Abstract
Polyhedra are ubiquitous in chemistry, biology, mathematics and other disciplines. Coordination-driven self-assembly has created molecules mimicking Platonic, Archimedean and even Goldberg polyhedra, however, nesting multiple polyhedra in one cluster is challenging, not only for synthesis but also for determining the alignment of the polyhedra. Here, we synthesize a nested Ag90 nanocluster under solvothermal condition. This pseudo-Th symmetric Ag90 ball contains three concentric Ag polyhedra with apparently incompatible symmetry. Specifically, the inner (Ag6) and middle (Ag24) shells are octahedral (Oh), an octahedron (a Platonic solid with six 3.3.3.3 vertices) and a truncated octahedron (an Archimedean solid with twenty-four 4.6.6 vertices), whereas the outer (Ag60) shell is icosahedral (Ih), a rhombicosidodecahedron (an Archimedean solid with sixty 3.4.5.4 vertices). The Ag90 nanocluster solves the apparent incompatibility with the most symmetric arrangement of 2- and 3-fold rotational axes, similar to the arrangement in the model called Kepler’s Kosmos, devised by the mathematician John Conway.
Similar content being viewed by others
Introduction
Encouraged by masterpieces of self-assembly in biology1,2,3,4,5, some seminal metal clusters6 of nanometer size have been assembled from simple components7,8,9,10,11,12, such as Al7713, Gd14014, Pd14515, Ag37416, and Au24617. Among families of metal clusters, silver nanoclusters benefit from the exceptional versatility of silver(I) atoms, which have flexible coordination preferences, a tendency to form argentophilic interactions and a susceptibility to reduction, all properties enriching the number of members and types of this family18,19,20. To obtain structures with polyhedral geometry21,22,23,24, silver polygons can form with the assistance of surface ligands, inner anion templates and argentophilic interactions. Nonetheless, most silver nanoclusters lack typical polyhedral features. Exceptionally, in 2017, we synthesized a buckyball-like Goldberg cage with 180 Ag atoms25.
Even more complex is a nested silver(I) nanocluster with two or more chemically bound metallic shells. Examples include Ag56 (Ag14 ⊂ Ag42)26, Ag60 (Ag12 ⊂ Ag48)27, Ag62 (Ag14 ⊂ Ag48)28, and Ag78 (Ag18 ⊂ Ag60)29, but even in these cases, most of the shells lack typical polyhedral features, and the number of silver shells is just two. We assembled the first three-shell Ag73 silver nanocluster with a central Ag atom in an Ag24 rhombicuboctahedron in an Ag48 octahedral Goldberg 2,0 polyhedron30. Although there has been progress, the synthetic challenges in making single polyhedral and nested polyhedral silver nanoclusters remain due to the difficulty of precisely shaping silver polygons and obtaining polyhedral shells with compatible symmetry. Here, we report the synthesis and characterization of nested Ag90 nanoclusters which show pseudo-Th symmetry and contain three concentric silver polyhedra with apparently incompatible symmetry. The nested silver shells can be expressed as Ag6 ⊂ Ag24 ⊂ Ag60 and belong to octahedron, truncated octahedron, and rhombicosidodecahedron, respectively. The Ag90 nanocluster solves the apparent incompatibility with the most symmetric arrangement of two- and threefold rotational axes.
Results
Structures of SD/Ag90a and SD/Ag90b
With a combination of anion-template and geometrical-polyhedron strategies31,32,33,34,35,36,37 and with careful choice of organic ligands (tBuSH and PhPO3H2) and anion templates (S2− and PO43−), we one-pot synthesized Ag90 nanoclusters under the solvothermal condition as dark brown or red rhombic crystals, depending on the polymorphs. They are stable under ambient conditions because the bulk sample of them can keep the color and morphology unchanged for at least one month. The PO43− ion was considered as anion-template mainly due to its high negative charge and Td symmetry. The former feature can aggregate more Ag+ ions to form high-nuclearity cluster through electrostatic attraction, whereas the latter can shape cluster with a T-related symmetry. The Ag90 nanoclusters can be crystallized into monoclinic P21/n or triclinic P-1 phases dictated by the silver salts used (Fig. 1), hereafter denoted as SD/Ag90a and SD/Ag90b, respectively. Although the different anions, PhCOO− and CH3SO3−, did not participate in the final structures of the Ag90 clusters, they may have influenced the crystallization process through supramolecular interactions such as hydrogen bonds, which causes the formation of the ultimate crystalline phases. Other common silver salts, such as AgBF4, CF3COOAg, CF3SO3Ag, and AgNO3, were tried in the above synthesis experiments, but none produced desired clusters. A series of characterization techniques such as single-crystal X-ray crystallography, infrared spectroscopy, 31P nuclear magnetic resonance, ultraviolet–visible absorption spectroscopy, fluorescence spectroscopy, and thermogravimetric analysis were used on these two nests (Supplementary Figs. 1–5, 13–17 and Supplementary Tables 1–3). The electrospray ionization mass spectrometry (ESI-MS) of SD/Ag90a dissolved in CH2Cl2 or CH3OH did not give useful data, which indicates that either (i) SD/Ag90a is fragmented during the ionization process or (ii) it is neutral and is hard to ionize under mass spectrometer conditions—even when we added CsOAc to aid in ionization38.
