Abstract
Cell fate decisions involved in vascular and hematopoietic embryonic development are still poorly understood. An ETS transcription factor Etv2 functions as an evolutionarily conserved master regulator of vasculogenesis. Here we report a single-cell transcriptomic analysis of hematovascular development in wild-type and etv2 mutant zebrafish embryos. Distinct transcriptional signatures of different types of hematopoietic and vascular progenitors are identified using an etv2ci32Gt gene trap line, in which the Gal4 transcriptional activator is integrated into the etv2 gene locus. We observe a cell population with a skeletal muscle signature in etv2-deficient embryos. We demonstrate that multiple etv2ci32Gt; UAS:GFP cells differentiate as skeletal muscle cells instead of contributing to vasculature in etv2-deficient embryos. Wnt and FGF signaling promote the differentiation of these putative multipotent etv2 progenitor cells into skeletal muscle cells. We conclude that etv2 actively represses muscle differentiation in vascular progenitors, thus restricting these cells to a vascular endothelial fate.
Similar content being viewed by others
Introduction
During embryonic development, the lateral plate mesoderm (LPM) gives rise to multiple different cell lineages which include vascular endothelial cells, hematopoietic lineages and cardiomyocytes1,2. The specification of these lineages occurs in parallel to the specification of adjacent skeletal muscle progenitors which are thought to originate in the somites from different progenitors than the LPM lineages3. Although many factors involved in different steps of LPM lineage specification and differentiation have been identified, the entire timeline, transitional steps, and changes in the global transcriptional program that occur during hematopoietic and cardiovascular differentiation in vivo are still poorly understood.
While it is challenging to study early cardiovascular development in mammalian embryos, the zebrafish has emerged as an advantageous model system to study early cell fate decisions during embryogenesis. The signaling pathways and transcriptional programs that regulate the specification and differentiation of the LPM lineages are highly conserved between zebrafish and other vertebrates4.
We and others have previously identified an ETS transcription factor Etv2/Etsrp which is one of the earliest markers of vascular and hematopoietic progenitor cells and functions as a key regulator of vascular and hematopoietic development in multiple vertebrates including mouse and zebrafish5,6,7. In zebrafish embryos, etv2 is expressed in vascular endothelial progenitor cells, as well as early myeloid and erythroid progenitors, and its expression is downregulated after cells undergo hematopoietic and vascular differentiation5,6. In the absence of Etv2 function, vascular endothelial and myeloid progenitors fail to differentiate. While some of them undergo apoptosis, others can acquire alternative cell fates and differentiate into cardiomyocytes, demonstrating fate flexibility of early progenitors8,9,10.
The relatively recent emergence of highly efficient and high-throughput single-cell transcriptomic technologies has facilitated extensive probing of cellular diversity and complex cell differentiation pathways both in vitro and in vivo. In recent years, several studies have been performed to delineate the transcriptional diversity of vascular cell types, and to uncover lineage commitment trajectories during cardiovascular development11,12,13. However, the fate decisions of LPM-derived cells are still poorly understood.
Here, we report single-cell transcriptomic profiling of etv2-expressing cells in wild-type and etv2-deficient zebrafish embryos during early stages of vascular development. We identify the transcriptional profiles and novel transitional states of cells during different steps of vascular and hematopoietic differentiation. Our results show that in the absence of Etv2 function, vascular progenitors can acquire a skeletal muscle fate, arguing that Etv2 function is required to actively repress alternative cell fates in multipotent mesodermal progenitors. These findings will be important in understanding the ontogeny of different cardiovascular and hematopoietic lineages and will help in designing more efficient in vitro and in vivo cell differentiation strategies to generate different types of progenitors for therapeutic purposes.
Results
Single-cell RNA-seq of vascular and blood progenitors
To analyze the diversity of hematovascular progenitors, we performed single-cell RNA-seq of GFP-positive cells sorted by fluorescence-activated cell sorting (FACS) from heterozygous and homozygous etv2ci32Gt; UAS:GFP zebrafish embryos at the 20-somite stage. This reporter line, recently generated by CRISPR mediated homology-independent repair, has an insertion of the gal4 reporter within the etv2 coding sequence14. As described previously, heterozygous etv2ci32Gt embryos recapitulate the endogenous expression pattern of etv2 in vascular endothelial progenitors and differentiated vascular endothelial cells, while homozygous embryos show profound defects in vascular development due to the interruption of the etv2 coding sequence14 (Supplementary Fig. 1).
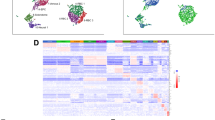
Transcriptomes of 2049 and 588 cells were obtained from heterozygous and homozygous etv2ci32Gt embryos, respectively, using the Chromium system (10× Genomics) which employs a microdroplet technology to isolate individual cells, followed by the next-generation seuencing. The relative frequency of GFP+ cells out of the total number of cells was similar in heterozygous and homozygous embryos (1.89% and 1.98%, respectively). Transcriptomes from heterozygous and homozygous embryos were pooled and clustered using Seurat15, resulting in 12 distinct cell clusters which were visualized using the t-distributed stochastic neighbor embedding (t-SNE) approach16 (Fig. 1a–d). We subsequently assigned cell identities based on marker genes which were significantly enriched in each cluster (Supplementary Table 1, Supplementary Datas 1 and 2). Two different clusters (#2 and #3) corresponded to vascular endothelial cells and were thus labeled as EC1 and EC2. The EC1 cluster showed expression of multiple known vascular endothelial markers, including cdh5, cldn5b, egfl7, sox7, ecscr and others, while the top genes expressed in EC2 cells included crip2, tpm4a, fli1a, and erg, all known to label vascular endothelial cells (Supplementary Table 1 shows the top marker genes for each cluster, while t-SNE and violin plots for selected marker genes are shown in Fig. 1c and Supplementary Figs. 2 and 3). There was a large overlap in marker expression between the EC1 and EC2 groups. It is currently unclear whether EC1 and EC2 cells represent distinct cellular identities or different stages of endothelial differentiation. Cluster #4 corresponded to the vascular endothelial progenitor group (EPC) which showed enriched expression of tal1, lmo2, etv2, tmem88a, egfl7 (Fig. 1c, d, Supplementary Figs. 2 and 4, Supplementary Table 1). Although some of these genes (tal1, lmo2, etv2) are known to label both vascular and hematopoietic progenitors5,17,18, other markers specific to this group (egfl7, sox7, fli1b) label vascular and not hematopoietic cells19,20,21, arguing that this population corresponds to vascular endothelial progenitors. Two groups of cells with a strictly hematopoietic gene signature were identified. Cluster #7 showed specific expression of cebpb, spi1b, cebpa, cxcr3.2, all known to be specifically enriched in macrophages (Fig. 1c, d, Supplementary Figs. 2 and 5, Supplementary Table 1, Supplementary Data 1). Cluster #11 showed strong expression of multiple hemoglobin genes, including hbbe3, hbbe1.3, hbae1.3, as well as klf17, blf and other genes that are specific to red blood cells (Fig. 1d, Supplementary Figs. 2 and 5, Supplementary Table 1, Supplementary Data 1). Although etv2 in zebrafish does not show significant expression in zebrafish blood cells, etv2:GFP expression has been previously observed in myeloid and erythroid cells22, likely due to the expression of etv2 in hematopoietic progenitors, which becomes downregulated as they differentiate. Cluster #10 had very few significantly enriched genes, which included a novel protein si:ch211–11n16.3, and genes tubb2b and hmg2b which are likely to be ubiquitously expressed. Apoptosis and cell cycle regulators pmaip1/noxa and ccng123,24 also showed relatively strong expression in this cell population (Fig. 1d, Supplementary Fig. 6, Supplementary Table 1, Supplementary Data 1), suggesting that this group is likely to include pro-apoptotic cells, which were observed in etv2ci32Gt embryos. Previous studies have established that the loss of etv2 function results in increased apoptosis of vascular or hematopoietic cells21,25. Cluster #12 showed strong expression of hoxa11b, hoxb7a, cdx4, apoeb, which are all expressed in the posterior mesenchyme either in zebrafish or mouse embryos26,27,28 (Fig. 1c, d, Supplementary Figs. 2 and 6, Supplementary Table 1, Supplementary Data 1). The tailbud progenitors have been shown to include multipotent cells which can give rise to endothelial, somitic and neural lineages29. Therefore this group may include early stage endothelial progenitors located in the tailbud. Indeed, many cells from the tailbud population exhibit noticeable expression of EPC markers (Fig. 1d, Supplementary Fig. 4). Another intriguing population of etv2+ cells (cluster #6) showed expression of BMP response genes id3 and id1, as well as twist1a, pitx3, and prrx1a (Fig. 1c, d, Supplementary Figs. 2 and 4, Supplementary Table 1, Supplementary Data 1). Our subsequent analysis (see below) demonstrated that all analyzed genes exhibited expression in the LPM area; therefore we annotated this cluster as LPM cells. Additional cell populations identified in our analysis included cells with neural, epidermal, cardiomyocyte, and skeletal muscle-specific gene signatures (Fig. 1c, d, Supplementary Figs. 2, 7, and 8, Supplementary Table 1, Supplementary Data 1). Observation of GFP-positive cardiomyocyte and skeletal muscle populations in both etv2ci32Gt heterozygous and homozygous embryos was somewhat unexpected. Cardiomyocytes in wild-type embryos do not express etv2, while our previous study has shown that etv2-deficient endothelial progenitors can differentiate as cardiomyocytes8. The presence of GFP-positive cardiomyocytes in etv2ci32Gt heterozygous embryos could be explained by the loss of one functional etv2 allele, which resulted in some etv2+ cells differentiating as cardiomyocytes. However, differentiation of hematovascular progenitors into skeletal muscle cells has not been previously reported.
