Abstract
Hypothalamic neurons expressing the anorectic peptide Pro-opiomelanocortin (Pomc) regulate food intake and body weight. Here, we show that Steroid Receptor Coactivator-1 (SRC-1) interacts with a target of leptin receptor activation, phosphorylated STAT3, to potentiate Pomc transcription. Deletion of SRC-1 in Pomc neurons in mice attenuates their depolarization by leptin, decreases Pomc expression and increases food intake leading to high-fat diet-induced obesity. In humans, fifteen rare heterozygous variants in SRC-1 found in severely obese individuals impair leptin-mediated Pomc reporter activity in cells, whilst four variants found in non-obese controls do not. In a knock-in mouse model of a loss of function human variant (SRC-1L1376P), leptin-induced depolarization of Pomc neurons and Pomc expression are significantly reduced, and food intake and body weight are increased. In summary, we demonstrate that SRC-1 modulates the function of hypothalamic Pomc neurons, and suggest that targeting SRC-1 may represent a useful therapeutic strategy for weight loss.
Similar content being viewed by others
Introduction
Transcriptional coactivators and corepressors regulate the ability of nuclear hormone receptors (NRs) and transcription factors (TFs) to enhance/suppress the expression of target genes by facilitating the assembly of the transcription complex at target gene promoters1. Understanding the molecular mechanisms by which coactivators and corepressors alter gene expression to modulate physiological processes may provide insights into disease mechanisms and highlight potential therapeutic targets.
Steroid receptor coactivator (SRC)-1 belongs to a family of coactivators (SRC-1, -2, and -3) that mediate NR-dependent or TF-dependent transcription2. Global deletion of SRC-1 in mice leads to obesity3; however, to date, the molecular mechanisms involved are incompletely understood. SRC-1 is abundantly expressed in the hypothalamus, including neurons within the arcuate nucleus of the hypothalamus (ARH)4, which play a key role in mediating the weight-reducing effects of the adipocyte-derived hormone leptin5,6. Leptin is a signal of nutrient deprivation, with a fall in leptin levels triggering a set of responses that seek to restore energy homeostasis by increasing food intake and decreasing energy expenditure7. In the fed state, an increase in leptin levels leads to the activation of neurons expressing the anorectic peptide Pro-opiomelanocortin (POMC) leading to a reduction in food intake8. Specifically, leptin binding to its receptor phosphorylates the transcription factor STAT3 which dimerizes and translocates to the nucleus where it stimulates the expression of POMC9,10,11. Leptin-induced STAT3 activation also stimulates expression of Socs3 (suppressor of cytokine signaling-3) which acts to inhibits leptin signaling12,13.
In this study, we sought to investigate the central mechanisms by which SRC-1 modulates energy homeostasis. SRC family members bind to STAT transcription factors in cells14. Thus, we first examined the effects of SRC-1 on STAT3 transcriptional activity and Pomc expression. We then characterized metabolic phenotypes in mice lacking SRC-1 in Pomc neurons and explored the underlying mechanisms. Additionally, we examined the potential functional consequences of rare human variants in SRC-1 identified in severe childhood-onset obesity. Finally, we generated a knock-in mouse model of the most severe loss of function human SRC-1 variant and characterized the metabolic consequences of these mutant mice.
Results
SRC-1 interacts with pSTAT3 to stimulate Pomc expression
We found that global SRC-1-KO mice15 had lower Pomc but normal Socs3 mRNA levels in the hypothalamus compared to control littermates (Fig. 1a). Using Chromatin-immunoprecipitation (ChIP) assays, we found that leptin-stimulated pSTAT3 binding to Pomc promoters was decreased in the hypothalamus of SRC-1-KO mice compared to control mice (Fig. 1b). In keeping with these findings, SRC-1 overexpression potentiated STAT3-induced Pomc transcription but had no effect on Socs3 transcription in Neuro2A cells and HEK293 cells (Fig. 1c, d; Supplementary Figure 1a-b). Similar effects of SRC-1 were observed in SRC-1-KO MEFs cells, although STAT3 alone could stimulate Pomc expression in these cells devoid of endogenous SRC-1 (Supplementary Figure 1c-d). These results indicate that SRC-1, while not required for STAT3 transcriptional activity, can facilitate STAT3-induced Pomc expression.
SRC-1 potentiates STAT3-induced Pomc expression. Numbers of mice/repeats in each group are indicated; data are presented as mean ± SEM and compared using T-tests or two-way ANOVA followed by post hoc Sidak tests (#). a Pomc and Socs3 mRNA levels in hypothalami from 16-week old SRC-1-KO and WT control littermates (n = 7/8); **P < 0.01. b ChIP assays detecting pSTAT3 binding on Pomc promoters in hypothalami from male SRC-1-KO and control littermates 30 min after leptin injections (5 mg/kg, i.p.): site 1, −998 to −989; site 2, −361 to −353; site 3, −76 to −68 upstream of Pomc (n = 3/4); *P < 0.05. c, d Effects of overexpressed constitutively active STAT3 and SRC-1 on Pomc- (c) or Socs3-luciferase activity (d) in Neuro2A cells (n = 5–9 independent experiments). ***P < 0.001 vs. empty vectors; ###P < 0.001 vs. STAT3 alone (#). e Change (∆) in body weight after male control and pomcSRC-1-KO mice were switched onto a HFD at day 97 (n = 6/9); *P < 0.05 and **P < 0.01 (#). f Fat mass and lean mass measured 28 days after HFD feeding (n = 6/9); *P < 0.05. g Energy intake measured by CLAMS chambers in 12-week old male mice matched for body weight, lean mass, and fat mass. Mice were subjected to a 2-day-chow–2-day-HFD protocol, and chow was replaced by HFD before the onset of dark cycle on day 3. Energy intake was averaged for 2-day chow feeding period and for 2-day HFD feeding period (n = 7/8); *P < 0.05. h Cumulative HFD intake measured in 12-week old male mice singly housed in home cages (n = 10/14); *P < 0.05 (#). i Change in body weight after control and MpomcSRC-1-KO mice were switched on a HFD at the age of day 84 (n = 8); *P < 0.05 (#). j Fat mass and lean mass measured 30 days after HFD feeding (n = 8); *P < 0.05. k Cumulative HFD intake measured in 12-week old male mice (n = 6/7); *P < 0.05 (#), **P < 0.01. Source data are provided as Source Data Fig. 1
SRC-1 in Pomc neurons regulates energy homeostasis
To test whether SRC-1 in Pomc neurons plays a functionally significant role in energy homeostasis, we crossed SRC-1lox/lox mice with Pomc-Cre mice to generate mice lacking SRC-1 selectively in Pomc lineage cells (pomcSRC-1-KO, Supplementary Figure 1e). On a standard chow diet, the body weight of male pomcSRC-1-KO mice was comparable to control littermates (SRC-1lox/lox) (Supplementary Figure 1f), whilst female pomcSRC-1-KO mice showed significant weight gain (Supplementary Figure 1g). This sexual dimorphism may be explained by our earlier observations that global SRC-1 deficiency blunts the weight-reducing effects of estrogen4. On a high fat diet (HFD), male pomcSRC-1-KO mice gained significantly more weight compared to control littermates (Fig. 1e) due to an increase in fat mass (Fig. 1f). In weight-matched mice, we observed a significant increase in HFD intake in pomcSRC-1-KO mice vs. controls (Fig. 1g, h); measurements of energy expenditure were comparable (Supplementary Figure 1h–j).
A caveat of the regular Pomc-Cre mouse line is that, during the early development, Cre recombinase is transiently expressed in a broader population of neurons and some of these Pomc lineage cells mature into orexigenic Npy/Agrp neurons with opposing effects on food intake16. To address this concern, we crossed a Pomc-CreER transgene17 onto the SRC-1lox/lox mouse allele. Tamoxifen induction at 9 weeks of age resulted in the deletion of SRC-1 in mature Pomc neurons (MpomcSRC-1-KO; Supplementary Figure 1k-l). When fed with a HFD, MpomcSRC-1-KO mice displayed increased weight gain and fat mass, associated with increased food intake compared to control littermates (Fig. 1i–k), which recapitulated the phenotypes observed in pomcSRC-1-KO mice. Collectively, these results indicate that SRC-1 in mature Pomc neurons is required to defend against diet-induced obesity.
