Abstract
Roughly a third (~30 ppm) of the carbon dioxide (CO2) that entered the ocean during ice ages is attributed to biological mechanisms. A leading hypothesis for the biological drawdown of CO2 is iron (Fe) fertilisation of the high latitudes, but modelling efforts attribute at most 10 ppm to this mechanism, leaving ~20 ppm unexplained. We show that an Fe-induced stimulation of dinitrogen (N2) fixation can induce a low latitude drawdown of 7–16 ppm CO2. This mechanism involves a closer coupling between N2 fixers and denitrifiers that alleviates widespread nitrate limitation. Consequently, phosphate utilisation and carbon export increase near upwelling zones, causing deoxygenation and deeper carbon injection. Furthermore, this low latitude mechanism reproduces the regional patterns of organic δ15N deposited in glacial sediments. The positive response of marine N2 fixation to dusty ice age conditions, first proposed twenty years ago, therefore compliments high latitude changes to amplify CO2 drawdown.
Similar content being viewed by others
Introduction
As much as 30 ppm of the total glacial-interglacial difference in atmospheric CO2 is attributed to marine biological mechanisms1. The most prominent biological mechanism is the fertilisation of Fe-limited high latitude regions, namely the Southern Ocean2 and subarctic Pacific3, with dust-borne Fe under dusty glacial conditions4,5. Today, phytoplankton that inhabit these high latitude regions are unable to consume all available macronutrients, which allows CO2 to escape to the atmosphere as deep waters mix into surface layers. Iron fertilisation of the high latitude glacial ocean therefore stands as a leading hypothesis to explain a more efficient biological carbon (C) pump and the associated drawdown of atmospheric CO2. Yet, modelling focussed on the high latitudes has sequestered less than 10 ppm of atmospheric CO2 via Fe fertilisation5,6,7 and indicates that additional biological mechanisms are required.
There are good reasons to accommodate the lower latitudes in our search for additional mechanisms. First, the region is enormous. Surface waters between 40°S and 40°N represent over two thirds of CO2 outgassing to the atmosphere8 and more than half of global C export9,10. Second, unconsumed phosphate (PO4) at concentrations in excess of 0.1 to 0.2 mmol m−3 exists in surface waters across the tropics, which is evidence for unrealised biological CO2 fixation. Third, tropical oceans produce organic matter that is enriched in C because tropical phytoplankton are adapted to fix more C per unit phosphorus (P) under P scarcity11. Fourth, oxygen-deficient waters in the tropical Pacific, Indian and Atlantic allow organic matter to sink deeper into the ocean interior10,12,13. If these mechanisms are combined, the co-occurrence of more complete PO4 utilisation and the production of C-enriched organic matter near to oxygen-deficient zones would constitute an effective pathway of CO2 drawdown.
Enabling greater PO4 utilisation and CO2 drawdown in the lower latitudes, however, requires simultaneously relieving Fe limitation in upwelling zones14, nitrate (NO3) limitation in the tropics14,15 and their co-limitation at the boundary of both regimes16. An aeolian Fe-induced stimulation of dinitrogen (N2) fixation is therefore an obvious candidate to alleviate low latitude nutrient limitation. Originally proposed by Falkowski17, this mechanism is now supported by many independent lines of evidence. N2 fixers are highly sensitive to the aeolian supply of Fe18,19, they represent up to half of primary production and C export in oligotrophic waters20,21,22,23,24, they are physiologically adapted to P scarcity25,26, produce organic matter that is enriched in C27,28,29, and previous modelling has demonstrated the potential of N2 fixation to draw CO2 into the ocean30. Dinitrogen fixation is also inextricably linked to suboxic zones (dissolved oxygen (O2) < 10 mmol m−3) where denitrification strips NO3 from the waters that upwell at the equator, creating a potential niche for N2 fixers across the wide expanse of the lower latitudes. The strength of N2 fixation, which strengthens PO4 utilisation, whole community C:P ratios and C export20, is thus tied to the strength of denitrification, which in turn strengthens N2 fixation.
In this study, we use an ocean model to demonstrate that aeolian Fe supply to the tropical oceans under glacial conditions31,32 relieves low latitude nutrient limitation14,15,16 by stimulating N2 fixation, which in turn drives PO4 consumption, suboxic zone expansion, the acceleration of the nitrogen (N) cycle and a more efficient C export to the interior ocean. Furthermore, we estimate the contribution of this mechanism to CO2 drawdown and reveal evidence of its existence within glacial-interglacial sedimentary records of N isotopes (δ15Norg).
Results
A low latitude pathway
Inspired by these insights, we undertook multi-millennial simulations using a global ocean biogeochemical model to explore the link between Fe fertilisation, N2 fixation and CO2 drawdown. The ocean biogeochemical model is part of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) Mark 3L—Carbon of the Ocean, Atmosphere and Land (Mk3L-COAL)33. The model is designed for long-term, global oceanographic studies. It resolves multi-millennial timescales and so produces equilibrium circulation states under a given set of atmospheric conditions. It is equipped with prognostic C, PO4, NO3, 15NO3, and Fe cycles34 (see Methods), and includes a dynamic ecosystem component where phytoplankton alter their nutrient requirements, stoichiometry and remineralisation rates according to their environment33 (Supplementary Fig. 1). We increased the supply of aeolian Fe to the ocean model from its modern35 to glacial rate5 (see Methods; Supplementary Fig. 2) under preindustrial physical conditions (Mk3Lmild state in Table 1; Supplementary Note 1; Supplementary Figs. 3 and 4; Supplementary Table 1) with an atmospheric CO2 held at 280 ppm, and assessed changes to elemental cycling. To isolate the response of the lower latitudes, we nudged subsurface Fe concentrations to 0.6 μmol m−3 on a yearly timescale, which ensured that Fe was near non-limiting in regions of strong mixing, like the Southern Ocean and subarctic Pacific.
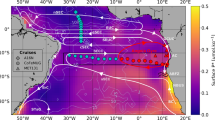
The glacial aeolian Fe supply increased the global rate of N2 fixation by 26 Tg N yr−1 and caused a large-scale change in its distribution (Fig. 1a). Dinitrogen fixers exhibited a closer coupling to regions of strong upwelling in the tropics (solid contour in Fig. 1a) that are co-located with areas of denitrification (dots in Fig. 1a). The greatest changes were observed in the Pacific. Dinitrogen fixation decreased in the Northwest Pacific and increased in the Eastern Tropical Pacific, which hosted low rates of N2 fixation under modern Fe supply (Supplementary Fig. 5a). As a result, surface PO4 was reduced throughout the tropical Pacific by between 0.1 and 0.2 mmol m−3 (Fig. 1b; Supplementary Fig. 5b). Pacific PO4 utilisation increased the C:P ratio of exported organic matter by an average of ~14 units (contours in Fig. 1b), which elevated local C export (Fig. 1c; Supplementary Fig. 5c), caused a vertical expansion of suboxia (dots in Fig. 1c), and enabled the permanent accumulation of 244 Pg of respired C in the eastern Pacific (Fig. 1d; Supplementary Fig. 5d). As a result, 11.6 ppm of CO2 was permanently sequestered (see “Quantifying CO2 drawdown” and Fig. 2).