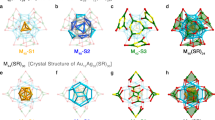
As deduced from crystallography, SD/Ag90a (Fig. 2a) and SD/Ag90b (Supplementary Fig. 6a) have the same molecular structure and composition of [(PO4)8@Ag90S6(tBuS)24(PhPO3)12(PhPO3H)6], but they crystallize into different space groups (Supplementary Table 1) due to different cluster packing in their unit cells (Supplementary Figs. 7 and 8). The straightforward single-crystal X-ray diffraction (SCXRD) of crystals at 100 K unambiguously established ball-shaped structures for the Ag90 nanoclusters (Fig. 2b–e; Supplementary Figs. 6b–e, 9, 10). Due to the similarity of their molecular structures, we take SD/Ag90a as representative for discussions in detail below.
a Total cluster structure of SD/Ag90a incorporating Van der Waals surfaces. Hydrogen atoms are removed for clarity. Color legend: Ag, purple; P, brown; S, yellow; O, red; C, gray. b The ball-and-stick mode of the triply nested polyhedral silver skeleton, viewed down an axis through the front silver 5-gon. The three different shells are individually colored. c The icosahedral Ag60 rhombicosidodecahedron. d The octahedral Ag24 truncated octahedron. e The octahedral Ag6 octahedron. f The interactions between eight PO43− and three different shells. g The detailed coordination of PO43− towards different silver polygons in three different shells. All PO43− ions are shown as yellow tetrahedra. h Six μ8-S2− ions intercalate the aperture between Ag24 and Ag60 shells of SD/Ag90a by linking two 4-gons up and down from these shells, respectively.
SD/Ag90a crystallized into the monoclinic P21/n space group and conformed to pseudo-Th symmetry. SD/Ag90a is a neutral cluster with all Ag(I) atoms in 3- or 4-coordination with S and/or O atoms. The all-silver framework (Fig. 2b) is composed of three concentric nested polyhedra, an outer Ag60 rhombicosidodecahedron with 60 3.4.5.4 vertices (Fig. 2c), a middle Ag24 truncated octahedron with 24 4.6.6 vertices (Fig. 2d), and an inner Ag6 octahedron with 6 3.3.3.3 vertices (Fig. 2e), where the numbers 5, 4, and 3 represent faces around that vertex, respectively, 5-gons, 4-gons, and 3-gons. Of note, the outer Ag60 shell is geometrically reminiscent of the third shell (Pd60) in Dahl’s Pd145 cluster15. All vertices on these three polyhedra are Ag(I) atoms, and all edges are built from the connection of adjacent two Ag atoms. The Ag–Ag edge lengths in outer, middle, and inner shells range from 2.96 to 4.03, 3.02 to 3.48, and 3.51–3.61 Å, respectively (Supplementary Fig. 11 and Supplementary Table 3). Some of these Ag···Ag edge lengths, shorter than 3.44 Å, twice the Van der Waals radius of silver(I) ion, can be deemed as argentophilic interactions that contribute to the stability of the silver shells. Of note, the long Ag···Ag edges in the Ag6 octahedron also rule out a subvalent nature, which usually produces short Ag···Ag distances approximating to 2.88 Å39. By measuring distances between inversion-related pairs of Ag atoms in the same shell, the diameters of outer, middle and inner shells are determined to be 1.5, 1.0, and 0.5 nm, respectively.
