Abstract
Negative symptoms, such as amotivation and anhedonia, are a major cause of functional impairment in schizophrenia. There are currently no licensed treatments for negative symptoms, highlighting the need to understand the molecular mechanisms underlying them. Mu-opioid receptors (MOR) in the striatum play a key role in hedonic processing and reward function and are reduced post-mortem in schizophrenia. However, it is unknown if mu-opioid receptor availability is altered in-vivo or related to negative symptoms in schizophrenia. Using [11 C]-carfentanil positron emission tomography (PET) scans in 19 schizophrenia patients and 20 age-matched healthy controls, here we show a significantly lower MOR availability in patients with schizophrenia in the striatum (Cohen’s d = 0.7), and the hedonic network. In addition, we report a marked global increase in inter-regional covariance of MOR availability in schizophrenia, largely due to increased cortical-subcortical covariance.
Similar content being viewed by others
Introduction
Schizophrenia affects about one in 100 people and is a leading cause of disease burden1. Patients with schizophrenia commonly experience negative symptoms, which include amotivation, anhedonia and asociality. Moreover, negative symptoms are associated with poor functional outcomes2. Antipsychotics only marginally improve negative symptoms3, and there are no currently licensed treatments specifically for negative symptoms. In view of this, and the marked impact of negative symptoms on recovery, there is considerable interest in identifying potential treatment targets for these symptoms. However, the neurobiological mechanisms underlying negative symptoms remain unclear4.
Impairments in hedonic function are thought to be a key mechanism underlying negative symptoms2, conceptualised as reduced motivation to pursue reward (anticipatory anhedonia) and reduced hedonic response to reward itself (consummatory anhedonia). Studies assessing these features in schizophrenia have reported deficits in either consummatory5, or anticipatory6, or both components of hedonic function7.
Converging lines of evidence indicate that MOR play a central role in hedonic function. Specifically, MOR knock out mouse models show anhedonic phenotypes such as decreased conditioned social place preference8, and reduced nose pokes for sucrose pellets9. Moreover, pharmacological MOR blockade induces conditioned place aversion10 and reduces social novelty preference11. Deficits in both sucrose preference12 and progressive ratio response paradigms9 suggest that mu-opioid system is involved in both the anticipatory and consummatory components of the anhedonic response. In contrast, MOR stimulation increases motivation to seek reward13,14, and increases food palatability15. Consistent with these findings, human studies report blockade of MOR decreases the pleasantness of palatable foods16 and sexual stimuli17, whilst pharmacological stimulation of the opioid system increases the hedonic evaluation and motivation for viewing rewarding images18.
The MOR system also modulates social interaction. Specifically, MOR knockout pups emit fewer ultrasonic vocalisations when removed from their mother, suggesting reduced maternal attachment behaviour19. Human molecular imaging studies using [11C]-carfentanil, a MOR agonist tracer, have shown that MOR activation in the amygdala, insula, and striatum are linked to increased social acceptance and interaction20. In summary, both preclinical and human studies show a potential role of MOR in mediating anhedonia, amotivation and asociality.
Opioid dysregulation in schizophrenia was first suggested in the 1980s21 based on findings of elevated beta-endorphin levels in the cerebrospinal fluid of schizophrenia patients22,23,24 and clinical studies of naloxone, a MOR antagonist treatment showing a reduction of psychotic symptoms in some patients25. Post-mortem studies have found elevated MOR mRNA levels in the frontal cortex of patients26, but lower binding of the MOR selective agonist, [3H]-DAMGO, in the cingulate gyrus and caudate-putamen of schizophrenia patients who died by suicide compared to healthy controls and patients who had died by non-suicide causes27. The inconsistency in the findings of post mortem studies could be due to confounders such as the inclusion of more people who had died by suicide in the patient group given that brain MOR mRNA and protein levels have been found to be altered in suicide victims compared to healthy controls27,28,29.
Despite the preclinical, human studies and evidence from post-mortem and peripheral measures of the potential role of MOR in schizophrenia, there have not been, to our knowledge, any previous PET studies of MOR availability in vivo in schizophrenia. [11C]-carfentanil is a selective MOR tracer with over two orders of magnitude higher affinity for MOR than other receptors [Ki(µ) = 0.024 nM, Ki(δ) = 3.28 nM, Ki(κ) = 43.1nM30], shows excellent reproducibility (variability < 10%, intraclass correlation coefficients >0.93 in test-retest studies)31 and kinetic properties, making it a good tracer to evaluate the MOR in vivo in neuropsychiatric disorders30.
Here we show that patients with schizophrenia have reduced MOR availability in striatum and brain regions implicated in hedonic responses compared to healthy controls. In addition, we report a highly significant global increase in MOR connection strength in schizophrenia patients relative to controls, largely due to increased cortical-subcortical connectivity.
Results
Subject charecteristics
Twenty individuals with schizophrenia and 20 healthy control subjects were studied. One patient had a structural abnormality on the MRI scan and was excluded from further analysis. Demographic details for all participants are given in Table 1. All patients were taking an antipsychotic at the time of the scan (listed in Supplementary Table 1). There was no significant difference between groups in age, sex, radioactive dose and injected mass per body weight (µgm/kg) received. There were more smokers in the patient group compared to healthy control group, and patients smoked significantly more cigarettes per day than controls (number of cigarettes smoked per day (mean ± SEM): patients = 8.2 ± 2.1 vs. controls = 0.9 ± 0.6; p < 0.001).
There were significant group differences in anhedonia and BMI, with higher anhedonia ratings in patients (Table1). There was no significant difference in the rs1799971 genotype frequency between groups. The gene frequency in our sample is consistent with previous studies which reported a A:A genotype frequency of 75%32.