a, b t-SNE plots and cell clustering analysis. Twelve different clusters were identified. Card cardiomyocytes, EC endothelial cells, EPC endothelial progenitor cells, Epid epidermal cells, LPM lateral plate mesoderm, Macr macrophages, Myoc myocytes, Neur neural, Apo putative pro-apoptotic cells, RBC red blood cells, Tailbud tailbud progenitors. c t-SNE plots showing selected top markers for different cell types. d Heatmap of marker gene expression in different cell populations. e The proportions of GFP+ cell types in etv2ci32Gt+/− and etv2ci32Gt−/− embryos. Note a great reduction in EC1, EC2, EPC, and macrophage populations and an increase in LPM, cardiomyocyte, myocyte, RBC, and Apo populations in etv2 mutants. ***p < 0.001, NS not significant, chi-square test. p Values: Card—5.0 × 10−26, EC1— 6.2 × 10-18, EC2—1.8 × 10−14, EPCs—2.4 × 10−11, Epid—0.66, LPM—8.4 × 10−27, Macr—2.5 × 10−16, Myoc—3.4 × 10−4, Neur—1.0, Apo—1.9 × 10−7, RBC—1.1 × 10−4, Tailbud—0.21. Totally, 2049 and 588 cells total from etv2ci32Gt+/− and etv2ci32Gt−/− embryos, respectively, were analyzed in a single scRNA-seq experiment. f Pseudotime analysis graph of cells in LPM, EPC, EC1, EC2, and myocyte populations in etv2ci32Gt homozygous and heterozygous embryos.
scRNA seq analysis of etv2 loss-of-function embryos
We then analyzed the cell clusters present in etv2ci32Gt homozygous embryos, which exhibit severe defects in vascular differentiation. Both EC1 and EC2 populations, EPCs and macrophages were nearly completely absent in etv2ci32Gt homozygous embryos (Fig. 1b, e), consistent with the known requirement of etv2 in the formation of these lineages5,6. In contrast, percentage of RBC cells was slightly but significantly increased in etv2ci32Gt homozygous embryos (Fig. 1b, e). To confirm this result, we analyzed expression of RBC-specific markers using data from the recently reported global transcriptome study of etv2ci32Gt heterozygous and homozygous embryos14. Indeed, expression of multiple RBC-specific genes, including hbaa1, vwf, hbae3 and others was significantly upregulated in etv2ci32Gt homozygous embryos (Supplementary Table 2). Furthermore, differential expression analysis of scRNA-seq data between RBC cell populations in heterozygous and homozygous embryos revealed an upregulation of several RBC-specific genes in homozygous embryos, including alas2, hbae3, hbae1.3, and hbbe1.1, while several vascular endothelial specific genes including tpm4, ecscr and an EPC specific gene lmo2 were downregulated (Supplementary Data 3). For additional confirmation, we knocked down etv2 function using previously validated MO5 in gata1:dsRed transgenic embryos which express fluorescent reporter in RBCs. Etv2 knockdown embryos were morphologically normal and showed previously reported vascular defects that were similar to the etv2 mutant phenotype. Subsequently, cells were disaggregated at 23 hpf and analyzed by FACS sorting. A statistically significant increase in the number of dsRed-positive cells was observed in etv2 knockdown embryos (Supplementary Fig. 9). This suggests that the erythropoiesis pathway is upregulated in etv2-deficient embryos, although exact mechanism requires further investigation.
The tailbud, epidermal and neural populations were not significantly affected in etv2-deficient embryos. Interestingly, the LPM, cardiomyocyte, and skeletal muscle (myocyte) populations were greatly increased in the homozygous embryos (Fig. 1b, e). While an increase in cardiomyocyte population was expected because etv2+ cells are known to differentiate as cardiomyocytes in etv2-inhibited embryos8, the presence of a myocyte population was intriguing. These results suggested that some etv2-deficient vascular progenitors may acquire skeletal muscle fate. Pseudotime analysis of EPCs, ECs, LPM cells and myocytes showed the distribution of EPCs which appear to originate from the cluster of LPM cells and progress along a differentiation trajectory towards either EC-1 or EC-2 states, while the myocyte pool branches off from the main trajectory at an earlier time point (Fig. 1f). Cells in the homozygous embryos showed an increase in the LPM and myocyte populations, and a loss of EC populations. We hypothesized that the LPM group represented EPCs at an earlier stage of differentiation, and that the myocyte cells were an alternative cell fate for etv2+ progenitors when they could not differentiate into the vascular endothelial lineage.
Identification of an endocardial subcluster
In an attempt to identify additional heterogeneity within the endothelial cells, which may be missed in the global cell clustering approach, we performed subclustering of endothelial cell populations EC1 and EC2 identified in etv2ci32Gt+/− embryos using Seurat (see Methods). The subclustering of EC1 resulted in the identification of two subpopulations of cells (Fig. 2a–c), while the subclustering of EC2 did not yield meaningful subpopulations. One of the two EC1 sub-clusters showed enriched expression of cdh6, gata5, fn1a, and drl, among the other markers (Fig. 2d, e, Supplementary Data 4). gata5 has been previously known to be enriched in endocardial cells30. In situ hybridization (ISH) analysis confirmed that expression of fn1a and drl was also enriched in the endocardial cells and largely absent from other vascular endothelial cells (Fig. 2f, g). In contrast, genes enriched in the remaining EC1 subpopulation (designated as EC1a) which included etv2 and lmo2, were largely absent from endocardial progenitors at the 20-somite stage (Fig. 2h, i). This strongly suggests that the first subcluster represents a population of endocardial progenitors which possess a unique transcriptomic signature even prior to the formation of the heart tube.
Subclustering of the EC1 population in etv2ci32Gt+/− embryos identified a novel endocardial subpopulation (while the remaining endothelial cells are designated as EC1a). a A section of a heatmap showing marker gene expression in endocardial, endothelial (EC1a and EC2), endothelial progenitor (EPC), and cardiomyocyte populations. b A portion of the global t-SNE plot showing the endocardial subpopulation. c Heatmap for subclustering of EC1 population showing genes enriched in endocardial and the remaining endothelial (EC1a) cells. d, e t-SNE and violin plots for selected endocardial genes. f–i In situ hybridization analysis at the 20-somite stage for selected endocardial-enriched genes fn1a and drl (f, g arrows point to endocardial expression) and endocardial-excluded genes etv2 and lmo2 (bilateral expression is present in cranial endothelial progenitors but is largely absent from the endocardial cluster). Flat-mounted embryos, ventral view, anterior is to the left. The number of embryos displaying the representative phenotype out of the total number of embryos obtained from two replicate experiments is shown.
Vascular progenitors convert into muscle in etv2 mutants
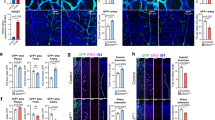
To confirm if GFP expression was in fact present in skeletal muscle cells, as suggested by scRNA-seq analysis, we analyzed etv2ci32Gt; UAS:GFP embryos by confocal microscopy. Indeed, GFP expression was observed in several myocytes in etv2ci32Gt heterozygous embryos (3 ± 2.5 myocytes per embryo, ± refers to standard deviation, n = 8 embryos), while multiple GFP-positive cells (18.5 ± 5.4 per embryo, n = 10) were present within the somitic muscle of etv2ci32Gt homozygous embryos at 25 hpf (Fig. 3a–e). These cells displayed an elongated shape, similar to other skeletal muscle cells, and were positive for expression of skeletal muscle actin actc1b:GFP (Fig. 3f–h). GFP expression in skeletal muscle overlapped with antibody staining for fast muscle but not slow muscle cells (Supplementary Fig. 10). When crossed into the previously generated etv2:mCherry reporter line31, these muscle cells were also positive for mCherry expression (Fig. 3i–k), indicating that the reporter fluorescence is unlikely to be an artifact of transgene misexpression in unrelated tissues. To confirm that etv2ci32Gt; UAS:GFP expression in the skeletal muscle is caused by the loss of etv2 function, we crossed the etv2ci32Gt line with etv2ci33 loss-of-function mutants. The etv2ci33 allele, generated using CRISPR/Cas9 mutagenesis, contains a 13 bp insertion in exon 5 of the etv2 gene (see “Methods”). Homozygous etv2ci33 mutants show vascular defects similar to the previously reported etv2y11 allele, which is likely to be null21 (Supplementary Fig. 11a–d). Double heterozygous etv2ci32Gt/ci33 embryos displayed a significant increase in GFP+ myocytes compared to etv2ci32Gt+/− embryos (Fig. 3l–n, Supplementary Fig. 11e–h). In addition, GFP expression in skeletal muscle cells was also observed in etv2y11−/− mutant embryos crossed into the previously established Tg(-2.3 etv2:GFP) reporter line but not in wild-type Tg(-2.3 etv2:GFP)14,32 embryos (Fig. 3o). Collectively, these results argue that etv2-expressing progenitors differentiate as skeletal muscle in the absence of Etv2 function.