Biogeochemical response to an Fe-induced stimulation of N2 fixers. Change in a N2 fixation rate, b surface PO4 concentration, c carbon export rate and d total respired carbon through the water column. The changes shown in both the coloured shading and the headings above each panel show the effect of increasing Fe deposition from the modern flux to the glacial flux5. The headings describe the integrated change (Δtotal) or the average change (Δave) of each property, calculated as the volume or area weighted sum/average of each property. Dots in (a) represent active water column denitrification. Contours in (a) define upwelling where ideal water age >25 years at 80 metres depth. Contours in (b) are the change in C:P ratios of exported organic matter. Dots (dashes) in (c) define a vertical expansion (shrinking) of suboxia >500 metres
N2 fixation and its relationship with atmospheric CO2. The atmospheric reservoir of CO2 was made responsive to oceanic uptake and release of C by the ocean (see methods). Coloured edges of markers correspond to ocean states as described in the figure legend. Star markers represent oceans with carbon cycles equilibrated to the modern Fe deposition36 under atmospheric CO2 of −280 ppm. The plus symbol in each ocean state represents the change due to a glacial increase in Fe supply6, which represents a global integrated increase of 500% over the modern supply but is regionally variable (Supplementary Fig. 2). Triangles represent changes in the half-saturation constant for Fe limitation of N2 fixers, emulating changes in Fe supply to only N2 fixers. Note the diminishing gains in CO2 drawdown between 500 and 2500% iron deposition scenarios, consistent with Fe-saturation and PO4 limitation. Regression lines and their slopes (gradient ± standard deviation (SD)) represent the linear relationship between N2 fixation and atmospheric CO2, with a multi-ocean mean of 0.58 ± 0.03 ppm CO2 sequestered for every additional Tg N yr−1. Source data are provided in the source data file
Dust-borne Fe fertilisation therefore involved a set of biogeochemical feedbacks, not possible by increasing the NO3 inventory (Supplementary Note 2; Supplementary Fig. 6), that enabled CO2 drawdown. Tropical upwelling zones are highly productive regions that drive strong subsurface O2 depletion, which in turn stimulates denitrification and strips upwelling waters of NO3. Consequently, the tropical Pacific hosts low NO3:PO4 ratios (Supplementary Fig. 7a), which provides a competitive niche for N2 fixers. Today, the low supply of aeolian Fe to the tropical Pacific31,35 prevents N2 fixers from inhabiting this niche24, and allows excess, unconsumed PO4 (>0.2 mmol m−3) to spill 10–15° either side of the equator36 (Supplementary Fig. 7b). In contrast, the glacial Fe supply allowed N2 fixers to inhabit the low NO3:PO4 waters at the boundary to upwelling zones where local Fe-N co-limitation prevails today14,16. This shift in N2 fixation initiated strong biogeochemical feedbacks that encouraged PO4 utilisation, C export, suboxic expansion, denitrification, and a local NO3 supply via N2 fixation. Two consequences of this simulated feedback, the local increase in C export and a vertical expansion of suboxia, enabled the transfer of large amounts of C-rich organic matter deep within the interior of the Pacific basin (Fig. 1d).
Quantifying CO2 drawdown
We sought to quantify the sensitivity of atmospheric CO2 drawdown to the physical conditions of the ocean, as glacial conditions were distinct from pre-industrial37. We produced four different ocean states that can be considered broadly representative of glacial-interglacial conditions, encompassing warm to cold, well-mixed to stratified, and thus interglacial to glacial (Table 1; Supplementary Fig. 8). Dust-borne Fe supply was varied to 50, 80, 100, 500% (glacial) and 2500% of the modern rate35 over these four ocean states (see Methods) to fully encompass the glacial-interglacial range in conditions. Both high Fe deposition scenarios (500 and 2500%) are based on the climatology of Lambert5, meaning that the delivery of Fe is not uniformly greater everywhere (Supplementary Fig. 2). The tropical Pacific, for instance, receives roughly 2-fold more Fe than under modern conditions consistent with recent estimates31,32. In addition, we increased and decreased the Fe requirements of N2 fixers without varying aeolian Fe deposition (See methods), which emulated variations in Fe supply but only to N2 fixers. If similar changes occurred via both methods, then N2 fixation could be considered the primary driver of CO2 drawdown.
The ocean states were GFDLwarm, Mk3Lmild (control state used previously), HadGEMcool and Mk3Lcold. GFDLwarm was the warmest, youngest (see δ14C), most deoxygenated, NO3-deplete and PO4-rich ocean, with a rapid overturning circulation dominated by the upper cell. Mk3Lmild and HadGEMcool were cooler, fresher and formed greater quantities of Antarctic Bottom Water than GFDLwarm. The key difference between Mk3Lmild and HadGEMcool was the rate of North Atlantic Deep Water formation, which was stronger for Mk3Lmild and elevated surface PO4, C export, O2 consumption and denitrification rates. Mk3Lcold represented full glacial conditions. It was the coldest, saltiest, and oldest ocean state, featuring strong vertical density gradients that restricted PO4 supply and a greatly expanded lower overturning cell consistent with glacial conditions38.
An increase in Fe supply drew between 6.7 and 16 ppm of atmospheric CO2 into the ocean (compare star and plus symbols in Fig. 2). Different ocean states (colours in Fig. 2) therefore absorbed different quantities of CO2. However, all states developed a positive, linear relationship between N2 fixation and CO2 drawdown (coloured lines in Fig. 2). A consistent relationship between N2 fixation and CO2 drawdown suggested that all states absorbed atmospheric CO2 via the same low latitude pathway described in previously. Approximately 0.58 ± 0.03 ppm of CO2 was absorbed by the ocean for every additional Teragram of N fixed per year (Tg N yr−1). The linear relationship was generated as N2 fixation responded to variations in Fe supply (circles) and as N2 fixation was made more or less sensitive to the modern supply of Fe (triangles). Similar responses occurred via both methods (altered Fe deposition and Fe requirements) and strongly implicated N2 fixation as the driver of CO2 drawdown.
The sensitivity of each ocean state to changes in N2 fixation was fundamentally linked to the strength of equatorial upwelling. The greatest sensitivity was found in GFDLwarm, which featured strong upwelling, and therefore high surface PO4 and large suboxic zones (Table 1). Consequently, large regions of the tropical ocean were low in NO3:PO4, which enabled large gains in N2 fixation (88 Tg N yr−1) and CO2 drawdown (43 ppm) as Fe supply increased from 50 to 2500% of its modern rate. In contrast, Mk3Lcold featured the weakest rates of upwelling, lowest surface concentrations of PO4, the smallest suboxic zones (Table 1), and thus the weakest sensitivity. Phosphate availability therefore emerged as the ultimate control on biological CO2 drawdown by setting N2 fixation potential, while Fe supply modulated the extent to which this potential was realised.
