Abstract
Thermochemical oxidation of methane (TOM) by high-valence metal oxides in geological systems and its potential role as a methane sink remain poorly understood. Here we present evidence of TOM induced by high-valence metal oxides in the Junggar Basin, located in northwestern China. During diagenesis, methane from deeper source strata is abiotically oxidized by high-valence Mn(Fe) oxides at 90 to 135 °C, releasing 13C-depleted CO2, soluble Mn2+ and Fe2+. Mn generally plays the dominant role compared to Fe, due to its lower Gibbs free energy increment during oxidation. Both CO2 and metal ions are then incorporated into authigenic calcites, which are characterized by extremely negative δ13C values (−70 to −22.5‰) and high Mn content (average MnO = 5 wt.%). We estimate that as much as 1224 Tg of methane could be oxidized in the study area. TOM is unfavorable for gas accumulation but may act as a major methane sink in the deep crustal carbon cycle.
Similar content being viewed by others
Introduction
Methane (CH4) is an economically important fossil fuel, and by 2014 about 4.3 × 1014 m3 or 4.7 × 105 Tg of proven natural gas reserves, including shale gas, had been identified in sedimentary basins worldwide1,2. However, methane is also a significant greenhouse gas, second only to CO2 in the contribution to the current global greenhouse effect3,4; during certain intervals of the geologic past, such as the Archean, it was perhaps the most significant greenhouse gas in the atmosphere5,6. Since the late Holocene pre-industrial era, the concentration of atmospheric methane had increased sharply from an average of 680 p.p.b.v. (parts per billion volume) during AD 800–1600 to 1799 p.p.b.v. by the year 20104,7. Furthermore, the current emission flux of methane from sedimentary basins to the atmosphere is estimated to be 10 to 25 Tg yr−1, which is a key to understanding the global carbon cycle and climate change8,9.
Oxidation reactions are the key mechanism by which methane is consumed before emission into the atmosphere. Methane can theoretically be oxidized via two distinct geological processes. The first is anaerobic oxidation of methane (AOM), where methane oxidation is mediated by sulfate- or metal-reducing bacteria in an anoxic environment, typically resulting in the precipitation of authigenic carbonate cements10,11,12. The second is thermochemical oxidation of methane (TOM) at high temperatures by sulfate or high-valence metal oxides. Like the bacterially-mediated AOM reaction, this process typically results in the formation of authigenic carbonate13,14,15,16.
Natural gas reservoirs in sedimentary basins are typically buried to depths of several kilometers, and experience temperatures from 60 to 150 °C17, while the metabolism of the archaeal groups responsible for AOM are mainly active at temperatures below 80 °C18,19, limiting the depth at which archaea dominate the conversion of CH4 to CO2. Therefore, TOM is potentially the predominant methane oxidation process in the strata with high formation temperature (especially > 80 °C) 13,14,15,20.
Thermochemical sulfate reduction (TSR) consuming methane has been observed to occur in geological systems13,14,15,20. Its reaction conditions, as well as geological and geochemical responses, have been deeply explored13,14,15,20,21. In contrast, almost no geological evidence has been reported for the oxidation of methane with high-valence metal oxides, except for the oxidation of methane by Fe3+ in detrital biotite at temperatures above 270 °C during metamorphism16,22,23. To date, the process has only been simulated in the laboratory setting at high temperatures (350 to 650 °C) 24,25,26,27. Thus, both the extent of TOM by high-valence metal oxides in geological systems and its role in the methane sink remain poorly understood.