a–d Trunk region of etv2ci32Gt; UAS:GFP embryos at 25 hpf. Maximum intensity projections of selected confocal slices are shown in (c, d). Note the absence of intersegmental vessels (ISVs) and elongated GFP-positive skeletal muscle fibers (arrows, c, d). The embryos were obtained from an incross of heterozygous etv2ci32Gt carriers; embryo numbers (lower right) correspond to the expected Mendelian ratio. e Quantification of GFP+ myocytes in the trunk region of 8 etv2ci32Gt+/− and 10 etv2ci32Gt−/−; UAS:GFP embryos at 25 hpf, analyzed in 2 replicate experiments. The bars show median values. f–h Co-expression of etv2ci32Gt+/−; UAS:NTR-mCherry and muscle-specific actc1b:GFP in mCherry-positive muscle cells (arrows, g, h). i–k The Tg(etv2:mCherry) line shows mCherry expression in skeletal muscle cells when crossed to etv2ci32Gt+/−; UAS:GFP carriers. l, m Multiple GFP+ skeletal muscle cells are apparent in the progeny of etv2ci32Gt+/−; UAS:GFP zebrafish crossed with the etv2ci33+/− line, which carries a loss-of-function mutation in etv2. Note that the expected frequency of double heterozygous embryos in (m) is 50%. n Quantification of GFP+ myocytes in the trunk region of etv2ci32Gt/ci33 and etv2ci32Gt+/−; scl MO embryos shown in (m, p) obtained in two replicate experiments. The graphs show median and SD values. o GFP+ skeletal muscle cells observed in etv2y11−/− embryos crossed into the Tg(-2.3 etv2:GFP) reporter line. p Multiple GFP + skeletal muscle cells are apparent in etv2ci32Gt+/−; UAS:GFP embryos injected with scl MO. See graph n for quantification. q–s Ectopic myocytes observed in scl MO-injected etv2ci32Gt+/−; UAS:NTR-mCherry embryos are positive for muscle-specific actc1b:GFP expression at 24 hpf. t etv2 RNA overexpression inhibits myod expression (arrow). Dorsal view, anterior is to the left. u qPCR analysis of myf5, myod, and myog expression in etv2 RNA-injected and uninjected control embryos at the 10-somite stage. Mean values ± SEM is shown. RNA was purified from groups of ten embryos analyzed in two replicate experiments. In all graphs, two-tailed Student’s t test was used. The number of embryos displaying the representative phenotype out of the total number of embryos obtained from two replicate experiments is shown.
To investigate the mechanism of the conversion of these etv2-deficient progenitors into skeletal muscle in greater detail, we analyzed the functional role of bHLH transcription factor scl/tal1 which functions downstream of etv2 during vascular development and myelopoiesis6. Previous research has demonstrated that the combined knockdown of scl and etv2 results in ectopic myocardial differentiation of etv2+ progenitors in zebrafish10, and that scl represses myocardial differentiation in mouse embryos33. However, its role in repressing skeletal muscle differentiation has not been investigated. The number of myocytes, positive for etv2ci32Gt+/−; UAS:GFP expression, was significantly increased in the etv2ci32Gt+/− embryos injected with the previously validated scl morpholino (MO)33, compared to the control etv2ci32Gt+/− embryos (Fig. 3n, p). Furthermore, these ectopic myocytes were positive for expression of the skeletal muscle actin actc1b (Fig. 3q-s), arguing that scl knockdown results in increased skeletal muscle differentiation of etv2ci32Gt+/−; UAS:GFP cells. However, injection of scl mRNA into etv2ci32Gt; UAS:GFP embryos did not have a significant effect on the number of ectopic GFP-positive muscle cells (Supplementary Fig. 12), suggesting that etv2 and scl genes may not function in a simple linear pathway to repress muscle differentiation in etv2+ progenitor cells.
Heat-shock inducible etv2 expression has been demonstrated to convert differentiated skeletal muscle into vascular endothelial cells34. To test if etv2 is sufficient to inhibit early specification or differentiation of myocytes, we overexpressed etv2 mRNA in zebrafish embryos. We have previously shown that such overexpression induces strong ectopic expression of vascular endothelial markers5. Conversely, expression of the early regulators of muscle differentiation myod, myf5, and myog was greatly inhibited upon etv2 overexpression (Fig. 3t, u).
Vascular endothelial progenitors are known to originate in the LPM region. However, the somitic origin of vascular progenitors has also been suggested35. In order to analyze the origin of GFP+ myocytes, we performed time-lapse imaging of etv2ci32Gt; UAS:GFP embryos. The majority of EPCs in heterozygous embryos originated in the LPM region and subsequently migrated to the midline where they coalesced into the DA and the PCV (Fig. 4a–f, Supplementary Fig. 13a–f, Supplementary Movies 1 and 2). Occasional cells failed to migrate to the midline and elongated to form somitic muscle. In contrast, GFP+ cells in homozygous embryos failed to migrate to the midline. Some of these cells underwent apoptosis while others elongated and incorporated into the somitic muscle at approximately 14–18-somite stages (Fig. 4g–m, Supplementary Fig. 13g–l, Supplementary Movies 3–5). Thus, etv2ci32Gt; UAS:GFP cells that give rise to the skeletal muscle cells in the homozygous embryos appear to be derived from the initial pool of etv2+ cells in the LPM region. To analyze when GFP+ positive cells initiate muscle marker expression, we performed fluorescent ISH for myod expression combined with GFP fluorescence analysis at the 8–10-somite stages in etv2ci32Gt+/−; UAS:GFP embryos. GFP and myod co-expressing cells were positioned at the posterior edge of the somites next to GFP+ cells in the LPM (Fig. 5). In wild-type embryos, etv2-expressing vascular progenitors are known to migrate between the somites and coalesce into axial vasculature starting at the 8–10-somite stage36. In contrast, some GFP+ cells migrate into the somites instead, and initiate myod expression in etv2ci32Gt embryos.
Lateral view is shown, anterior is to the left. a–f In etv2ci32Gt+/−; UAS:GFP embryos, bilaterally located vascular and hematopoietic progenitors within the lateral plate mesoderm (LPM, arrowheads) migrate toward the midline and coalesce into the axial vasculature (arrows). Note that some cells remain in the lateral position and elongate into muscle cells (myoc). DA progenitors of the dorsal aorta, ISV intersegmental vessels. Time frames are selected from the Supplementary Movie 1. g–l In etv2ci32Gt−/−; UAS:GFP embryos, cells initiate migration (blue arrows) but fail to coalesce into the axial vasculature. Instead, many cells either undergo apoptosis (apo, red arrowheads point to round apoptotic cells) or differentiate into myocytes (white arrows, myoc). Time frames are selected from the Supplementary Movie 3. m Higher magnification view showing differentiation of a GFP+ progenitor cell initially positioned in the LPM into a myocyte (arrowhead points to the same cell). Note that the cell migrates dorsally from the LPM into the somite and then elongates as it undergoes differentiate into the muscle. Time frames are selected from the Supplementary Movie 4. Representative embryos are shown out of the total of seven heterozygous and four homozygous etv2ci32Gt; UAS:GFP embryos that were imaged in two replicate experiments.
In situ hybridization was performed using hybridization chain reacion (HCR) at the 8–10-somite stages. a–c Maximum intensity projection is shown; d–f An area boxed in a was imaged at higher magnification; maximum intensity projection of three confocal slices is shown. Note that most GFP-expressing cells are positioned in the lateral plate mesoderm, while some cells are starting to migrate toward the midline (arrow, d, e, points to a migrating angioblast which does not have myod expression). GFP and myod co-expressing cells (arrowheads, d, e) are apparent at the posterior edge of the somite. The punctate myod expression pattern is due to the nature of the HCR probe. Dorsolateral view, anterior is to the left. The numbers in the lower left corner display the number of embryos showing the phenotype out of the total number of embryos analyzed in two replicate experiments.
Wnt and FGF pathways are required for ectopic muscle
Wnt and FGF signaling pathways have been previously implicated in somitic differentiation and myogenesis37. We tested if these pathways were involved in the differentiation of etv2ci32Gt; UAS:GFP+ progenitors into ectopic muscle cells. To inhibit Wnt signaling we induced expression of Wnt inhibitor dkk1 at the 8-somite stage. As it has been previously demonstrated29, Wnt inhibition at this stage resulted in reduced myod expression in the tailbud region, while myod expression in differentiated somitic cells was largely unaffected (Supplementary Fig. 14a, b). Heat-shock inducible expression of Wnt inhibitor dkk1 greatly reduced the number of etv2+ myocytes observed in heterozygous etv2ci32Gt; UAS:GFP embryos (Fig. 6a–c). Dkk1 overexpression did not affect the total number of cells positive for the UAS reporter expression in etv2ci32Gt embryos (Supplementary Fig. 14c). This suggests that Wnt signaling promotes muscle differentiation in multipotent etv2+ progenitors.
a–c Heat-shock inducible expression of Wnt inhibitor Dkk1 greatly reduces GFP-positive muscle cells in hsp70:dkk1; etv2ci32Gt+/−; UAS:GFP embryos. Heat-shock (HS) was performed at the 8-somite stage, and GFP fluorescence imaged at 24–26 hpf. Sixteen embryos were analyzed in each group in two replicate experiments. d–f etv2ci32Gt−/−; UAS:GFP embryos treated with FGF inhibitor SU5402 starting at the 50% epiboly stage show a greatly reduced number of GFP-positive cells at the 22-somite stage. Nine control DMSO-treated and 15 SU5402-treated embryos were analyzed in two replicate experiments. g–i Heat-shock inducible expression of dnFGFR1 results in a significant reduction of mCherry-positive muscle cells in etv2ci32Gt+/−; UAS:NTR-mCherry; hsp70:dnFGFR1 embryos. Twenty each control and HS hsp70:dnFGFR1 embryos were analyzed in two replicate experiments. Median ± SD values are shown in all graphs, two-tailed Student’s t test was used for statistical analysis.
To investigate the role of FGF signaling, we treated etv2ci32Gt−/− embryos with SU5402, a chemical inhibitor of FGF signaling38. SU5402-treated embryos showed a significant decrease in the number of ectopic GFP+ muscle cells (Fig. 6d–f). To confirm the requirement of FGF signaling in the ectopic muscle differentiation, etv2ci32Gt+/−; UAS:NTR-mCherry carriers were crossed with a heat-shock inducible hsp70:dnFGFR1 line39, and the expression of dnFGFR1 was induced at the 8-somite stage. A significant reduction in the number of ectopic muscle cells was observed in FGF-inhibited embryos while no significant defects in vascular development were apparent (Fig. 6g–i). The overall number of UAS reporter-positive cells was not affected in FGF-inhibited etv2ci32Gt+/− embryos (Supplementary Fig. 14c). Together, these results suggest that FGF signaling is required for muscle-specific differentiation of multipotent etv2+ progenitors.