A central role for N2 fixers
The previous experiments showed that N2 fixers responded to Fe addition leading to reduced atmospheric CO2. To elucidate the mechanisms through which this occurred we considered several additional experiments with the Mk3Lmild ocean state subject to variations in aeolian Fe supply. First, we removed N2 fixers and denitrification completely, thereby holding the NO3 reservoir constant. Second, we reinstated N2 fixers (NO3 supply) and a marine N cycle (active denitrification), but removed their C export by setting their C:P ratio equal to zero. Third, we decreased their C:P ratio to 165:1, half its default of 331:139. Fourth, we reinstated their default C:P ratio of 331:1, but increased their PO4 half-saturation coefficient \(\left( {K_{PO_4}^D} \right)\) to 0.1 mmol m−3, which is the same as the general phytoplankton group and so removed their competitive advantage for PO4.
These experiments revealed that N2 fixers were essential for C accumulation via the low latitudes. If N2 fixers were removed and the NO3 reservoir remained constant, greater Fe supply did not cause respired C storage (ones in Fig. 3a). Insensitivity to Fe supply was due to widespread NO3 limitation of lower latitude ecosystems15. The simple addition of N2 fixers without changes in Fe increased NO3 supply to surface waters (Supplementary Fig. 9) and increased PO4 utilisation between 40°S and 40°N by 7%. Dinitrogen fixers were, therefore, able to provide significant gains to the oceanic C store over millennia, which extends insights of in situ studies20,21,22,23 and prior modelling30 to the scale of the glacial cycles, as originally proposed by Falkowski17.
How N2 fixation enables CO2 storage in the ocean. The response of the respired C inventory to aeolian Fe deposition experiments for five different representations of N2 fixers, with a demonstrating the relationship between respired C and Fe availability and b demonstrating the relationship between respired C and N2 fixation. The marker numbers refer to how N2 fixers are represented. Ones: no N2 fixers and no active N cycle. Twos: N2 fixers and N cycle (i.e., denitrification) reinstated but no N2 fixer carbon export (C:P = 0:1). Threes: N2 fixers C:P ratio increased to 165:1, half its default value. Fours: N2 fixers PO4 limitation enforced by increasing their half saturation coefficient \(\left( {K_{PO_4}^D} \right)\) from 10−10 to 0.1 mmol m−3. Fives: N2 fixers with default parameterisation. Colour shading of the markers indicates the mean surface PO4 concentration of the experiment. Background shading in b is a qualitative indicator of the transition from NO3 to PO4 limitation, where dark green to yellow indicates NO3 limitation and light blue indicates PO4 limitation. Source data are provided in the source data file
Dinitrogen fixers were therefore essential for oceanic C storage for the simple reason that they supplied fixed N to the upper ocean. Fixed N supply was responsible for 70% of the C gains (~5–11 ppm) and responsible for the increase in PO4 utilisation as Fe supply increased (compare ones and twos in Fig. 3a). Included within this C storage was the increase in C:P ratios of Pacific Ocean phytoplankton as PO4 concentrations declined (see Fig. 1b). The final 30% (~2–5 ppm CO2) of additional C gain was mostly due to export of N2 fixer’s C-rich organic matter as the ocean became PO4-limited (compare ones, threes and fives in Fig. 3a), while their efficient utilisation of PO4 provided a small benefit to oceanic C storage (compare fours and fives in Fig. 3a). As N2 fixers already inhabit a niche of low NO3:PO420, they are already at a competitive advantage over non-N2 fixing phytoplankton for available PO4.
The linear relationship between N2 fixation and CO2 drawdown, which was robust across different ocean states (Fig. 2), was therefore built on two phases. The first phase (green-yellow shading in Fig. 3b) occurred in a NO3-limited ocean, where N2 fixation increased the supply of NO3 to surface communities and thereby allowed excess PO4 to be consumed. The second phase (light blue shading in Fig. 3b) occurred in a PO4-limited ocean. As N2 fixers consumed proportionally more of the remaining PO4, their C export became more important for overall C export (compare ones, threes and fours/fives Fig. 3b). The slope of the linear relationship presented in Fig. 2, therefore, rested on a C:P ratio of N2 fixer organic matter equal to 331:139. While there is significant variation around this number, under PO4-limiting conditions the C:P ratio tends to increase, exceeding 500:1 among Trichodesmium species28. Therefore, the C:P of N2 fixers could rise as PO4-limiting conditions develop and steepen the linear relationship to enable greater CO2 drawdown above that suggested here.
Glacial δ 15N records
To test our proposed mechanism of low latitude CO2 drawdown against observations, we simulated the response of the isotopic composition of organic N (δ15Norg) to a glacial increase in Fe supply, and compared this response to a global compilation of glacial δ15N records (Supplementary Data 1). These experiments were completed within each ocean state presented in Table 1, so as to isolate the effect of Fe fertilisation from the effects of physical changes. In the following, we discuss the response using the Mk3Lmild ocean state, but each ocean state gave a similar response (Supplementary Fig. 10).
An Fe-induced coupling of N2 fixers to the upwelling zones of the eastern tropical Pacific increased δ15Norg in the west and decreased it in the east, which broadly reproduced patterns of glacial-interglacial change throughout the Pacific basin (Fig. 4). The increase in the western part of the basin was due to local decreases in N2 fixation and sedimentary denitrification, both of which lower δ15N. Our simulation of higher δ15Norg in the west Pacific, therefore, supports the interpretation of a recent foraminifera-bound record in the South China Sea40 (star marker). However, the simulated decrease in the δ15N of the eastern Pacific was not caused by a decrease in water column denitrification as suggested by numerous studies since the seminal paper of Ganeshram et al.41. Instead, our simulated decrease in eastern δ15Norg was caused by increases in both sedimentary denitrification and N2 fixation (Fig. 1a).
Seeking evidence from sedimentary δ15Norg records. Shading depicts the simulated change in δ15N of organic matter as a result of increasing aeolian Fe supply from the modern to a glacial rate. Solid and dashed contour lines mark simulated differences of 1 and −1. Circles mark locations of sediment cores where bulk organic matter was analysed for δ15N, while stars mark locations where either foraminifera- or diatom-bound δ15N was measured. The colour of the markers is an estimate of the glacial (Last Glacial Maximum: 20,000–26,000 BCE) minus interglacial (Late Holocene: 0–5000 BCE) difference in δ15Norg, with red colours representing higher values and blues representing lower values in the glacial ocean. Sedimentary record data are provided in Supplementary Data 1
However, poor agreement was found in other regions, namely in the tropical western Atlantic and Southern Ocean where an increase in δ15Norg was not simulated. In the west Atlantic, Straub et al.42 presented a compelling relationship between δ15N and orbital precession, leading the authors to surmise a dependence on the upwelling of PO4 via changes in the circulation. In the Southern Ocean, a glacial increase in δ15N in Subantarctic2 and Antarctic zones43,44 is explained by a weaker physical delivery of NO3 to the mixed layer combined with Fe fertilisation. We therefore expected and found no response in both regions in these experiments (Supplementary Fig. 10) because the only change was an increase in dust-borne Fe and the Southern Ocean was made insensitive to increases in Fe supply.