SRC-1 in Pomc neurons is required for the anorectic effects of leptin
Several studies have shown that STAT3 signaling is a mediator of leptin’s effects on body weight10,18. In HFD-fed pomcSRC-1-KO mice, we observed a 5–6-fold increase in circulating leptin levels in HFD-fed pomcSRC-1-KO mice (Fig. 2a), whilst adiposity only increased 2-fold (Fig. 1f). Thus, we hypothesized that SRC-1 is downstream of leptin action and loss of SRC-1 in Pomc neurons may impair leptin signaling. Supporting this possibility, we found that intra-peritoneal administration of leptin to control mice rapidly increased the hypothalamic SRC-1-pSTAT3 interaction (Fig. 2b, c). Leptin administration significantly reduced 1-hour (1h) food intake in control mice but not in pomcSRC-1-KO mice (Fig. 2d), despite increased leptin-induced pSTAT3 in the arcuate nucleus (Fig. 2e, f). These results suggest that the SRC-1-pSTAT3 interaction is downstream of leptin-STAT3 signaling, and contributes to the acute anorectic effects of leptin. Notably, the effects of leptin on 4 and 24h food intake were not significantly altered in pomcSRC-1-KO mice (Supplementary Figure 2a-b), presumably because the anorectic effects of leptin after the first hour are mediated by other leptin-responsive neurons or other signaling pathways19,20.
SRC-1 mediates leptin signaling. Numbers of mice/experiments/neurons are indicated; data are presented as mean ± SEM and compared using T-tests or one- or two-way ANOVA followed by post hoc Sidak tests (#). a Serum leptin levels 42 days after HFD feeding (n = 5/8); *P < 0.05. b Time course of hypothalamic SRC-1-pSTAT3 interaction in C57Bl6 wild type mice that received i.p. injections of leptin (5 mg/kg). c Quantification of the hypothalamic SRC-1-pSTAT3 interaction. *P < 0.05 (#). d Two-hour fasted mice (12 weeks of age) received i.p. injections of saline or leptin (5 mg/kg) 15 min prior to refeeding and food intake was recorded for 1 h afterwards (n = 7/9); **P < 0.01 (#). e Representative pSTAT3 immunohistochemical staining in the ARH and VMH of control and pomcSRC-1-KO mice receiving a single bolus i.p. injection of leptin (0.5 mg/kg, 90 min). Scale bar = 50 μm. 3V the 3rd ventricle, ARH arcuate nucleus, VMH ventromedial hypothalamic nucleus. f Quantification of pSTAT3 (+) neurons in the ARH (n = 5); ***P < 0.001. g Representative traces of leptin-induced depolarization, in the presence of TTX, CNQX, DAP-5, and bicuculline, in mature Pomc neurons from control mice vs. from MpomcSRC-1-KO mice after 1-week HFD feeding. h Responsive ratio (depolarization is defined as >2 mV elevations in resting membrane potential) (n = 39/43); P = 0.002 in χ2 tests. i Quantification of leptin-induced depolarization in two groups (n = 39/43); **P < 0.01. j Representative traces of action potentials in untreated mature Pomc neurons from control mice vs. from MpomcSRC-1-KO mice. k, l Quantification of firing frequency (k) and resting membrane potential (l) in two groups (n = 29–36); *P < 0.05. m Representative traces of mIPSC in untreated mature Pomc neurons from control mice vs. from MpomcSRC-1-KO mice. n, o Quantification of amplitude (n) and frequency (o) of mIPSC in two groups (n = 13/14); ***P < 0.001. Source data are provided as Source Data Fig. 2
Leptin also depolarizes a subset of Pomc neurons to exert its anorectic effects8, although recent fiber photometry studies failed to detect acute effects of leptin on calcium dynamics in Pomc neurons21. Thus, we examined leptin-induced depolarization in TOMATO-labeled mature Pomc neurons from MpomcSRC-1-KO mice and tamoxifen-treated controls after 1-week HFD feeding. We recorded leptin-induced changes in resting membrane potential (RM) in the presence of tetrodotoxin (TTX), which blocks action potentials, and a mixture of fast synaptic inhibitors which block the majority of presynaptic inputs. We found that 26/39 (67%) of Pomc neurons from control mice were depolarized (>2 mV elevations in RM) by leptin (Fig. 2g, h). In contrast, only 14/43 (33%) of Pomc neurons from MpomcSRC-1-KO mice were depolarized by leptin (P = 0.002) and the amplitude of leptin-induced depolarization was significantly reduced in these Pomc neurons (Fig. 2g–i). Interestingly, in the absence of TTX and synaptic inhibitors, leptin-induced depolarization and increases in firing frequency were comparable between the two groups (Supplementary Figure 2c–f), suggesting that indirect effects of leptin through presynaptic terminals22,23 were not affected by the loss of SRC-1 in Pomc neurons. Notably, the baseline firing frequency was significantly decreased in mature Pomc neurons from MpomcSRC-1-KO mice compared to those from control mice, whereas the baseline RM remained unchanged (Fig. 2j–l). We found that the amplitude, but not the frequency, of miniature inhibitory postsynaptic currents (mIPSC) was significantly higher in mature Pomc neurons from MpomcSRC-1-KO mice than those from control mice (Fig. 2m–o). The frequency of mIPSC is thought to reflect presynaptic events (e.g., GABA release), while mIPSC amplitude is largely determined by the responsiveness of postsynaptic neurons. Thus we suggest that SRC-1 also regulates the responsiveness of Pomc neurons to GABA-ergic inputs via a leptin-independent mechanism.
Rare SRC-1 variants found in obese humans impairs SRC-1 functions
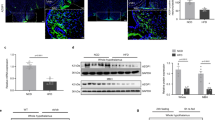
We next investigated the potential role of SRC-1 in humans by interrogating exome sequencing and targeted resequencing data on 2548 European ancestry individuals with severe, early-onset obesity (mean body mass index [BMI] standard deviation score = 3; age of onset < 10 years) and 1117 ancestry-matched controls24. Eleven rare heterozygous variants in SRC-1 were identified; another 8 variants were identified in an earlier data release (total n = 19). Fifteen SRC-1 variants were identified only in obese cases (N1212K was found in two unrelated obese individuals); the other 4 variants were found in controls (Fig. 3a). Compared to WT SRC-1, six of seven randomly selected SRC-1 mutants found in obese cases (except for S738L) were significantly impaired in their interaction with pSTAT3 in leptin-treated HEK293 cells (Fig. 3b, c, Supplementary Figure 3a–c). To test whether heterozygous SRC-1 variants exerted a dominant negative effect to inhibit the interaction between WT SRC-1 and pSTAT3, we overexpressed SRC-1 mutants in HEK293 cells which endogenously express SRC-1. After leptin treatment, an anti-pSTAT3 antibody was used to pull down the immunocomplex from cell lysates, followed by immunoblotting with an anti-SRC-1 antibody to examine the interaction between pSTAT3 and total SRC-1. Overexpression of SRC-1 mutants found in obese cases (6 of 7 tested mutants) significantly decreased the interaction between pSTAT3 and the total SRC-1, suggesting that these SRC-1 mutants can impair the ability of WT SRC-1 to interact with pSTAT3 (Fig. 3d, e and Supplementary Figure 3d-e). This dominant negative effect was not seen when testing the 4 mutants found in controls (Fig. 3e and Supplementary Figure 3d). We used a POMC-luciferase reporter assay to examine the effects of leptin on Pomc expression. We found that WT SRC-1 significantly enhanced leptin-induced Pomc-luciferase reporter activity, but co-expression of a dominant negative form of STAT3 abolished this effect (Supplementary Figure 3f-g), suggesting that the interaction with STAT3 is required for the observed effects of SRC-1 on Pomc transcription. Fourteen of fifteen SRC-1 mutants found in severely obese cases (except for S738L) significantly impaired leptin-induced Pomc expression, whereas the 4 control mutants exhibited WT-like responses in this assay (Fig. 3f). Interactions with estrogen receptor-α, vitamin D receptor, glucocorticoid receptor, thyroid hormone receptor-β, and peroxisome proliferator-activated receptor γ (PPARγ) were comparable to those seen for WT SRC-1 (Supplementary Figure 4) in co-immunoprecipitation assays.