Identification of putative multipotent LPM progenitors
Based on scRNA-seq analysis, cell cluster #6 was significantly increased in etv2ci32Gt−/−; UAS:GFP embryos. Marker genes for this population included BMP response genes id3 and id1 (Supplementary Table 1, Supplementary Data 1), which have been recently shown to function upstream of etv2 during vascular differentiation of tailbud-derived neuromesodermal progenitors40. Another marker gene for this population, twist1a, has been implicated in developmental angiogenesis41, although its mechanistic role in this process has not been investigated. Other genes enriched in this cluster included transcription factors prrx1a, foxd1, and foxd2, which have been implicated in diverse processes, including epithelial–mesenchymal transition and craniofacial development42,43. However, their function or relevance to vascular development has yet to be elucidated. We decided to investigate the identity of the cells from cluster #6 in greater detail. Expression of transcription factors twist1a, prrx1a, and foxd2 was present in the LPM region at the 18-somite stage (Fig. 7a–n). Based on ISH analysis of etv2ci32Gt+/−; UAS:GFP embryos, prrx1a and twist1a expression was absent from the midline GFP+ cell population which correspond to vascular endothelial cells that are coalescing into axial vessels, while it partially overlapped with GFP expression in the most lateral vascular progenitors. The expression of these genes was not significantly affected in etv2ci32Gt−/−; UAS:GFP embryos (Fig. 7d–f, j–n). Similar overlap of prrx1a or twist1a expression with GFP fluorescence in the most lateral vascular progenitors was also observed in wild-type TgBAC(etv2:GFP) embryos (Supplementary Fig. 15). Because vascular progenitors originate within the LPM prior to their migration to the midline, such gene expression pattern suggests that this cell cluster corresponds to the LPM cells which may include the earliest vascular progenitors (Fig. 7o, p).
a–l Fluorescent in situ hybridization using hybridization chain reaction for prrx1a (a–f) and twist1a (g–l) expression combined with GFP fluorescence in etv2ci32Gt; UAS:GFP heterozygous or homozygous embryos at the 18-somite stage. Maximum intensity projections of selected confocal z-stacks are shown. Note that both prrx1a and twist1a are expressed bilaterally, and their expression partially overlaps with GFP in the most lateral cells (arrows). GFP+ cells fail to coalesce into axial vasculature in etv2ci32Gt−/− embryos. DA precursor vessel for the dorsal aorta, LDA lateral dorsal aortae. m, n Expression of foxd2 in the trunk region in etv2ci32Gt heterozygous and homozygous embryos at the 18-somite stage. Note expression in the LPM region (arrows). Expression in the somites is also apparent. o, p In situ hybridization for GFP expression in etv2ci32Gt+/− and etv2ci32Gt−/−; UAS:GFP embryos at the 17–18-somite stage. GFP expression is observed in endothelial cell (EC, combined EC1 and EC2), endocardial (Endoc), endothelial progenitor cell (EPC), lateral plate mesoderm (LPM), tailbud and red blood cell (RBC) populations. Note the absence of EC and endocardial expression in (p). In all panels, the numbers display the number of embryos showing the expression pattern out of the total number of embryos analyzed in two replicate experiments.
Transcriptional profiling of etv2:GFP cells using Fluidigm
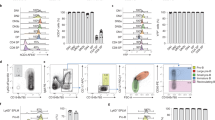
Although the Chromium-based scRNA-seq analysis identified novel cell populations, we were not able to distinguish endothelial subpopulations such as arterial and venous progenitors which are known to separate early during vasculogenesis. We decided to supplement our analysis by sequencing fewer cells at a higher coverage. For this approach, we used a previously established Tg(-2.3 etv2:GFP) line31 which shows specific GFP expression in vascular endothelial cells (Fig. 8a–c). Cells from embryos at 16–20-somite stages were dissociated and sorted using the Fluidigm C1 system. Totally, 96-cells were subjected to scRNA-seq analysis. After cell clustering using AltAnalyze software44, 8 distinct cell populations were identified. They included macrophage, erythroid, tailbud progenitor, and nonspecific epidermal cell populations (Fig. 8d–g). Four different vascular endothelial cell populations were identified. The vascular endothelial progenitor population displayed high expression of etv2, lmo2 and tal1, as well as multiple genes that have not been previously associated with EPCs (Fig. 8d–g, Supplementary Datas 5 and 6). Many genes overlapped between EPC populations identified in the Chromium and Fluidigm analyses, suggesting that the same populations of cells were identified using both approaches. Cluster 5 was enriched in venous-specific vascular endothelial genes such as flt4 and stab1l, while cluster 6 showed high expression of known arterial-specific markers, including dll4, efnb2a, cldn5b (Fig. 8g, Supplementary Data 5). Cluster 7 included many genes with known expression in vascular endothelium such as col4a1, ldb2a, or hapln3. Validation by ISH analysis confirmed expected expression patterns of EPC, arterial and venous markers and showed that expression of the cluster 7 marker ldb2a was also primarily enriched in the arterial vasculature (Fig. 8i–l). The biological difference between the clusters 6 and 7 is unclear, and it will require further investigation. Notably, marker genes for cluster 7 (also labeled as EC2) are largely different from the EC2 cluster identified using the Chromium platform.
a–c GFP expression in live embryos, maximum intensity projection is shown. Arrows label vascular endothelial cells and their progenitors. Ten embryos were imaged in two independent experiments and a representative embryo is shown. a lateral view; b anterior view, c dorsal view. A anterior, P posterior. d Heatmap view of marker gene expression in different cell clusters. A complete list of differentially expressed genes is presented in Supplementary Data 5. e, f 2-D and 3-D principal component analysis plots of different cell clusters. Cluster names are the same as in (d). g Relative marker gene expression in different cell clusters. Vertical bars depict log-normalized gene expression. h An Arteriovenous (A-V) index of different endothelial cells. Note that many cells are positive for both arterial and venous marker expression. i–l ISH expression analysis of key marker genes for EPC (etv2), venous (flt4), arterial (cldn5b), and EC-2 (ldb2a) populations in the trunk region at the 20-somite stage. Black arrows label the DA and white arrowheads label venous progenitors which are starting to coalesce into the PCV. Note that flt4 is enriched in the PCV while cldn5b and ldb2a label the DA. m–r Two color ISH analysis for the expression of venous dab2 and arterial cldn5b at the 20-somite and 24 hpf stages. Arrows label the DA while arrowheads mark the PCV or its progenitors. Note that dab2 and cldn5b are co-expressed in the DA progenitors at the 20-somite stage but not at 24 hpf. In all panels, the numbers in the lower right corner display the number of embryos showing the expression pattern out of the total number of embryos analyzed in two replicate experiments.
Expression of venous markers in arterial progenitors
Previous studies have suggested that arterial and venous progenitors originate at distinct spatial regions and at different times36. However, it has been controversial as to when cells acquire distinct arterial and venous identities45,46,47. We used the expression of well-established arterial and venous markers to calculate an arteriovenous index for each vascular endothelial cell (see “Methods”). While some cells showed a very clear arterial or venous signature, many cells co-expressed both arterial and venous markers (Fig. 8h). Fluorescent ISH analysis for venous dab2 and arterial marker cldn5b expression confirmed that many arterial progenitors co-express both markers at the 20-somite stage (Fig. 8m–o). In contrast, arterial dab2 expression becomes downregulated by 24 hpf and is restricted to the PCV (Fig. 8p–r). Intriguingly, ventrally located progenitors of the PCV mostly displayed venous marker expression and showed very little, if any, arterial cldn5b expression (Fig. 8m–o). Thus, arterial progenitors co-express both arterial and venous markers at early stages of vascular development while venous progenitors show largely venous-specific marker expression.
Discussion
In this study we used scRNA-seq analysis to identify transcriptional signatures and investigate transitional states of cell populations during the differentiation of vascular endothelial and hematopoietic lineages. Our results suggest that cells in the LPM are multipotent and that the same progenitors can give rise to hematopoietic, vascular, myocardial, and even skeletal muscle lineages. Wnt and FGF signaling pathways promote skeletal muscle development in etv2-negative multipotent progenitors (Fig. 9). Etv2 overexpression was sufficient to inhibit muscle differentiation and instead promoted hematovascular fates. Our results argue that in addition to activating vasculogenic and hematopoietic programs, etv2 also actively represses myogenesis, possibly through the transcription factor scl/tal1. Previous work has demonstrated that etv2 can similarly repress myocardial differentiation in both zebrafish and murine embryos8,9. Scl, similar to Etv2, also represses myocardial fates by occupying primed enhancers of myocardial specific genes and preventing their activation by cardiac factors48. It is tempting to speculate that Etv2 also inhibits muscle specification by a similar mechanism, where Scl functions downstream of Etv2 and binds to muscle-specific promoters, thus preventing their activation by Myod. Alternatively, Scl and Etv2 may form a protein complex and/or function in parallel pathways to repress myocardial differentiation. Because scl overexpression failed to repress the formation of ectopic myocytes observed in etv2-deficient embryos, we speculate that scl may not function in a simple linear pathway downstream of etv2 to inhibit myogenesis. While the precise molecular pathways remain to be established, they will likely involve epigenetic mechanisms to repress the transcriptional activation of the myogenic program when the hematovascular program is activated.
Wnt and FGF signaling promotes myocyte differentiation of multipotent progenitors in the lateral plate mesoderm (LPM). BMP signaling through its downstream effectors id1 and id3 promotes vascular endothelial differentiation. Additional LPM markers that include prrx1a, foxd1, foxd2 and others may be involved in this process, although their role is purely speculative at this point. Etv2 promotes vascular endothelial differentiation while directly or indirectly repressing myogenesis.