Region of interest analysis
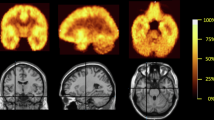
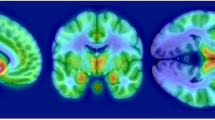
The Region of interest (ROI) analysis showed significantly lower MOR availability in the striatum of patients with schizophrenia relative to controls (patients vs. controls (mean ± SEM): 1.54 ± 0.06 vs. 1.7 ± 0.05, Cohen’s d = 0.7 t = −2.2, df (37), p = 0.037) (Fig. 1). These changes were significant in the dorsal striatum (patients vs. controls (mean ± SEM): 1.35 ± 0.06 vs. 1.53 ± 0.05, p = 0.03), but no significant differences were seen in the ventral striatum (patients vs. controls (mean ± SEM): 2.6 ± 0.08 vs. 2.69 ± 0.07, p = 0.45). There was no correlation between striatal MOR availability and negative symptom severity (Supplementary Fig. 1; PANSS-negative symptom subscale- r: 0.07, p = 0.78, SANS-25 total score- r: −0.151, p = 0.54), or social, physical, anticipatory and consummatory anhedonia measures in patients and controls (all p > 0.05). Similarly, there was no association between dorsal or ventral striatal MOR availability and negative symptom or anhedonia severity (all p > 0.05). Secondary analysis revealed a significant effect of both group (F (5, 222) = 334.5, p < 0.05) and ROI (F (1, 222) = 5.654 p < 0.05) on BPND measures in the hedonic network (Fig. 2). The group × ROI interaction was not significant (F (5, 222) = 0.2167, p > 0.05).
In view of the significant group differences in BMI and smoking, we conducted an exploratory analysis of the relationship between them and MOR. This showed there was no significant correlation between striatal MOR availability and BMI in patients (Pearson r = −0.35, p = 0.1) or controls (Pearson r = −0.33, p = 0.15). There was no association between number of tobacco cigarette smoked per day and MOR availability in striatum (patients: r = −0.047 p = 0.85; controls: r = 0.27, p = 0.25).
The area under the curve of the standardised uptake values for the occipital cortex did not differ between the groups (Supplementary Fig. 2, p > 0.05), consistent with the assumption of no difference in reference tissue MOR signal between the groups. To explore if antipsychotic treatment could influence our MOR findings, we calculated the chlorpromazine equivalent (CPZ equivalent) dose of antipsychotic treatment in all patients using the method described by Leucht et al.33,34, and investigated if there was a relationship between antipsychotic dose and MOR. There was no association between striatal MOR availability and antipsychotic dose (r = 0.06, p = 0.82) (Supplementary Fig. 3). Furthermore, none of the antipsychotics has significant affinity for MOR (see Supplementary Table 2 for affinities).
There was no significant difference in the total grey matter volume between groups (patients vs. control: mean ± SEM (mm3) 1171573 ± 16661 vs. 1217443 ± 21157, respectively, p = 0.1). Moreover, there were no significant differences in the volume of the striatum between groups (patients vs. controls: mean ± SEM (mm3) 13019 ± 302 vs. 12937 ± 327, respectively, p = 0.86). There was a significant difference between groups in the volumes of the regions in the hedonic network (Supplementary Table 3). However, there were no relationships between grey matter volume and MOR binding potential in the striatum or any of the other regions of interest (all r −0.2 to 0.3, p > 0.05).
Mu opioid receptor covariance network analysis
Z-matrices for both patients and controls are shown in Fig. 3. Binding potentials throughout the cortex show moderate to high correlations with binding potentials in other cortical regions in both groups. However, qualitatively, the schizophrenia group shows more regions with high positive correlations relative to healthy controls (see Fig. 3). Quantitatively, we found a highly significant (p < 0.00001) increase in the connection strength of MOR BPND in 1791 out of 7750 edges in schizophrenia patients relative to healthy controls with a threshold of p < 0.05 corrected for multiple comparisons using the network-based statistics algorithm (see Fig. 4a). This represents MOR correlations across the whole brain region, suggesting a global increased connectivity in patients compared to controls. To investigate the most significantly different networks between groups, we applied a more constrained primary threshold of p < 0.001 (corrected for multiple comparison using the network-based statistics algorithm). This identified increased MOR BPND covariation in 99 out of 7750 edges in the schizophrenia patient group compared to controls in a cerebello-thalamo-cortical covariance network (see Fig. 4b).
Matrices showing region-by-region correlations, represented as Z scores, throughout the whole brain (125 ROIs) by group. X and Y axes represent BPND values across the 125 ROIs defined by the Clinical Imaging Centre (CIC) atlas. a shows data for controls. b shows data for patients. The colour bar represents the strength of correlation expressed as a Z-score (red = high positive correlation, blue = high negative correlation)
Regions identified as showing significantly greater covariance in schizophrenia compared to healthy controls. Using a primary threshold of p < 0.05 (false discovery rate corrected), a global increase in connectivity was shown in schizophrenia (left-hand side). A more conservative (p < 0.001 false discovery rate corrected) threshold identified the most significant components to be in a cerebello-thalamo-cortical network (b, right-hand side)
Discussion
MOR availability was reduced in vivo in the striatum with a large effect size (Cohen’s d = 0.7) in schizophrenia patients compared to healthy controls, consistent with our main hypothesis. Our secondary analyses showed significantly reduced mu-opioid receptor availability across other brain regions involved in hedonic processes, comprising the orbitofrontal cortex, cingulate cortex, insular cortex, midbrain, and amygdala, in schizophrenia. Furthermore, the covariance network analysis showed significantly elevated cortical-to-subcortical correlations between mu-opioid receptor levels in schizophrenia patients relative to controls. However, MOR availability was not linked to negative symptom or anhedonia severity ratings.
Our findings extend post-mortem findings of reduced MOR density measured with DAMGO, a MOR selective agonist, by showing reduced MOR availability in vivo and without the potential confound of suicide27. MOR mRNA levels have been reported to be elevated in the frontal cortex of patients with schizophrenia26. Studies in healthy volunteers have shown that MOR levels measured with PET are not closely correlated with mRNA levels, likely reflecting the fact the MOR undergoes post-transcriptional modification35. Thus, our findings taken with evidence of elevated MOR mRNA levels in the frontal cortex of patients26, could suggest there is increased post-transciptional modification and/or internalisation or breakdown of mu-opioid receptors in schizophrenia.
To our knowledge, this is the first study investigating MOR availability in vivo in schizophrenia. In our sample, all subjects were treated with antipsychotics. However, none of the antipsychotics taken by the patients has significant affinity for the MOR (all Ki values > 1000)36,37,38,39. Moreover, it is important to note that non-human primate studies show that neither haloperidol nor olanzapine leads to appreciable alterations in the MOR availability, indicating that antipsychotic treatment does not significantly alter MOR26. Thus, it is unlikely that antipsychotic treatment is a significant confounder. Nevertheless, several preclinical studies have shown that alteration in dopaminergic activity can affect opioidergic neurotransmission40. Future studies in drug naïve subjects are needed to address this potential confounding effect.