Missense variants in SRC-1 disrupt leptin signaling. Numbers of experiments are indicated; data are presented as mean ± SEM and compared using one-way ANOVA followed by post hoc Sidak tests unless mentioned otherwise. a Rare variants identified in individuals with severe early onset obesity (above) and in controls (below). b, c HEK293 cells were co-transfected with leptin receptor vector and human STAT3 vector. Cells were treated with leptin (200 ng/ml, 15 min) to induce phosphorylation of STAT3. pSTAT3 was pulled down using anti-pSTAT3 sepharose beads; beads were then aliquoted equally and incubated with the same amount of the long isoform of human SRC-1-HA (WT/mutant) and interactions between the pSTAT3 and SRC-1 were determined by CoIP experiments using anti-pSTAT3 and anti-HA antibodies. b Representative blots showing interactions between pSTAT3 and SRC-1 (WT/mutant), and inputs of pSTAT3 and SRC-1-HA. c Quantification for WT and SRC-1 mutants. Comparative folds were calculated as the ratios of HA blots and HA inputs (n = 3–5); *P < 0.05. d, e SRC-1 mutants inhibit the interaction between STAT3 and WT SRC-1. HEK293 cells were co-transfected with leptin receptor vector, STAT3 vector, and mutant SRC-1 vector (or empty vector). Cells were treated with leptin (200 ng/ml, 15 min) to induce phosphorylation of STAT3 and interactions between pSTAT3 and total SRC-1 were determined by CoIP experiments using anti-pSTAT3 and anti-SRC-1 antibodies. d Representative blots showing interactions between pSTAT3 and SRC-1 variants found in obese cases and inputs of pSTAT3. e Quantification. Comparative folds were calculated as the ratios of SRC-1-pSTAT3 interaction blots and pSTAT3 inputs (n = 4–12); *P < 0.05. f SRC-1 variants impair POMC expression. Neuro2A cells were co-transfected with leptin receptor vector, SRC-1 (WT or mutant) and a POMC luciferase expression reporter construct. Cells were stimulated with 200 ng/ml leptin for 15 min and then incubated for 6 h, following which luminescence was measured. Results were normalized to empty vector-induced expression (n = 3–16); *P < 0.05. Source data are provided as Source Data Fig. 3
A mouse model of the human SRC-1 variant L1376P is obese
To directly test whether rare human SRC-1 variants contribute to Pomc neuron function and/or energy homeostasis, we generated a knock-in mouse model of a human variant which results in a severe loss of function in cells, SRC-1L1376P (Fig. 4a). Heterozygous mutant mice (SRC-1L1376P/+) fed a HFD exhibited increased weight gain, adiposity and food intake, associated with reduced Pomc mRNA levels compared to WT controls (Fig. 4b–e). We recorded leptin-induced depolarization in Pomc neurons in control vs. SRC-1L1376P/+ mice 1 week after HFD feeding. In control mice, 13/19 (68%) Pomc neurons were depolarized by leptin, whilst only 5/18 (26%) Pomc neurons from SRC-1L1376P/+ mice were depolarized by leptin (P = 0.022) and the amplitude of leptin-induced depolarization was significantly reduced in these Pomc neurons (Fig. 4f–h). Baseline firing frequency and resting membrane potential were both significantly decreased in Pomc neurons from SRC-1L1376P/+ mice compared to those from control mice (Fig. 4i–k). Further, the amplitude, but not the frequency, of the mIPSC was significantly higher in Pomc neurons from SRC-1L1376P/+ mice than those from control mice (Fig. 4l–n). Thus, these data indicate that the SRC-1L1376P variant causes obesity in mice, associated with decreased Pomc expression and decreased Pomc neuron excitability through both leptin-dependent and independent mechanisms.
SRC-1L1376P/+ mice are obese. Numbers of mice in each group are indicated; data are presented as mean ± SEM and compared using T-tests or two-way ANOVA followed by post hoc Sidak tests (#). a The PCR products (121 bp) around the L1376 were amplified from genomic DNA extracts of a WT and two SRC-1L1376P/+ mutant mice and incubated with or without Sau3AI. Control reaction (WT) resulted in a single large fragment (121 bp) and DNAs from the two SRC-1L1376P/+ mutant mice were cut into two fragments (70 and 51 bp) as expected. b Change in body weight after male control and SRC-1L1376P/+ mice were fed on a HFD (n = 5/6); *P < 0.05 (#). c Fat mass and lean mass measured 7 weeks after HFD feeding (n = 5/6); ***P < 0.001. d Cumulative HFD intake measured (n = 5/6); *P < 0.05 or **P < 0.01 (#). e Pomc mRNA levels in hypothalami from 20-week old HFD-fed male control and SRC-1L1376P/+ mice (n = 12/16); *P < 0.05. f Representative traces of leptin-induced depolarization, in the presence of TTX, CNQX, DAP-5, and bicuculline, in Pomc neurons from control mice vs. from SRC-1L1376P/+ mice after 1-week HFD feeding. g Responsive ratio (depolarization is defined as >2 mV elevations in resting membrane potential) (n = 19); P = 0.022 in χ2 tests. h Quantification of leptin-induced depolarization in two groups (n = 19); ***P < 0.001. i Representative traces of action potentials in untreated Pomc neurons from control mice vs. from SRC-1L1376P/+ mice. j, k Quantification of firing frequency (j) and resting membrane potential (k) in two groups (n = 22–28); ***P < 0.001. l Representative traces of mIPSC in untreated Pomc neurons from control mice vs. from SRC-1L1376P/+ mice. m, n Quantification of amplitude (m) and frequency (n) of mIPSC in two groups (n = 10/12); ***P < 0.001. Source data are provided as Source Data Fig. 4
Discussion
In this study, we demonstrated that in the hypothalamus, the coactivator SRC-1 modulates the ability of leptin to regulate the expression of the anorectic peptide POMC by directly interacting with phosphorylated STAT3, a known product of leptin-receptor activation. In mice, disruption of SRC-1 in Pomc neurons led to increased food intake, weight gain on a HFD and impaired the acute anorectic response to leptin administration demonstrating the physiological relevance of this molecular interaction. The modest degree of obesity in these mice was comparable to that seen with inactivation of STAT3 in Pomc neurons9 and studies demonstrating that direct leptin action on Pomc neurons accounts for a proportion of leptin’s effects on body weight19,20,25,26. The obesity seen in SRC-1 deletion or mutant mice was less severe than that see in mice deficient in Pomc27 or melanocortin 4 receptor28 in keeping with SRC-1’s role as a modulator of Pomc expression. Additionally, leptin-responsive Agrp neurons have been shown to play a major role in energy homeostasis20.
We identified 15 rare heterozygous variants in SRC-1 in 16 severely obese individuals and 4 rare variants in controls. Notably, there are several low frequency and many rare variants in this gene in publically available databases (http://gnomad.broadinstitute.org/). Some of these low frequency variants have been shown to have functional consequences, for example, P1272S (MAF: 3.16% in cases, 3.45% in controls; 1.66% in gnomAD) disrupts a putative glycogen synthase 3 (GSK3)β phosphorylation site and has been shown to exhibit reduced ability to co-activate Estrogen Receptor in multiple cell lines29. Genetic studies in larger numbers of cases and controls with functional studies of all variants identified will be needed to establish whether variants that result in a loss of function when tested in cells are more likely to be found in severely obese individuals than in controls. In this study, the variants found in obese individuals, but not those found in controls, were associated with impaired interaction with pSTAT3 and reduced POMC reporter activity in cells, predominantly through a dominant negative effect. Given the challenges associated with studying such rare variants, and to directly test whether rare human SRC-1 variants contribute to Pomc neuron function and/or energy homeostasis, we generated a knock-in mouse model of a human variant which results in a severe loss of function in cells, SRC-1L1376P. The increased food intake and weight gain in heterozygous knock-in mice carrying a severe loss of function human SRC-1 variant supports the potential importance of the mechanism identified here in humans.
Recent evidence indicates that loss of leptin receptors in Pomc neurons does not affect body weight in chow-fed mice19,20. In line with these reports, we show that loss of SRC-1 in Pomc neurons produced minor effects on energy balance in chow-fed male mice. These suggest that the physiological consequences of disrupting this interaction in normal weight animals are small and/or may be compensated for by increased signaling through non-POMC expressing leptin-responsive neurons30 and/or signaling via phosphoinositide-3-kinase (PI3K)31, mTOR/S6K32 and/or AMPK pathways33,34. We showed that SRC-1 deletion in Pomc neurons attenuated the acute anorectic response (1 h) to leptin but not the late phase (4–24 h). Cumulatively, these findings indicate that leptin-mediated POMC expression (modulated by the SRC-1-pSTAT3 interaction) primarily contributes to the acute anorectic response to leptin. In keeping with this finding, we demonstrated that the hypothalamic SRC-1-pSTAT3 interaction was enhanced by leptin. Consumption of HFD leads to sustained positive energy balance and an increase in leptin levels. The resulting increase in pSTAT3 would be expected to stimulate POMC expression and reduce food intake, a response that we have shown is modulated by the interaction between pSTAT3 and SRC-1. We suggest that in the absence of functional SRC-1, pSTAT3 is less effective at stimulating POMC expression, which manifests as a relative increase in food intake and weight gain when mice are challenged with HFD. In this way, we conclude that SRC-1 acts as a positive regulator of leptin sensitivity in hypothalamic Pomc neurons.