Each μ12-κ3:κ3:κ3:κ3 PO43− ion (as anion-template) penetrates the hexagonal windows of the Ag24 shell to connect all three silver shells (Ag-O: 2.3–2.6 Å) by linking two silver 3-gons of the Ag6 and Ag60 shells and one 6-gon of the Ag24 shell (Fig. 2f, g). Six μ8-S2− ions from in situ decomposition of tBuSH intercalate the aperture between Ag24 and Ag60 shells (Ag-S: 2.43–2.89 Å) by linking two 4-gons up and down from these shells, respectively (Fig. 2h)28. Based on the above analysis, we found that the tetrahedral PO43− ion has a special role in shaping silver 3-gons and 6-gons, essential elements to construct the rhombicosidodecahedron and the truncated octahedron, respectively. As for the spherical S2− ion, it assists in fabricating the silver 4-gon, an essential element for both the rhombicosidodecahedron and the truncated octahedron. Both inorganic anions act not only as templates to shape the silver polyhedra by defining the essential polygon elements but also function as glue to consolidate the overall nested silver shells.
The ligand coverage on the surface of the outer Ag60 shell is polygon selective with 24 tBuS− and 6 PhPO3H− on thirty 4-gons and 12 PhPO32− on twelve 5-gons. There are no ligands capping the twenty 3-gons. The PhPO3H2 ligand exhibits two kinds of deprotonated forms, PhPO32− and PhPO3H−, that respectively coordinate with twelve 5-gons (μ5-κ2:κ2:κ1) and six 4-gons (μ4-κ2:κ2) on the surface of the Ag60 shell (Ag-O: 2.2–2.6 Å). The 31P NMR (nuclear magnetic resonance) of the digestion solution of SD/Ag90a shows two sharp peaks with chemical shifts at δ = −1.07 and 15.62 ppm (Supplementary Fig. 2), corresponding to H3PO4 and PhPO3H2, respectively, which clearly verify the existence of two different P-containing chemicals in SD/Ag90a. From the ligation modes of each coordinative component, we suggest that all of them play roles to shape different silver polygons, paving the way for further construction of polyhedral silver nanoclusters. The overall structure is reinforced by a combination of argentophilic interactions (<3.44 Å) and the scaffolding provided by all other coordination bonds. The remarkable structure of SD/Ag90a has not been previously observed in the family of silver nanoclusters.
Alignment of shells with compatible point-group symmetry
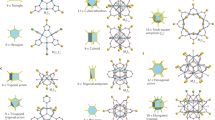
We now ask about the alignment of the icosahedral and octahedral cages in the SD/Ag90a nest. For the dodecahedron and its dual (the icosahedron), both Platonic solids with icosahedral (Ih) symmetry and thus “compatible”, nesting may be based on alignment of all of the five-, three- and twofold axes of rotational symmetry (Fig. 3a; Supplementary Table 4). The same full alignment could obtain for nests of any icosahedral structures, including the six icosahedral Archimedean solids and an infinity of other icosahedral structures. Likewise, nesting of octahedral shells like the cube and its dual (the octahedron)—both Platonic solids—with each other (Fig. 3b) and of tetrahedral shells like the tetrahedron and its self-dual (the tetrahedron) (Fig. 3c) with each other may be based on alignment of all rotational axes (Supplementary Table 4). Indeed, the Ag73 cited above is just such a symmetry-compatible nesting of octahedral silver cages30.
The solids, arbitrarily scaled, are labeled by their vertex descriptions: the icosahedral dodecahedron (555) and icosahedron (33333), the octahedral cube (444) and octahedron (3333), and the tetrahedral tetrahedron (333), where 5, 4, and 3 represent faces, respectively, 5-gons, 4-gons, and 3-gons. a Arrangement of the icosahedral solids with alignment along five-, three- and twofold rotational axes. b Arrangement of the octahedral solids with alignment along four-, three- and twofold axes. c Arrangement of the tetrahedron with its self-dual, another tetrahedron, with alignment along threefold axes with a face in front, threefold axes with a vertex in front, and twofold axes.