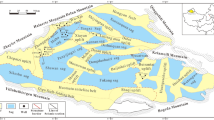
The Junggar Basin is a major petroliferous basin in northwestern China, with an area of approximately 1.3 × 105 km2 28. The Lower Triassic Baikouquan Formation (T1b) is located on the western slope of the Mahu Sag, a structure forming part of the northwestern margin of the Junggar Basin (Fig. 1a). The Mahu Sag is the richest hydrocarbon reservoir in the Junggar Basin, with approximately two billion tons of proven crude oil reserves29. Continuous oil and gas reservoirs were recently discovered in the Baikouquan Formation (Fig. 1a), with the hydrocarbons believed to be derived largely from the dark, organic-rich mudstones of the underlying Lower Permian Fengcheng Formation (see Supplementary Note 1 and Supplementary Fig. 1 for detailed descriptions)28,30.
Schematic geologic map of the Mahu Sag, and generalized stratigraphy of the Lower Triassic Baikouquan Formation. a The location of the Junggar Basin in China. b The study area is located in the west slope of the Mahu Sag adjacent to the northwestern marginal thrust faults of the Junggar Basin. c The sun-shape symbols within the Resistivity logging column indicate reservoir beds rich oil and gas
The Baikouquan Formation can be divided into three members, referred to as T1b1, T1b2, and T1b3 in ascending stratigraphic order. The maximum burial depth of the unit from west to east across the Mahu Sag increases from 2850 m to 4300 m. The formation consists of interlayered mudstone and sandy conglomerate, deposited in a succession of alluvial fan-deltaic systems with abundant gravity flow deposits31,32. The brown or greyish-green conglomeratic layers contain pebble conglomerates, sandy gravel conglomerates, and sandstones, whereas the mudstone layers are dominated by massive brown silty mudstones with minor gray laminations (Fig. 1b). The abundance of brown mudstone increases up-section in three members, which is interpreted to reflect an arid-semiarid climate regime and oxic lacustrine environment during deposition31,33.
The T1b sediments are derived from two provenances, i.e., the basement, comprising granite and mafic-ultramafic igneous rocks, and the sedimentary strata from the Carboniferous to the Permian28,31. The weathering of mafic-ultramafic rocks and tuffaceous components in sedimentary rocks forms abundant high-valence Fe-Mn oxides during T1b deposition interval (see Supplementary Note 2 for provenance analysis)34.
In this study, we report the first geological evidence for extensive TOM induced by high-valence metal oxides in the Lower Triassic reservoir strata of the Junggar Basin. We find that methane was abiotically oxidized by high-valence Mn(Fe) oxides at high temperatures (90–135 °C) in the deep burial strata, ultimately yielding extremely 13C-depleted authigenic calcites with high manganese contents. In the process thousands of Tg of CH4 were consumed, implying TOM may act as a major CH4 sink in the deep carbon cycles.
Results
Distribution and geochemistry of calcite cements
Authigenic calcite in the sandy conglomerates of the Baikouquan Formation occurs mainly as coarsely crystalline cement (Fig. 2a, b), sometimes within feldspar along cleavage (Fig. 2a). Based on microscopic observations, the total content of calcite cement in the sandy conglomerates ranges from 0 to 6% by area, with an average of ~2.5%. There are two stages of precipitation: the early-stage calcite occurs as xenomorphic crystals filling dissolved pores or poorly-connected interparticle pores (Fig. 2a), whereas the late-stage cements appear as coarse crystals, with well-developed crystal planes in the central portions of large interparticle pores (Fig. 2a, b).
Back scattered electron microscopic images of sandy conglomerates from the Baikouquan Formation, with crossplots of in situ δ13C and Mn contents of authigenic calcite. a, c Sandy conglomerate from the T1b3 member (3824.2 m in Well M18), with two generations of calcite cement. The early-stage cements have consistently lower MnO content than the late-stage cements. b, d Sandy conglomerate from the T1b2 member (3866.9 m in Well M18), cemented by calcite with higher MnO content. Circles indicate measurements of early-stage calcite, diamonds represent measurements of late-stage cements. The overall values of late-stage calcite δ13C are lower than those of the early-stage calcite. The error bars, referring to the standard error of the mean (SEM) in this paper, is smaller than the symbol size in the cross-plots
Electron probe micro-analysis (EPMA) reveals that these authigenic calcites are enriched in manganese, with MnO content ranging from 0.79 to 14.67 wt.% (average = 5.05 wt.%; Fig. 3a). In detail, the MnO content of early-stage calcite cements is generally lower than 4.00 wt.%, while late-stage cements typically have MnO content greater than 5.00 wt.%, some up to 11.00–15.00 wt.% (Fig. 3a). In contrast, the FeO content of the calcite cements is very low, ranging from<0.01 to 0.79 wt.%, with an average of 0.12 wt.% (Fig. 3b).
