catal. 346, 1728–1732 (2004).
Zhao, K. et al. Enhanced reactivity by torsional strain of cyclic diaryliodonium in Cu-catalyzed enantioselective ring-opening reaction. Chem 4, 599–612 (2018).
Li, B., Chao, Z., Li, C. & Gu, Z. Cu-catalyzed enantioselective ring opening of cyclic diaryliodoniums toward the synthesis of chiral diarylmethanes. J. Am. Chem. Soc. 140, 9400–9403 (2018).
Zhu, K. et al. Enantioselective synthesis of axially chiral biaryls via Cu-catalyzed acyloxylation of cyclic diaryliodonium salts. ACS Catal. 9, 4951–4957 (2019).
Deng, R., Xi, J., Li, Q. & Gu, Z. Enantioselective carbon-carbon bond cleavage for biaryl atropisomers synthesis. Chem 5, 1834–1846 (2019).
Ramesh, R. & Reddy, D. S. Quest for novel chemical entities through incorporation of Silicon in drug scaffolds. J. Med. Chem. 61, 3779–3798 (2018).
Barraza, S. J. & Denmark, S. E. Synthesis, reactivity, functionalization, and ADMET properties of Silicon-containing Nitrogen heterocycles. J. Am. Chem. Soc. 140, 6668–6684 (2018).
Toyama, H. et al. Altered activity profile of a tertiary silanol analog of multitargeting nuclear receptor modulator T0901317. Bioorg. Med. Chem. Lett. 26, 1817–1820 (2016).
Min, T., Fettinger, J. C. & Franz, A. K. Enantiocontrol with a hydrogen-bond directing pyrrolidinylsilanol catalyst. ACS Catal. 2, 1661–1666 (2012).
Beemelmanns, C., Husmann, R., Whelligan, D. K., Özҫubukҫu, S. & Bolm, C. Planar-chiral bis-silanols and diols as H-bonding asymmetric organocatalysts. Eur. J. Org. Chem. 2012, 3373–3376 (2012).
Hatanaka, Y. & Hiyama, T. Cross-coupling of organosilanes with organic halides mediated by a palladium catalyst and tris(diethylamino)sulfonium difluorotrimethylsilicate. J. Org. Chem. 53, 918–920 (1988).
Denmark, S. E., Wehrli, D. & Choi, J. Y. Convergence of mechanistic pathways in the palladium(0)-catalyzed cross-coupling of alkenylsilacyclobutanes and alkenylsilanols. Org. Lett. 2, 2491–2494 (2002).
Yamane, M., Uera, K. & Narasaka, K. Rhodium-catalyzed acylation of vinylsilanes with acid anhydrides: application to the transformation of α-acyloxy vinylsilanes to unsymmetrical 1,2-diketones. Chem. Lett. 33, 424–425 (2004).
Yamane, M., Uera, K. & Narasaka, K. Rhodium-catalyzed acylation of vinylsilanes with acid anhydrides. Bull. Chem. Soc. Jpn. 78, 477–486 (2005).
Wang, L. & Duan, Z. Formation of silacycles via metal-mediated or catalyzed Si-C bond cleavage. Chin. Sci. Bull. 58, 307–315 (2013).
Tobisu, M., Onoe, M., Kita, Y. & Chatani, N. Rhodium-catalyzed coupling of 2-silylphenylboronic acids with alkynes leading to benzosiloles: catalytic cleavage of the carbon-silicon bond in trialkylsilyl groups. J. Am. Chem. Soc. 131, 7506–7507 (2009).
Liang, Y., Zhang, S. G. & Xi, Z. Palladium-catalyzed synthesis of benzosilolo[2,3-b] indoles via cleavage of a C(sp3)–Si bond and consequent intramolecular C(sp2)–Si coupling. J. Am. Chem. Soc. 133, 9204–9207 (2011).
Mochida, K., Shimizu, M. & Hiyama, T. Palladium-catalyzed intramolecular coupling of 2-[(2-pyrrolyl)silyl]aryl triflates through 1,2-silicon migration. J. Am. Chem. Soc. 131, 8350–8351 (2009).
Wang, K. et al. Selective manganese-catalyzed oxidation of hydrosilanes to silanols under neutral reaction conditions. Angew. Chem. Int. Ed. 58, 6380–6384 (2019).
Wang, J. et al. Metal-free visible-light-mediated aerobic oxidation of silanes to silanols. Sci. China Chem. 61, 1594–1599 (2018).
Unni, A. K., Takenaka, N., Yamamoto, H. & Rawal, V. H. (2005). Axially chiral biaryl diols catalyze highly enantioselective hetero-Diels-Alder reactions through hydrogen bonding. J. Am. Chem. Soc. 127, 1336–1337 (2005).
Zaranek, M. et al. Iridium-catalysed desilylative acylation of 1-alkenylsilanes. J. Mol. Catal. A Chem. 426, 75–78 (2017).
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (no. 21871241, 21901236), Strategic Priority Research Program of the Chinese Academy of Sciences (XDB20000000), and the Fundamental Research Funds for the Central Universities (WK2060190086). We thank the Supercomputing Center of University of Science and Technology of China for providing the computational resources.
Author information
Authors and Affiliations
Contributions
J.F. designed the approach and performed the experiments, analyzed the experimental data, and prepared the Supplementary Information. X.B. modified the reaction conditions, expanded the scope of the substrates. J.F. and X.B. contributed equally to this project. X.X. performed the calculation. N.L. and L.S. were involved in the preparation of substrates. Z.G. directed the investigations and prepared the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Fengzhi Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Feng, J., Bi, X., Xue, X. et al. Catalytic asymmetric C–Si bond activation via torsional strain-promoted Rh-catalyzed aryl-Narasaka acylation. Nat Commun 11, 4449 (2020). https://doi.org/10.1038/s41467-020-18273-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-18273-3
This article is cited by
-
Pd-catalyzed intermolecular Si–O formation via Si–C activation
Science China Chemistry (2024)
-
Recent advances in the construction of axially chiral arylpyrroles
Science China Chemistry (2023)
-
Sila-spirocyclization involving unstrained C(sp3)−Si bond cleavage
Nature Communications (2022)
-
Enantioselective alkylative cross-coupling of unactivated aromatic C–O electrophiles
Nature Communications (2022)
-
Organocatalytic dynamic kinetic resolution of N-arylindole lactams: atroposelective construction of axially chiral amino acids bearing a C-N chiral axis
Science China Chemistry (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.