Alignment of shells with incompatible point-group symmetry
Although it might be assumed that only cages with compatible symmetry (e.g., icosahedral with icosahedral) could be nested, for the Zometool toy the mathematician John Conway created a model called “Kepler’s Kosmos”, a model that aligns the five Platonic solids, the two with icosahedral symmetry, the two with octahedral, and the one with tetrahedral40. As its name suggests, the inspiration for this model dates back to Johannes Kepler. In 1596, in his Mysterium Cosmographicum (The Secret of the Universe)41, Kepler hypothesized that the orbits of the six known planets corresponded with six spheres, five circumscribing the five Platonic solids and one inscribing the smallest. To test his hypothesis, Kepler became mathematical assistant to Tycho Brahe in 1600 and gained access to more than 30 years of astronomical observations. By 1605, Kepler had shown that the orbit of Mars was an ellipse, culminating by 1619 with his discovery of the three laws of planetary motion42, 43 and subsequently Isaac Newton’s discovery of the law of universal gravity44.
Kepler’s Kosmos provides one possible answer to the question of how to align icosahedral polyhedra with fivefold rotational axes but no fourfold, octahedral polyhedra with fourfold axes but no fivefold, and tetrahedral polyhedra with neither. Conway’s arrangement (Fig. 4a; Supplementary Fig. 12a and Supplementary Table 4) has these properties: (i) None of the six fivefold axes of the dodecahedron (or icosahedron) is aligned with any rotational axis of an octahedral or tetrahedral polyhedron. (ii) Four of the ten threefold axes of the dodecahedron (or icosahedron) are aligned with all four threefold axes of the cube (or octahedron) and all four threefold axes of the tetrahedron (Fig. 4b–e). (iii) Three of the fifteen twofold axes of the dodecahedron (or icosahedron) are aligned with all three (orthogonal) fourfold axes of the cube (or octahedron) and all three (orthogonal) twofold axes of the tetrahedron (Fig. 4f–i). This quintuple nest itself has tetrahedral (Td) symmetry.
a A view of the quintuple nest, slightly off a twofold axis, and b along a common threefold axis. c Views along the threefold axis of the double 555 and 444 nest, d the double 555 and 333 nest, and e the double 444 and 333 nest. f A view of the quintuple nest along four- and twofold axes. g Views of the double 555 and 444 nest along two- and fourfold axes, h the double 555 and 333 nest along twofold axes, and i the double 444 and 333 nest along four- and twofold axes. The solids are arbitrarily scaled.
However, other alignments are possible. Here, we devise an alternative alignment with three different combinations of four- and twofold axes to produce a nest with just three different, orthogonal, twofold axes (Fig. 5; Supplementary Fig. 12b, Supplementary Table 4, and Supplementary Movie 1), producing a quintuple nest with lower (D2) symmetry. Of course, it is also possible to align none of the rotational axes, producing a quintuple nest with trivial (C1) symmetry.
a Views of the quintuple nest, arbitrarily scaled, slightly off a twofold axis, b along four- and twofold axes, c along twofold axes (with edges at right angles), and d along twofold axes (with edges parallel). e Views of the double 555 and 444 nest along two- and fourfold axes, f along twofold axes (with edges at right angles, corresponding to c), and g along twofold axes (with edges parallel, corresponding to d). h Views of the double 444 and 333 nest along four- and twofold axes and i along twofold axes. With just three (orthogonal) twofold axes, each one different from the other, but with no mirrors, this regular quintuple nest has D2 symmetry.
Alignment of shells in SD/Ag90a
Given different symmetric arrangements of the three shells—as in the Kepler’s Kosmos with point-group Td (Fig. 4), alignment of only three different orthogonal twofold axes with point-group D2 (Fig. 5) and no alignment of rotational axes (thus C1)—we ask how the shells in SD/Ag90a align. The icosahedral outer shell of SD/Ag90a has fivefold axes, but these are absent in the octahedral middle and inner shells (Fig. 2b). The octahedral inner and middle shells compatibly align all of their four-, three- and twofold axes, as in Fig. 3b. The icosahedral outer shell aligns four of its threefold axes with all four of the threefold axes of the octahedral shells (Fig. 6a) and three of its twofold axes with all three fourfold axes of the octahedral shells (Fig. 6b). Of note, the interstitial anions of S2− and PO43− also have important influences on aligning the three shells. Specifically, the threefold axes of the Ag6, Ag24, and Ag60 shells pass through the PO43− ions, and the twofold axis of Ag60 shell and fourfold axis of Ag24 shell pass through the S2− ions, thus dictating the alignment of three different shells in the unique fashion discussed above. The alignment of these approximate polyhedra is nearly as good as in the same nest with regular polyhedra (Fig. 6c, d). Thus, the arrangement of the icosahedral and octahedral shells in the Ag90 triple nest is the same as in Kepler’s Kosmos (Fig. 4).
