Abstract
Physical activity and sleep duration are established risk factors for many diseases, but their aetiology is poorly understood, partly due to relying on self-reported evidence. Here we report a genome-wide association study (GWAS) of device-measured physical activity and sleep duration in 91,105 UK Biobank participants, finding 14 significant loci (7 novel). These loci account for 0.06% of activity and 0.39% of sleep duration variation. Genome-wide estimates of ~ 15% phenotypic variation indicate high polygenicity. Heritability is higher in women than men for overall activity (23 vs. 20%, p = 1.5 × 10−4) and sedentary behaviours (18 vs. 15%, p = 9.7 × 10−4). Heritability partitioning, enrichment and pathway analyses indicate the central nervous system plays a role in activity behaviours. Two-sample Mendelian randomisation suggests that increased activity might causally lower diastolic blood pressure (beta mmHg/SD: −0.91, SE = 0.18, p = 8.2 × 10−7), and odds of hypertension (Odds ratio/SD: 0.84, SE = 0.03, p = 4.9 × 10−8). Our results advocate the value of physical activity for reducing blood pressure.
Similar content being viewed by others
Introduction
Physical inactivity is a global public health threat and is estimated to cost health-care systems ~$50 billion per year worldwide1,2. It is associated with a range of common diseases including multiple cardiometabolic outcomes such as obesity, type 2 diabetes, and cardiovascular diseases3. Alterations in sleep duration also associate with negative health outcomes, including cardiometabolic diseases4 and psychiatric disorders5. In response, recent Canadian public health guidelines recommend how much physical activity and sleep duration youths should engage in for health benefit6.
Twin and family studies have shown that self-reported daily physical activity is heritable, but with a large degree of heterogeneity in measurement methods and sample size (h2 range: 0–78%)7. Recent GWAS studies have indicated a genetic contribution to physical activity, finding three variants8 associated with standard UK Biobank activity metrics9. Recent GWAS have also reported common variants that are associated with sleep duration in UK Biobank10,11,12. However, GWAS of behavioural traits such as physical activity and sleep have mostly relied on self-reported data that are prone to measurement error and thus have limited statistical power. It is also possible that self-reported measures capture how well individuals perceive what they do, rather than what they actually do. Consequently, our understanding of the genes and biological pathways that underpin physical activity and sleep behaviours is still limited.
Here, we report a GWAS in 91,105 UK Biobank participants who were asked to wear activity trackers over a 7-day period. Our use of statistical machine learning, to derive objective measures of physical activity and sleep duration from raw device data, helps identify 14 genetic loci. The associated loci reveal pathways in the central nervous system that are associated with sleep duration and activity behaviours. We also show that physical activity might causally lower diastolic blood pressure and odds of hypertension.
Results
Statistical machine learning of behaviours from sensor data
We examined data from 101,307 UK Biobank participants who agreed to wear a wrist-worn accelerometer for 7 days9 and additionally underwent genome-wide genotyping and imputation13. As part of the UK Biobank Accelerometer Working Group, we generated a continuous phenotype representing overall activity time9 (Methods). Additionally, we applied a machine-learning model, using balanced random forests with Markov confusion matrices14, to identify which one of four activity states {sleep, sedentary, walking, moderate intensity} an individual was in at any given time (Supplementary Data 1). We trained and evaluated the model in 153 free-living individuals (mean age = 42, female n = 100) to distinguish between activity states, evaluated against reference wearable camera, time-use, and sleep diary information sources14. Our model achieved an overall classification score of kappa = 0.68 in correctly predicting what activity state an individual is in for any given 30-s time period (Supplementary Table 1). As detailed in a previous publication14 this model was considered suitable for population inference in UK Biobank as the performance was not materially affected by age and sex characteristics (difference in kappa score: sex < 0.0001; age < 0.05). Therefore, our model was applied to over 100,000 UK Biobank participants (Fig. 1), where the machine-learned phenotypes demonstrated face validity14 with clear and expected differences in sleep duration by self-reported chronotype status (Supplementary Fig. 1). These machine-learned phenotypes provided orthogonal information to the traditional overall activity time phenotype (Supplementary Fig. 2).
Loci associated with physical activity and sleep duration
After quality control, 91,105 participants of European descent and 9,926,106 single-nucleotide polymorphisms (SNPs) remained for subsequent genetic association analysis (Methods). To maximise statistical power, we performed our analyses using the BOLT-LMM software tool15, which applies a linear mixed model to the data, allowing for inclusion of related individuals and those of varying genetic ancestry16. We additionally included assessment centre, genotyping array, age, age squared, and season of wear as covariates.
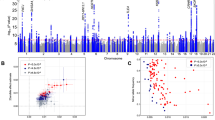
Our analysis identified 14 significant loci (p < 5 × 10−9) that were separated by at least 400 kilobases (kb) (Table 1, Fig. 1, and Supplementary Fig. 3). We empirically derived this threshold (p < 5 × 10−9) for genome-wide significance that considers multiple testing in densely imputed data17. Seven loci were novel and these include: one for overall activity (rs564819152 near SKIDA1, p = 1.9 × 10−9); two for sleep duration (rs2416963 near MAPKAP1, p = 2.3 × 10−10; and rs2006810 near AUTS2, p = 3.9 × 10−9); four for sedentary time (rs26579 near MEF2C-AS2, p = 2.6 × 10−9; rs25981 near EFNA5, p = 3.0 × 10−9; rs1858242 near LOC105377146, p = 3.1 × 10−9; and rs34858520 near CALN1, p = 4.2 × 10−9); while no loci were identified for walking or moderate intensity activity (Table 1 and Supplementary Fig. 4). The remaining associations were known: two for overall and moderate intensity activity8 (near KANSL1-AS1 and SYT4) and six for sleep duration12,18,19,20,21,22 (near PAX8-AS1, MEIS1, LOC101928519, MAPK8IP1P2, OLFM2, and DPYD). Setting genome-wide significance at the conventional threshold of p < 5 × 10−8 would have revealed 13 additional loci, 11 of which are novel (Supplementary Data 2).