Discussion
Our study confirms that N2 fixation is a key component of the global C cycle. We extend a theoretical proposal made over 20 years ago17 to a quantifiable mechanism of CO2 drawdown. The main biogeochemical feedbacks are illustrated in Fig. 5, where a coupling of N2 fixers to upwelling zones is the catalyst that drives CO2 drawdown.
Scenarios of Fe supply to the tropical Pacific. In the low iron scenario, analogous to the modern climate, N2 fixation (yellow zone and dots) is concentrated in the Northwest and Southwest subtropical Pacific where aeolian dust deposition is greatest. Non-limiting PO4 concentrations (green zone and dots) exist within the tropics and spread laterally from the area of upwelling near the Americas and at the equator (blue zone). In the high Fe scenario, analogous to the glacial climate, N2 fixation couples to the upwelling zones in the east Pacific, enabling strong utilisation of PO4, the vertical expansion of suboxic zones (grey bubbles) and a deeper injection of carbon-enriched organic matter (downward squiggly arrows)
The importance of N2 fixation for CO2 drawdown is relevant when assessing prior modelling work. Simulations of the glacial climate have struggled to explain the full drawdown of roughly 90 ppm45, unless they make manual, and therefore non-mechanistic, changes to biological functioning38,46. Furthermore, model studies that explore Fe fertilisation without considering variable stoichiometry and remineralisation rates6,7 have struggled to sequester more than 10 ppm of CO2. The permanent sequestration of 7–16 ppm solely via the low latitudes, therefore, represents a new and complementary pathway to explain the glacial CO2 drawdown. Thinking conservatively given the stratified and therefore PO4-limited conditions of a glacial ocean37,42, we propose that one third, or 10 ppm, of the 30 ppm attributed to Fe fertilisation1 can be explained by a closer coupling of N2 fixation to tropical upwelling zones.
It is important to recognise, however, that our simulations rendered eutrophic regions insensitive to Fe fertilisation. Consequently, we neglect the response of Fe-limited regions like the Southern Ocean that not only have demonstrated potential for CO2 drawdown5,6,7,38,45, but also influence low latitude biogeochemistry through mode and intermediate waters47. This work should therefore not be interpreted as a globally integrated response to Fe fertilisation. Instead, it isolates the response of the lower latitudes and offers important lessons. First, that the debated32,48,49 CO2 drawdown via the tropics is possible. Second, that this drawdown can accompany and thus complement high latitude mechanisms of CO2 drawdown. Third, that this drawdown requires simultaneous relief from both Fe and NO3 limitation14,15,16, which is plausibly achieved by stimulating N2 fixers with dust-borne Fe.
Our confidence in this N2 fixer-mediated mechanism is bolstered by our simulation of the glacial-interglacial changes in δ15Norg within the Pacific basin. However, both the drawdown of CO2 and the reproduction of the δ15Norg patterns in our study hinge on an acceleration of N cycling in the Eastern Tropical Pacific. By acceleration of N cycling, we mean an acceleration of the rates of N2 fixation and denitrification. Such an acceleration conflicts with a long-assumed deceleration of N cycling. Since Ganeshram et al.41, glacial records of low δ15Norg are interpreted to reflect a massive deceleration of water column denitrification, which must have exceeded a deceleration of sedimentary denitrification caused by a loss of shelf area50. Instead, our simulations produced an increase in sedimentary denitrification under Fe fertilisation. While both possibilities can explain the trends in Pacific δ15Norg because they both involve more sedimentary over water column denitrification, they diverge in the inferred intensity of N cycling.
New evidence questions a glacial deceleration of the N cycle in the Eastern Tropical Pacific. Recent work has revealed a vertical expansion of Pacific suboxic zones32,51, a feature reproduced by our Fe fertilisation simulations. While it is not well known whether sedimentary or water column denitrification is more sensitive to increases in suboxia, it seems unlikely that both would decrease as suboxic zones expanded. In fact, it seems more likely that sedimentary denitrification was stimulated as waters overlying the sediment became deoxygenated52 and as more organic carbon was buried within sediments53, while water column denitrification, which is centred within the thermocline54, was reduced in line with reduced rates of particle export55. If suboxic zones did expand vertically32,51,52 and local N cycling accelerated, then the coupling of N2 fixers to eastern upwelling zones and subsequent CO2 drawdown is legitimate.
The legitimacy of our proposal then requires explaining another apparent inconsistency in glacial records: how could less particle export55 in the tropical Pacific coincide with more C export? Our results suggest that an answer may be found in the combination of variable stoichiometry and deoxygenation. Strong PO4 utilisation and aeolian Fe supply enriches the C content of exported organic matter11,56, while deoxygenation enables a strong transfer of particles to depth10,12,13. If both features were present during glacial periods, then lower rates of particle export55 do not preclude more C export, and therefore CO2 drawdown.
Today, there are compelling signs that N2 fixation has strengthened within the Pacific since the industrial revolution57,58 and that suboxic zones are expanding59,60. Our experiments suggest that these changes are symptomatic of a stronger biological C pump, but even so, we propose that gains in N2 fixation remain unrealised. Evidence that N2 fixation is operating well below full capacity can be found in the excess PO4 that spreads 10–15° outwards from tropical upwelling zones36 (Supplementary Fig. 7b) and the spatial decoupling of N2 fixation from denitrification24. Realising the full potential of N2 fixation appears primarily dependent on the delivery of aeolian Fe to the surface ocean. Like the high latitudes2,3, we find that the strength of the lower latitude biological C pump demonstrates a strong link to the Fe cycle. However, how the oceanic Fe cycle will change in the future is uncertain61, and undermines our ability to predict the ocean’s role in atmospheric CO2 drawdown in the coming centuries.
Methods
Model
Model simulations were performed using the ocean component of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) Mark 3L—Carbon of the Ocean, Atmosphere and Land (Mk3L-COAL) Earth system model. The ocean component is comprised of an ocean general circulation model (OGCM) described in Phipps et al.62 and an ocean biogeochemical model (OBGCM) described in Buchanan et al.33 and Buchanan et al.34. A more specific description of the N cycle and Fe cycle are presented in the supplement. The ocean model has a horizontal resolution of 2.8° in longitude by 1.6° in latitude, with 21 vertical levels. It is a coarse resolution, z-coordinate OGCM, allowing millennial timescales to be resolved.