a A view through the common threefold axes of SD/Ag90a. There are four of these rotational axes. b A view through a twofold axis of the outer icosahedral Ag shell of SD/Ag90a, which corresponds with a fourfold axis through the middle and inner octahedral Ag shells. There are three of these rotational axes, arranged orthogonally. c, d The corresponding triple nest with regular polyhedral shells viewed along the three- and twofold axes of the nest.
However, without a tetrahedral shell, SD/Ag90a is a subset of Kepler’s Kosmos, with just Ih (3.4.5.4) and Oh (4.6.6 and 3.3.3.3) shells. As both Ih and Oh structures have inversion symmetry, their combination in SD/Ag90a also has inversion symmetry. Thus, the regular version of SD/Ag90a (Fig. 6c, d), with four threefold axes, three twofold axes, mirrors, inversion, and a symmetry order of 24, has Th symmetry (Supplementary Table 4).
Optical properties of SD/Ag90a
The solid-state ultraviolet–visible (UV/Vis) absorption spectra of SD/Ag90a and [tBuSAg]n were measured at room temperature. As shown in Fig. 7a, SD/Ag90a exhibits a wide absorption range spanning UV and Vis regions with an absorption maximum at 419 nm. Compared with the absorption of [tBuSAg]n at 280 nm, the absorption edge is significantly red-shifted in SD/Ag90a, which should be caused by the ligand-to-metal charge transfer (LMCT) or/and cluster-centered (CC) transitions. Based on the Kubelka–Munk function of (αhυ)1/2 = κ(hυ − Eg) (Eg is the band gap (eV), h is Planck’s constant (J·s), υ is the light frequency (s−1), κ is the absorption constant, and α is the absorption coefficient)45, the band gaps of SD/Ag90a and [tBuSAg]n precursor were determined as 0.69 and 2.34 eV, respectively (Supplementary Fig. 3), which indicates that the aggregation of multiple silver atoms into a cluster structure has an important influence on the HOMO–LUMO gap.
We further studied the photocurrent responses of SD/Ag90a and [tBuSAg]n driven by visible-light in a typical three-electrode system (ITO glass as the working electrode, platinum wire as the assisting electrode and Ag/AgCl as the reference electrode) and keeping the bias voltage at 0.6 V. Upon on–off cycling irradiation with LED light (λ = 420 nm; 50 W; intervals of 10 s), clear photocurrent responses were observed for SD/Ag90a and [tBuSAg]n (Fig. 7b). The photocurrent density of SD/Ag90a (0.9 μA cm−2) was five times larger than that of [tBuSAg]n (0.17 μA cm−2), indicating that SD/Ag90a has better generation and separation efficiency of photoinduced electrons/holes pairs in ITO electrodes46. The generation of photocurrent may involve photoinduced charge migration from S 3p to the Ag 5s orbits.
The solid state emission spectra of SD/Ag90a were recorded as a function of temperature from 293 to 93 K with 40 K as an interval, showing luminescence thermochromic behavior (Fig. 8). The luminescence of SD/Ag90a originates from the ligand-to-metal charge transfer transition with a charge transfer from S 3p to Ag 5s orbitals and/or mixed with a cluster-centered transition related to Ag···Ag interactions47. SD/Ag90a shows gradually blue-shifted emissions from 700 to 684 nm (λex = 468 nm) upon cooling, possibly related to enhanced molecular rigidity at lower temperatures. During the cooling process, the emission intensity shows a nearly tenfold increase from 293 to 93 K, which is attributed to reduction of the nonradiative decay at low temperature48,49,50. The emission lifetime of SD/Ag90a (Supplementary Fig. 4), falling in the microsecond scale (τSD/Ag90a = 17.80 μs) at 93 K, suggests a triplet phosphorescence origin.