We found concordance for loci previously reported using UK Biobank data for overall (near PML) and moderate intensity activity8 (near LINC02210-CRHR1, and SYT4), sleep duration12,19,23,24 (near PUM1, CHCHD5, SAMMSON, BTBD9-AS1, LRP12, EBLN3P, BSX, KCNH5, PRKCB, and RPGRIP1L), and sedentary time (near LINC01930 and LY6H) (Supplementary Data 3). No other findings were confirmed from previous GWAS that relied on self-report measures of physical activity and sleep duration. To account for the likely non-linear U-shape relationship between sleep and disease outcomes25, we ran additional GWAS on short and long sleep, defined as those in the bottom or top quintiles for sleep duration, respectively. We found three loci for short sleep (near PAX8-AS1, MEIS1, and MAPK8IP1P2) that were already identified for sleep duration. We found one locus for long sleep duration (near PAX8-AS1) that was also already identified for sleep duration. This indicates that our sleep duration results are not driven by individuals who are very short or long sleepers.
Genomic control lambda across the GWAS ranged from 1.1 to 1.20, indicating modest deviation in the test statistics compared with the expectation. Estimation of the LD Score intercepts, using LD score regression26, revealed intercepts near 1.00 (range: 0.98–1.02), indicating that the genomic inflation was due to a polygenic architecture rather than inflation due to uncorrected population structure. We performed approximate conditional analysis in each locus reaching genome-wide significance using Genome-wide Complex Trait Analysis27 (GCTA) but did not identify secondary signal in any locus. Gene-based analysis using FUMA28, with input SNPs mapped by position to 18,232 protein-coding genes, identified an additional locus for walking (near HP1BP3) and moderate activity (near KCNA6) (Supplementary Data 4).
Genetic architecture of physical activity and sleep duration
Using heritability estimates from BOLT-LMM15 (Methods), we found all traits to have modest heritability, ranging from 10% for moderate intensity activity to 21% for overall activity (Supplementary Fig. 3). Genome-wide significant loci account for 0.06% (overall activity) to 0.33% (sleep duration) of phenotypic variance across the traits (Supplementary Fig. 3). Additional analyses testing for polygenic trait architecture indicate that SNPs with p-values above genome-wide significance add significantly to the phenotypic variance explained. For example, explained phenotypic variance ranged from 12.9% for sedentary behaviour to 18.1% for sleep duration when we selected independent SNPs (r2 < 0.1) with p < 5 × 10−3 and distance > 250 kb from index SNPs.
We applied partitioned heritability29 analysis for tissue and functional categories using LD score regression26. We identified significant tissue enrichments (p < 1 x 10−3, accounting for ten tissue types and five traits) in the central nervous system for all traits apart from moderate activity. We also identified significant enrichments for adrenal/pancreatic and skeletal muscle tissues for overall activity and sleep duration traits (Supplementary Fig. 5). Regions of the genome annotated as conserved across mammals were enriched (p < 4.7 × 10−4, accounting for 21 functional categories and five traits) for overall activity, sleep duration, and sedentary behaviour traits. These findings support a role for physical activity and sleep behaviours throughout mammalian evolution.
For all sleep duration and activity phenotypes, we used FINEMAP30 to identify credible sets of causal SNPs (meeting a log10 Bayes Factor > 2) in a 1 Mb window around each index SNP, on the assumption that there is a maximum of five causal variants per locus. Three loci contained plausible causal variants in regions spanning < 15 kb (Supplementary Table 2). One locus contained a single plausible causal variant (rs113851554) near MEIS1 in chromosome 2 for sleep duration.
Sexual dimorphism in physical activity and sleep duration
Given that known sex differences exist for physical activity levels9 and muscle and fat mass distribution31, we investigated genetic sexual dimorphisms in our activity traits. Genetic correlations32 of trait architectures as measured using LD score regression26 showed strong correlation between men and women; correlations ranged from 0.92 for overall activity to 0.97 for sleep duration (Supplementary Fig. 6). In addition, there was no evidence of sexual dimorphism using strict criteria for heterogeneity of effects between men and women (p < 5 x 10−9).
Heritability in women was higher than that in men for overall activity (23% vs. 20%, p = 1.5 × 10−4) and sedentary behaviours (18% vs. 15%, p = 9.7 × 10−4) (Supplementary Fig. 6). We found no evidence for sexually dimorphic heritability in sleep duration (p = 0.19), walking (p = 0.54), and moderate intensity activities (p = 0.69). We found some evidence of sexually dimorphic variance explained in 4 of the 5 traits, likely due to the greater sample size of female participants (Supplementary Fig. 6).
Association of physical activity and sleep with other traits
While physical activity and sleep duration are established risk factors for multiple diseases from observational epidemiology, the extent to which their underlying genetic architectures are shared with disease phenotypes is unknown7. We conducted various analyses that included: (1) genome-wide genetic correlations between our traits and those from publicly available GWAS data using LD score regression26 on the LD-Hub web resource33; (2) phenome-wide association (pheWAS) analysis for index SNPs using the Oxford Brain Imaging Genetics Server34 (big.stats.ox.ac.uk); and (3) a lookup of the NHGRI GWAS catalogue35 for related SNPs associated with other diseases and traits (within 400 kb and r2 > 0.2). Activity and sleep traits showed genetic associations with many other independent traits in LD score regression (n = 155), pheWAS (n = 71), and GWAS catalogue lookup (n = 11) analyses (Supplementary Data 5–7). Associations with anthropometric and cognitive health traits were particularly common (Supplementary Note 1). In general, increases in overall activity and walking phenotypes were genetically correlated with improved health status, while increases in sleep duration were negatively correlated with fluid intelligence scores and health status. Increases in sedentary time were genetically correlated with increased fluid intelligence scores, but decreased health status (Supplementary Note 1 and Supplementary Data 5).