Our findings suggest that SRC-1 facilitates but is not required for pSTAT3 to regulate Pomc expression and that this effect is target-specific as SRC-1 does not modulate the ability of pSTAT3 to regulate Socs3. The mechanisms underlying such specificity remain unclear at present. The molecular interaction between SRC-1 and pSTAT3 enhances pSTAT3-mediated transcriptional activity, presumably by stabilizing pSTAT3 binding to the POMC promoter, although we cannot exclude the possibility that recruitment of other co-coactivators or histone acetyltransferase activity of SRC-1 also may be involved35. Further studies of the molecular mechanisms that modulate leptin signaling are emerging36,37,38,39,40,41. For example, Chen et al showed that the nuclear receptor Nur77 facilitates STAT3 acetylation by recruiting acetylase p300 and disassociating deacetylase histone deacetylase 1 (HDAC1) to enhance the transcriptional activity of STAT342. In findings that parallel our studies, they showed that Nur77 deficiency reduced the expression of Pomc in the hypothalamus and attenuated the response to leptin in mice fed on a HFD42.
Transcriptional coactivators such as SRC-1 facilitate the signaling mediated by multiple NRs and/or TFs factors2. Several NRs/TFs have been shown to affect energy homeostasis through their actions in the brain43, including FoxO144,45,46,47, ERα48,49, PPARγ50,51, and THR52 and thus could contribute to the body weight phenotype seen with SRC-1 disruption in mice and loss of function variants in humans. In addition to the central actions of SRC-1 on energy homeostasis, SRC-1 is expressed in brown adipose tissue, where it appears to compete with SRC-2 to interact with the PPARγ-PGC1α complex. Picard et al showed that SRC-1-KO mice had reduced rectal temperatures upon cold exposure and reduced oxygen consumption although they did not quantify food intake in this study3. Notably, we did not observe any changes in energy expenditure in mice lacking SRC-1 in Pomc neurons, consistent with the notion that SRC-1 in other tissues may also contribute to the regulation of energy expenditure3. Whilst we found that SRC-1 variants detected in obese patients did not affect the interactions with a number of NRs, these results do not exclude the potential impact of SRC-1 variants on the signaling of these NRs which need to be explored in more detail using tissue-specific conditional knockout mouse models.
Targeting specific coactivator-mediated interactions has emerged as a potential therapeutic strategy to enhance signaling in some tissues while inhibiting signaling in others53,54. For example, Selective Estrogen Receptor Modulators (SERMs) are effective in modulating the growth of hormone-responsive tumors (e.g., Tamoxifen in breast cancer) by impacting on coactivator stability and activity55. As such, compounds that target the interaction between SRC-1 and STAT3 at specific sites may potentially be used to modulate (i.e., enhance) leptin signaling. Could this approach be efficacious in the treatment of obesity? Studies in mice and humans have consistently demonstrated that leptin sensitivity is greatest in those with no/very low endogenous circulating leptin levels56,57. Whether enhancing leptin sensitivity in the context of common obesity, which is associated with elevated leptin levels, may be clinically beneficial, is the subject of much debate18,58,59,60. The finding that some compounds (e.g., the amylin derivative pramlintide) can augment the effects of leptin61,62, suggests that it may be possible to increase the sensitivity of some individuals to therapeutic leptin administration and that this approach may lead to weight loss. These observations and our findings on SRC-1 suggest that pharmacological approaches based on the modulation of leptin sensitivity could represent a potential therapeutic strategy for the treatment of obesity-associated metabolic disease.
Methods
Contact for reagent and resource sharing
Further information and requests for resources and reagents should be directed to and will be fulfilled by Yong Xu (yongx@bcm.edu) and Sadaf Farooqi (isf20@cam.ac.uk).
Experimental model and subject details
Mice: We crossed regular Pomc-Cre transgenic mice26 and SRC-1lox/lox mice63. This cross produced pomcSRC-1-KO mice (those that are homozygous for SRC-1lox/lox and also carry the Pomc-Cre transgene) and control mice (those that are homozygous for SRC-1lox/lox but do not carry the Pomc-Cre transgene). These littermates were used to characterize the metabolic profile.
In addition, we also crossed inducible Pomc-CreER mice17 with SRC-1lox/lox mice to generate MpomcSRC-1-KO mice (those that are homozygous for SRC-1lox/lox and also carry the Pomc-CreER transgene) and control mice (those that are homozygous for SRC-1lox/lox but do not carry the POMC-CreER transgene). Both these mice received tamoxifen injections (0.2 mg/g, i.p., twice at 9 weeks of age). These littermates were used to characterize the metabolic profile. For electrophysiological recordings, we crossed the inducible Pomc-CreER and the Rosa26-tdTOMATO mouse alleles onto SRC-1lox/lox mice, to produce MpomcSRC-1-KO mice with mature Pomc neurons labeled by TOMATO; as controls, we crossed inducible Pomc-CreER mice and Rosa26-tdTOMATO mice to generate Pomc-CreER/Rosa26-tdTOAMTO mice. In parallel, we also crossed the Npy-GFP mouse allele23 and the Rosa26-tdTOMATO allele onto inducible Pomc-CreER mice. This cross produced Pomc-CreER/Rosa26-tdTOAMTO/Npy-GFP mice, which were subjected to histology validation for the inducible Pomc-CreER mice.
To generate the SRC-1L1376P/+ knock-in mice, a single-guide RNA (sgRNA) sequence was selected overlap amino acid residue L1382 (equivalent to human L1376) in SRC-1 (sgRNA 5′-CATCTGCGTCTGTTTTGAGAagg chr12:4253665-4253687; GRCm38/mm10) using the CRISPR Design Tool (Ran et al. 2013). A DNA templates for in vitro transcription of the sgRNA was produced using overlapping oligonucleotides in a high-fidelity PCR reaction64, and sgRNA was transcribed using the MEGAshortscript T7 kit (ThermoFisher, Waltham, MA). Cas9 mRNA was purchased from ThermoFisher. The donor DNA template to introduce the L1382P point mutation, as well as a silent mutation D1381D to introduce a novel restriction site for Sau3AI, was purchased as an Ultramer from IDT (Coralville, IA). The sequence of ssODN is as follows (complementary to non-target strand): 5′ TGAAAATCTG CTCTTTTGTT TATCCTTAAT AGATGAATGA TCCAGCACTG AGACACACAG GCCTCTACTG CAACCAGCTC TCGTCCACTG ATCCCCTCAA AACAGACGCA GATGGAAACC AGGTCAGTAA GAAA, where the homology arms are in bold. The mutations introduced in the donor sequence disrupt base 20 of the sgRNA and the PAM site to prevent additional mutagenesis. The BCM Genetically Engineered Mouse (GEM) Core microinjected Cas9 mRNA (100 ng/μl), Ultramer ssDNA (100 ng/µl), and sgRNA (20 ng/μl) into the cytoplasm of 200 pronuclear stage C57Bl/6J embryos. Cytoplasmic injections were performed using a microinjection needle (1 mm outer and 0.75 mm inner) with a tip diameter of 0.25–0.5 μm, an Eppendorf Femto Jet 4i to set pressure and time to control injection volume (0.5–1 pl per embryo). Injections were performed under a 200–400× magnification with Hoffman modulation contrast for visualizations. Founder animals (F0) were identified by PCR-based restriction digestion to detect the CRISPR generated point mutations in SRC-1. PCR product was first amplified with the primer pairs: 5′-CCTCACTTGTGGCAATGTGA and 5′-TCGTGGCAGTTCTGTAGTCAC; and then amplified with 2nd pairs: 5′-CACTGAGACACACAGGCCTC and 5′-ATCGAATCTGCCAGCTCTGC. The 121 bp PCR products were then digested with Sau3AI. 70 and 51 bp products after digest could be detected only for the mutated SRC-1 PCR products. Three independent lines were sequenced for the further confirmation of the point mutation. One of these lines was crossed to C57Bl6j to produce cohorts comprised of SRC-1L1376P/+ and wild-type control mice. In some breedings, the Pomc-CreER/Rosa26-tdTOAMTO alleles were introduced to allow specific labeling of Pomc neurons.
In parallel, we crossed heterozygous SRC-1-KO mice65 to heterozygous SRC-1-KO mice to produce homozygous SRC-1-KO and wild-type littermates. All the breeders have been backcrossed to C57Bl6 background for more than 12 generations. In addition, some C57Bl6 mice were purchased from the mouse facility of Baylor College of Medicine.