A recent study has demonstrated two opposing activities of FGF and BMP signaling in specifying somitic progenitors and the LPM40. FGF signaling through bHLH factors myf5, myod and msgn1 induced medial fate such as skeletal muscle in tailbud-derived neuromesodermal progenitors while BMP signaling induced blood and endothelium marker expression, and etv2 expression in particular, through transcriptional activation of id genes, including id1 and id3. Loss of msgn1/myod/myf5 function or activation of hs:id3 expression resulted in a dramatic expansion of etv2 expression into the paraxial mesoderm40. Our results show that a loss of etv2 expression results in the opposite phenotype, and endothelial progenitors differentiate as skeletal muscle cells. FGF signaling was required for this differentiation, consistent with its role in promoting muscle fate. Similar loss of endothelial cells and expansion of skeletal muscle was observed upon inhibition of BMP signaling using the HS:dnBMPR line40, suggesting that BMP inhibits paraxial mesoderm development through etv2 function.
Intriguingly, based on our scRNA-seq analysis, id1 and id3 genes were enriched not in the population of vascular progenitors, but in the LPM cells, together with twist1a, prrx1a, and foxd1/d2 homologs. This suggests that BMP signaling is active in the multipotent progenitors in the LPM and activates etv2 expression through id1 and id3, which are then downregulated as cells differentiate into endothelial or hematopoietic lineages (Fig. 9). Based on our results, etv2 mutant embryos showed a nearly complete absence of vascular endothelial and progenitor cells while the LPM population was increased, suggesting that many LPM cells are arrested in the progenitor stage unable to differentiate further, while others undergo differentiation into skeletal muscle.
A previous study has suggested that id1 and id3 genes function upstream of etv2 during embryogenesis40. Here, we provide evidence that prrx1a and foxd1/d2 are co-expressed with etv2 in the LPM, and are involved in regulating its expression during vasculogenesis. This further supports the model that the LPM population includes early progenitors that can give rise to the vascular endothelial lineage and suggests that additional factors which function upstream of etv2 may be present within this cell population.
While etv2ci32Gt−/− mutant embryos displayed UAS:GFP expression in multiple skeletal muscle cells, some muscle cells were also present in the heterozygous etv2ci32Gt+/− embryos. Because no GFP expression in skeletal muscle cells was observed in wild-type Tg(-2.3etv2:GFP) embryos, this phenotype is likely caused by the loss of a single etv2 allele in heterozygous etv2ci32Gt+/− embryos. Intriguingly, vascular defects observed in etv2ci32Gt−/− mutant embryos were more severe compared to etv2y11 and etv2ci33 mutants, both of which are expected to be null alleles. A possible explanation is that the insertion of a rather large Gal4-containing construct into the etv2 locus may have interrupted the expression of a related ETS transcription factor fli1b, which is positioned adjacent to etv2 on the same chromosome and may share common regulatory elements with etv2. The combined loss of Etv2 and Fli1b function results in a similar (albeit more severe) loss of vascular endothelial differentiation as seen in homozygous etv2ci32Gt−/− embryos49.
Overall, scRNA-seq of etv2ci32Gt and Tg(-2.3 etv2:GFP) embryos using the Chromium and Fluidigm platforms, respectively, identified some of the same cell populations, including endothelial progenitor cells, vascular endothelial cells, red blood cells, macrophages and presumptive tailbud progenitors. The cell populations which were observed in etv2ci32Gt heterozygous embryos but not in Tg(-2.3 etv2:GFP) embryos, include cardiomyocytes, skeletal muscle cells, apoptotic cells, and LPM progenitors. We have not observed any GFP expression in cardiomyocytes, skeletal muscle or apoptotic cells in Tg(-2.3 etv2:GFP) or TgBAC(etv2:GFP) embryos, therefore the presence of these populations is likely caused by a partial loss of etv2 function in etv2ci32Gt+/− embryos. Indeed, loss of Etv2 function is known to result in EC apoptosis21,49, and we have previously demonstrated that etv2-positive cells can differentiate into cardiomyocytes in etv2-inhibited embryos8. There are a couple of reasons as to why LPM progenitors were not identified by scRNA-seq of Tg(-2.3 etv2:GFP) embryos. It is possible that a low number of LPM cells could not be separated into a distinct cluster because only 96 cells were sequenced using the Fluidigm C1 instrument. Also, this population may have low-GFP expression as these cells may have just initiated etv2 expression, making them particularly difficult to detect by FACS. The etv2ci32Gt; UAS:GFP line is significantly brighter than the Tg(-2.3 etv2:GFP) line, likely due to the amplification of GFP expression by the Gal4:UAS system14. Therefore low GFP cells may have been missed when sorting cells from the Tg(-2.3 etv2:GFP) line.
The precise developmental timepoint at which vascular progenitors acquire either arterial or venous identities has been the subject of debate. Some experiments have suggested that arterial and venous identities are prespecified at very early stages during gastrulation or early somitogenesis45,46. In contrast, our results show that many arterial and venous genes are co-expressed in the same cells as late as the 20-somite stage, when arterial progenitors have begun assembling into vascular cords. These results support the model where vascular progenitors are initially bipotent and can differentiate as either arterial or venous cells. Intriguingly, venous progenitors which are known to originate more laterally and later in development than arterial progenitors36, do not show significant expression of arterial markers. This suggests differences in the mechanisms involved in the early specification of arterial and venous progenitors.
In summary, our work suggests that cells in the vascular endothelial lineage arise from multipotent progenitors in the LPM which can differentiate into skeletal muscle in the absence of Etv2 function. The role of the key transcription factor Etv2 and the molecular mechanisms governing vascular development are highly conserved between many vertebrates, and therefore it is highly likely that our findings will also be relevant for mammalian embryos. Gaining insight into the fundamental mechanisms of cell fate commitment will not only provide us with a deeper understanding of complex developmental processes but will also be important for stem cell differentiation into highly diverse and specific lineages for regenerative therapies.
Methods
Zebrafish lines
The following zebrafish lines were used in the study: Tg(5xUAS:EGFP)50, Tg(UAS-E1B:NTR-mCherry)51, Tg(etv2:mCherry)zf37331, Tg(-2.3 etv2:GFP)zf37231, TgBAC(etv2:GFP)ci122, etv2y1121, Tg(fli1a:GFP)y152, Tg(gata1a:dsRed)53, Tg(actc1b:GFP)32, Tg(hsp70l:dkk1b-GFP)54, abbreviated further as hsp70:dkk1, Tg(hsp70l:dnfgfr1a-EGFP)39, abbreviated as hsp70:dnfgfr1a. The etv2Gt(2A-Gal4)ci32 gene trap line (further abbreviated as etv2ci32Gt) was generated by performing a knock-in of a construct with gal4-pA sequence into exon 5 of the etv2 gene by CRISPR/Cas9 mediated homology-independent repair mechanism14. The etv2ci33 mutant line was generated by injecting Cas9 mRNA and etv2 gRNA mixture (gRNA targets the following sequence within the exon 5 of etv2: GGGAAAGGCCCAAGTCACAGAGG, PAM sequence is underlined) into embryos from the kdrl:mCherryci5 line22. The line contains a 13 bp insertion in the region targeted by the gRNA, designated in capital letters:…ggcccaagtcaGCTCTGCTGCCTGcagagg.
Tg (5xUAS:loxP-mCerulean-pA-loxP-NTR-2A-YFP-pA)ci46 line (abbreviated as UAS:mCer) was made using Tol2-mediated transgenesis approach. The line showed mCerulean expression in all vascular endothelial cells in non-mosaic fashion when mated to etv2ci32Gt line.
Throughout the study, embryos were typically incubated at 28.5 °C, except for the embryonic stages prior to 24 hpf, where embryos were incubated for part of the time at 23.5–24 °C to slow down their development. Embryos were staged using previously established criteria55.
MO injections
Totally, 10 ng (per embryo) of previously validated scl splice-blocking MO was used for injections: AATGCTCTTACCATCGTTGATTTCA56. To knockdown etv2, 10 ng mixture (5 ng each MO) of two previously described etv2 (etsrp) MOs was used: MO1, TTGGTACATTTCCATATCTTAAAGT and MO2, CACTGAGTCCTTATTTCACTATATC (Gene Tools, Inc.)5.
Skeletal muscle cell counts
To count GFP- or mCherry-positive cells within the skeletal muscle, embryos were mounted in 0.6% low-melting-point agarose and the trunk region was imaged using a Nikon A1 confocal microscope at the CCHMC Confocal Imaging Core. Fluorescent cells within the skeletal muscle were counted manually within the entire trunk region by analyzing confocal z-stacks.
In situ hybridization
To perform ISH, embryos were fixed overnight in BT-Fix (4% paraformaldehyde in 1× phosphate-buffered saline (PBS)), dehydrated in sequential ethanol series and stored at −20 °C. Embryos were rehydrated and washed 3× in PBT (1× PBS, 0.2% bovine serum albumin, 0.2% Tween 20). Embryos were incubated in the prehybridization buffer (50% formamide, 5× SSC, saline sodium citrate, 50 µg/ml heparin, 5 mM EDTA, 0.5 mg/ml rRNA, 0.1% Tween 20, citric acid added to pH 6.0) for 2 h at 65 °C. DIG-labeled RNA probe solution was added at approximately 1 µg/ml concentration in prehybridization buffer (prehyb) and incubated overnight at 65 °C with shaking. Embryos were washed at 65 °C with subsequent solutions: 75% prehyb/25% 2× SSC; 50% prehyb, 50% 2× SSC; 25% prehyb, 75% 2× SSC; 2× SSC; twice with 0.2× SSC. Subsequently, embryos were washed at room temperature in the following solutions: 75% 0.2× SSC, 25% PBT; 50% 0.2× SSC, 50% PBT, 25% 0.2× SSC, 75% PBT, and 100% PBT. Afterwards embryos were incubated overnight in 1× PBT with 2% lamb serum and 1:4000 dilution of anti-digoxigenin-AP antibody (Sigma-Aldrich, cat No. 11093274910). Embryos were washed 6× in PBT solution and incubated in AP buffer (100 mM NaCl, 50 mM MgCl2, 100 mM Tris-Cl, pH 9.5, 0.1% Tween 20) in the presence of 4-nitrotetrazolium blue chloride (0.225 mg/ml) and 5-Bromo-4-chloro-3-indolyl phosphate disodium salt (0.175 mg/ml). Staining was stopped with PBT washes.