We used the SRTM model with the occipital cortex as a reference region to estimate MOR availability. We used lower layers of the occipital cortex as the reference region as this has negligible MOR levels and has been validated by previous studies41,42. In addition, the area under the curve of the standardised uptake values for the occipital cortex, representing non-specific tracer accumulation in this region, did not differ between the groups, indicating that there are no significant group differences in tracer binding in this region.
Previous studies have reported altered MOR availability in morbidly obese subjects with BMI values of 38–4243. In our cohort, patients had a higher BMI (range 17.5–38) compared to controls (range 18.5–35), although not in the morbidly obese range. Moreover, there was no correlation between BMI and striatal MOR availability in our samples, suggesting the BMI difference is unlikely to account for our findings. Nevertheless, it would be useful in future studies to investigate this further.
Studies have shown that alcohol, opioid, stimulant, and tobacco smoking could affect the opioid signaling44,45. However, none of our patients and controls met criteria for present or past substance use disorder, indicating this is unlikely to be a confound for the MOR availability measurements. Although, patients smoked more cigarretes than controls, there was no correlation between MOR availability and a number of tobacco cigarettes smoked, suggesting smoking is not a major confound. However, given that tobacco smoking may affect opioid signalling44, it is possible that group differences in cigarette smoking could have influenced our findings.
There was no significant difference in the total gray matter and striatal volume between patients and controls. Further, there was no association between gray matter volume and BPND, suggesting partial volume effects are unlikely to be a significant confounder. However, there was a difference in the volumes between groups in the regions in the hedonic network, which could indicate that partial volume effects contribute to our findings in these regions. Notwithstanding this, we did not find any significant relationships between gray matter volume in the striatum or any other region of interest, indicating that partial volume effects are unlikely to have had a major effect on our findings.
Previous post-mortem studies have reported no alteration in delta and kappa opioid receptors in schizophrenia26. Combined with the high selectivity of [¹¹C]-carfentanil for the MOR (Ki = 0.0024 nM) over delta opioid (Ki = 3.28 nM) and kappa opioid receptors (Ki = 43.1 nM), our data indicate a lower MOR availability in schizophrenia patients46.
The reduced BPND could be due to receptor down-regulation, or neuronal loss30, whilst our finding of increased cross-correlations between MOR levels across the brain in schizophrenia suggests a global dysregulation. The downregulation of the MOR could be due to long-term overstimulation of MOR by beta-endorphins and Leu-enkephalin, which both have high affinity for MOR (Ki values of 0.3 and 1, respectively)47. This is consistent with the findings of elevated beta-endorphin23 and enkepalin48 levels in CSF from schizophrenia patients, although post mortem studies have not reported alteration in enkephalin mRNA levels in schizophrenia26,49. MOR activation inhibits release of GABA in the cortex50 and, in the striatum, leads to dopamine release51 and is associated with increased dopamine synthesis capacity52. Thus, increased beta-endorphin levels could lead to reduced cortical inhibition, leading to cognitive impairments, and striatal dopamine disinhibition in schizophrenia, leading to psychotic symptoms due to dopamine dysregulation53. However, as we did not measure beta-endorphin levels or dopamine function, it needs to be determined if lower MOR availability in the striatum in schizophrenia is due to elevated beta-endorphin levels or linked to dopamine dysfunction. [11C]-carfentanil is sensitive to endogenous opioid levels, which compete with the tracer to reduce its binding to the MOR42,54. Thus, the reduction in MOR BPND could be due to either reduced receptor availability or increased endogenous opioid release, or a combination of both. Future studies, using pharmacological challenges that release endogenous opioids, such as amphetamine, would be useful to determine if the lower levels of [11C]-carfentanil binding we observed are due to altered endogenous opioid release or reduced MOR levels41,42.
It is surprising that there was no significant association between MOR availability and either total negative symptoms or anhedonia severity (social, physical, anticipatory and consummatory anhedonia), given the lower MOR availability in patients. Our secondary analyses found that MOR availability was significantly lower in the dorsal but not ventral striatum in patients relative to controls, suggesting that the finding of lower striatal MOR in patients was driven by differences in the dorsal striatum. Striatal MOR blockade reduces the motivation to seek food13 and sexual pleasure in animals55. In addition, there is some evidence that this particularly involves the dorsal striatum, including findings that endogenous opioids released in the dorsal striatum during food consumption are associated with motivation to eat but not with the hedonic orofacial response to food13 and blockade of MOR in the dorsal striatum abolished formation of partner preference without evoking partner aversion55. In contrast, ventral striatal MOR blockade has generally been linked to anhedonia56. Thus, these findings indicate that our results of lower dorsal striatal MOR availability may contribute to the amotivation rather than anhedonic component of negative symptoms in schizophrenia, and the lack of major differences in ventral striatal MOR availability could indicate that another mechanism underlies anhedonia seen in schizophrenia. However, as we did not measure motivation, the association between dorsal striatal MOR and amotivation requires testing in patients. Alternatively, the lack of association between negative symptoms and MOR could indicate they are linked to other neurotransmitter dysfunction rather than MOR2,57,58, or, given that our patients had been ill for a number of years, that MOR alterations underlie the development of negative symptoms but not their maintenance in chronic patients.
The mean PANSS in our cohort was 60 and the mean PANSS-negative symptom score in our cohort was 21, and the highest was 30 (total possible score = 49). The total severity rating is lower than typically reported in studies of acute relapses, but is consistent with recent randomised control trials of treatments for negative symptoms, where mean PANSS total scores were 47–80 and mean negative scores were 17–2259,60,61. Consistent with these studies and recommendations for studies of negative symptoms, we recruited subjects with predominant negative symptom without acute positive symptoms (no more than PANSS positive subscale score of 4) as these can confound the assessment of negative symptoms62. Thus, our study, in common with others in the literature59,60,61, largely recruited patients with moderate symptom severity, which could affect generalisability to patients with more severe symptoms.
It has been suggested that negative symptoms can be considered as primary, that is intrinsic to the disease process, or secondary to other factors such as acute psychosis, comorbid depression, antipsychotic effects or other factors63. We excluded marked psychosis and comorbid depression in our sample, indicating that negative symptoms are unlikely to be due to these factors. We also applied the deficit schizophrenia criteria, which are used to identify patients with schizophrenia showing primary and enduring negative symptoms64. This suggests the negative symptoms in our patient sample are likely to be predominantly primary in nature. However, we can not exclude some contribution from secondary factors in the negative symptoms in our patients, which, if these have alternative mechanisms, may have reduced our ability to detect a relationship between MOR and negative symptoms. Future longitudinal studies in early course, untreated patients would be helpful to confirm our findings in patients at the onset of negative symptoms.