Discussion
We have successfully synthesized an Ag90 nanocluster with overall pseudo-Th symmetry. This silver nanocluster is divided into three shells as Ag6@Ag24@Ag60 from inner to outer. The Ag6 inner shell is an octahedron (a Platonic solid with 6 3.3.3.3 vertices), the Ag24 middle shell is a truncated octahedron (an Archimedean solid with 24 4.6.6 vertices), and both have octahedral (Oh) symmetry. However, the Ag60 outer shell is a rhombicosidodecahedron (an Archimedean solid with 60 3.4.5.4 vertices and icosahedral (Ih) symmetry). The SD/Ag90a nanocluster solves the apparent incompatibility among their symmetry groups with the most symmetric arrangement of two- and threefold axes. Creation of a nest with all three of the polyhedral symmetries, icosahedral, octahedral and tetrahedral—and resembling Kepler’s Kosmos even more closely—remains an exciting challenge.
Methods
Syntheses of SD/Ag90a and SD/Ag90b
[tBuSAg]n (9.9 mg, 0.05 mmol), PhCOOAg (11.5 mg, 0.05 mmol), Na3PO4·12H2O (5 mg, 0.013 mmol), and PhPO3H2 (4.7 mg, 0.03 mmol) were mixed in 6.5 mL MeOH/MeCN/DMF (v:v:v = 6:6:1). The resulting suspension was sealed and heated at 65 °C for 2000 min. After cooling to room temperature, dark brown crystals of SD/Ag90a were formed (yield: 30%). Combustion elementary analysis calculated (experimental) for SD/Ag90a: (C204H312Ag90O86P26S30): C, 15.69 (15.71%); H, 2.01 (1.99%). Selected IR peaks (cm−1) of SD/Ag90a: 3664 (w), 2983 (w), 2894 (w), 1150 (w), 1104 (w), 1040 (m), 1007 (m), 928 (s), 747 (m), 715 (w), 689 (m), and 553 (s).
SD/Ag90b was synthesized similarly to SD/Ag90a but using CH3SO3Ag (10.2 mg, 0.05 mmol) instead of PhCOOAg. SD/Ag90b precipitated as red crystals from the evaporation of filtrate after solvothermal reaction for 1–2 weeks (yield: 19%).
Data availability
The X-ray crystallographic coordinates for structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC: 1913186–1913187. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
References
Bale, J. B. et al. Accurate design of megadalton-scale two-component icosahedral protein complexes. Science 353, 389–394 (2016).
He, Y. et al. Hierarchical self-assembly of DNA into symmetric supramolecular polyhedra. Nature 452, 198–201 (2008).
Douglas, S. M. et al. Self-assembly of DNA into nanoscale three-dimensional shapes. Nature 459, 414–418 (2009).
Lawson, D. M. et al. Solving the structure of human H ferritin by genetically engineering intermolecular crystal contacts. Nature 349, 541–544 (1991).
Caspar, D. L. D. & Klug, A. Physical principles in construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 27, 1–24 (1962).
Fujita, D. et al. Self-assembly of tetravalent Goldberg polyhedra from 144 small components. Nature 540, 563–566 (2016).
Bai, J. F., Virovets, A. V. & Scheer, M. Synthesis of inorganic fullerene-like molecules. Science 300, 781–783 (2003).
Moses, M. J., Fettinger, J. C. & Eichhorn, B. W. Interpenetrating As20 fullerene and Ni12 icosahedra in the onion-skin [As@Ni12@As20]3- ion. Science 300, 778–780 (2003).
Kong, X.-J., Long, L.-S., Zheng, Z., Huang, R.-B. & Zheng, L.-S. Keeping the ball rolling: fullerene-like molecular clusters. Acc. Chem. Res. 43, 201–209 (2010).
Desireddy, A. et al. Ultrastable silver nanoparticles. Nature 501, 399–402 (2013).
Chen, Y. et al. Crystal structure of barrel-shaped chiral Au130(p-MBT)50 nanocluster. J. Am. Chem. Soc. 137, 10076–10079 (2015).
Du, W. et al. Ag50(Dppm)6(SR)30 and its homologue AuxAg50-x(Dppm)6(SR)30 alloy nanocluster: seeded growth, structure determination, and differences in properties. J. Am. Chem. Soc. 139, 1618–1624 (2017).
Ecker, A., Weckert, E. & Schnockel, H. Synthesis and structural characterization of an Al77 cluster. Nature 387, 379–381 (1997).
Zheng, X. Y. et al. A gigantic molecular wheel of Gd140: a new member of the molecular wheel family. J. Am. Chem. Soc. 139, 18178–18181 (2017).