The following probes were used: etv257, cldn5b58, flt418, fn159, drl60. To synthesize a gfp probe, a gfp fragment was amplified by PCR from the myl7-hand2-IRES-GFP vector61 using the following primers: ATGGTGAGCAAGGGCGAGGAG and ATTATGCTGAGTGATATCCCTTACTTGTACAGCTCGTCC followed by RNA synthesis using T7 RNA polymerase (Promega) and DIG-labeling mix (Sigma-Aldrich). To synthesize a ldb2a probe, a cDNA clone from GE Dharmacon, cat. No. MDR1734-202728743, which contains ldb2a cDNA in pME18S-FL3 vector, was used as a template for PCR amplification with primers 18S-1 (CTTCTGCTCTAAAAGCTGCG) and 18S-T7 (CCTTTAATACGACTCACTATAGGGCCGCGACCTGCAGCTCG). DIG-labeled RNA was transcribed from the PCR product using T7 RNA polymerase (Promega). A foxd2 cDNA fragment was amplified by PCR from zebrafish embryonic cDNA using primers TCGGACAGTTCTGCTCTGTC (Forward) and TAATACGACTCACTATAGGGCTTGCTTCGGCCACGAACCA (Reverse with T7 sequence), followed by RNA synthesis using T7 RNA polymerase. T3 and T7 primers were used for PCR amplification of lmo2 cDNA in pBK-CMV vector18, followed by in vitro probe synthesis using T7 RNA polymerase.
Fluorescent ISH for prrx1a, twist1a, dab2, cldn5b, and myod expression was performed using hybridization chain reaction (HCR, version 3)62. HCR probes and fluorescent hairpins were synthesized by Molecular Technologies at Beckman Institute, California Institute of Technology, Pasadena, CA. Embryos were fixed, dehydrated and rehydrated using standard ISH protocol as described above. Embryos were prehybridized in hybridization buffer (30% formamide, 5× SSC, 9 mM citric acid (pH 6.0), 0.1% Tween 20, 50 µg/ml heparin, 1× Denhardt’s solution, 10% dextran sulfate) for 30 min at 37 °C. Two picomole of each HCR probe was added to 50 µl of hybridization buffer and incubated overnight at 37 °C. Embryos were washed 4 × 15 min in 30% formamide, 5× SSC, 9 mM citric acid (pH 6.0), 0.1% Tween 20, 50 µg/ml heparin solution at 37 °C. Samples were then washed 3 × 5 min with 5× SSCT (5× SSC and 0.1% Tween 20) at room temperature, followed by 30 min incubation in amplification buffer (5× SSC, 0.1% Tween 20, 10% dextran sulfate) at room temperature. Thirty picomole of each fluorescently labeled hairpin by snap cooling 10 µl of 3 µM stock solution. Hairpin solutions were added to embryo samples in amplification buffer and incubated overnight in the dark at room temperature. Samples were washed 5× with 5× SSCT solution, and stored at 4 °C until imaging. GFP fluorescence was still apparent after HCR. Embryos were mounted in 0.6% low-melting point agarose and imaged using a Nikon A1 confocal microscope at the CCHMC Confocal Imaging Core.
Immunostaining
F310 (1:50 dilution) and S58 (1:10) primary antibodies (Developmental Studies Hybridoma Bank, Iowa City, IA) were used for whole-mount immunostaining of etv2ci32Gt; UAS:GFP embryos at 24 hpf. No immunostaining was performed for GFP which was visible in fixed embryos. Anti-mouse IgG, CF594 antibody (Sigma-Aldrich, SAB4600098) was used as a secondary antibody.
Microscopy imaging and image processing
Embryos were whole mounted in slide chambers with 0.6% low-melting poing agarose. To image stained embryos after ISH, images were captured using a 10× objective on an AxioImager Z1 (Zeiss) compound microscope with an Axiocam ICC3 color camera (Zeiss). Images in multiple focal plans were captured individually and combined using the Extended Focus module within Axiovision software (Zeiss). For confocal imaging, embryos were mounted in 0.6% low-melting point agarose and imaged using a 10×, 20×, or 40× objective on a Nikon A1R confocal microscope. Denoising (Nikon Elements software) was performed for some of the images with weak signal to reduce noise. Images were assembled in Adobe Photoshop CS6 software package. Non-linear level adjustment was used to increase contrast and reduce background. In all cases, images of control and experimental embryos were adjusted similarly.
etv2 overexpression and quantitative RT-PCR
etv2 mRNA was synthesized as previously described5. To analyze expression of muscle markers, approximately 75 pg of zebrafish etv2 mRNA was injected into fli1a:GFP embryos at the 1-cell stage. Embryos were screened for ectopic GFP expression at the 10-somite stage and then frozen for qPCR analysis. Groups of 10 embryos were analyzed in two independent experiments. An RNAqueous 4-PCR kit (ThermoFisher) was used to extract RNA. cDNA synthesis was performed using Superscript IV cDNA synthesis kit (ThermoFisher). Quantitative real-time PCR was performed using SYBR Green Master Mix (ThermoFisher) and StepOne Software v2.3 (Applied Biosystems). The following primers were used:
ef1α (TCACCCTGGGAGTGAAACAGC) and (ACTTGCAGGCGATGTGAGCAG),
myf5 (GGTTGACTGCAACAGTCCTG) and (GCGTTGGCCTGAGGCATCTT),
myog (GCATAACGGGAACAGAGGCA) and (CAGCCTTCCTGACTGCCTTA),
myod (CCAGCATCGTGGTGGAGCGAATT) and (GGTCGGATTCGCCTTTTTCT).
The results were analyzed using StepOne Software v2.3 (Applied Biosystems). Two biological and two technical replicates were obtained for each sample (four biological replicates were available for control uninjected embryos). Relative expression values were normalized for ef1α expression. Statistical analysis was performed using Prism 8 software (GraphPad Software).
Single-cell RNA sequencing using the Chromium platform
Approximately, 100–150 of etv2ci32Gt+/−; UAS:GFP and 75–100 etv2ci32Gt−/−; UAS:GFP embryos were collected at the 20-somite stage and dissociated into a single-cell suspension using the previously reported protocol63. Briefly, the embryos were manually dechorionated, and transferred into the deyolking buffer (55 mM NaCl, 1.8 mM KCl, and 1.25 mM NaHCO3). Embryos were pipetted up and down on ice using p1000 pipettor until yolk was dissolved. Embryos were centrifugated at 300 G for 1 min, and supernatant was removed, while the pellet was resuspended in 0.5× Danieau solution (1× Danieau: 58 mM NaCl, 0.7 mM KCl, 0.4 mM MgSO4, 0.6 mM Ca(NO3)2, 5 mM HEPES, pH 7.6). Centrifugation and removal of supernatant was repeated again, followed by another centifugation at 300 G. The pellet was resuspended in FACSmax solution cell dissociation solution, which was then passed through a cell strainer. Cells were centrifugated at 300 G and suspended in a buffer of 1× PBS containing 1 mM EDTA and 2% fetal bovine serum (FBS). Fluorescence-activated cell sorting was then used to collect GFP+ cells in a solution of 50% FBS in 1× PBS (see Supplementary Fig. 16a for FACS gating strategy). Single cells were captured and processed for RNA-seq using the Chromium instrument (10× Genomics) at the CCHMC Gene Expression Core facility. RNA-seq was performed at the CCHMC DNA Sequencing core on Illumina HiSeq2500 sequencer using one flow cell of paired-end 75 bp reads, generating 240–300 million total reads.
Cell Ranger version 2.0.0 was utilized for processing and de-multiplexing raw sequencing data. Raw basecall files were first converted to the fastq format, and subsequently the sequences were mapped to the Danio rerio genome (version zv10) to generate single-cell feature counts (using STAR alignment). Downstream analysis of the gene count matrix generated by CellRanger was performed in R version 3.5.2 using Seurat version 2.3.464,65 and Tidyverse packages. The gene counts matrix was loaded into Seurat and created by filtering cells which only expressed more than 200 genes and genes that were expressed in at least 3 cells. In addition, as an extra quality-control step, cells were filtered out (excluded) based on the following criteria: <200 or >3500 unique genes expressed, or >5% of counts mapping to the mitochondrial genome. This resulted in 2049 cells in the etv2ci32Gt heterozygous sample and 588 cells in the etv2ci32Gt homozygous sample. For further downstream analysis, data from the heterozygous and homozygous samples were merged, resulting in a combined dataset of 2637 cells. Raw read counts were normalized by the “LogNormalize” function that normalizes gene expression levels for each cell by the total expression, multiplies the value by a scale factor of 104 and then log-transforms the result. Highly variable genes were determined by calculating the average expression and dispersion for each gene, placing the genes into bins and then calculating a z-score for dispersion within each bin. Based on the parameters selected to remove outliers, 3707 genes were calculated as being highly variable in expression and these genes were used for downstream analysis. Prior to dimensionality reduction, a linear transformation was performed on the normalized data. Unwanted cell-cell variation driven by mitochondrial gene expression and the number of detected molecules (nUMIs) was removed by regressing out these variables during the scaling of data.