To investigate whether activity and sleep might contribute causally to disease outcomes, we performed Mendelian randomisation (MR) analysis in 278,374 UK Biobank participants who were not included in our discovery analysis, and other publicly available GWAS summary data from the MR-Base web platform36,37 (Methods). Rather than selecting all significant traits from the previous genetic correlation analyses, we decided to only focus on major diseases and risk factors. These included cardiovascular disease (CHD, stroke, heart failure), type 2 diabetes, Alzheimer’s, cancer, blood pressure, and anthropometric traits (BMI and body fat %). For the MR analysis of each trait, we selected instrument variables at loci with p < 5 × 10−6, as they explain more phenotypic variance than traditional genome-wide loci (p < 5 × 10−8) with negligible effects on horizontal pleiotropy (Supplementary Fig. 7). We then followed a number of steps to denote potential causality with disease outcomes (Methods, Supplementary Note 2). These included: maximum likelihood estimates38, leave-one-out analyses, robustness against classic confounders and pleiotropy via median and mode estimators, MR-Egger, and bi-directional tests39. We found consistent evidence of inverse relationships for overall activity with body mass index (beta SD of BMI per SD higher overall activity: −0.14, SE = 0.015, p = 8.7 × 10−20), diastolic blood pressure (beta mm Hg per SD: −0.91, SE = 0.18, p = 8.2 × 10−7), and hypertension (Odds ratio per SD: 0.84, SE = 0.03, p = 4.9 × 10−8). (Supplementary Data 8–10 and Supplementary Fig. 8). None of these results appear affected by horizontal pleiotropy or a single variant or association with classic confounders (Supplementary Data 11). However, there was evidence of a bi-directional relationship between adiposity and overall activity (Supplementary Data 12), which is considered plausible40,41 despite some evidence of horizontal pleiotropy.
Potential functional and biological mechanisms
We anticipated the use of objective measures would not only identify activity and sleep associated variants, but also provide insight on potential functional and biological mechanisms for these traits. First, we used DEPICT42 at suggestive loci with p < 1 × 10−5 to identify gene sets enriched for accelerometer-defined phenotype associations; and tissues and cell types in which genes from associated loci are highly expressed. This analysis did not yield any significant results (FDR < 0.05). Secondly, we used the FUMA28 web platform to identify tissues and gene pathways enriched for genetic signals (Methods). Here, we found brain tissues to be enriched (p < 1.8 × 10−4), in particular at the cerebellum, frontal cortex, brain cortex, and anterior cingulate cortex for at least two accelerometer traits (Supplementary Fig. 9). We also identified pathways enriched in neurological disease, brain structure, and cognitive function, among other traits (Supplementary Fig. 10).
Discussion
Our analysis of 91,105 individuals identified 14 loci significantly associated with objectively measured physical activity and sleep traits. While these loci alone account for less than 0.39% of phenotypic variation, our analyses suggest that as much as 18% of physical activity and sleep duration variation might be accounted for by common (minor allele frequency > 1%) genetic variation. We found heritability was higher in women than in men for overall activity and sedentary behaviour, but no differences were found for sleep, walking, and moderate intensity activity time. As anticipated, physical activity and sleep duration have polygenic architectures and share biology with multiple traits including intelligence, education, obesity, and cardiometabolic diseases.
Our results advocate the value of physical activity for the reduction of blood pressure. Until now, it has been difficult to discern whether associations between activity and disease are truly causal or biased due to reverse causality, confounding, and self-report measurement error associated with traditional observational studies43. Ascertainment bias is also an issue in randomised controlled trials of physical activity, due to difficulties in blinding participants to behavioural interventions44. Debate on whether obesity is a determinant of physical activity, or vice-versa, has previously lacked information due to a paucity of instrument variables for physical activity7,40. Our MR analyses indicate that activity and adiposity share a bi-directional relationship. In summary, our MR analyses suggest that higher levels of physical activity might causally lower diastolic blood pressure, and odds of hypertension.
We observed an important role for the central nervous system with respect to physical activity and sleep, consistent with other studies that have used UK Biobank data8. In addition to these findings from heritability partitioning, enrichment and pathway analyses; our gene-set and pheWAS analyses showed genetic overlap with neurodegenerative diseases, mental health wellbeing, and brain structure. Intriguingly, a recent GWAS of BMI highlighted a key role for neurological pathways in overall fat distribution45, while a GWAS of fat distribution as measured by waist-to-hip ratio highlight adipose pathways46. It is, therefore, possible that our findings might be driven through obesity, as we observed strong genetic correlations between activity and obesity traits, and the strength of association for some activity loci was attenuated when including BMI as an additional covariate. However, we have likely only begun to understand the complex interplay between activity, cardiometabolic phenotypes, and neurologic health. Animal models might be informative to answer such questions, as our heritability partitioning analysis supports a role for physical activity and sleep behaviours in regions conserved in mammals.
Our work demonstrates the utility of device measures to identify genetic associations with activity phenotypes. For example, we found that previous self-reported literature has significantly underestimated heritability for physical activity8 (~ 5% vs. ~ 20% for accelerometer-measured traits). The larger-sample sizes that are currently available for self-reported phenotypes results in more identified loci8, however, heritability and potential explained variance appears higher for the objective traits. Recall, comprehension, and social-desirability bias are prevalent in the measurement of behavioural traits such as physical activity, where phenotypic agreement is generally poor between subjective and objective measures (typically r < 0.2)47. It is likely that combination of self-report and objective measures will be needed to further our understanding of the genes and biological pathways that underpin physical activity.
This study has many strengths including: objectively measured behavioural phenotypes validated in free-living environments, use of linear mixed models, and large sample sizes all key to maximise power in genetic discovery analyses; and Mendelian Randomisation analyses, for delineating causal relationships. Given the cohort age range (45–80 years) and inclusion of European ancestry individuals only, we cannot necessarily assume our results generalise to other age groups or ancestral populations. Therefore, replication of our findings in non-European populations with both genetic and accelerometer-measures of sleep duration and physical activity will be important in the future. The high polygenicity of these traits makes it difficult to precisely estimate replication sample sizes. Assuming the replication p-value is set at 0.05/15 loci = 3.3 × 10−3, and variance explained by a SNP ranges between 1.5–5.0 × 10−4, we estimate between ~ 36 and ~ 119 k participants are necessary to provide 90% power to replicate the loci listed in Table 1. Replication in other cohorts would also help refine estimates of phenotypic variance explained by genome-wide significant loci, which are currently calculated in the discovery dataset reported in this paper. To account for the likely U-shape relationship between sleep and disease outcomes25, rather than a linear relationship as assumed in current models, future work will be also required for MR sleep analysis. To develop machine-learned models of sleep duration in free-living environments, we did not use traditional cumbersome polysomnography methods, and instead relied on self-reported sleep diaries which can be prone to measurement error. While the participants in our machine learning free-living training dataset are not a random subsample of the UK Biobank study, the size of our training dataset has helped provide a diverse set of representative behaviours, rather than individuals, which is important for model development14.
In summary, our GWAS of device-measured physical activity and sleep duration has identified 14 associated genetic loci. Our analysis shows shared genetic pathways with multiple traits including intelligence-related phenotypes and cardiometabolic disease. In support of national and international clinical guidance, this study provides strong evidence for physical activity in the prevention of blood pressure.