The application of the network-based statistical analysis to MOR is, to our knowledge, novel in both healthy volunteers and schizophrenia. Nevertheless, we applied an approach that is well-established in the fMRI literature and has been applied to PET studies of other receptors65,66,67, a conservative p-value threshold (p < 0.001) and false discovery rate correction to control the type-1 error rate. This analysis found stronger [11C]-carfentanil BPND correlations across cerebello-thalamo-cortical regions in patients compared to healthy control. There are two main mechanisms that could account for this. One is it could be a consequence of increased CSF beta-endorphin levels in schizophrenia23. Beta-endorphin synthesised in pro-opiomelanocortin (POMC) neurons in the hypothalamus diffuses through cerebrospinal fluid to act on MOR throughout the brain by volume transmission68. Thus, given that [11C]-carfentanil is displaced by endogenous MOR ligands, increased brain beta-endorphin levels in schizophrenia could lead to reduced variability of MOR, as is seen with exogenous opiate block, which will increase inter-regional correlations as the range of possible BPND values is lower69. Alternatively, the higher inter-regional correlations could be due to altered genetic regulation of MOR expression. Supporting this, a preclinical study has shown that deletion of the mu opioid receptor gene (Oprm1) results in disrupted whole brain resting state functional connectivity70. Notwithstanding this evidence, it is important to recognise that the physiological significance of the increased MOR covariance network in schizophrenia remains to be determined. Combined MOR and functional imaging studies in patients and healthy volunteers would be useful to test this.
In conclusion, mu opioid receptor availability is reduced in the striatum and other brain regions involved in hedonic processes and shows increased cortical-subcortical correlations in schizophrenia. However, it is not associated with negative symptom severity or anhedonia measures. These findings provide in vivo evidence for altered brain opioid signaling in schizophrenia.
Methods
Subject recruitment
The study protocol was approved by London-Camberwell St Giles Research Ethics Committee and approval to administer radioactive material was granted by Administration of Radioactive Substances Advisory Committee (ARSAC, UK). All participants provided written informed consent to participate after receiving a description of the study.
We recruited 20 patients with schizophrenia from secondary mental health services. All patients met DSM-IV criteria for schizophrenia. They were required to have a minimum score ≥4 on atleast one domain of positive and negative symptom scale (PANSS) negative symptom sub-scale71 OR two or more negative symptoms with a score ≥3 on the PANSS-negative symptom sub-scale to ensure current negative symptoms. All patients were required to meet criteria for deficit syndrome defined, in accordance with guidelines64,72, as (a) the presence of at least two out of six of the following negative symptoms: restricted affect (referring to observed behaviours rather than to the patient’s subjective experience); diminished emotional range (i.e., reduced range of the patient’s subjective emotional experience); poverty of speech; curbing of interests; diminished sense of purpose; diminished social drive; and (b) a combination of two or more of the above symptoms have been present for the preceding 12 months and were always present during periods of clinical stability; and (c) the above symptoms are not secondary to other factors, including anxiety, drug effects, psychotic symptoms, mental retardation, or depression; and (d) the patient meets DSM criteria for schizophrenia. The secondary factors associated with negative symptoms were excluded based on clinical interview. All patients were required to be on a stable dose of an antipsychotic for at least four weeks before the scan (see Supplementary Table 1 for a list of treatments).
Twenty healthy volunteers were recruited from the same local catchment area through public advertisement. Inclusion criteria included no psychiatric morbidity as assessed by the Structured Clinical Interview for DSM IV (SCID), and no family history of psychosis. Exclusion criteria for all subjects were: history or current substance use disorder (other than to tobacco) as assessed by clinical interview, history of head injury or neurological abnormality, present or recent (1 month) use of opiates, antidepressants or other psychoactive medications including antiepileptics, or significant physical comorbidity (minor self-limiting illnesses were permitted) as assessed by history and physical examination and contraindications to PET or MRI scanning.
Subjects underwent a screening assessment, which included medical and psychiatric history as well as the history of alcohol, tobacco and other substance use and a physical examination. A urine drug screen was carried out on the scan day to exclude psychoactive drug use. Negative symptom severity was assessed using the scale for the Assessment of Negative Symptoms (SANS)73. In addition, the physical and social anhedonia rating scale74, and temporal experience of pleasure scale (TEPS) were administered to the participants to assess anhedonia. The Calgary depression scale was used to assess depression75.
Genotyping
Previous studies have shown that carriers of the OPRM1 G allele (rs1799971) show reduced [11C]-carfentanil binding44,76. In view of this, venous blood samples were taken for genotyping for the OPRM1 A118G polymorphism and were analysed by LGC Limited (Middlesex, UK). DNA was extracted, normalised and underwent SNP-specific KASPTM assay mix. Loci with a call rate < 90% were not included. Subjects were categorised as a G-allele carrier (G: A/ G: G) or not (A: A).
Structural MRI acquisition
High resolution T1 weighted volumes were acquired using a 3T MR scanner (Magneton Trio Syngo MR B13 Siemens 3T; Siemens AG, Germany) and a magnetisation prepared rapid gradient echo (MPRAGE) sequence (TR = 2300 ms, TE = 2.98 ms, TI = 900 ms, flip angle = 9°, field of view = 256 mm, image matrix = 240 × 256) with a resolution of 1 mm isotropic. For the volume, 160 abutting straight sagittal slices were collected in an interleaved right to left manner, resulting in whole head coverage. Parallel imaging using Generalized Auto calibrating Partially Parallel Acquisition (GRAPPA) with an acceleration factor of 2 was performed.
PET acquisition
[11C]-carfentanil, a selective MOR agonist, was synthesised by labelling its des-methyl precursor (4-Piperidinecarboxylic acid, 4-[(1-oxopropyl)phenylamino]-1-(2-phenylethyl), sodium salt; ABX Advanced Biochemical Compounds, Radeberg, Germany/ Pharmasynth, Tartu, Estonia), with carbon-11 using a modification of a previously described method77 on a semi-automated Modular Lab Multifunctional Synthetic Module (Eckert & Ziegler, Berlin, Germany). The final product was reformulated in sterile 0.9% saline containing ∼10% ethanol (v/v) and satisfied quality control criteria for specific activity and purity before being injected intravenously as a slow bolus over ∼20 s. Following a transmission CT scan, a maximum of 300MBq of [¹¹C]-carfentanil was administered. PET emission data were collected for 90 min in 26 frames (8 × 15 s, 3 × 60 s, 5 × 120 s, 5 × 300 s and 5 × 600 s, to a total of 5400 s). PET scans were acquired on a Siemens HiRez 6 PET/computed tomography scanner (Siemens Healthcare, Erlangen, Germany).