Dimensionality reduction was performed on the entire dataset using principal component analysis using the list of highly variable genes generated above. The top 13 principal components which explained more variability (than expected by chance) were identified based on PC heatmaps, the JackStrawPlot and PCElbowPlot. These 13 components were used as input for generating clusters (using the default SLM algorithm), with a resolution of 0.6. t-SNE16 was utilized to reduce the dimensionality of the data to two dimensions (for visualization purposes). Following clustering, genes differentially expressed in each of the clusters were determined using a method of differential expression analysis based on the non-parametric Wilcoxon rank sum test. Cells in each cluster were compared against cells of all other clusters. Genes were then filtered based on being detected in ≥25% of cells within a cluster and a Bonferroni adjusted p value < 0.05. Based on the lists of differentially expressed genes ordered by average log fold change, clusters were assigned specific cell identities. Visualization of specific gene expression patterns across groups on t-SNE and violin plots was performed using functions within the Seurat package.
To subcluster endothelial cells, prior to merging data from etv2ci32Gt heterozygous and homozygous cells, clustering was performed on the two datasets independent of each other. For the etv2 heterozygous dataset, 15 significant principal components were identified and used as input for t-SNE dimensionality reduction, using a resolution of 1.2 for clustering. An endothelial cell population was identified based on differentially expressed genes in that cluster. To identify heterogeneity within the endothelial cell population, the endothelial cells were subsetted into a separate Seurat object and highly variable genes were calculated. A linear transformation was performed again whilst removing unwanted variation driven by mitochondrial gene expression and nUMI by regressing out these variables during the scaling of data. Fourteen significant principal components were selected for t-SNE dimensionality reduction, using a resolution of 1.0 for clustering. Based on these parameters, there appeared to be two transcriptionally distinct populations of endothelial cells, which was also confirmed by a heatmap of gene expression of the two groups.
Pseudotime analysis
To analyze endothelial cell lineage pathways, we used Monocle version 2.8.0 that implements reversed graph embedding on gene expression data to determine single-cell developmental trajectories in an entirely unsupervised manner66. The following populations of cells (from merged heterozygous and homozygous data) were subsetted and imported to Monocle: LPM, EPCs, EC1, EC2, Myocytes. Cells with a UMI count > 106 were excluded from the data to exclude potential multiplets. Furthermore, upper and lower bounds on nUMI were set at two standard deviations above and below the mean UMI to remove low-quality cells. Only cells falling within these boundaries were included for further downstream analysis Filtering of genes was performed by keeping genes expressed in ≥10 cells. Gene dispersion values and average expression was calculated and genes with a mean expression ≥0.1 were subsetted. The top seven significant principal components were used to reduce the dimensionality of the data using t-SNE. Cells were then clustered, setting num_clusters = 8. A differential gene test was performed to determine significant differentially expressed genes and the top 425 genes were arranged by q value to create a list of ordering genes. An ordering filter was then applied to the cells based on the list of genes created in the previous step to label whether a cell is used for ordering or not. In order to determine cell differentiation trajectories, dimensionality reduction to two components was performed using the “DDRTree” method and then assignment of pseudotime and state to each of the cells was carried out using the OrderCells function. The trajectories of cells were plotted on a pseudotime graph and colored by cell identities previously determined using clustering in Seurat.
Single-cell RNA sequencing using Fluidigm C1
Sequencing was performed on cells from zebrafish embryos at 16–18 somite stages and at 20 somite stages. Approximately, 300 Tg(-2.3 etv2:GFP) embryos were collected at each stage, dissociated into a single-cell suspension as described above and suspended in a buffer of 1× PBS containing 1 mM EDTA and 2% FBS. Fluorescence-activated cell sorting was then used to collect GFP+ cells in a solution of 50% FBS in 1× PBS (Supplementary Fig. 16b). Cells were then counted on a hemocytometer, spun down at 300 × G and resuspended at a concentration of approximately 30,000 cells per ml. Each suspension was then loaded onto a 96-well Fluidigm C1 chip for capture of single cells at the CCHMC Gene Expression core. Forty-eight individual cells from each stage group were selected for library synthesis, barcoding, and pooled paired-end sequencing using a single lane of a PE75 flow cell at 1.5–2 million reads per cell, performed at the CCHMC DNA Sequencing core. Data analysis were performed using AltAnalyze44. Reads were aligned to Ensembl release 72 of the Zv9 assembly of the zebrafish genome and gene counts were quantified using Kallisto67. Iterative Clustering and Guide-gene Selection (ICGS) was used to identity de novo clusters/groups of cell68. ICGS was performed using cosine similarity with an expression fold cutoff of six for a minimum of five cells using conservative filtering for cell cycle effects. From ICGS, group specific enriched genes were identified automatically using Markerfinder69. All heatmaps and principal component analyses were created using AltAnalyze.
For the enriched marker table for each cluster, log-normalized fold change values were acquired directly from the AltAnalyze MarkerFinder analysis. Averages of log-normalized fold-change were generated for each marker per cluster. P-value was determined by a two-tailed Mann–Whitney U test comparing fold change in the cluster of interest to fold change in all other cells. The table of average expression values for genes in each cluster was created directly from a text file generated from AltAnalyze. These values represent the average of expression of all cells in that cluster calculated as reads for a specific gene that have been normalized to the total reads per cell and a scale factor of 10,000× applied.
To obtain arteriovenous index, the average of the log2 expression was calculated first for the following known arterial and venous markers: cldn5b, efnb2a, dll4, hey2, dlc, notch3 (arterial), and flt4, dab2, stab1l, ephb4a, mrc1a (venous). Subsequently, the average arterial expression was divided by the average venous expression to generate an arteriovenous identity index (AV index).
Fluorescent cell counting
Percentage of fluorescent cells which express etv2 reporter was estimated in etv2ci32Gt+/−; UAS:mCerulean (mCer) embryos alone or crossed with hsp70:Dkk1-GFP and hsp70:dnFGFR1-GFP lines. Cells from 10 to 25 embryos in each group were disaggregated at 24 hpf as described above and mCer-positive cells were counted using BD FACS Aria II at the CCHMC FACS core facility.
To count gata1:dsRed-positive cells in wild-type and etv2 MO-injected embryos, cells from 50 to 70 embryos in each group were disaggregated at approximately 23 hpf. dsRed-positive cells were counted at the CCHMC FACS core facility using BD FACS Canto II. FACS gating strategy for both fluorescent lines is shown in Supplementary Fig. 16c, d.
Ethics approval
Zebrafish embryo experiments were performed under animal protocol. IACUC2016-0039, approved by the Institutional Animal Care and Use Committee at the. Cincinnati Children’s Hospital Medical Center.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data that support the findings of this study are available from the corresponding author upon reasonable request. scRNA-seq data generated in the study using the Chromium (10× Genomics) platform have been deposited in the NCBI GEO database under the accession code GSE143750. scRNA-seq data generated in the study using the Fluidigm platform have been deposited in the NCBI GEO database under the accession code GSE142484.
References
Ishitobi, H. et al. Molecular basis for Flk1 expression in hemato-cardiovascular progenitors in the mouse. Development 138, 5357–5368 (2011).
Kattman, S. J., Huber, T. L. & Keller, G. M. Multipotent flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev. Cell 11, 723–732 (2006).
Chal, J. & Pourquie, O. Making muscle: skeletal myogenesis in vivo and in vitro. Development 144, 2104–2122 (2017).
Matsuoka, R. L. & Stainier, D. Y. R. Recent insights into vascular development from studies in zebrafish. Curr. Opin. Hematol. 25, 204–211 (2018).
Sumanas, S. & Lin, S. Ets1-related protein is a key regulator of vasculogenesis in zebrafish. PLoS Biol. 4, e10 (2006).
Sumanas, S. et al. Interplay among Etsrp/ER71, Scl, and Alk8 signaling controls endothelial and myeloid cell formation. Blood 111, 4500–4510 (2008).
Lee, D. et al. ER71 acts downstream of BMP, Notch, and Wnt signaling in blood and vessel progenitor specification. Cell Stem Cell 2, 497–507 (2008).
Palencia-Desai, S. et al. Vascular endothelial and endocardial progenitors differentiate as cardiomyocytes in the absence of Etsrp/Etv2 function. Development 138, 4721–4732 (2011).
Rasmussen, T. L. et al. ER71 directs mesodermal fate decisions during embryogenesis. Development 138, 4801–4812 (2011).
Schoenebeck, J. J., Keegan, B. R. & Yelon, D. Vessel and blood specification override cardiac potential in anterior mesoderm. Dev. Cell 13, 254–267 (2007).
Lescroart, F. et al. Defining the earliest step of cardiovascular lineage segregation by single-cell RNA-seq. Science 359, 1177–1181 (2018).
Sabbagh, M. F. et al. Transcriptional and epigenomic landscapes of CNS and non-CNS vascular endothelial cells. Elife 7, e36187 (2018).
Gong, W. et al. Dpath software reveals hierarchical haemato-endothelial lineages of Etv2 progenitors based on single-cell transcriptome analysis. Nat. Commun. 8, 14362 (2017).
Chestnut, B. & Sumanas, S. Zebrafish etv2 knock-in line labels vascular endothelial and blood progenitor cells. Dev. Dyn. 249, 245–261 (2020).
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F. & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
van der Maaten, L. & Hinton, G. Visualizing data using t-SNE. J. Mach. Learn. Res. 9, 2579–2605 (2008).
Liao, E. C. et al. SCL/Tal-1 transcription factor acts downstream of cloche to specify hematopoietic and vascular progenitors in zebrafish. Genes Dev. 12, 621–626 (1998).
Thompson, M. A. et al. The cloche and spadetail genes differentially affect hematopoiesis and vasculogenesis. Dev. Biol. 197, 248–269 (1998).