Methods
Participants
Study participants were from the UK Biobank13 where a subset of 103,712 participants agreed to wear a wrist-worn accelerometer for a 7-day-period between 2013 and 20159. Participants who were slightly more likely to consent included: women, those aged 55–74 years, those with higher socio-economic status, better physical health status, and less time since the baseline assessment (all odds ratios < 1.1). There were no significant differences in consent by self-reported physical activity status. This study (UK Biobank project #9126) was covered by the general ethical approval for UK Biobank studies from the NHS National Research Ethics Service on 17th June 2011 (Ref 11/NW/0382). For the development and free-living evaluation of accelerometer machine-learning methods, we used a validation set of 153 participants recruited to the CAPTURE-24 and ENERGY-24 studies where adults aged 18–91 were recruited from the Oxford region in 2014–201514,48. Participants were asked to wear a wrist-worn accelerometer (same as in UK Biobank) for a 24-h period and then given a £20 voucher for taking part in this study that received ethical approval from University of Oxford (Inter-Divisional Research Ethics Committee (IDREC) reference number: SSD/CUREC1A/13–262).
Device
Participants wore an Axivity AX3 wrist-worn triaxial accelerometer on their dominant hand at all times over a 7-day-period. It was set to capture triaxial acceleration data at 100 Hz with a dynamic range of + −8g. This device has demonstrated equivalent output49 to the GENEActiv accelerometer, which has been validated using both standard laboratory and free-living energy expenditure assessment methods50,51. For data pre-processing we followed procedures that we developed as part of the UK Biobank accelerometer data processing expert group9, that included device calibration, resampling to 100 Hz, and removal of noise and gravity.
Development and validation of machine-learned phenotypes
To create a reference set of labels for sleep, sitting/standing, and walking time, participants in the validation study also wore cameras in natural free-living, rather than constrained laboratory, environments. Wearable cameras automatically take photographs every ~ 20 s, have up to 16 h battery life and storage capacity for over 1 week’s worth of images. When worn, the camera is reasonably close to the wearer’s eye line and has a wide-angle lens to capture everything within the wearer’s view. Each image is time-stamped so duration of sedentary behaviour, waking, and a range of other physical activity behaviours can be captured52. To extract sleep information, participants were asked to complete a simple sleep diary, as used in the Whitehall study, which consisted of two questions53: ‘what time did you first fall asleep last night?’ and ‘what time did you wake up today (eyes open, ready to get up)?’. Participants were also asked to complete a Harmonised European Time Use Survey diary54, and sleep information from here was extracted in cases where data was missing from the simple sleep diary. An overview of the distribution of activity behaviours in this training dataset is provided in Supplementary Fig. 11.
For every non-overlapping 30-s time window, we then extracted a 126-dimensional feature vector representing a range of time and frequency domain features14. For activity classification we used random forests55 which offer a powerful nonparametric discriminative method for classification that offers state-of-the-art performance56. Random forests are able to classify data points, but do not have an understanding of our data as having come from a time series. Therefore we then used a hidden Markov model57 (HMM) to smooth our predictions. This smoothing corrects erroneous predictions from the random forest, such as where the error is a blip of one activity surrounded by another and the transitions between those two classes of activity are rare. This allowed us to train a model using all free-living ground truth data, achieving 79% accuracy (kappa = 0.68) across classes of interest14 (Supplementary Table 1).
Physical activity and sleep duration in UK Biobank
To predict sleep, sedentary, walking, and moderate intensity activity time, we applied our machine-learning method to predict behaviour for each 30-s epoch in 103,712 UK Biobank participants’ accelerometer data. A sedentary behaviour is one which has a MET (Metabolic Equivalent of Task) energy expenditure score of ≤ 1.5 and occurs in a sitting, lying, or reclining posture58. As an exception to this rule we also categorised the following annotations as sedentary behaviour: driving (code 16010) where assigned MET score is 2.5, and some instances of non-desk work (code 21010 rather than 5080). The complete list of image derived annotations and the phenotype class mappings are available in Supplementary Data 1. For any given time window (e.g., 1 h, 1 day, etc.) the probability of a participant engaging in a specific behaviour type was expressed as the number-of-epoch-predictions-for-class divided by the number-of-epochs. For overall activity levels, we selected average vector magnitude for each 30-s epoch, which is the recommended variable for activity analysis9. This variable has been shown to account for 44–47% explained variance vs. combined sensing heart-rate + trunk-acceleration (a proxy for free-living physical activity energy expenditure) in 1695 UK adults51.
Device non-wear-time was automatically identified as consecutive stationary episodes lasting for at least 60 min9. These non-wear segments of data were imputed with the average of similar time-of-day data points, for each behaviour prediction, from different days of the measurement. We excluded participants whose data could not be calibrated (n = 0), had too many clipped values (n = 3), had unrealistically high values (average vector magnitude > 100 mg) (n = 32), or who had poor wear-time (n = 6860). We defined minimum wear-time criteria as having at least 3 days (72 h) of data and also data in each 1-h period of the 24-h cycle9.
Genotyping and quality control
The UK Biobank provides ~ 92 million variants, including imputation based on UK10K haplotype, 1000 Genomes Phase 3, and Haplotype Reference Consortium (HRC) reference panels59. We excluded SNPs with MAF < 0.1% (~ 83 M) and imputation R2 < 0.3 (~9 k). We removed participants who self-reported as being non-white (n = 3192), had abnormal genetic versus self-reported sex mismatches (n = 36) or sex chromosome aneuploidy (n = 71). This left 91,105 participants of European descent (39,968 men; 51,137 women), and 9,926,106 SNPs, for subsequent genetic association analysis.
Identifying loci associated with physical activity and sleep
To improve our power to detect associations, we used BOLT-LMM15 to perform linear mixed models analysis. As this adjusts for population structure and relatedness, we could include many additional individuals (n = 13,320) than if following the common practice of analysing a reduced set of unrelated white British individuals using linear regression60. We included assessment centre, genotyping array, age, age squared, and season as covariates. Sensitivity analysis using LD score regression26 on the overall activity trait showed we did not need to include principal components of ancestry as covariates in our linear mixed models (lambda = 1.20 vs. 1.20, LD intercept = 1.0085 vs. 1.0092, genome-wide genetic correlation (Rg) = 0.99). We decided against including too many covariates to avoid the introduction of any possible collider bias that could have an effect on downstream analyses. In addition, we found little difference with the inclusion of: (1) sex as an additional covariate (RG = 0.99); (2) BMI as an additional covariate (RG = 0.93); and (3) both sex and BMI as additional covariates (RG = 0.93). Supplementary Data 2 reports a sensitivity analysis for each locus with sex and BMI included as additional covariates, where all attenuations were driven by BMI alone. These results should be cautiously interpreted due to the potential for collider bias when adjusting for BMI.