Tran, N. T., Powell, D. R. & Dahl, L. F. Nanosized Pd145(CO)x(PEt3)30 containing a capped three-shell 145-atom metal-core geometry of pseudo icosahedral symmetry. Angew. Chem. Int. Ed. 39, 4121–4125 (2000).
Yang, H. et al. Plasmonic twinned silver nanoparticles with molecular precision. Nat. Commun. 7, 12809 (2016).
Zeng, C., Chen, Y., Kirschbaum, K., Lambright, K. J. & Jin, R. Emergence of hierarchical structural complexities in nanoparticles and their assembly. Science 354, 1580–1584 (2016).
Young, A. G. & Hanton, L. R. Square planar silver(I) complexes: a rare but increasingly observed stereochemistry for silver(I). Coord. Chem. Rev. 252, 1346–1386 (2008).
Schmidbaur, H. & Schier, A. Argentophilic interactions. Angew. Chem. Int. Ed. 54, 746–784 (2015).
Bhattarai, B. et al. Chemistry and structure of silver molecular nanoparticles. Acc. Chem. Res. 51, 3104–3113 (2018).
Coxeter, H. S. M., Longuet-Higgins, M. S. & Miller, J. C. P. Uniform polyhedra. Philos. Trans. R. Soc. Ser. A-Math. Phys. Sci. 246, 401–450 (1954).
Baker, T. S., Olson, N. H. & Fuller, S. D. Adding the third dimension to virus life cycles: three-dimensional reconstruction of icosahedral viruses from cryo-electron micrographs. Microbiol. Mol. Biol. Rev. 63, 862–922 (1999).
Liu, C. W. et al. An eleven-vertex deltahedron with hexacapped trigonal bipyramidal geometry. Chem. Commun. 47, 5831–5833 (2011).
Rais, D. et al. Anion-templated syntheses of rhombohedral silver-alkynyl cage compounds. Angew. Chem. Int. Ed. 40, 3464–3467 (2001).
Wang, Z. et al. Assembly of silver trigons into a buckyball-like Ag180 nanocage. Proc. Natl Acad. Sci. USA 114, 12132–12137 (2017).
Sun, D. et al. Two birds with one stone: anion templated ball-shaped Ag56 and disc-like Ag20 clusters. Dalton Trans. 42, 6281–6284 (2013).
Wang, Z. et al. Johnson solids: anion-templated silver thiolate clusters capped by sulfonate. Chem. Eur. J. 24, 1640–1650 (2018).
Li, G., Lei, Z. & Wang, Q.-M. Luminescent molecular Ag-S nanocluster [Ag62S13(SBut)32](BF4)4. J. Am. Chem. Soc. 132, 17678–17679 (2010).
Cheng, L.-P. et al. Small size yet big action: a simple sulfate anion templated a discrete 78-nuclearity silver sulfur nanocluster with a multishell structure. Chem. Commun. 54, 2361–2364 (2018).
Su, Y.-M. et al. Three silver nests capped by thiolate/phenylphosphonate. Chem. Eur. J. 24, 15096–15103 (2018).
Wang, Q.-M., Lin, Y.-M. & Liu, K.-G. Role of anions associated with the formation and properties of silver clusters. Acc. Chem. Res. 48, 1570–1579 (2015).
Liu, H. et al. Acid-base-triggered structural transformation of a polyoxometalate core inside a dodecahedrane-like silver thiolate shell. Angew. Chem. Int. Ed. 55, 3699–3703 (2016).
Xie, Y.-P., Jin, J.-L., Lu, X. & Mak, T. C. W. High-nuclearity silver thiolate clusters constructed with phosphonates. Angew. Chem. Int. Ed. 54, 15176–15180 (2015).
Li, S. et al. Atom-precise modification of silver(I) thiolate cluster by shell ligand substitution: a new approach to generation of cluster functionality and chirality. J. Am. Chem. Soc. 140, 594–597 (2018).
Wang, Z. et al. Trapping an octahedral Ag6 kernel in a seven-fold symmetric Ag56 nanowheel. Nat. Commun. 9, 2094 (2018).
Liu, X., Yang, H., Zheng, N. & Zheng, L. Bromide-induced formation of a highly symmetric silver thiolate cluster containing 36 silver atoms from an infinite polymeric silver thiolate. Eur. J. Inorg. Chem. 2010, 2084–2087 (2010).