Parker, L. H. et al. The endothelial-cell-derived secreted factor Egfl7 regulates vascular tube formation. Nature 428, 754–758 (2004).
Pendeville, H. et al. Zebrafish Sox7 and Sox18 function together to control arterial-venous identity. Dev. Biol. 317, 405–416 (2008).
Pham, V. N. et al. Combinatorial function of ETS transcription factors in the developing vasculature. Dev. Biol. 303, 772–783 (2007).
Proulx, K., Lu, A. & Sumanas, S. Cranial vasculature in zebrafish forms by angioblast cluster-derived angiogenesis. Dev. Biol. 348, 34–46 (2010).
Oda, E. et al. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 288, 1053–1058 (2000).
Okamoto, K. & Prives, C. A role of cyclin G in the process of apoptosis. Oncogene 18, 4606–4615 (1999).
Casie Chetty, S. et al. Vegf signaling promotes vascular endothelial differentiation by modulating etv2 expression. Dev. Biol. 424, 147–161 (2017).
Small, K. M. & Potter, S. S. Homeotic transformations and limb defects in Hox A11 mutant mice. Genes Dev. 7, 2318–2328 (1993).
Davidson, A. J. & Zon, L. I. The caudal-related homeobox genes cdx1a and cdx4 act redundantly to regulate hox gene expression and the formation of putative hematopoietic stem cells during zebrafish embryogenesis. Dev. Biol. 292, 506–518 (2006).
Thisse, B. et al. Expression of the zebrafish genome during embryogenesis. ZFIN Direct Data Submission http://zfin.org (2001).
Martin, B. L. & Kimelman, D. Canonical Wnt signaling dynamically controls multiple stem cell fate decisions during vertebrate body formation. Dev. Cell 22, 223–232 (2012).
Reiter, J. F. et al. Gata5 is required for the development of the heart and endoderm in zebrafish. Genes Dev. 13, 2983–2995 (1999).
Veldman, M. B. & Lin, S. Etsrp/Etv2 is directly regulated by Foxc1a/b in the zebrafish angioblast. Circ. Res. 110, 220–229 (2012).
Higashijima, S., Okamoto, H., Ueno, N., Hotta, Y. & Eguchi, G. High-frequency generation of transgenic zebrafish which reliably express GFP in whole muscles or the whole body by using promoters of zebrafish origin. Dev. Biol. 192, 289–299 (1997).
Van Handel, B. et al. Scl represses cardiomyogenesis in prospective hemogenic endothelium and endocardium. Cell 150, 590–605 (2012).
Veldman, M. B. et al. Transdifferentiation of fast skeletal muscle into functional endothelium in vivo by transcription factor Etv2. PLoS Biol. 11, e1001590 (2013).
Nguyen, P. D. et al. Haematopoietic stem cell induction by somite-derived endothelial cells controlled by meox1. Nature 512, 314–318 (2014).
Kohli, V., Schumacher, J. A., Desai, S. P., Rehn, K. & Sumanas, S. Arterial and venous progenitors of the major axial vessels originate at distinct locations. Dev. Cell 25, 196–206 (2013).
Aulehla, A. & Pourquie, O. Signaling gradients during paraxial mesoderm development. Cold Spring Harb. Perspect. Biol. 2, a000869 (2010).
Mohammadi, M. et al. Structures of the tyrosine kinase domain of fibroblast growth factor receptor in complex with inhibitors. Science 276, 955–960 (1997).
Lee, Y., Grill, S., Sanchez, A., Murphy-Ryan, M. & Poss, K. D. Fgf signaling instructs position-dependent growth rate during zebrafish fin regeneration. Development 132, 5173–5183 (2005).
Row, R. H. et al. BMP and FGF signaling interact to pattern mesoderm by controlling basic helix-loop-helix transcription factor activity. Elife 7, e31018 (2018).
Mahmoud, M. M. et al. TWIST1 integrates endothelial responses to flow in vascular dysfunction and atherosclerosis. Circ. Res. 119, 450–462 (2016).
Ocana, O. H. et al. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell 22, 709–724 (2012).
Xu, P. et al. Fox proteins are modular competency factors for facial cartilage and tooth specification. Development 145, dev165498 (2018).
Emig, D. et al. AltAnalyze and DomainGraph: analyzing and visualizing exon expression data. Nucleic Acids Res. 38, W755–W762 (2010).
Zhong, T. P., Childs, S., Leu, J. P. & Fishman, M. C. Gridlock signalling pathway fashions the first embryonic artery. Nature 414, 216–220 (2001).
Quillien, A. et al. Distinct Notch signaling outputs pattern the developing arterial system. Development 141, 1544–1552 (2014).
Herbert, S. P. et al. Arterial-venous segregation by selective cell sprouting: an alternative mode of blood vessel formation. Science 326, 294–298 (2009).
Org, T. et al. Scl binds to primed enhancers in mesoderm to regulate hematopoietic and cardiac fate divergence. EMBO J. 34, 759–777 (2015).
Craig, M. P. et al. Etv2 and fli1b function together as key regulators of vasculogenesis and angiogenesis. Arterioscler Thromb. Vasc. Biol. 35, 865–876 (2015).
Asakawa, K. et al. Genetic dissection of neural circuits by Tol2 transposon-mediated Gal4 gene and enhancer trapping in zebrafish. Proc. Natl Acad. Sci. USA 105, 1255–1260 (2008).
Davison, J. M. et al. Transactivation from Gal4-VP16 transgenic insertions for tissue-specific cell labeling and ablation in zebrafish. Dev. Biol. 304, 811–824 (2007).
Lawson, N. D. & Weinstein, B. M. In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev. Biol. 248, 307–318 (2002).
Traver, D. et al. Transplantation and in vivo imaging of multilineage engraftment in zebrafish bloodless mutants. Nat. Immunol. 4, 1238–1246 (2003).
Stoick-Cooper, C. L. et al. Distinct Wnt signaling pathways have opposing roles in appendage regeneration. Development 134, 479–489 (2007).
Kimmel, C. B., Ballard, W. W., Kimmel, S. R., Ullmann, B. & Schilling, T. F. Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253–310 (1995).
Dooley, K. A., Davidson, A. J. & Zon, L. I. Zebrafish scl functions independently in hematopoietic and endothelial development. Dev. Biol. 277, 522–536 (2005).
Sumanas, S., Jorniak, T. & Lin, S. Identification of novel vascular endothelial-specific genes by the microarray analysis of the zebrafish cloche mutants. Blood 106, 534–541 (2005).
Rost, M. S. & Sumanas, S. Hyaluronic acid receptor Stabilin-2 regulates Erk phosphorylation and arterial-venous differentiation in zebrafish. PLoS ONE 9, e88614 (2014).
Wong, K. S. et al. Hedgehog signaling is required for differentiation of endocardial progenitors in zebrafish. Dev. Biol. 361, 377–391 (2012).
Herbomel, P., Thisse, B. & Thisse, C. Ontogeny and behaviour of early macrophages in the zebrafish embryo. Development 126, 3735–3745 (1999).
Palencia-Desai, S. et al. Myocardium and BMP signaling are required for endocardial differentiation. Development 142, 2304–2315 (2015).
Choi, H. M. T. et al. Third-generation in situ hybridization chain reaction: multiplexed, quantitative, sensitive, versatile, robust. Development 145, dev165753 (2018).
Manoli, M. & Driever, W. Fluorescence-activated cell sorting (FACS) of fluorescently tagged cells from zebrafish larvae for RNA isolation. Cold Spring Harb. Protoc. 2012, pdb.prot069633 (2012).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Macosko, E. Z. et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214 (2015).
Trapnell, C. et al. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat. Biotechnol. 32, 381–386 (2014).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Olsson, A. et al. Single-cell analysis of mixed-lineage states leading to a binary cell fate choice. Nature 537, 698–702 (2016).
Sigdel, T. K. et al. The identification of novel potential injury mechanisms and candidate biomarkers in renal allograft rejection by quantitative proteomics. Mol. Cell Proteom. 13, 621–631 (2014).
Acknowledgements
This research was supported by the awards from National Insitute of Health: NIH R01 HL134815 and R21 AI128445 to S.S., and NIH F31 HL135986 to A.L.K. We thank Nathan Salomonis and Praneet Chaturvedi for their assistance with scRNA-seq analysis, Steve Potter for critical comments, and CCHMC Gene expression and FACS cores for their assistance with the project.
Author information
Authors and Affiliations
Contributions
B.C. generated etv2ci32Gt line and performed most of experiments in the study, S.C.C. performed scRNA-seq analysis of the Chromium data and edited the paper, A.L.K. performed all experiments using the Fluidigm platform and subsequent data analysis, as well as the cell disaggregation and initial data processing using the Chromium platform, S.S. initiated and supervised the entire study, contributed to data analysis, and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Peng Jiang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chestnut, B., Casie Chetty, S., Koenig, A.L. et al. Single-cell transcriptomic analysis identifies the conversion of zebrafish Etv2-deficient vascular progenitors into skeletal muscle. Nat Commun 11, 2796 (2020). https://doi.org/10.1038/s41467-020-16515-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-16515-y
This article is cited by
-
Canalizing cell fate by transcriptional repression
Molecular Systems Biology (2024)
-
Plant biotechnology research with single-cell transcriptome: recent advancements and prospects
Plant Cell Reports (2024)
-
ETV2/ER71, the key factor leading the paths to vascular regeneration and angiogenic reprogramming
Stem Cell Research & Therapy (2023)
-
Transgenic IDH2R172K and IDH2R140Q zebrafish models recapitulated features of human acute myeloid leukemia
Oncogene (2023)
-
X-box binding protein 1 as a key modulator in “healing endothelial cells”, a novel EC phenotype promoting angiogenesis after MCAO
Cellular & Molecular Biology Letters (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.