PET image analysis
Image pre-processing and PET modeling were carried out using MIAKAT™ software (www.miakat.org). Dynamic PET data were corrected for attenuation and scatter, and for motion by frame-by-frame realignment to frame 16. Each scan was rigid-body coregistered to the structural MRI. ROIs were defined using a neuroanatomical atlas78, applied to the PET image by non-linear deformation parameters derived using unified segmentation of the structural MRI using statistical parametric mapping software (SPM 12). The template and atlas fits were confirmed visually for each participant. [11C]-carfentanil binding potential (BPND) values were quantified using the simplified reference tissue model (SRTM) with occipital lobe grey matter as the reference41,79. This approach shows good agreement on comparison with the arterial input function derived volume of distrbution31,46, and the occipital cortex has negligible MOR availability42,80,81,82,83.
We focussed on striatum as our primary region of interest because it plays a key role in the hedonic response, is rich in MOR84, and studies in schizophrenia show striatal hypoactivation in response to reward85, and reduced striatal MOR ligand binding post-mortem27. In addition, studies have shown that a hedonic network, comprising the orbitofrontal cortex, cingulate cortex, insular cortex, midbrain, and amygdala as well as the striatum, is activated in response to hedonic stimuli86, including to social reward20,87. Moreover, a meta-analysis of fMRI studies which focussed on neural correlates of anhedonia identified hypo-activation of the cingulate cortex, orbitofrontal cortex, as well as the striatum, during reward tasks in schizophrenia relative to controls88. In view of this, we conducted a secondary analysis to test the hypothesis that MOR availability was reduced in schizophrenia in the hedonic network consisting of the orbitofrontal cortex, cingulate cortex, insular cortex, midbrain, and amygdala.
To assess if [11C]-carfentanil uptake in the reference tissue differed between groups, and thus affected global BPND calculations, standardised uptake values (SUV) were calculated by dividing the tissue radioligand uptake (calculated using the 10–90 min summed image) by injected dose per body weight.
The gray matter volume of the whole brain and the volumes of the ROIs were obtained from the individual’s structural MRI scans after tissue segmentation as follows. The CIC atlas was non-linearly warped into subject space, and normalised to the subject’s T1 weighted MRI images using SPM12 (SPM; https://www.fil.ion.ucl.ac.uk/spm/software/spm12/). After tissue segmentation also using SPM12, the grey matter volume for each ROI in the CIC atlas was then extracted as the volume of each ROI weighted by the grey matter probability.
MOR covariance analysis
In addition to regional alterations, it remains unknown if global brain MOR organisation is altered in schizophrenia. To assess this, we performed a correlation analysis for the whole brain and compared this for the two groups89. Within each group, the correlation coefficients between [11C]-carfentanil BPND at each ROI with all other ROIs (125 ROIs defined in the Clinical Imaging Centre atlas78) were calculated and z values derived using the Fisher z-transformation to derive a global correlation matrix. This matrix was considered as a covariance network, where nodes are the ROIs and interregional correlations are the edges. The matrix consisted of 7750 edges. To determine the difference in the strength of connectivity of the edges between patient and control groups, each edge within these networks was compared between groups using permutation testing in MATLAB (100,000 permutations of group labels). To correct for the large number of partially dependent comparisons, the network-based statistic was used, with a primary threshold of alpha = 0.0590. To identify primary contributors to the effect between groups, we conducted a further analysis using a more conservative threshold, alpha = 0.001, consistent with the approach used in previous imaging studies using network-based stastistics35,89.
Statistical analysis
Statistical analysis was performed with SPSS (version 20) for MAC OS X and Graph pad prism version 7.04. Normality of distribution was tested using the Shapiro-Wilk test. The main hypothesis that there was a group difference in the MOR availability in the striatum was tested using an independent sample t-test. To determine whether there was an effect of group on BPND values of the hedonic network, we performed two way-ANOVA with BPND as the dependent variable and group (patient or control) as the independent variable. We examined correlations between PET and clinical data using Pearson’s r. All data are presented as mean ± SEM, and the level α was set for all comparisons at P < 0.05.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data and code used in the production of this manuscript are available on request.
References
Howes, O. D. & Murray, R. M. Schizophrenia: an integrated sociodevelopmental-cognitive model. Lancet (Lond., Engl.) 383, 1677–1687 (2014).
Galderisi, S., Mucci, A., Buchanan, R. W. & Arango, C. Negative symptoms of schizophrenia: new developments and unanswered research questions. lancet Psychiatry 5, 664–677 (2018).
Leucht, S. et al. Sixty years of placebo-controlled antipsychotic drug trials in acute schizophrenia: systematic review, bayesian meta-analysis, and meta-regression of efficacy predictors. Am. J. Psychiatry 174, 927–942 (2017).
Remington, G. et al. Treating negative symptoms in schizophrenia: an update. Curr. Treat. Options Psychiatry 3, 133–150 (2016).
Strauss, G. P., Wilbur, R. C., Warren, K. R., August, S. M. & Gold, J. M. Anticipatory vs. consummatory pleasure: what is the nature of hedonic deficits in schizophrenia? Psychiatry Res. 187, 36–41 (2011).
Gard, D. E., Kring, A. M., Gard, M. G., Horan, W. P. & Green, M. F. Anhedonia in schizophrenia: distinctions between anticipatory and consummatory pleasure. Schizophrenia Res. 93, 253–260 (2007).
Mann, C. L., Footer, O., Chung, Y. S., Driscoll, L. L. & Barch, D. M. Spared and impaired aspects of motivated cognitive control in schizophrenia. J. Abnorm. Psychol. 122, 745–755 (2013).
Cinque, C. et al. Modeling socially anhedonic syndromes: genetic and pharmacological manipulation of opioid neurotransmission in mice. Transl. Psychiatry 2, e155 (2012).
Papaleo, F., Kieffer, B. L., Tabarin, A. & Contarino, A. Decreased motivation to eat in mu-opioid receptor-deficient mice. Eur. J. Neurosci. 25, 3398–3405 (2007).