Care of all animals and procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Baylor College of Medicine Animal Facility, and all experimental procedures in animals complied with all relevant ethical regulations. Mice were housed in a temperature-controlled environment in groups of 2–5 at 22–24 °C using a 12 h light/12 h dark cycle. Some cohorts were singly housed to measure food intake. The mice were fed either standard chow (6.5% fat, #2920, Harlan-Teklad, Madison, WI), or a 60% HFD (60% fat, #D12492, Research Diets). Water was provided ad libitum.
Studies in mice
Validation of genomic deletion of SRC-1 in Pomc cells: Control mice, pomcSRC-1-KO mice or MpomcSRC-1-KO mice (after tamoxifen inductions) were anesthetized with inhaled isoflurane, and sacrificed. Various tissues, as detailed in the figures, were collected. Genomic DNAs were extracted using the REDExtract-N-Amp Tissue PCR Kit (#XNATS; Sigma-Aldrich, St Louis, MO), followed by PCR amplification of the floxed or recombined alleles. We used primers: forward-CAGTAAGGAATAGCAGATGTC and reverse-TGGCATCTATAACCAAATGTGTA TCA to detect the wild-type allele (a 560 bp band) and the floxed SRC-1 allele (a 630 bp band); and combined the reverse primer (mentioned above) with another forward primer: GTCGTACCATCTATGCCTCCTATAT to detect the recombined SRC-1 allele (a 320 bp band).
Histology: To validate specificity of the inducible Pomc-CreER transgene, Pomc-CreER/Rosa26-tdTOAMTO/Npy-GFP mice received tamoxifen injections (0.2 mg/g, i.p., twice) at 9 weeks of age, and then were perfused 1 week later. Brain sections were cut at 25 μm (1:5 series) and subjected to direct visualization of GFP and TOMATO signals using a Leica DM5500 fluorescence microscope with OptiGrid structured illumination configuration.
To examine the effects of leptin on STAT3 phosphorylation in vivo, control and pomcSRC-1-KO mice (5 or 6 per group) were fasted overnight and then received a single bolus injection of saline or leptin (0.5 mg/kg, i.p.). Ninety minutes after the bolus injections, mice were anesthetized with inhaled isofluorane, and quickly perfused with 10% formalin, and brain sections were cut at 25 μm. The brain sections were pretreated (1% H2O2, 1% NaOH, 0.3% glycine, 0.03% SDS), blocked (3% goat-anti-rabbit serum for 1 h), incubated with rabbit anti-pSTAT3 antibody (1:2000; #9145, Cell Signaling) on shaker at room temperature for 24 h and then put in 4 °C for 48 h, followed by biotinylated anti-rabbit secondary antibody (1:1000; Vector) for 2 h. Sections were then incubated in the avidin–biotin complex (1:500, ABC; Vector Elite Kit) and incubated in 0.04% 3,3′-diaminobenzidine and 0.01% hydrogen peroxide. After dehydration through graded ethanol, the slides were then immersed in xylene and cover-slipped. Images were analyzed using a brightfield Leica microscope. The numbers of pSTAT3-positive neurons in the ARH were counted by blinded investigators. For each mouse, pSTAT3-positive neurons were counted in 3–5 consecutive brain sections containing ARH, and the average was treated as the data value for that mouse. Five or six mice were included in each group for statistical analyses.
Body weight study: pomcSRC-1-KO mice and their control littermates were weaned at week 4. These mice were group housed and maintained on the standard chow (6.5% fat, #2920, Harlan-Teklad). At the age of day 97, mice were switched to the HFD (60% fat, #D12492, Research Diets) for 6 weeks. Body weight was measured every 4 days since weaning. Body composition was determined using quantitative magnetic resonance (QMR) on 28 days after HFD feeding. On day 42 after HFD feeding, the mice were deeply anesthetized with inhaled isoflurane and sacrificed. Blood was collected and processed to measure serum leptin using the mouse leptin ELISA kit (#90030, Crystal Chem, Inc.). Serum samples with hemolysis (one from each group) were excluded from leptin ELISA assay. The gonadal white adipose tissue, the inguinal white adipose tissue, and the interscapular brown adipose tissue were isolated and weighed.
Similarly, MpomcSRC-1-KO mice and their control littermates were weaned at week 4. These mice were singly housed and maintained on the standard chow (6.5% fat, #2920, Harlan-Teklad). All mice received tamoxifen injections (0.2 mg/g, i.p., twice) at 9 weeks of age. At the age of day 84, mice were switched to the HFD (60% fat, #D12492, Research Diets) for 30 days. Body weight and food intake were measured every 4 days. Body composition was determined using QMR on 30 days after HFD feeding.
Food intake and energy expenditure: To further characterize the food intake and energy expenditure of pomcSRC-1-KO mice, an independent male cohort (pomcSRC-1-KO mice and their control littermates) was weaned on the standard chow. At the age of 12 weeks, these mice were acclimated into the Comprehensive Laboratory Animal Monitoring System (CLAMS). Mice were housed individually at room temperature (22 °C) under an alternating 12:12-h light-dark cycle. After adaptation for 3 days, mice were subjected to a 2-day-chow–2-day-HFD protocol. Chow was replaced by HFD before the onset of dark cycle on day 3. Note that, the body weight and body composition were measured before the mice entered the CLAMS metabolic cages, and no difference was observed in body weight, fat mass, and lean mass.
Another male cohort (pomcSRC-1-KO mice and their control littermates) was weaned on the standard chow. At the age of 11 weeks, these mice were singly housed and at week 12, the chow diet was replaced by HFD. HFD intake was measured every 2 days for 10 days.
Leptin-induced anorexia: Male pomcSRC-1-KO mice and their control littermates (chow-fed) were briefly fasted for 2 h prior to the onset of dark cycle. These mice received intraperitoneal injections of saline or leptin (5 mg/kg in saline in a volume of 0.01 ml/g body weight) at 15 min prior to the dark cycle. The standard chow was provided at the onset of dark cycle. Food intake was measured 1, 4, and 24 h after food provision. Each mouse was tested with saline and leptin, administered in a counterbalanced order, with 4-day interval between the treatments.
Electrophysiology: For electrophysiological studies, Pomc-CreER/Rosa26-tdTOMATO (control) mice and Pomc-CreER/Rosa26-tdTOMATO/SRC-1lox/lox (MpomcSRC-1-KO) mice received tamoxifen inductions (0.2 mg/g, i.p., twice at 9 weeks of age) and fed on HFD for 1 week. Pomc-CreER/Rosa26-tdTOMATO/SRC-1L1376P/+ and their control littermates (Pomc-CreER/Rosa26-tdTOMATO) were also fed on HFD for 1 week followed by electrophysiology recording as described below. Briefly, at 9:00–9:30 am, these mice were deeply anesthetized with isoflurane and transcardially perfused with a modified ice-cold artificial cerebral spinal fluid (aCSF, in mM: 10 NaCl, 25 NaHCO3, 195 Sucrose, 5 Glucose, 2.5 KCl, 1.25 NaH2PO4, 2 Na pyruvate, 0.5 CaCl2, 7 MgCl2)47. The mice were then decapitated, and the entire brain was removed. Brains was quickly sectioned in ice-cold aCSF solution (in mM: 126 NaCl, 2.5 KCl, 1.2 MgCl2, 2.4 CaCl2, 1 NaH2PO4, 11.1 Glucose, and 21.4 NaHCO3)23 saturated with 95% O2 and 5% CO2. Coronal sections containing the ARH (250 µm) was cut with a Microm HM 650V vibratome (Thermo Scientific). Then the slices were recovered in the aCSF23 at 34 °C for 1 h.
Whole-cell patch clamp recordings were performed in the TOMATO-labeled mature Pomc neurons in the ARH visually identified by an upright microscope (Eclipse FN-1, Nikon) equipped with IR-DIC optics (Nikon 40× NIR). Signals were processed using Multiclamp 700B amplifier (Axon Instruments), sampled using Digidata 1440A and analyzed offline on a PC with pCLAMP 10.3 (Axon Instruments). The slices were bathed in oxygenated aCSF23 (32–34 °C) at a flow rate of approximately 2 ml/min. Patch pipettes with resistances of 3–5 MΩ were filled with solution containing 126 mM K gluconate, 10 mM NaCl, 10 mM EGTA, 1 mM MgCl2, 2 mM Na-ATP and 0.1 mM Mg-GTP (adjusted to pH 7.3 with KOH).