The OBGCM is equipped with 13 prognostic tracers that can be grouped into carbon chemistry fields, oxygen fields, nutrient fields and age tracers. Carbon chemistry and air-sea gas exchange is parameterised according to the latest ocean model requirements63. Nitrogen isotope routines are described in Buchanan et al.34. The cycling of organic matter considers three forms of phytoplankton. These are a general phytoplankton group (G), N2 fixers (otherwise known as diazotrophs; D) and calcifiers. The general phytoplankton group is controlled by dynamic equations for organic matter production, remineralisation and stoichiometry according to the study of Buchanan et al.33. These equations allow the general phytoplankton group to represent variations in the biogeochemical properties of the marine ecosystem, which has positive effects on the simulation of global ocean biogeochemistry, particularly the N cycle. Meanwhile, N2 fixers and calcifiers follow more static equations. N2 fixers have fixed nutrient limitation functions and stoichiometry based on laboratory studies, but are also remineralised according to community composition. Remineralisation of both forms of organic matter is also conserved and passed to deeper grid boxes if oxygen is not sufficient. The calcifying group, which only interacts with DIC and ALK species, produces particulate inorganic carbon at 8% of the rate at which the general phytoplankton group produces organic carbon. Its remineralisation rate is also fixed according to an e-folding depth-dependent decay, which transfers a large fraction of particulate inorganic carbon to the deep ocean.
Nitrogen cycle
Nitrate is introduced to the ocean through atmospheric deposition and N2 fixation. Atmospheric deposition adds 11.3 Tg N to the surface ocean each year using a prescribed monthly climatology64.
The addition of NO3 by N2 fixation is calculated by considering marine N2 fixers as a unique group of phytoplankton. N2 fixers consume PO4 and Fe at the surface ocean, and release PO4, Fe and NO3 at depth during remineralisation. The stoichiometry of N2 fixers is static, with a C:N:P:Fe ratio of 331:50:1:0.00064 according to physiological studies39,65,66. With this stoichiometry, we apply Orem:P and Nrem:P requirements of 431 and 294.8, respectively, using the equations of Paulmier et al.67.
The export of phosphorus by N2 fixers \((P_{exp}^D)\) is calculated using a maximum growth rate μD(T) that is temperature dependent68, limitation terms dependent on the availability of PO4, NO3 and Fe, and minimum thresholds to account for cold water N2 fixation69. These terms are applied against an export:production ratio \((S_{E:P}^D)\) in units of mmol P m−3 day−1. \(P_{exp}^D\) is calculated via:
where,
The Fe half saturation coefficient (\(K_{Fe}^D\)) was kept at 0.3 μmol m−3, 3× that of other phytoplankton, unless otherwise clearly defined as another value in our discussion of the results below. The PO4 half saturation coefficient (\(K_{PO_4}^D\)) was 10−10 unless otherwise clearly defined as another value to emulate N2 fixers efficient utilisation of P25,26. Light was also not considered as a limiting factor. A dependency on light was omitted because of the strong correlation between incident radiation and sea surface temperature70 and its negligible effect on N2 fixation in the Atlantic Ocean71. Finally, the fractional area coverage of sea ice (ico) is included to ensure that no cool-water N2 fixation69 occurs under ice. The remineralisation of N2 fixer export occurs at the same rate as other labile organic matter produced by the general phytoplankton group.
Two processes remove NO3 from the ocean model: water column and sedimentary denitrification. Water column denitrification occurs when O2 concentrations are less than a particular threshold \((R_{lim}^{O_2})\), which is set at 7.5 mmol O2 m−3. We calculate the fraction of organic matter (Porg) that is remineralised by water column denitrification via:
and then apply the appropriate stoichiometric requirements of NO3 to this fraction of Porg:
Following this, the strength of water column denitrification is reduced if the ambient concentration of NO3 is deemed to be limiting. Water column denitrification depletes NO3 towards concentrations between 15 and 40 mmol m−3 in modern suboxic zones36. Without this additional constraint, here defined as rden, NO3 concentrations quickly go to zero in simulated suboxic zones. We calculate rden by prescribing a lower limit at which NO3 can no longer be consumed \((R_{lim}^{NO_3})\), which was set to 30 mmol NO3 m−3:
Sedimentary denitrification was calculated using the paramaterisation of Bohlen et al.72, where the removal of NO3 is dependent on the rain rate of organic carbon to the sediments (Corg) and the ambient concentrations of O2 and NO3.
The α term was 0.08, while the β term was halved compared the original value of Bohlen et al.72 to β = 0.1 in an attempt to increase the deep NO3 inventory. The availability of NO3 for sedimentary denitrification was accounted for according to the equation:
Thus, sedimentary denitrification was relaxed towards zero as NO3 concentrations became low.
If NO3 was limiting, the remaining organic matter was remineralised using O2, so long as the environment was sufficiently oxygenated. The availability of oxygen in the sediments was estimated to be two-thirds of the overlying bottom water concentration, based on observations of transport across the diffusive boundary layer by Gundersen and Jorgensen73. Furthermore, an additional limitation was set for sediments underlying hypoxic waters (O2 < 40 mmol m−3), where aerobic remineralisation was diminished towards zero according to the hyperbolic tangent function:
If both NO3 and O2 were limiting, the remaining organic matter was assumed to be remineralised via sulfate reduction.
Subgrid-scale bathymetry
A large amount of sedimentary remineralisation was not included using these parameterisations because the coarse resolution OGCM enables it to resolve only the largest continental shelves. Many small areas of raised bathymetry in pelagic environments were also unresolved. To address this insufficiency, we coupled a sub-grid scale bathymetry to the course resolution OGCM following the methodology of Somes et al.74 and using the ETOPO5 \(\frac{1}{{12}}^{th}\) of a degree dataset. For each latitude by longitude grid point, we calculated the fraction of area that would be represented by shallower levels in the OGCM if this finer resolution bathymetry were used. At each depth level above the OGCM’s deepest level, the fractional area represented by sediments on the sub-grid scale bathymetry was used to remineralise all forms of organic matter via the sedimentary processes defined above.
Iron cycle
Our simulated Fe cycle involves a prescribed external source via the aeolian deposition of dust35, and an internal control in water masses in contact with the ocean floor. The internal control relaxes Fe concentrations to a set concentration given in the control file, which is set to 0.6 μmol m−3 over a period of 1 year. The iron cycle, therefore, considers an atmospheric source, internal cycling via organic matter, and deep ocean sources and sinks via the sediments.
Simulations
All experiments were simulated for 10,000 years to achieve steady-state solutions of major biogeochemical tracers. Unless clearly defined otherwise, all experiments were run under preindustrial conditions, Mk3Lmild, driven by monthly climatologies of surface conditions over an annual cycle. Surface climatologies required to force the OGCM and OBGCM under Mk3Lmild conditions were generated by a 10,000 year pre-industrial (PI) control run of the flux corrected CSIRO Mk3L v1.2 climate system model in fully coupled mode62.
We forced the OGCM with three sets of additional boundary conditions to generate cold, cool and warm ocean states in addition to Mk3Lmild. The glacial ocean state (Mk3Lcold) was generated by forcing the CSIRO Mk3L climate system model with glacial conditions as simulated in Buchanan et al.38. Warm and mild conditions of GFDLwarm and HadGEMcool, respectively, were provided by the pre-industrial control runs of the GFDL-ESM2G and HadGEM2-CC climate system models from the Climate Model Inter-comparison Project phase 5 (CMIP5) multi-model ensemble75. More thorough physical analyses of these ocean states are contained in Buchanan et al.38 and Buchanan et al.33.