We used PLINK61 (version 1.9) to exclude candidate variants (p < 1 × 10−5) that deviated from Hardy-Weinberg equilibrium (p < 1 × 10−7). To identify genome-wide-significant (GWS) loci, we defined a distance criterion of + −400 kb surrounding each GWS peak (p < 5 × 10−9). We empirically derived this threshold for genome-wide significance that considers multiple testing and densely imputed data17. For reporting, we also identified loci using a traditional GWS value of p < 5 × 10−8.
To identify novel loci, we considered index SNPs falling outside 400 kb of a SNP previously associated with one of the following: self-report sleep duration or physical activity from the NHGRI-EBI GWAS catalogue35, CHARGE21 and CARe62 consortia; an analysis of our accelerometer-measured overall and moderate intensity activity variables9 with loci reported in 91,000 UK Biobank participants8; accelerometer-measured sleep duration genes tested for replication in recent bioRxiv papers that used ~ 85,000 UK Biobank participants22,23; self-reported overall activity loci reported in bioRxiv papers using ~ 377,000 and ~ 120,000 UK Biobank participants, respectively8,63; self-reported sleep duration GWAS results in recent bioRxiv papers that used up to ~ 446,000 UK Biobank participants12,24; and sleep duration (field #1160), television watching time as a proxy for sitting (field #1070), and walking (field #874) GWAS results in ~ 330,000 UK Biobank participants published by the Neale lab at the Broad Institute.
To identify additional signals in regions of association, we performed approximate joint and conditional SNP association analysis in each locus using the Genome-wide Complex Trait Analysis27 (GCTA) tool. Any lead SNPs identified in a known high-LD area64 between 43.5 and 45.5 mb at chromosome 17 were treated as a single large locus in GCTA analysis.
Gene-based analysis were performed with MAGMA65 v1.6, as implemented on the FUMA28 web platform. Input SNPs were mapped by position to genes obtained from 18,232 protein-coding genes obtained via Ensembl66 build 85. Bonferroni-correction was used to define genome-wide significance. To identify novel loci, we looked for genes more than 400 kb away from loci identified in the above SNP association analysis.
Genetic architecture of physical activity and sleep duration
To estimate heritability of each trait, we used BOLT-LMM15 for computational efficiency, after sensitivity analysis showed negligible differences in estimates between BOLT-LMM and restricted maximum likelihood analysis67 implemented using the BOLT-REML software tool. We applied partitioned heritability29 analysis across the phenotypes by tissue and functional category using LD score regression26 with the LDSC tool. Significant enrichments for individual traits were identified using thresholds of p < 1 × 10−3 for tissues (i.e., p < 0.05/10 cell types/5 traits) and p < 3.57 × 10−4 for functional categories (i.e., p < 0.05/28 categories/5 traits). We also investigated enrichments in the median value across all accelerometer-measured traits.
To calculate the explained phenotypic variance for each trait, we used the PRSice68 tool to generate polygenic risk scores for the lead SNP in GWS loci and also for all SNPs with p < 5 × 10−3, distance > 250 kb from index SNPs, and r2 < 0.1. The same participant inclusion criteria were used as for association analysis.
To identify plausible causal SNPs associated with sleep and activity phenotypes, we used the FINEMAP30 software. Configurations of plausible causal SNPs from a 1 Mb window around each genome-wide significant locus were calculated on the assumption that there were a maximum of five causal variants per locus. Across all loci, we defined plausible causal SNPs as those meeting a log10 Bayes Factor > 2.
Sexual dimorphism in physical activity and sleep duration
To investigate potential sources of sex heterogeneity, we ran the aforementioned genome-wide association analyses for men and women independently. Genetic correlations32 of trait architectures between men and women were measured using LD score regression26 for each phenotype. We also tested for heterogeneity of effect estimates31 between men and women for all SNPs using the EasyStrata69 tool, with p < 5 × 10−9 to assess significance. Heritability differences between men and women for each trait were assessed by extracting a two-tailed p-value from the z-score in equation (1), where var indicates variance.
To estimate genetic contributions to phenotypic variance for men and women separately, we selected index SNPs from loci with a less stringent p < 5 × 10−7, due to smaller sample sizes within strata.
Association of physical activity and sleep with other traits
To estimate genetic correlations between our accelerometer-defined phenotypes and other complex traits and diseases, we used LD score regression26 implemented in the LD-Hub web resource33. To assess significance, we corrected for 4160 tests (5 traits x 832 phenotypes available on LD-Hub) with p < 1.2 × 10−5.
To examine whether the genome-wide significant SNPs identified in our analysis affected other traits, we used the Oxford Brain Imaging Genetics Server (big.stats.ox.ac.uk34) to perform a phenome-wide association study (PheWAS) on almost 4000 traits in UK Biobank participants. This included GWAS results of ~ 2000 phenotypes in ~ 330,000 UK Biobank participants published by the Neale lab at the Broad Institute, and the remaining traits were brain imaging-derived phenotypes measured on a subset of ~ 10,000 UK Biobank participants34. To assess significance, we corrected for 40,000 tests (10 loci x 4000 traits) with p < 1.25 × 10−6. Additionally, we extracted previously reported GWAS associations within 400 kb and r2 > 0.2 of accelerometer index SNPs from the NHGRI GWAS catalogue35.