Bootharaju, M. S. et al. A new class of atomically precise, hydride-rich silver nanoclusters co-protected by phosphines. J. Am. Chem. Soc. 138, 13770–13773 (2016).
Chen, T., Yao, Q., Nasaruddin, R. R. & Xie, J. Electrospray ionization mass spectrometry: a powerful platform for noble-metal nanocluster analysis. Angew. Chem. Int Ed. 58, 11967–11977 (2019).
Yonesato, K. et al. Controlled assembly synthesis of atomically precise ultrastable silver nanoclusters with polyoxometalates. J. Am. Chem. Soc. 141, 19550–19554 (2019).
Baer, S., Vorthmann, S., Wildung, A. & Hildebrandt, P. Kepler’s Kosmos. http://www.zometool.com/products/keplers-kosmos.html (2009).
Kepler, J. Mysterium Cosmographicum (The Secret of the Universe) (Georg Gruppenbach, Tübingen, (1596).
Kepler, J. Astronomia Nova (New Astronomy) (Voegelin, Heidelberg, 1609).
Kepler, J. Harmonices Mundi (The Harmony of the World) (Johann Planck, Linz, Austria, 1619).
Newton, I. Philosophiae Naturalis Principia Mathematica (Royal Society, London, 1687).
Chaki, N. K. et al. Controlling band gap energies in cluster-assembled ionic solids through internal electric fields. ACS Nano 4, 5813–5818 (2010).
Wu, T. et al. Monocopper doping in Cd-In-S supertetrahedral nanocluster via two-step strategy and enhanced photoelectric response. J. Am. Chem. Soc. 135, 10250–10253 (2013).
Yam, V. W.-W., Au, V. K.-M. & Leung, S. Y.-L. Light-emitting self-assembled materials based on d8 and d10 transition metal complexes. Chem. Rev. 115, 7589–7728 (2015).
Jiang, X.-F., Hau, F. K.-W., Sun, Q.-F., Yu, S.-Y. & Yam, V. W.-W. From {AuI···AuI}-coupled cages to the cage-built 2-D {AuI···AuI} arrays: AuI···AuI bonding interaction driven self-assembly and their AgI sensing and photo-switchable behavior. J. Am. Chem. Soc. 136, 10921–10929 (2014).
Allendorf, M. D., Bauer, C. A., Bhakta, R. K. & Houk, R. J. T. Luminescent metal-organic frameworks. Chem. Soc. Rev. 38, 1330–1352 (2009).
Yam, V. W. W. et al. Synthesis and luminescence behaviour of mixed-metal rhenium(I)-copper(I) and -silver(I) alkynyl complexes. Coord. Chem. Rev. 245, 39–47 (2003).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant Nos. 91961105, 21822107, and 21571115), the Natural Science Foundation of Shandong Province (Nos. ZR2019ZD45, JQ201803, and ZR2017MB061), the Taishan Scholar Project of Shandong Province of China (Nos. tsqn201812003 and ts20190908), the Qilu Youth Scholar Funding of Shandong University, Project for Scientific Research Innovation Team of Young Scholar in Colleges, and Universities of Shandong Province (2019KJC028).
Author information
Authors and Affiliations
Contributions
D.S. conceived and designed the experiments; Y.-M.S. and Z.W. conducted synthesis and characterization; Y.-M.S., Z.W., and D.S. performed research and analyzed data; C.-H.T. contributed to scientific discussion; S.S. analyzed the mathematics of the nested polyhedral structure; D.S. and S.S. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Su, YM., Wang, Z., Schein, S. et al. A Keplerian Ag90 nest of Platonic and Archimedean polyhedra in different symmetry groups. Nat Commun 11, 3316 (2020). https://doi.org/10.1038/s41467-020-17198-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-17198-1
This article is cited by
-
Temperature-dependent chloride-mediated access to atom-precise silver thiolate nanoclusters
Science China Chemistry (2022)
-
A stable magnetic core–shell Cu12@Au30Pt12 molecule
Theoretical Chemistry Accounts (2022)
-
Revealing the chirality origin and homochirality crystallization of Ag14 nanocluster at the molecular level
Nature Communications (2021)
-
Interdependence between nanoclusters AuAg24 and Au2Ag41
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.