Iron deposition experiments that varied Fe supply to the surface ocean involved altering the field of Mahowald et al.35 with constant factors to achieve 25, 50, 75, 80, 90, 100, 125, 150, 200, 300, and 400% of the modern flux. Higher fluxes representative of the glacial field were undertaken using the dust deposition fields of Lambert et al.5 assuming 3.5% Fe content and 0.4 and 2% solubility, respectively, to achieve 500 and 2500% of the modern Fe supply rate (Supplementary Fig. S2). The glacial dust deposition rate referred to in the main text is the 500% version of the Lambert et al.5 field (Supplementary Fig. 2). These rates of Fe deposition were applied to the Mk3Lmild state and discussed in “A central role for N2 fixers”, while a subset of these Fe deposition experiments, as well as variations in the Fe half-saturation constant for N2 fixers (see Supplementary description of the N cycle), were undertaken in with multiple physical states discussed in “Quantifying CO2 drawdown”.
For those experiments with a freely evolving atmospheric CO2 concentration (within section “Quantifying CO2 drawdown”), we initialised each with the near-equilibrium solution produced by holding atmospheric pCO2 at 280 ppm and with the modern Fe deposition (stars in Fig. 2), such that experiments with modern Fe deposition maintained atmospheric pCO2 near to 280 ppm. Altering Fe deposition then caused changes in air-sea CO2 exchange that altered the atmospheric and oceanic C reservoirs. The atmospheric C reservoir was calculated assuming a constant atmospheric weight of 5.1 × 1021 g and a mean molecular weight of air of 28.97 g mol−1.
All experiments involved a relaxation of deep ocean Fe to values of 0.6 μmol m−3 over a period of 365 days. Areas of connection between the deep and surface ocean, such as the high latitudes and deep upwelling zones, were therefore either non-Fe limited or almost non-Fe limited. This parameterisation rendered the high latitudes insensitive to greater Fe supply, while stratified lower latitudes were sensitive to Fe supply but NO3-limited.
δ 15Norg records
Glacial minus interglacial values of δ15Norg records were calculated by averaging values during the Last Glacial Maximum, defined as between 20 and 26 kya, and the Late Holocene, defined as between 0–5 kya. The early Holocene was ignored due to transient changes in the δ15N records since the deglaciation. The global compilation of δ15Norg was composed of bulk sediment and diatom- and foraminifera-bound measurements, and is available in the supplementary material. A slight correction to simulated δ15Norg was applied to correct for diagenetic effects that increase with depth in the water column. The addition of 0.9 per 1000 metres to the raw, simulated δ15Norg values was applied and substantially improves comparisons between simulated and coretop values34.
Data availability
The model output data that support the findings of this study are available for download from Australia’s National Computing Infrastructure (NCI) at https://researchdata.ands.org.au/marine-nitrogen-fixers-output-v10/1385710 with the identifier https://doi.org/10.25914/5d730c40c2729. Source data underlying Figs. 2 and 3 are provided in the Supplementary information as a source data file. Glacial-interglacial differences in δ15Norg are held in Supplementary Data 1. Code for making Figs. 1–4 is freely available at https://github.com/pearseb/Marine-nitrogen-fixers-paper-python-code.
Code availability
The source code for CSIRO Mk3L-COAL is shared via a repository located at http://svn.tpac.org.au/repos/CSIRO_Mk3L/branches/CSIRO_Mk3L-COAL/. Access to the repository may be obtained by following the instructions at https://www.tpac.org.au/csiro-mk3l-access-request/. Access to the source code is subject to a bespoke license that does not permit commercial usage, but is otherwise unrestricted. An “out-of-the-box” run directory is also available for download with all files required to run the model in the configuration used in this study, although users will need to modify the runscript according to their computing infrastructure. Any queries may be directed to the lead author.
References
Kohfeld, K. E. Role of marine biology in glacial-interglacial CO2 cycles. Science 308, 74–78 (2005).
Martinez-Garcia, A. et al. Iron fertilization of the subantarctic ocean during the last ice age. Science 343, 1347–1350 (2014).
Brunelle, B. G. et al. Glacial/interglacial changes in nutrient supply and stratification in the western subarctic North Pacific since the penultimate glacial maximum. Quat. Sci. Rev. 29, 2579–2590 (2010).
Martin, J. H. Glacial-interglacial CO2 change: the iron hypothesis. Paleoceanography 5, 1–13 (1990).
Lambert, F. et al. Dust fluxes and iron fertilization in Holocene and Last Glacial Maximum climates. Geophys. Res. Lett. 42, 6014–6023 (2015).
Tagliabue, a., Aumont, O. & Bopp, L. The impact of different external sources of iron on the global carbon cycle. Geophy. Research Lett. 41, https://doi.org/10.1002/2013GL059059 (2014).
Muglia, J., Somes, C. J., Nickelsen, L. & Schmittner, A. Combined effects of atmospheric and seafloor iron fluxes to the glacial ocean. Paleoceanography 32, 1204–1218 (2017).
Takahashi, T. et al. Global sea-air CO2 flux based on climatological surface ocean pCO2, and seasonal biological and temperature effects. Deep-Sea Res. Part II: Topical Stud. Oceanogr. 49, 1601–1622 (2002).
Emerson, S., Quay, P., Karl, D. M., Winn, C. & Tupas, L. M. Experimental determination of the organic carbon flux from open-ocean surface waters. Nature 389, 951–954 (1997).
DeVries, T. & Weber, T. The export and fate of organic matter in the ocean: new constraints from combining satellite and oceanographic tracer observations. Glob. Biogeochem. Cycles 31, 535–555 (2017).
Garcia, C. A. et al. Nutrient supply controls particulate elemental concentrations and ratios in the low latitude eastern Indian Ocean. Nat. Commun. 9, 4868 (2018).
Pavia, F. J. et al. Shallow particulate organic carbon regeneration in the South Pacific Ocean. Proc. Natl Acad. Sci. USA 116, 201901863 (2019).
Cavan, E. L., Trimmer, M., Shelley, F. & Sanders, R. Remineralization of particulate organic carbon in an ocean oxygen minimum zone. Nat. Commun. 8, 14847 (2017).
Saito, M. A. et al. Multiple nutrient stresses at intersecting Pacific Ocean biomes detected by protein biomarkers. Science 345, 1173–1177 (2014).
Moore, C. M. et al. Processes and patterns of oceanic nutrient limitation. Nat. Geosci. 6, 701–710 (2013).
Browning, T. J. et al. Nutrient co-limitation at the boundary of an oceanic gyre. Nature 551, 242–246 (2017).
Falkowski, P. G. Evolution of the nitrogen cycle and its influence on the biological sequestration of CO2 in the ocean. Nature 387, 272–275 (1997).
Rubin, M., Berman-Frank, I. & Shaked, Y. Dust-and mineral-iron utilization by the marine dinitrogen-fixer Trichodesmium. Nat. Geosci. 4, 529–534 (2011).