Skoubis, P. D. & Maidment, N. T. Blockade of ventral pallidal opioid receptors induces a conditioned place aversion and attenuates acquisition of cocaine place preference in the rat. Neuroscience 119, 241–249 (2003).
Smith, C. J. W., Wilkins, K. B., Li, S., Tulimieri, M. T. & Veenema, A. H. Nucleus accumbens mu opioid receptors regulate context-specific social preferences in the juvenile rat. Psychoneuroendocrinology 89, 59–68 (2018).
Ostlund, S. B., Kosheleff, A., Maidment, N. T. & Murphy, N. P. Decreased consumption of sweet fluids in mu opioid receptor knockout mice: a microstructural analysis of licking behavior. Psychopharmacology 229, 105–113 (2013).
DiFeliceantonio, A. G., Mabrouk, O. S., Kennedy, R. T. & Berridge, K. C. Enkephalin surges in dorsal neostriatum as a signal to eat. Curr. Biol.: CB 22, 1918–1924 (2012).
Smith, K. S. & Berridge, K. C. Opioid limbic circuit for reward: interaction between hedonic hotspots of nucleus accumbens and ventral pallidum. J. Neurosci.: Off. J. Soc. Neurosci. 27, 1594–1605 (2007).
Wassum, K. M., Ostlund, S. B., Maidment, N. T. & Balleine, B. W. Distinct opioid circuits determine the palatability and the desirability of rewarding events. Proc. Natl Acad. Sci. USA 106, 12512–12517 (2009).
Barbano, M. F. & Cador, M. Differential regulation of the consummatory, motivational and anticipatory aspects of feeding behavior by dopaminergic and opioidergic drugs. Neuropsychopharmacol.: Off. Publ. Am. Coll. Neuropsychopharmacol. 31, 1371–1381 (2006).
Murphy, M. R., Checkley, S. A., Seckl, J. R. & Lightman, S. L. Naloxone inhibits oxytocin release at orgasm in man. J. Clin. Endocrinol. Metab. 71, 1056–1058 (1990).
Chelnokova, O. et al. Rewards of beauty: the opioid system mediates social motivation in humans. Mol. Psychiatry 19, 746–747 (2014).
Moles, A., Kieffer, B. L. & D’Amato, F. R. Deficit in attachment behavior in mice lacking the mu-opioid receptor gene. Science 304, 1983–1986 (2004).
Hsu, D. T. et al. It still hurts: altered endogenous opioid activity in the brain during social rejection and acceptance in major depressive disorder. Mol. Psychiatry 20, 193–200 (2015).
Schmauss, C. & Emrich, H. M. Dopamine and the action of opiates: a reevaluation of the dopamine hypothesis of schizophrenia. With special consideration of the role of endogenous opioids in the pathogenesis of schizophrenia. Biol. Psychiatry 20, 1211–1231 (1985).
Lindstrom, L. H., Besev, G., Gunne, L. M. & Terenius, L. CSF levels of receptor-active endorphins in schizophrenic patients: correlations with symptomatology and monoamine metabolites. Psychiatry Res. 19, 93–100 (1986).
Brambilla, F., Facchinetti, F., Petraglia, F., Vanzulli, L. & Genazzani, A. R. Secretion pattern of endogenous opioids in chronic schizophrenia. Am. J. Psychiatry 141, 1183–1189 (1984).
Wiegant, V. M., Verhoef, C. J., Burbach, J. P. & de Wied, D. Increased concentration of alpha- and gamma-endorphin in post mortem hypothalamic tissue of schizophrenic patients. Life Sci. 42, 1733–1742 (1988).
Gunne, L. M., Lindstrom, L. & Terenius, L. Naloxone-induced reversal of schizophrenic hallucinations. J. Neural Transm. 40, 13–19 (1977).
Volk, D. W., Radchenkova, P. V., Walker, E. M., Sengupta, E. J. & Lewis, D. A. Cortical opioid markers in schizophrenia and across postnatal development. Cereb. Cortex (New Y., NY: 1991) 22, 1215–1223 (2012).
Scarr, E., Money, T. T., Pavey, G., Neo, J. & Dean, B. Mu opioid receptor availability in people with psychiatric disorders who died by suicide: a case control study. BMC Psychiatry 12, 126 (2012).
Gabilondo, A. M., Meana, J. J. & Garcia-Sevilla, J. A. Increased density of mu-opioid receptors in the postmortem brain of suicide victims. Brain Res. 682, 245–250 (1995).
Gross-Isseroff, R., Dillon, K. A., Israeli, M. & Biegon, A. Regionally selective increases in mu opioid receptor density in the brains of suicide victims. Brain Res. 530, 312–316 (1990).
Henriksen, G. & Willoch, F. Imaging of opioid receptors in the central nervous system. Brain: A J. Neurol. 131, 1171–1196 (2008).
Hirvonen, J. et al. Measurement of central mu-opioid receptor binding in vivo with PET and [11C]carfentanil: a test-retest study in healthy subjects. Eur. J. Nucl. Med. Mol. imaging 36, 275–286 (2009).
Sery, O., Prikryl, R., Castulik, L. & St’astny, F. A118G polymorphism of OPRM1 gene is associated with schizophrenia. J. Mol. Neurosci.: MN 41, 219–222 (2010).
Woods, S. W. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J. Clin. Psychiatry 64, 663–667 (2003).
Leucht, S., Samara, M., Heres, S. & Davis, J. M. Dose equivalents for antipsychotic drugs: the DDD method. Schizophrenia Bull. 42, S90–S94 (2016).
Berridge, K. C., Robinson, T. E. & Aldridge, J. W. Dissecting components of reward: ‘liking’, ‘wanting’, and learning. Curr. Opin. Pharmacol. 9, 65–73 (2009).
Abbas, A. I. et al. Amisulpride is a potent 5-HT7 antagonist: relevance for antidepressant actions in vivo. Psychopharmacology 205, 119–128 (2009).
Kalkman, H. O., Subramanian, N. & Hoyer, D. Extended radioligand binding profile of iloperidone: a broad spectrum dopamine/serotonin/norepinephrine receptor antagonist for the management of psychotic disorders. Neuropsychopharmacol.: Off. Publ. Am. Coll. Neuropsychopharmacol. 25, 904–914 (2001).
Schotte, A. et al. Risperidone compared with new and reference antipsychotic drugs: in vitro and in vivo receptor binding. Psychopharmacology 124, 57–73 (1996).