To investigate whether activity and sleep might causally contribute to disease outcomes, we performed Mendelian Randomisation (MR) analysis. Rather than selecting all significant traits identified in other correlation analysis, we decided to concentrate on major diseases and peripheral risk factors. These included vascular disease (CHD, stroke, heart failure), diabetes, Alzheimer’s, major cancer subtypes, blood pressure, and anthropometric traits (BMI and body fat %). Disease phenotypes were prepared following similar procedures as used for UK Biobank variables in the LD-Hub web resource33. We defined hypertensive cases as individuals with systolic blood pressure of > 140 mm Hg, or a diastolic blood pressure of > 90 mm Hg, or the report of blood pressure medication usage. For the analysis of systolic and diastolic blood pressure, we corrected blood pressure measures in people on antihypertensive drugs by adding 15 mm Hg to systolic and 10 mm Hg to diastolic blood pressure, in keeping with the approach taken by genome-wide association studies70. Similar to the LD-Hub web resource33, we used linear regression analyses with sex and the first 10 principal components as covariates. For linear regression, we used the bgenie tool59. We removed participants from the accelerometer discovery sample (n = 91,112), those who self-reported as being non-white British (n = 68,428), had abnormal genetic vs. self-reported sex mismatches (n = 192) or sex chromosome aneuploidy (n = 652). We also removed 48,658 participants due to relatedness. This left 278,374 UK Biobank participants who were not included in our discovery analysis, and other publicly available GWAS summary data from the MR-Base web platform36. Where possible, estimates were meta-analysed using a fixed effects model (inverse variance weighted average).
For analysis we retained index SNPs with p < 5 × 10−6 that were pruned for LD (r2 < 0.001) and more than 10,000 kb apart. These loci (p < 5 × 10−6) explain more phenotypic variance than traditional genome-wide loci (p < 5 × 10−8) with negligible effects on horizontal pleiotropy (Supplementary Fig. 7). We then followed a number of steps to denote potential causality with disease outcomes (Supplementary Note 2). We used the maximum likelihood-based approach as our primary source of MR estimates. This is based on published simulation results suggesting that causal estimates obtained from summarised data using a likelihood-based model are almost as precise as those obtained from individual-level data38. Only likelihood-based risk estimates that were significant after Bonferroni-correction were considered. The potential effect of pleiotropy was evaluated by three complementary approaches, namely weighted median and weighted mode estimation71,72, and the regression intercept from the MR-Egger method73. The sensitivity of causal inference to any individual genetic variant was tested by leave-one-out analysis. The Steiger test was used to provide evidence for the causal direction of the effect estimates74. Sensitivity analyses was also conducted to test whether instrument variables were associated with the following observational ‘classic’ confounders: income, smoking, area deprivation, and years of education75. Potential associations with confounders were subsequently followed-up by multivariate mendelian randomisation analyses76 to investigate the robustness of these associations to adjustment for potential confounders. Additionally, for MR associations that appeared robust to sensitivity and pleiotropy analyses, bi-directional MR was conducted to assess the direction of the causal estimate. Instrument variables for bi-directional analyses that relied on estimates generated from UK Biobank were re-estimated on participants not in our discovery GWAS accelerometer dataset. All MR analyses were conducted in R using the TwoSampleMR package36.
Investigating functional and biological mechanisms
To investigate potential biological mechanisms underlying physical activity and sleep, we used DEPICT42 at suggestive loci with p < 1 × 10−5 to identify: the most likely causal gene; reconstituted gene sets enriched for accelerometer-defined phenotype associations; and tissues and cell types in which genes from associated loci are highly expressed. Next, we used the FUMA web platform28 to perform tissue enrichment analysis where the full distribution of SNPs was tested with 53 specific tissue types, based on GTEx77 data. To identify significant enrichments, we accounted for multiple testing across 53 tissues and five traits (p < 1.88 x 10−4). To then identify pathways implicated by the activity and sleep associated loci, we used FUMA to perform hypergeometric tests on genes from these loci to investigate over-representations in genes predefined from the GWAS catalogue.
Data availability
The summary phenotype variables that we have constructed will be made available as a part of the UK Biobank Returns Catalogue at http://biobank.ctsu.ox.ac.uk/crystal/docs.cgi?id = 1. All accelerometer data processing, feature extraction, and machine learning code are available at https://github.com/activityMonitoring. GWAS summary statistics, including with and without adjustment for BMI and sex as covariates, can be downloaded from https://doi.org/10.5287/bodleian:yJp6zZmdj.
References
Ding, D. et al. The economic burden of physical inactivity: a global analysis of major non-communicable diseases. Lancet 388, 1311–1324 (2016).
Althoff, T. et al. Large-scale physical activity data reveal worldwide activity inequality. Nature 547, 336–339 (2017).
Lee, I. -M. et al. Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy. Lancet 380, 219–229 (2012).
Cappuccio, F. P., Cooper, D., D’Elia, L., Strazzullo, P. & Miller, M. A. Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. Eur. Heart J. 32, 1484–1492 (2011).
Wulff, K., Gatti, S., Wettstein, J. G. & Foster, R. G. Sleep and circadian rhythm disruption in psychiatric and neurodegenerative disease. Nat. Rev. Neurosci. 11, 589–599 (2010).
Tremblay, M. S., Carson, V. & Chaput, J. -P. Introduction to the Canadian 24-hour movement guidelines for children and youth: An integration of physical activity, sedentary behaviour, and sleep. Appl. Physiol. Nutr. Metab. 41, iii–iv (2016).
Bauman, A. E. et al. Correlates of physical activity: why are some people physically active and others not? Lancet 380, 258–271 (2012).
Klimentidis, Y. C. et al. Genome-wide association study of habitual physical activity in over 377,000 UK Biobank participants identifies multiple variants including CADM2 and APOE. Int. J. Obes. 42, 1161–1176 (2018).
Doherty, A. et al. Large scale population assessment of physical activity using wrist worn accelerometers: The UK Biobank Study. PLoS ONE 12, e0169649 (2017).
Hammerschlag, A. R. et al. Genome-wide association analysis of insomnia complaints identifies risk genes and genetic overlap with psychiatric and metabolic traits. Nat. Genet. 49, 1584–1592 (2017).
Lane, J. M. et al. Genome-wide association analysis identifies novel loci for chronotype in 100,420 individuals from the UK Biobank. Nat. Commun. 7, 10889 (2016).
Jansen, P. R. et al. Genome-wide analysis of insomnia (N=1,331,010) identifies novel loci and functional pathways. bioRxiv 214973, https://doi.org/10.1101/214973 (2018).
Sudlow, C. et al. UK Biobank: An open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS. Med. 12, e1001779 (2015).
Willetts, M., Hollowell, S., Aslett, L., Holmes, C. & Doherty, A. Statistical machine learning of sleep and physical activity phenotypes from sensor data in 96,220 UK Biobank participants. Sci. Rep. 8, 7961 (2018).