Polyviou, D. et al. Desert dust as a source of iron to the globally important diazotroph Trichodesmium. Front. Microbiol. 8, 1–12 (2018).
Karl, D. et al. The role of nitrogen fixation in biogeochemical cycling in the subtropical North Pacific Ocean. Nature 388, 533–538 (1997).
Karl, D. M., Church, M. J., Dore, J. E., Letelier, R. M. & Mahaffey, C. Predictable and efficient carbon sequestration in the North Pacific Ocean supported by symbiotic nitrogen fixation. Proc. Natl Acad. Sci., USA 109, 1842–1849 (2012).
Shiozaki, T. et al. Linkage between dinitrogen fixation and primary production in the oligotrophic south pacific ocean. Glob. Biogeochem. Cycles 32, 1028–1044 (2018).
Ko, Y. H. et al. Carbon-based estimate of nitrogen fixation-derived net community production in N-depleted ocean gyres. Global Biogeoche. Cy. 32, 1241–1252 (2018).
Wang, W. L., Moore, J. K., Martiny, A. C. & Primeau, F. W. Convergent estimates of marine nitrogen fixation. Nature 566, 205–211 (2019).
Dyhrman, S. T. et al. Phosphonate utilization by the globally important marine diazotroph Trichodesmium. Nature 439, 68–71 (2006).
Landolfi, A., Koeve, W., Dietze, H., Kähler, P. & Oschlies, A. A new perspective on environmental controls of marine nitrogen fixation. Geophys. Res. Lett. 42, 4482–4489 (2015).
White, A. E., Spitz, Y. H., Karl, D. M. & Letelier, R. M. Flexible elemental stoichiometry in Trichodesmium spp. and its ecological implications. Limnol. Oceanogr. 51, 1777–1790 (2006).
Nuester, J., Vogt, S., Newville, M., Kustka, A. B. & Twining, B. S. The unique biogeochemical signature of the marine diazotroph Trichodesmium. Front. Microbiol. 3, 1–15 (2012).
Fu, F. X. et al. Differing responses of marine N2 fixers to warming and consequences for future diazotroph community structure. Aquat. Microb. Ecol. 72, 33–46 (2014).
Moore, K., Doney, S. C., Lindsay, K., Mahowald, N. & Michaels Anthony, F. A. F. Nitrogen fixation amplifies the ocean biogeochemical response to decadal timescale variations in mineral dust deposition. Tellus, Ser. B: Chem. Phys. Meteorol. 58, 560–572 (2006).
Kienast, S. S., Winckler, G., Lippold, J., Albani, S. & Mahowald, N. M. Tracing dust input to the global ocean using thorium isotopes in marine sediments: ThoroMap. Glob. Biogeochem. Cycles 30, 1526–1541 (2016).
Loveley, M. R. et al. Millennial-scale iron fertilization of the eastern equatorial Pacific over the past 100,000 years. Nat. Geosci. 10, 760–764 (2017).
Buchanan, P., Matear, R., Chase, Z., Phipps, S. & Bindoff, N. Dynamic Biological Functioning Important for Simulating and Stabilizing Ocean Biogeochemistry. Global Biogeoche.Cy. 32, 565–593 (2018).
Buchanan, P. J., Matear, R. J., Chase, Z., Phipps, S. J. & Bindoff, N. L. Ocean carbon and nitrogen isotopes in CSIRO Mk3L-COAL version 1.0: a tool for palaeoceanographic research. Geoscientific Model Dev. 12, 1491–1523 (2019).
Mahowald, N. M. et al. Atmospheric global dust cycle and iron inputs to the ocean. Global Biogeoche. Cy. 19, https://doi.org/10.1029/2004GB002402 (2005).
Garcia, H. E. et al. World Ocean Atlas 2013. Vol. 4: Dissolved Inorganic Nutrients (phosphate, nitrate, silicate). (ed. S. Levitus; Technical Ed. A. Mishonov). Tech. Rep (2013).
Sigman, D. M., Hain, M. P. & Haug, G. H. The polar ocean and glacial cycles in atmospheric CO2 concentration. Nature 466, 47–55 (2010).
Buchanan, P. J. et al. The simulated climate of the Last Glacial Maximum and insights into the global marine carbon cycle. Climate 12, 2271–2295 (2016).
Karl, D. M. & Letelier, R. M. Nitrogen fixation-enhanced carbon sequestration in low nitrate, low chlorophyll seascapes. Mar. Ecol. Prog. Ser. 364, 257–268 (2008).
Ren, H. et al. Impact of glacial/interglacial sea level change on the ocean nitrogen cycle. Proc. Natl Acad. Sci. USA 114, E6759–E6766 (2017).
Ganeshram, R. S. et al. Large changes in oceanic nutrient inventories from glacial to interglacial periods. Nature 376, 755–758(1995).
Straub, M. et al. Changes in North Atlantic nitrogen fixation controlled by ocean circulation. Nature 501, 200–203 (2013).
Francois, R. et al. Contribution of Southern Ocean surface-water stratification to low atmospheric CO2 concentrations during the last glacial period. Nature 389, 929–935 (1997).
Studer, A. S. et al. Antarctic Zone nutrient conditions during the last two glacial cycles. Paleoceanography 30, 845–862 (2015).
Muglia, J., Skinner, L. C. & Schmittner, A. Weak overturning circulation and high Southern Ocean nutrient utilization maximized glacial ocean carbon. Earth. Planet. Sci. Lett. 496, 47–56 (2018).
Schmittner, A. & Somes, C. J. Complementary constraints from carbon (13C) and nitrogen (15N) isotopes on the glacial ocean’s soft-tissue biological pump. Paleoceanography 31, 669–693 (2016).
Sarmiento, J. L., Gruber, N., Brzezinski, Ma & Dunne, J. P. High-latitude controls of thermocline nutrients and low latitude biological productivity. Nature 427, 56–60 (2004).
Jacobel, A. W. et al. No evidence for equatorial Pacific dust fertilization. Nat. Geosci. 12, 154–155 (2019).
Marcantonio, F., Loveley, M. R., Schmidt, M. W. & Hertzberg, J. E. Reply to: No evidence for equatorial Pacific dust fertilization. Nat. Geosci. 12, 156–156 (2019).
Christensen, J. P., Murray, J. W., Devol, A. H. & Codispoti, La Denitrification in continental shelf sediments has major impact on the oceanic nitrogen budget. Glob. Biogeochem. Cycles 1, 97 (1987).
Hoogakker, B. A. et al. Glacial expansion of oxygen-depleted seawater in the eastern tropical Pacific. Nature 562, 410–413 (2018).
Anderson, R. F. et al. Deep-sea oxygen depletion and ocean carbon sequestration during the last ice age. Glob. Biogeochem. Cycles 33, 301–317 (2019).
Cartapanis, O., Bianchi, D., Jaccard, S. L. & Galbraith, E. D. Global pulses of organic carbon burial in deep-sea sediments during glacial maxima. Nat. Commun. 7, 1–7 (2016).