Shapiro, D. A. et al. Aripiprazole, a novel atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacol.: Off. Publ. Am. Coll. Neuropsychopharmacol. 28, 1400–1411 (2003).
Chen, J. F., Aloyo, V. J. & Weiss, B. Continuous treatment with the D2 dopamine receptor agonist quinpirole decreases D2 dopamine receptors, D2 dopamine receptor messenger RNA and proenkephalin messenger RNA, and increases mu opioid receptors in mouse striatum. Neuroscience 54, 669–680 (1993).
Colasanti, A. et al. Endogenous opioid release in the human brain reward system induced by acute amphetamine administration. Biol. Psychiatry 72, 371–377 (2012).
Mick, I. et al. Amphetamine induced endogenous opioid release in the human brain detected with [(1)(1)C]carfentanil PET: replication in an independent cohort. Int. J. Neuropsychopharmacol. 17, 2069–2074 (2014).
Karlsson, H. K. et al. Obesity is associated with decreased mu-opioid but unaltered dopamine D2 receptor availability in the brain. J. Neurosci.: Off. J. Soc. Neurosci. 35, 3959–3965 (2015).
Ray, R. et al. Human Mu Opioid Receptor (OPRM1 A118G) polymorphism is associated with brain mu-opioid receptor binding potential in smokers. Proc. Natl Acad. Sci. USA 108, 9268–9273 (2011).
Hermann, D. et al. Low mu-opioid receptor status in alcohol dependence identified by combined positron emission tomography and post-mortem brain analysis. Neuropsychopharmacol.: Off. Publ. Am. Coll. Neuropsychopharmacol. 42, 606–614 (2017).
Frost, J. J. et al. Imaging opiate receptors in the human brain by positron tomography. J. Computer Assist. Tomogr. 9, 231–236 (1985).
Raynor, K. et al. Pharmacological characterization of the cloned kappa-, delta-, and mu-opioid receptors. Mol. Pharmacol. 45, 330–334 (1994).
Iadarola, M. J. et al. Met5-enkephalin-Arg6-Gly7-Leu8 immunoreactivity in rat and human cerebrospinal fluid: influence of neuroleptic drugs and electroconvulsive shock. Brain Res. 474, 75–85 (1988).
Harrington, K. A., Augood, S. J., Faull, R. L., McKenna, P. J. & Emson, P. C. Dopamine D1 receptor, D2 receptor, proenkephalin A and substance P gene expression in the caudate nucleus of control and schizophrenic tissue: a quantitative cellular in situ hybridisation study. Brain Res. Mol. Brain Res. 33, 333–342 (1995).
Lupica, C. R. Delta and mu enkephalins inhibit spontaneous GABA-mediated IPSCs via a cyclic AMP-independent mechanism in the rat hippocampus. J. Neurosci.: Off. J. Soc. Neurosci. 15, 737–749 (1995).
Spreckelmeyer, K. N. et al. Opiate-induced dopamine release is modulated by severity of alcohol dependence: an [(18)F]fallypride positron emission tomography study. Biol. Psychiatry 70, 770–776 (2011).
Majuri, J., Joutsa, J., Arponen, E., Forsback, S. & Kaasinen, V. Dopamine synthesis capacity correlates with micro-opioid receptor availability in the human basal ganglia: a triple-tracer PET study. NeuroImage 183, 1–6 (2018).
Howes, O., McCutcheon, R. & Stone, J. Glutamate and dopamine in schizophrenia: an update for the 21st century. J. Psychopharmacol. (Oxf., Engl.) 29, 97–115 (2015).
Quelch, D. R., Katsouri, L., Nutt, D. J., Parker, C. A. & Tyacke, R. J. Imaging endogenous opioid peptide release with [11C]carfentanil and [3H]diprenorphine: influence of agonist-induced internalization. J. Cereb. Blood Flow. Metab.: Off. J. Int. Soc. Cereb. Blood Flow. Metab. 34, 1604–1612 (2014).
Resendez, S. L. et al. mu-Opioid receptors within subregions of the striatum mediate pair bond formation through parallel yet distinct reward mechanisms. J. Neurosci.: Off. J. Soc. Neurosci. 33, 9140–9149 (2013).
Ward, H. G., Nicklous, D. M., Aloyo, V. J. & Simansky, K. J. Mu-opioid receptor cellular function in the nucleus accumbens is essential for hedonically driven eating. Eur. J. Neurosci. 23, 1605–1613 (2006).
Howes, O. D., McCutcheon, R., Owen, M. J. & Murray, R. M. The Role of genes, stress, and dopamine in the development of schizophrenia. Biol. psychiatry 81, 9–20 (2017).
Galderisi, S., Merlotti, E. & Mucci, A. Neurobiological background of negative symptoms. Eur. Arch. psychiatry Clin. Neurosci. 265, 543–558 (2015).
Bugarski-Kirola, D. et al. Bitopertin in negative symptoms of schizophrenia-results from the phase III FlashLyte and DayLyte studies. Biol. Psychiatry 82, 8–16 (2017).
Chaudhry, I. B. et al. Minocycline benefits negative symptoms in early schizophrenia: a randomised double-blind placebo-controlled clinical trial in patients on standard treatment. J. Psychopharmacol. (Oxf., Engl.) 26, 1185–1193 (2012).
Deakin, B. et al. The benefit of minocycline on negative symptoms of schizophrenia in patients with recent-onset psychosis (BeneMin): a randomised, double-blind, placebo-controlled trial. The Lancet. Psychiatry 5, 885–894 (2018).
Kane, J., Honigfeld, G., Singer, J. & Meltzer, H. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch. Gen. Psychiatry 45, 789–796 (1988).
Kirschner, M., Aleman, A. & Kaiser, S. Secondary negative symptoms - A review of mechanisms, assessment and treatment. Schizophrenia Res. 186, 29–38 (2017).
Carpenter, W. T. Jr., Heinrichs, D. W. & Wagman, A. M. Deficit and nondeficit forms of schizophrenia: the concept. Am. J. Psychiatry 145, 578–583 (1988).
Cervenka, S., Varrone, A., Fransen, E., Halldin, C. & Farde, L. PET studies of D2-receptor binding in striatal and extrastriatal brain regions: Biochemical support in vivo for separate dopaminergic systems in humans. Synap. (New Y., NY) 64, 478–485 (2010).
Erritzoe, D. et al. A nonlinear relationship between cerebral serotonin transporter and 5-HT(2A) receptor binding: an in vivo molecular imaging study in humans. J. Neurosci.: Off. J. Soc. Neurosci. 30, 3391–3397 (2010).