Loh, P. -R. et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat. Genet. 47, 284–290 (2015).
Yang, J., Zaitlen, N. A., Goddard, M. E., Visscher, P. M. & Price, A. L. Advantages and pitfalls in the application of mixed-model association methods. Nat. Genet. 46, 100–106 (2014).
Pulit, S. L., de With, S. A. J. & de Bakker, P. I. W. Resetting the bar: Statistical significance in whole-genome sequencing-based association studies of global populations. Genet. Epidemiol. 41, 145–151 (2017).
Lane, J. M. et al. Genome-wide association analyses of sleep disturbance traits identify new loci and highlight shared genetics with neuropsychiatric and metabolic traits. Nat. Genet. 49, 274–281 (2016).
Jones, S. E. et al. Genome-wide association analyses in 128,266 individuals identifies new morningness and sleep duration loci. PLoS Genet. 12, e1006125 (2016).
Lane, J. M. et al. Genome-wide association analysis identifies novel loci for chronotype in 100,420 individuals from the UK Biobank. Nat. Commun. 7, 10889 (2016).
Gottlieb, D. J. et al. Novel loci associated with usual sleep duration: the CHARGE Consortium Genome-Wide Association Study. Mol. Psychiatry 20, 1232–1239 (2015).
Lane, J. M. et al. Biological and clinical insights from genetics of insomnia symptoms. bioRxiv 257956, https://doi.org/10.1101/257956 (2018).
Jones, S. E. et al. Genetic studies of accelerometer-based sleep measures in 85,670 individuals yield new insights into human sleep behaviour. bioRxiv 303925, https://doi.org/10.1101/303925 (2018).
Dashti, H. et al. GWAS in 446,118 European adults identifies 78 genetic loci for self-reported habitual sleep duration supported by accelerometer-derived estimates. bioRxiv 274977, https://doi.org/10.1101/274977 (2018).
Hirshkowitz, M. et al. National Sleep Foundation’s sleep time duration recommendations: methodology and results summary. Sleep Heal 1, 40–43 (2015).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nat. Genet. 44, 369–375 (2012).
Watanabe, K., Taskesen, E., van Bochoven, A. & Posthuma, D. Functional mapping and annotation of genetic associations with FUMA. Nat. Commun. 8, 1826 (2017).
Finucane, H. K. et al. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat. Genet. 47, 1228–1235 (2015).
Benner, C. et al. FINEMAP: efficient variable selection using summary data from genome-wide association studies. Bioinformatics 32, 1493–1501 (2016).
Randall, J. C. et al. Sex-stratified Genome-wide Association Studies including 270,000 individuals show sexual dimorphism in genetic loci for anthropometric traits. PLoS Genet. 9, e1003500 (2013).
Bulik-Sullivan, B. et al. An atlas of genetic correlations across human diseases and traits. Nat. Genet. 47, 1236–1241 (2015).
Zheng, J. et al. LD Hub: a centralized database and web interface to perform LD score regression that maximizes the potential of summary level GWAS data for SNP heritability and genetic correlation analysis. Bioinformatics 33, 272–279 (2017).
Elliott, L. T. et al. Genome-wide association studies of brain imaging phenotypes in UK Biobank. Nature 562, 210–216 (2018).
MacArthur, J. et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucl. Acids Res. 45, D896–D901 (2017).
Hemani, G. et al. The MR-Base platform supports systematic causal inference across the human phenome. eLife 7, e34408 (2018).
Lu, Y. et al. New loci for body fat percentage reveal link between adiposity and cardiometabolic disease risk. Nat. Commun. 7, 10495 (2016).
Burgess, S., Butterworth, A. & Thompson, S. G. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet. Epidemiol. 37, 658–665 (2013).
Bowden, J. et al. A framework for the investigation of pleiotropy in two-sample summary data Mendelian randomization. Stat. Med. 36, 1783–1802 (2017).
Richmond, R. C. et al. Assessing causality in the association between child adiposity and physical activity levels: a Mendelian randomization analysis. PLoS Med. 11, e1001618 (2014).
Wade, K. H., Richmond, R. C. & Davey Smith, G. Physical activity and longevity: how to move closer to causal inference. Br. J. Sports Med. 52, 890–891 (2018).
Pers, T. H. et al. Biological interpretation of genome-wide association studies using predicted gene functions. Nat. Commun. 6, 5890 (2015).
Smith, G. D. & Ebrahim, S. Data dredging, bias, or confounding. BMJ 325, 1437–1438 (2002).
Cornelissen, V. A. & Smart, N. A. Exercise training for blood pressure: a systematic review and meta-analysis. J. Am. Heart Assoc. 2, e004473 (2013).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 (2015).
Shungin, D. et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 518, 187–196 (2015).
Sabia, S. et al. Association between questionnaire- and accelerometer-assessed physical activity: The role of sociodemographic factors. Am. J. Epidemiol. 179, 781–790 (2014).
Topouzi, M., Grunewald, P., Gershuny, J. & Harms, T. Everyday household practices and electricity use: early findings from a mixed-method approach to assign demand flexibility. in 4th European Conference on Behaviour and Energy Efficiency (Springer Verlag, Berlin, 2016).
Rowlands, A. V. et al. Accelerometer-assessed physical activity in epidemiology: Are monitors equivalent? Med. Sci. Sports Exerc. 50, 257–265 (2018).
Esliger, D. W. et al. Validation of the GENEA Accelerometer. Med Sci Sports Exerc 43, 1085–1093 (2011).
White, T., Westgate, K., Wareham, N. J. & Brage, S. Estimation of physical activity energy expenditure during free-living from wrist accelerometry in UK adults. PLoS ONE 11, e0167472 (2016).
Doherty, A. R. et al. Wearable cameras in health: The state of the art and future possibilities. Am. J. Prev. Med. 44, 320–323 (2013).
van Hees, V. T. et al. A novel, open access method to assess sleep duration using a wrist-worn accelerometer. PLoS ONE 10, e0142533 (2015).
Eurostat. Harmonised European Time Use Surveys: 2008 Guidelines. Eurostat ISBN:978-92-79-07853-8 (European Commission, 2009).
Breiman, L. Random forests. Mach. Learn. 45, 5–32 (2001).
Fernández-Delgado, M., Cernadas, E., Barro, S. & Amorim, D. Do we need hundreds of classifiers to solve real world classification problems? J. Mach. Learn. Res. 15, 3133–3181 (2014).