Current clamp was engaged to test neural firing frequency and RM at the baseline and after puff application of leptin (300 nM, 1 s). In some experiments, the aCSF solution also contained 1 μM TTX and a cocktail of fast synaptic inhibitors, namely bicuculline (50 μM; a GABA receptor antagonist), DAP-5 (30 μM; an NMDA receptor antagonist) and CNQX (30 μM; an AMPA receptor antagonist) to block the majority of presynaptic inputs. The values for RM and firing frequency were averaged within 2-min bin at the baseline or after leptin puff. The RM values were calculated by Clampfit 10.3 using the “analysis → statistic” function of the software. A neuron was considered depolarized or hyperpolarized if a change in membrane potential was at least 2 mV in amplitude and this response was observed after leptin application and stayed stable for at least 2 min. For the miniature inhibitory postsynaptic current (mIPSC) recordings, patch electrodes were filled with a recording solution that contained (in mM): 153.3 CsCl, 1.0 MgCl2, 5.0 EGTA, and 10.0 HEPES, pH of 7.20 with CsOH. CsCl was included to block potassium currents. Mg-ATP (3 mM) was added to the intracellular solution before recording. Glutamate receptor-mediated synaptic currents were blocked by 30 μM D-AP-5 and 30 μM CNQX in the external solution, along with 1 μM tetrodotoxin in the external solution blocking action potentials. Neurons were voltage-clamped at −70 mV during the recording.
At the end of recordings, lucifer yellow dye was included in the pipette solution to trace the recorded neurons and the brain slices were fixed with 4% formalin overnight and mounted onto slides. Cells were then visualized with the Leica DM5500 fluorescence microscope to identify post hoc the anatomical location of the recorded neurons in the ARH.
Real-time PCR analyses: Total RNA was isolated using TRIzol Reagent (Invitrogen) according to the manufacturer’s protocol and reverse transcription reactions were performed from 2 μg of total RNA using a High-Capacity cDNA Reverse Transcription Kits (Invitrogen). cDNA samples were amplified on an CFX384 Real-Time System (Bio-Rad) using SsoADV SYBR Green Supermix (Bio-Rad). Correct melting temperatures for all products were verified after amplification. Results were normalized against the expression of house-keeping gene-Cyclophilin. Primer sequences were listed in Supplementary Table 1.
Immunoprecipitation (Co-IP) and immunoblotting: The harvested hypothalami were lysed in lysis buffer (50 mM Tris–HCl, pH 8.0, 50 mM KCl, 20 mM NaF, 1 mM Na3VO4, 10 mM sodium pyrophosphate, 5 mM EDTA, and 0.5% Nonidet P-40) supplemented with protease inhibitors (1 mm phenylmethylsulfonyl fluoride, and 20 μg/ml each of leupeptin, aprotinin, and pepstatin). Lysates were cleared by centrifugation at 18,000 ×g for 10 min and used for immunoprecipitation or directly for immunoblotting. Equal amounts of tissue lysates were incubated with anti-Phospho-STAT3 (Tyr705) (D3A7) XP-Sepharose beads (Cell Signaling) or with a rabbit monoclonal SRC-1 (128E7) antibody (Cell Signaling) after preclearing for overnight and pulled down with Protein A/G agarose beads (Santa Cruz), respectively. Beads were washed three times with lysis buffer, and proteins were released from beads in SDS-sample buffer and analyzed by immunoblotting. For immunoblotting, protein samples were loaded onto SDS-polyacrylamide gels and transferred to a nitrocellulose membrane. The blot was probed with a rabbit monoclonal SRC-1 (128E7) antibody at 1:3000 (Cell Signaling), a rabbit monoclonal phospho-STAT3 (Tyr705) (D3A7) XP antibody at 1:2000 (Cell Signaling), or a monoclonal anti-β-Actin antibody (AC-15) at 1:10000 (Sigma). The secondary antibody was rabbit anti-mouse IgG or goat anti-rabbit IgG (Jackson ImmunoResearch), both at a 1:10,000 dilution, followed by development with the SuperSignal West Pico Chemiluminescent Substrate (Pierce).
Chromatin immunoprecipitation assay (ChIP): Fresh isolated hypothalami were homogenized and cross-linked in 1% formaldehyde. Then, the cross-linked protein–DNA complexes were sonicated to a length between 200 and 500 bp. The total chromatin (1%) was saved as an “input” for later quantification. Complexes were pre-cleared and incubated with the Pierce Protein A/G Magnetic Beads (Thermo Scientific) and antibodies against STAT3 (sc-482; Santa Cruz) overnight at 4 °C. Subsequently, cross-linking was reversed by overnight incubation at 65 °C. DNAs were purified by phenol/chloroform extraction, ethanol precipitation and the enriched promoter fragments were measured by qPCR (primer sequences provided in Supplementary Table 1). Relative STAT3 promoter occupancy was adjusted to the background content of the negative control, and the initial chromatin input. The assays were repeated independently 3 times.
Generation of SRC-1 constructs and expression plasmids: The long form of SRC-1 containing a C-terminal Flag MYC tag was purchased from Origene (RC224812). The short form of SRC-1 was generated using the Q5 site-directed mutagenesis kit (NEB) using primers containing the sequence specific to the short form of SRC-1. The N-terminal HA tag was added using the Q5 site-directed mutagenesis kit (NEB) using primers containing the HA tag sequence. The short and long forms of SRC-1 was then cloned into the pCDNA3.1(+) vector using KpnI and XhoI restriction sites after PCR amplification of SRC-1 using primers flanking the Origene KpnI and XhoI sites. SRC-1 mutant constructs were generated using the Quickchange II XL site-directed mutagenesis kit (Agilent).
In vitro protein interaction: HEK293 (Human embryonic kidney 293) cells were transfected with either Flag-tagged transcriptional factor (hSTAT3 or hPPARγ), Flag-tagged human hormone receptor (ERα, VDR, THRβ or GR) or empty vector using lipofectamine 2000 (Invitrogen). Before harvest, cell were treated with leptin (at 200 ng/ml, 15 min, HARBOR-UCLA Research And Education Institute), or rosiglitazone (at 50 μM, ADIPOGEN), 17β-estradiol (at 0.2 μg/ml, Sigma, E2758), Vitamin D3 (Calcitriol at 0.2 μM, TOCRIS), dexamethasone (at 10 μM, Sigma, D4902) for 30 min. Cells were collected and lysed with cell lysis buffer: 50 mM Tris, 50 mM KCL, 10 mM EDTA, 1% NP-40, supplied with protease inhibitor cocktail (Roche) and phosphatase inhibitor cocktail A (Santa Cruz). The lysates were incubated with proper amount of anti-phospho-STAT3 sepharose beads (Cell Signaling, #4074) or anti-Flag-beads (Sigma) for 4 h at 4 °C. After wash, beads were aliquoted equally and incubated with comparable amounts of SRC-1 protein (wt or mutants) overnight, and the interacting protein was detected by Western-Blot. SRC-1 WT or mutants were expressed in HEK293 cells and the amount of the SRC-1 expressed was determined by Western-Blot before the protein interaction assay. Comparable amounts of SRC-1 (wt or mutants) in the same volume of cell lysates (compensated with the cell lysates from the cells transfected with empty vector) were used for the in vitro protein interaction. Except for the THRβ IP were equal amounts of total protein from SRC-1 WT and mutant lysates (determined by Bradford assay (Biorad)) were incubated with equal volumes of flag-tagged THRβ lysate overnight at 4 °C with 1 μM T3 thyroid hormone. THRβ was then immunoprecipitated using anti-Flag conjugated beads for 1 h which were washed 6 times with lysis buffer and eluted with LDS sample buffer before western blotting.
Luciferase transcription activation assays: To measure STAT3 activity on the POMC promoter, HEK293, Neuro 2A (mouse neuroblastoma cell line) and immortalized MEF cells (generated in J.X. lab) were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum (Atlanta), 100 IU/ml penicillin and 100 ng/ml streptomycin. Cells were seeded into a 24-well plate overnight and then transfected with 600 ng of the Pomc-luciferase reporter plasmid66 or 300 ng Socs3-luciferase 6T1 reporter plasmid12, combined with 100 ng of pRL-SV40 (Promega), 100 ng of pCR3.1-SRC-1 and/or 10 ng pRc/CMV-STAT3C plasmids or the control empty plasmids, according to the Lipofectamine LTX protocol (Invitrogen). Thirty hours post-transfection, the cells were lysed and the luciferase activity was measured using the Dual-Luciferase® Reporter Assay System (Promega) according to the manufacturer’s instruction.