Tuominen, L., Nummenmaa, L., Keltikangas-Jarvinen, L., Raitakari, O. & Hietala, J. Mapping neurotransmitter networks with PET: an example on serotonin and opioid systems. Hum. Brain Mapp. 35, 1875–1884 (2014).
Veening, J. G., Gerrits, P. O. & Barendregt, H. P. Volume transmission of beta-endorphin via the cerebrospinal fluid; a review. Fluids Barriers CNS 9, 16 (2012).
Weerts, E. M. et al. Differences in delta- and mu-opioid receptor blockade measured by positron emission tomography in naltrexone-treated recently abstinent alcohol-dependent subjects. Neuropsychopharmacol.: Off. Publ. Am. Coll. Neuropsychopharmacol. 33, 653–665 (2008).
Mechling, A. E. et al. Deletion of the mu opioid receptor gene in mice reshapes the reward-aversion connectome. Proc. Natl Acad. Sci. USA 113, 11603–11608 (2016).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophrenia Bull. 13, 261–276 (1987).
Kirkpatrick, B., Buchanan, R. W., McKenney, P. D., Alphs, L. D. & Carpenter, W. T. Jr. The schedule for the deficit syndrome: an instrument for research in schizophrenia. Psychiatry Res. 30, 119–123 (1989).
Andreasen, N. C. Negative symptoms in schizophrenia. Definition and reliability. Arch. Gen. Psychiatry 39, 784–788 (1982).
Eckblad M. C. L., et al. The revised social anhedonia scale. (University of Wisconsin, Madison, 1982) (Unpublished test)
Addington, D., Addington, J. & Schissel, B. A depression rating scale for schizophrenics. Schizophrenia Res. 3, 247–251 (1990).
Weerts, E. M. et al. Influence of OPRM1 Asn40Asp variant (A118G) on [11C]carfentanil binding potential: preliminary findings in human subjects. Int. J. Neuropsychopharmacol. 16, 47–53 (2013).
Jewett, D. M. A simple synthesis of [11C]carfentanil using an extraction disk instead of HPLC. Nucl. Med. Biol. 28, 733–734 (2001).
Tziortzi, A. C. et al. Imaging dopamine receptors in humans with [11C]-(+)-PHNO: dissection of D3 signal and anatomy. NeuroImage 54, 264–277 (2011).
Lammertsma, A. A. & Hume, S. P. Simplified reference tissue model for PET receptor studies. NeuroImage 4, 153–158 (1996).
Hiller, J. M. & Fan, L. Q. Laminar distribution of the multiple opioid receptors in the human cerebral cortex. Neurochem. Res. 21, 1333–1345 (1996).
Mick, I. et al. Blunted endogenous opioid release following an oral amphetamine challenge in pathological gamblers. Neuropsychopharmacology 41, 1742–1750 (2016).
Rabiner, E. A. et al. Pharmacological differentiation of opioid receptor antagonists by molecular and functional imaging of target occupancy and food reward-related brain activation in humans. Mol. Psychiatry 16, 826–835 (2011).
Turton S., et al. Blunted endogenous opioid release following an oral dexamphetamine challenge in abstinent alcohol-dependent individuals. Mol. Psychiatry (2018). https://doi.org/10.1038/s41380-018-0107-4. [Epub ahead of print].
Le Merrer, J., Becker, J. A., Befort, K. & Kieffer, B. L. Reward processing by the opioid system in the brain. Physiological Rev. 89, 1379–1412 (2009).
Radua, J. et al. Ventral striatal activation during reward processing in psychosis: a neurofunctional meta-analysis. JAMA Psychiatry 72, 1243–1251 (2015).
Berridge, K. C. & Kringelbach, M. L. Pleasure systems in the brain. Neuron 86, 646–664 (2015).
Hsu, D. T. et al. Response of the mu-opioid system to social rejection and acceptance. Mol. Psychiatry 18, 1211–1217 (2013).
Zhang, B. et al. Mapping anhedonia-specific dysfunction in a transdiagnostic approach: an ALE meta-analysis. Brain Imaging Behav. 10, 920–939 (2016).
Veronese, M. et al. Covariance statistics and network analysis of brain PET imaging studies. Sci. Rep. 9, 2496 (2019).
Zalesky, A., Fornito, A. & Bullmore, E. T. Network-based statistic: identifying differences in brain networks. NeuroImage 53, 1197–1207 (2010).
Acknowledgements
This study was funded by grants MC-A656-5QD30 from the Medical Research Council-UK, 666 from the Maudsley Charity, 094849/Z/10/Z from the Brain and Behavior Research Foundation, and Wellcome Trust to Dr Howes and King’s College London scholarship to Dr Ashok. Dr Myers is supported by the Rosetrees Trust and Stoneygate Trust. Dr Rabiner wishes to acknowledge the support and funding by the National Institute for Health Research (NIHR) Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London.
Author information
Authors and Affiliations
Contributions
A.H.A. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: A.H.A, O.D.H. Acquisition, analysis, or interpretation of data: all authors. Drafting of the paper: all authors. Critical revision of the paper for important intellectual content: all authors. Statistical analysis: A.H.A., J.M. Administrative, technical, or material support: A.H.A. Study supervision: E.A.R., O.D.H.
Corresponding author
Ethics declarations
Competing interests
A.H.A. conducts research funded by the Medical Research Council (UK) and King’s College London. O.D.H. conducts research funded by the Medical Research Council (UK), the National Institute of Health Research (UK) and the Maudsley Charity. O.D.H. has received investigator-initiated research funding from and/or participated in advisory/speaker meetings organised by Astra-Zeneca, BMS, Eli Lilly, Jansenn, Lundbeck, Lyden-Delta, Servier and Roche. Neither O.D.H. nor his family have been employed by or have holdings/a financial stake in any biomedical company. E.A.R. is an employee of Invicro, a Konica-Minolta company, and has a financial stake in GlaxoSmithKline. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the paper; and decision to submit the paper for publication. The remaining authors have no competing interests.
Additional information
Peer review information Nature Communications thanks René Kahn and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ashok, A.H., Myers, J., Reis Marques, T. et al. Reduced mu opioid receptor availability in schizophrenia revealed with [11C]-carfentanil positron emission tomographic Imaging. Nat Commun 10, 4493 (2019). https://doi.org/10.1038/s41467-019-12366-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-019-12366-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.