Rabiner, L. & Juang, B. An introduction to hidden Markov models. IEEE ASSP Mag. 3, 4–16 (1986).
Tremblay, M. S. et al. Sedentary Behavior Research Network (SBRN) – Terminology Consensus Project process and outcome. Int. J. Behav. Nutr. Phys. Act. 2017 141 14, 75 (2017).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209 (2018).
Loh, P. -R., Kichaev, G., Gazal, S., Schoech, A. P. & Price, A. L. Mixed-model association for biobank-scale datasets. Nat. Genet. 50, 906–908 (2018).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Cade, B. E. et al. Common variants in DRD2 are associated with sleep duration: the CARe consortium. Hum. Mol. Genet. 25, 167–179 (2016).
Tikkanen, E., Gustafsson, S. & Ingelsson, E. Associations of fitness, physical activity, strength, and genetic risk with cardiovascular disease: Longitudinal analyses in the UK biobank study. Circulation 137, 2583–2591 (2018).
Stefansson, H. et al. A common inversion under selection in Europeans. Nat. Genet. 37, 129–137 (2005).
de Leeuw, C. A., Mooij, J. M., Heskes, T. & Posthuma, D. MAGMA: generalized gene-set analysis of GWAS data. PLoS Comput. Biol. 11, e1004219 (2015).
Zerbino, D. R. et al. Ensembl 2018. Nucl. Acids Res. 46, D754–D761 (2018).
Yang, J. et al. Common SNPs explain a large proportion of the heritability for human height. Nat. Genet. 42, 565–569 (2010).
Euesden, J., Lewis, C. M. & O’Reilly, P. F. PRSice: Polygenic Risk Score software. Bioinformatics 31, 1466–1468 (2015).
Winkler, T. W. et al. EasyStrata: evaluation and visualization of stratified genome-wide association meta-analysis data. Bioinformatics 31, 259–261 (2015).
Kraja, A. T. et al. New blood pressure-associated loci identified in meta-analyses of 475 000 individuals. Circ. Cardiovasc. Genet. 10, e001778 (2017).
Bowden, J., Davey Smith, G., Haycock, P. C. & Burgess, S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet. Epidemiol. 40, 304–314 (2016).
Hartwig, F. P., Davey Smith, G. & Bowden, J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int. J. Epidemiol. 46, 1985–1998 (2017).
Bowden, J., Davey Smith, G. & Burgess, S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int. J. Epidemiol. 44, 512–525 (2015).
Hemani, G., Tilling, K. & Davey Smith, G. Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 13, e1007081 (2017).
Okbay, A. et al. Genome-wide association study identifies 74 loci associated with educational attainment. Nature 533, 539–542 (2016).
Burgess, S., Dudbridge, F. & Thompson, S. G. Re: “Multivariable Mendelian randomization: The use of pleiotropic genetic variants to estimate causal effects”. Am. J. Epidemiol. 181, 290–291 (2015).
GTEx Consortium, Gte. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Acknowledgements
This research has been conducted using the UK Biobank resource under application number 11867. We would like to thank all participants for agreeing to volunteer in this research. Tugce Karaderi and Sven Hollowell contributed to data preparation. Matthew Willetts and Louis Aslett contributed to the development of machine-learning methods to identify activity phenotypes. We would also like to thank Benjamin Neale at the Broad Institute for advice on statistical testing for heritability differences between stratified groups. For data collection at Oxford and annotation we would like to acknowledge the support of Jonathan Gershuny, the UK Economic and Social Research Council (grant number ES/L011662/1), Charlie Foster, Paul Kelly, Teresa Harms, Emma Thomas, Karen Milton, Nicole Grey, Wong Tsz Yan, Salma Haque, Marina Topouzi, Sarah Darby, and Philipp Grunewald. The UK Biobank Activity Project and the collection of activity data from participants was funded by the Wellcome Trust (https://wellcome.ac.uk/) and the Medical Research Council (http://www.mrc.ac.uk/). The analysis was supported by: the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre (BRC) [A.D., M.V.H., C.M.L.]; the British Heart Foundation Centre of Research Excellence at Oxford (http://www.cardioscience.ox.ac.uk/bhf-centre-of-research-excellence) [grant number RE/13/1/30181 to A.D.]; the Li Ka Shing Foundation (http://www.lksf.org/) [to A.D., C.M.L.]; Welcome Trust-SSI/John Fell funds Oxford [CML]; European Commission Horizon 2020 Widenlife [C.M.L.]; and NIH [grant number CRR00070 CR00.01 to C.M.L.]. M.V.H. works in a unit that receives funding from the UK Medical Research Council and is supported by a British Heart Foundation Intermediate Clinical Research Fellowship (FS/18/23/33512). We would also like to acknowledge the use of the University of Oxford Advanced Research Computing (ARC) facility in carrying out this work. http://dx.doi.org/10.5281/zenodo.22558 The MRC and Wellcome Trust played a key role in the decision to establish UK Biobank, and the accelerometer data collection. No funding bodies had any role in the analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
A.D., C.H., and C.M.L. conceived this study. A.D. developed and implemented the analyses, with input from T.F., and S.P. K.S.-B., and A.D. implemented the Mendelian Randomisation analysis with input from M.H. A.D. wrote the paper. A.D., C.M.L., S.P., M.H., C.H., T.F., and K.S.-B. were involved in interpreting results and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Doherty, A., Smith-Byrne, K., Ferreira, T. et al. GWAS identifies 14 loci for device-measured physical activity and sleep duration. Nat Commun 9, 5257 (2018). https://doi.org/10.1038/s41467-018-07743-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-07743-4
This article is cited by
-
Sleep patterns and risks of incident cardiovascular disease and mortality among people with type 2 diabetes: a prospective study of the UK Biobank
Diabetology & Metabolic Syndrome (2024)
-
Dietary factors in relation to hypertension: a mendelian randomization study
Journal of Health, Population and Nutrition (2024)
-
Causal links between sedentary behavior, physical activity, and psychiatric disorders: a Mendelian randomization study
Annals of General Psychiatry (2024)
-
NiMBaLWear analytics pipeline for wearable sensors: a modular, open-source platform for evaluating multiple domains of health and behaviour
BMC Digital Health (2024)
-
Self-supervised learning of accelerometer data provides new insights for sleep and its association with mortality
npj Digital Medicine (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.