For leptin-induced Pomc-luciferase reporter assay, a fragment of the human POMC promoter (−949 to +416, relative to the transcription start site) was cloned into the pGL3 Luciferase Reporter Vector by using the primer pairs: 5′-TGTTCTAGTTGGGGGAACAGC-3′ and 5′-GCGCCCTTACCTGTCTCGG-3′. Neuro 2A cells were cultured in 48-well plate for overnight and then transfected with 0.1 µg human Pomc-luciferase reporter plasmid, 0.025 µg LepR and 0.05 µg hSRC-1 plasmid. Forty hours post-transfection, the cells were treated with 0.2 µg/ml Leptin for 20 min and then kept cultured in fresh media for 6 h. To test the effect of dominant negative STAT3 on leptin-induced POMC-luciferase reporter activity, the above protocol was modified by cotransfecting 10 ng of the dominant negative form of STAT3 (Y705F).
Human studies: The Genetics of Obesity Study (GOOS) is a cohort of 7000 individuals with severe early-onset obesity; age of obesity onset is less than 10 years67,68. Severe obesity is defined as a body mass index (weight in kilograms divided by the square of the height in meters) standard deviation score greater than 3 (standard deviation scores calculated according to the UK reference population). All studies were conducted in accordance with ethical regulations. The study protocol was reviewed and approved by the Cambridge Local Research Ethics Committee and each subject (or their parent for those under 16 years) provided written informed consent; minors provided oral consent.
Exome sequencing and targeted resequencing was performed in 2548 European ancestry individuals of the GOOS cohort (referred to as SCOOP) and in 1117 ancestry-matched controls16. Eleven rare variants (minor allele frequency <1%) in SRC-1 were identified in this study16; another 8 variants were identified in an earlier data release. Fifteen of these rare variants were identified in severely obese cases and 4 in the control dataset.
Quantification and statistical analysis: The minimal sample size was pre-determined by the nature of experiments. The actual sample size was indicated in each figure legend. The data are presented as mean ± SEM. Statistical analyses were performed using GraphPad Prism to evaluate normal distribution and variations within and among groups. Methods of statistical analyses were chosen based on the design of each experiment and are indicated in figure legends. P < 0.05 was considered to be statistically significant.
Reporting summary
Further information on experimental design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All relevant data are available from the authors. The source data underlying Figs. 1–4 and Supplementary Figs. 1–4 are provided as Source Data files. A Reporting Summary for this Article is available as a Supplementary Information file.
References
Dasgupta, S., Lonard, D. M. & O’Malley, B. W. Nuclear receptor coactivators: master regulators of human health and disease. Annu. Rev. Med. 65, 279–292 (2014).
York, B. & O’Malley, B. W. Steroid receptor coactivator (SRC) family: masters of systems biology. J. Biol. Chem. 285, 38743–38750 (2010).
Picard, F. et al. SRC-1 and TIF2 control energy balance between white and brown adipose tissues. Cell 111, 931–941 (2002).
Zhu, L. et al. Steroid receptor coactivator-1 mediates estrogenic actions to prevent body weight gain in female mice. Endocrinology 154, 150–158 (2013).
Hill, J. W., Elmquist, J. K. & Elias, C. F. Hypothalamic pathways linking energy balance and reproduction. Am. J. Physiol. Endocrinol. Metab. 294, E827–E832 (2008).
Morton, G. J. & Schwartz, M. W. Leptin and the central nervous system control of glucose metabolism. Physiol. Rev. 91, 389–411 (2011).
Ahima, R. S. et al. Role of leptin in the neuroendocrine response to fasting. Nature 382, 250–252 (1996).
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Xu, A. W., Ste-Marie, L., Kaelin, C. B. & Barsh, G. S. Inactivation of signal transducer and activator of transcription 3 in proopiomelanocortin (Pomc) neurons causes decreased pomc expression, mild obesity, and defects in compensatory refeeding. Endocrinology 148, 72–80 (2007).
Bates, S. H. et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 421, 856–859 (2003).
Munzberg, H., Huo, L., Nillni, E. A., Hollenberg, A. N. & Bjorbaek, C. Role of signal transducer and activator of transcription 3 in regulation of hypothalamic proopiomelanocortin gene expression by leptin. Endocrinology 144, 2121–2131 (2003).
Auernhammer, C. J., Bousquet, C. & Melmed, S. Autoregulation of pituitary corticotroph SOCS-3 expression: characterization of the murine SOCS-3 promoter. Proc. Natl. Acad. Sci. U.S.A. 96, 6964–6969 (1999).
Baskin, D. G., Breininger, J. F. & Schwartz, M. W. SOCS-3 expression in leptin-sensitive neurons of the hypothalamus of fed and fasted rats. Regul. Pept. 92, 9–15 (2000).
Giraud, S. et al. Functional interaction of STAT3 transcription factor with the coactivator NcoA/SRC1a. J. Biol. Chem. 277, 8004–8011 (2002).
Xu, J. et al. Partial hormone resistance in mice with disruption of the steroid receptor coactivator-1 (SRC-1) gene. Science 279, 1922–1925 (1998).
Padilla, S. L., Carmody, J. S. & Zeltser, L. M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 16, 403–405 (2010).
Berglund, E. D. et al. Serotonin 2C receptors in pro-opiomelanocortin neurons regulate energy and glucose homeostasis. J. Clin. Invest. 123, 5061–5070 (2013).
Myers, M. G. Jr., Leibel, R. L., Seeley, R. J. & Schwartz, M. W. Obesity and leptin resistance: distinguishing cause from effect. Trends Endocrinol. Metab. 21, 643–651 (2010).
Caron, A. et al. POMC neurons expressing leptin receptors coordinate metabolic responses to fasting via suppression of leptin levels. eLife 7, e33710 (2018).
Xu, J. et al. Genetic identification of leptin neural circuits in energy and glucose homeostases. Nature 556, 505–509 (2018).
Beutler, L. R. et al. Dynamics of gut–brain communication underlying hunger. Neuron 96, 461–475 (2017).
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011).
Pinto, S. et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304, 110–115 (2004).
Hendricks, A. E. et al. Rare variant analysis of human and rodent obesity genes in individuals with severe childhood obesity. Sci. Rep. 7, 4394 (2017).
van de Wall, E. et al. Collective and individual functions of leptin receptor modulated neurons controlling metabolism and ingestion. Endocrinology 149, 1773–1785 (2008).
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42, 983–991 (2004).
Yaswen, L., Diehl, N., Brennan, M. B. & Hochgeschwender, U. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nat. Med. 5, 1066–1070 (1999).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Hartmaier, R. J. et al. A SNP in steroid receptor coactivator-1 disrupts a GSK3beta phosphorylation site and is associated with altered tamoxifen response in bone. Mol. Endocrinol. 26, 220–227 (2012).
Leshan, R. L., Greenwald-Yarnell, M., Patterson, C. M., Gonzalez, I. E. & Myers, M. G. Jr. Leptin action through hypothalamic nitric oxide synthase-1-expressing neurons controls energy balance. Nat. Med. 18, 820–823 (2012).
Morton, G. J. et al. Leptin regulates insulin sensitivity via phosphatidylinositol-3-OH kinase signaling in mediobasal hypothalamic neurons. Cell Metab. 2, 411–420 (2005).
Blouet, C., Ono, H. & Schwartz, G. J. Mediobasal hypothalamic p70 S6 kinase 1 modulates the control of energy homeostasis. Cell Metab. 8, 459–467 (2008).
Minokoshi, Y. et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428, 569–574 (2004).
Dagon, Y. et al. p70S6 kinase phosphorylates AMPK on serine 491 to mediate leptin’s effect on food intake. Cell Metab. 16, 104–112 (2012).
Spencer, T. E. et al. Steroid receptor coactivator-1 is a histone acetyltransferase. Nature 389, 194–198 (1997).
Zhang, X. et al. Hypothalamic IKKbeta/NF-kappaB and ER stress link overnutrition to energy imbalance and obesity. Cell 135, 61–73 (2008).
Ozcan, L. et al. Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab. 9, 35–51 (2009).
Kaneko, K. et al. Neuronal Rap1 regulates energy balance, glucose homeostasis, and leptin actions. Cell Rep. 16, 3003–3015 (2016).
Schneeberger, M. et al. Mitofusin 2 in POMC neurons connects ER stress with leptin resistance and energy imbalance. Cell 155, 172–187 (2013).
Diano, S. et al. Peroxisome proliferation-associated control of reactive oxygen species sets melanocortin tone and feeding in diet-induced obesity. Nat. Med. 17, 1121–1127 (2011).
Thaler, J. P. et al. Obesity is associated with hypothalamic injury in rodents and humans. J. Clin. Invest. 122, 153–162 (2012).