DeVries, T., Deutsch, C., Rafter, P. A. & Primeau, F. Marine denitrification rates determined from a global 3-D inverse model. Biogeosciences 10, 2481–2496 (2013).
Costa, K. M. et al. Productivity patterns in the equatorial Pacific over the last 30,000 years. Glob. Biogeochem. Cycles 31, 850–865 (2017).
Galbraith, E. D. & Martiny, A. C. A simple nutrient-dependence mechanism for predicting the stoichiometry of marine ecosystems. Proc. Natl Acad. Sci. USA 112, 201423917 (2015).
Sherwood, O. A., Guilderson, T. P., Batista, F. C., Schiff, J. T. & McCarthy, M. D. Increasing subtropical north Pacific Ocean nitrogen fixation since the Little Ice Age. Nature 505, 78–81 (2014).
McMahon, K. W., McCarthy, M. D., Sherwood, O. A., Larsen, T. & Guilderson, T. P. Millennial-scale plankton regime shifts in the subtropical North Pacific. Ocean. Sci. 350, 1530–1533 (2015).
Stramma, L., Johnson, G. C., Sprintall, J. & Mohrholz, V. Expanding oxygen-minimum zones in the tropical oceans. Science 320, 655–658 (2008).
Schmidtko, S., Stramma, L. & Visbeck, M. Decline in global oceanic oxygen content during the past five decades. Nature 542, 335–339 (2017).
Hutchins, D. A. & Boyd, P. W. Marine phytoplankton and the changing ocean iron cycle. Nat. Clim. Change 6, 1072–1079 (2016).
Phipps, S. J. et al. Paleoclimate data-model comparison and the role of climate forcings over the past 1500 years*. J. Clim. 26, 6915–6936 (2013).
Orr, J. C. et al. Biogeochemical protocols and diagnostics for the CMIP6 Ocean Model Intercomparison Project (OMIP). Geoscientific Model Dev. 10, 2169–2199 (2017).
Lamarque, J. F. et al. Multi-model mean nitrogen and sulfur deposition from the atmospheric chemistry and climate model intercomparison project (ACCMIP): Evaluation of historical and projected future changes. Atmos. Chem. Phys. 13, 7997–8018 (2013).
Kustka, A., Sañudo-Wilhelmy, S., Carpenter, E. J., Capone, D. G. & Raven, Ja A revised estimate of the iron use efficiency of nitrogen fixation, with special reference to the marine cyanobacterium Trichodesmium spp. (Cyanophyta). J. Phycol. 39, 12–25 (2003).
Mills, M. M. & Arrigo, K. R. Magnitude of oceanic nitrogen fixation influenced by the nutrient uptake ratio of phytoplankton. Nat. Geosci. 3, 412–416 (2010).
Paulmier, a, Kriest, I. & Oschlies, A. Stoichiometries of remineralisation and denitrification in global biogeochemical ocean models. Biogeosciences 6, 923–935 (2009).
Kriest, I. & Oschlies, A. MOPS-1.0: towards a model for the regulation of the global oceanic nitrogen budget by marine biogeochemical processes. Geosci. Model Dev. 8, 2929–2927 (2015).
Sipler, R. E. et al. Preliminary estimates of the contribution of Arctic nitrogen fixation to the global nitrogen budget. Limnol Oceanogr. Lett. 2, 159–166 (2017).
Luo, Y. W., Lima, I. D., Karl, D. M., Deutsch, C. A. & Doney, S. C. Data-based assessment of environmental controls on global marine nitrogen fixation. Biogeosciences 11, 691–708 (2014).
McGillicuddy, D. J. Do Trichodesmium spp. populations in the North Atlantic export most of the nitrogen they fix? Glob. Biogeochem. Cycles 28, 103–114 (2014).
Bohlen, L., Dale, A. W. & Wallmann, K. Simple transfer functions for calculating benthic fixed nitrogen losses and C:N:P regeneration ratios in global biogeochemical models. Global Biogeochem.Cy. 26, https://doi.org/10.1029/2011GB004198 (2012).
Gundersen, J. K. & Jorgensen, B. B. Microstructure of diffusive boundary layers and the oxygen uptake of the sea floor. Nature 345, 604–607 (1990).
Somes, C. J., Oschlies, A. & Schmittner, A. Isotopic constraints on the pre-industrial oceanic nitrogen budget. Biogeosciences 10, 5889–5910 (2013).
Taylor, K. E., Stouffer, R. J. & Meehl, G. A. An overview of CMIP5 and experimental design. Bull. Am. Meteorological Soc. 93, 485–498 (2012).
Acknowledgements
We wish to thank the Australian Research Council’s Centre of Excellence for Climate System Science, CSIRO Oceans and Atmosphere and the Tasmanian Partnership for Advanced Computing (TPAC) for facilitating the research. This research was partly supported by the CSIRO Decadal Climate Forecasting Project, the Australian Research Council’s Special Research Initiative for the Antarctic Gateway Partnership (Project ID SR140300001), and through funding from the Australian Governments National Environmental Science Program Earth Systems and Climate Change Hub, which was supported by the National Environmental Science Program. We acknowledge the World Climate Research Programme’s Working Group on Coupled Modelling, which is responsible for CMIP, and we thank the climate modeling groups (GFDL-ESM2G and HadGEM2-CC, listed in Table 1 of Buchanan et al.33) for producing and making available their model output. For CMIP the U.S. Department of Energy’s Program for Climate Model Diagnosis and Intercomparison provides coordinating support and led development of software infrastructure in partnership with the Global Organization for Earth System Science Portals. We thank Toño Gomez for his preparation of Fig. 5 and acknowledge the Australian-American Fulbright Commission for a postgraduate scholarship that allowed the lead author to complete this work.
Author information
Authors and Affiliations
Contributions
P.J.B. developed the biogeochemical model code, designed and executed the experiments, collated the δ15N data, analysed and interpreted the results, and prepared and edited the paper. Z.C. designed the experiments, interpreted the results and wrote the paper. R.J.M. performed key early development of biogeochemical model code, interpreted the results and edited the paper. S.J.P. performed key early development of the climate system model and edited the paper. N.L.B. interpreted the results and edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Jennifer Hertzberg and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Buchanan, P.J., Chase, Z., Matear, R.J. et al. Marine nitrogen fixers mediate a low latitude pathway for atmospheric CO2 drawdown. Nat Commun 10, 4611 (2019). https://doi.org/10.1038/s41467-019-12549-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-019-12549-z
This article is cited by
-
Dynamic diel proteome and daytime nitrogenase activity supports buoyancy in the cyanobacterium Trichodesmium
Nature Microbiology (2022)
-
Ambiguous controls on simulated diazotrophs in the world oceans
Scientific Reports (2022)
-
Deep Equatorial Pacific Ocean Oxygenation and Atmospheric CO2 Over The Last Ice Age
Scientific Reports (2020)
-
A new mechanism of atmospheric CO2 absorption promoted by iron-nitrogen coupling in low-latitude oceans during ice age
Science China Earth Sciences (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.