Chen, Y. et al. Enhancement of hypothalamic STAT3 acetylation by nuclear receptor Nur77 dictates leptin sensitivity. Diabetes 64, 2069–2081 (2015).
Xu, Y., O’Malley, B. W. & Elmquist, J. K. Brain nuclear receptors and body weight regulation. J. Clin. Invest. 127, 1172–1180 (2017).
Plum, L. et al. The obesity susceptibility gene Cpe links FoxO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. Nat. Med. 15, 1195–1201 (2009).
Kitamura, T. et al. Forkhead protein FoxO1 mediates Agrp-dependent effects of leptin on food intake. Nat. Med. 12, 534–540 (2006).
Kim, M. S. et al. Role of hypothalamic Foxo1 in the regulation of food intake and energy homeostasis. Nat. Neurosci. 9, 901–906 (2006).
Ren, H. et al. FoxO1 target Gpr17 activates AgRP neurons to regulate food intake. Cell 149, 1314–1326 (2012).
Xu, Y. et al. Distinct hypothalamic neurons mediate estrogenic effects on energy homeostasis and reproduction. Cell Metab. 14, 453–465 (2011).
Musatov, S. et al. Silencing of estrogen receptor alpha in the ventromedial nucleus of hypothalamus leads to metabolic syndrome. Proc. Natl. Acad. Sci. U.S.A. 104, 2501–2506 (2007).
Lu, M. et al. Brain PPAR-gamma promotes obesity and is required for the insulin-sensitizing effect of thiazolidinediones. Nat. Med. 17, 618–622 (2011).
Ryan, K. K. et al. A role for central nervous system PPAR-gamma in the regulation of energy balance. Nat. Med. 17, 623–626 (2011).
Lopez, M. et al. Hypothalamic AMPK and fatty acid metabolism mediate thyroid regulation of energy balance. Nat. Med. 16, 1001–1008 (2010).
Lonard, D. M. & O’Malley, B. W. Molecular pathways: targeting steroid receptor coactivators in cancer. Clin. Cancer Res. 22, 5403–5407 (2016).
Wang, L. et al. Characterization of a steroid receptor coactivator small molecule stimulator that overstimulates cancer cells and leads to cell stress and death. Cancer Cell 28, 240–252 (2015).
Shang, Y. & Brown, M. Molecular determinants for the tissue specificity of SERMs. Science 295, 2465–2468 (2002).
Farooqi, I. S. et al. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N. Engl. J. Med. 341, 879–884 (1999).
Oral, E. A. et al. Leptin-replacement therapy for lipodystrophy. N. Engl. J. Med. 346, 570–578 (2002).
Flier, J. S. & Maratos-Flier, E. Leptin’s physiologic role: does the emperor of energy balance have no clothes? Cell Metab. 26, 24–26 (2017).
Konner, A. C. & Bruning, J. C. Selective insulin and leptin resistance in metabolic disorders. Cell Metab. 16, 144–152 (2012).
Friedman, J. The long road to leptin. J. Clin. Invest. 126, 4727–4734 (2016).
Li, Z., Kelly, L., Heiman, M., Greengard, P. & Friedman, J. M. Hypothalamic amylin acts in concert with leptin to regulate food intake. Cell Metab. 22, 1059–1067 (2015).
Roth, J. D. et al. Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc. Natl. Acad. Sci. U.S.A. 105, 7257–7262 (2008).
Yamada, T. et al. SRC-1 is necessary for skeletal responses to sex hormones in both males and females. J. Bone Mineral Res. 19, 1452–1461 (2004).
Bassett, A. R., Tibbit, C., Ponting, C. P. & Liu, J. L. Highly efficient targeted mutagenesis of Drosophila with the CRISPR/Cas9 system. Cell Rep. 4, 220–228 (2013).
Winnay, J. N., Xu, J., O’Malley, B. W. & Hammer, G. D. Steroid receptor coactivator-1-deficient mice exhibit altered hypothalamic–pituitary–adrenal axis function. Endocrinology 147, 1322–1332 (2006).
Zhang, H., Zhang, G., Gonzalez, F. J., Park, S. M. & Cai, D. Hypoxia-inducible factor directs POMC gene to mediate hypothalamic glucose sensing and energy balance regulation. PLoS Biol. 9, e1001112 (2011).
Farooqi, I. S. et al. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N. Engl. J. Med. 348, 1085–1095 (2003).
Wheeler, E. et al. Genome-wide SNP and CNV analysis identifies common and low-frequency variants associated with severe early-onset obesity. Nat. Genet. 45, 513–517 (2013).
Acknowledgements
Studies in cells and animals were supported by grants from the NIH (R01DK093587, R01DK101379, and R01DK117281 to Y.X.; R01DK114279 to Q.T., R01CA112403 and R01CA193455 to J.X.; R01HD07857, R01HD008818, and P01DK059820 to B.W.O’M.; P0101DK113954 to Y.X., J.X., and B.W.O’M.), USDA/CRIS (6250-51000-059-04S to Y.X.), American Heart Association (to Q.T. and Y.X.), Chinese National Natural Science Foundation (NSFC 81873558 to L.Z.). This work was partially supported by the Cancer Prevention and Research Institute of Texas (RP170005) and the NCI Cancer Center Support Grant (P30CA125123). Measurements of body composition, food intake, and energy expenditure in mice were performed in the Mouse Metabolic Research Unit (MMRU) at the USDA/ARS Children’s Nutrition Research Center, Baylor College of Medicine, which is supported by funds from the USDA ARS (www.bcm.edu/cnrc/mmru). The Pomc-CreER mouse line was provided by Dr. Joel Elmquist (University of Texas Southwestern Medical Center); the SRC-1lox/lox mouse line was provided by Dr. Pierre Chambon (IGBMC/GIE-CERBM). The authors thank BCM Mouse Embryonic Stem Cell Core and Genetically Engineered Mouse Core for their assistance in generating the SRC-1L1376P knock-in mouse. The authors acknowledge the expert assistance of Mr. Firoz Vohra and the MMRU Core Director, Dr. Marta Fiorotto. Studies in humans were supported by the Wellcome Trust (A.A. v d K., T.M.C., I.B., I.S.F.) (098497/Z/12/Z; WT098051; 203513/Z/16/Z), NIHR Cambridge Biomedical Research Centre (I.S.F., I.B., S.O’R.), Bernard Wolfe Health Neuroscience Endowment (I.S.F.). T.M.C. was supported by a Philip Greenwood Clinical Research Fellowship and a Wellcome Trust Research Training Fellowship. The authors are indebted to the participants and their families for their participation and to the Physicians involved in the Genetics of Obesity Study (GOOS) (www.goos.org.uk); whole-exome sequencing was performed as part of the UK10K Consortium (a full list of investigators who contributed to the generation of the human genetics data as part of the UK10K consortium is available from www.UK10K.org.uk).
Author information
Authors and Affiliations
Consortia
Contributions
Y.Y., A.A. v d K., Y.X., and I.S.F. conceived and designed the studies. Y.X. and I.S.F. supervised the project. Y.Y. and L.Z. performed and C.W., P.X., K.S., A.H.J., X.Y., L.L., and J.X. contributed to the experiments involving animals. Y.H. performed electrophysiological studies. Q.T. and B.W.O’M. participated in the design of the animal studies. A.A. v d K., T.M.C., J.M.K., and E.H. contributed to the human studies and with S.O’R. and I.S.F. to the recruitment of the GOOS cohort. T.M.C., L.K.J.S., and M.C.B. performed the molecular studies of human mutations. A.E.H., E.G.B., V.M., K.L.L., I.B., and I.S.F. contributed to the human genetic studies as part of work supported by the UK10K Consortium.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A full list of consortium members appears at the end of the paper.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, Y., van der Klaauw, A.A., Zhu, L. et al. Steroid receptor coactivator-1 modulates the function of Pomc neurons and energy homeostasis. Nat Commun 10, 1718 (2019). https://doi.org/10.1038/s41467-019-08737-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-019-08737-6
This article is cited by
-
The multifaceted therapeutic value of targeting steroid receptor coactivator-1 in tumorigenesis
Cell & Bioscience (2024)
-
Molecular and functional mapping of the neuroendocrine hypothalamus: a new era begins
Journal of Endocrinological Investigation (2024)
-
Updates on Rare Genetic Variants, Genetic Testing, and Gene Therapy in Individuals With Obesity
Current Obesity Reports (2024)
-
Quality of life improvements following one year of setmelanotide in children and adult patients with Bardet–Biedl syndrome: phase 3 trial results
Orphanet Journal of Rare Diseases (2023)
-
Hypothalamic Grb10 enhances leptin signalling and promotes weight loss
Nature Metabolism (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.