Manganese and iron contents in authigenic calcites. a Histogram of MnO content in early-stage and late-stage calcite cements from T1b sandy conglomerates. b Crossplot of MnO and FeO content in early-stage and late-stage calcite cements from T1b sandy conglomerates. The error bar (±SEM) is smaller than the symbol size. Source data are provided as Supplementary Data 1
Authigenic calcite cements in the T1b reservoir rocks are very negative in δ13C. Whole-rock carbonate δ13C values range from −69.8 to −22.5‰, though most samples fall within the −50.0 to −36.0‰ range (Fig. 4a). The calcite cements have δ18O values ranging from −22.6 to −12.8‰ (Fig. 4b).
Carbon and oxygen isotopic compositions in authigenic calcites. a Histogram of δ13C values, of calcite cements in T1b reservoirs, and calcite veins in P2w source rocks. b Crossplot of bulk rock δ13C and δ18O values in T1b calcite cements. The error bar (±SEM) is smaller than the symbol size. Source data can be seen in Supplementary Data 2
In situ carbon isotope analyses reveal that the two generations of calcite cements have markedly different δ13C values, sometimes by 10‰ (Fig. 2c, d). In contrast, the difference in δ18O between the two generations of cement is relatively small, with a maximum offset of 1.9‰ (Supplementary Table 1). To better constrain the sources of the carbon and oxygen incorporated into calcite cements in the T1b reservoir, we also measured the isotopic composition of calcite in the underlying source rocks: −22.4 to −13.5‰ for δ13C; and −18.5 to −12.8‰ for δ18O.
Geochemistry of hydrocarbons
In most natural gas samples more than 95 mol. % hydrocarbons are light components, i.e., C1–C7. CH4 is the most abundant hydrocarbon gas, with an aridity coefficient of 0.77–0.95. The non-hydrocarbon component consists mainly of N2, with minor CO2 and no detectable H2S. The ranges of δ13C values in methane, ethane, propane, and butane are −39.3 to −46.8‰, −27.5 to −31.9‰, −27.2 to −30.2‰, and −26.0 to −28.8‰, respectively (Supplementary Table 2).
Gas chromatography of crude oil samples from T1b reveals a complete sequence of n-alkanes (Fig. 5), especially ~C8–C12 which are preferentially removed in the earliest stages of biodegradation19. This indicates that the hydrocarbons have not experienced microbial degradation. The absence of 25-norhopanes and other representative values of key parameters also show no evidence for biodegradation (Supplementary Table 3). Biomarker analyses are described in details in the Methods section.
Chromatogram and mass spectrogram of a representative T1b crude oil sample (No. M18-O-1). a In the total ion chromatogram (TIC) Pr and Ph indicate pristane and phytane, respectively. b D represents demethylated hopanes in the m/z 177 mass spectrogram. c TT is tricyclic terpane and H is hopane in the m/z 191 mass spectrogram
Discussion
The extremely negative δ13C values of authigenic calcite can be interpreted to be derived from organic matter35,36. In organic matter-enriched geological systems, both oxidation of organic matter and thermal decarboxylation of organic acids can generate significantly 13C-depleted CO2, which may then be incorporated into authigenic calcite11,12,13,27,35. The δ13C value of authigenic calcite formed via the oxidation of hydrocarbons can be as low as −125‰11,12,13,36, whereas calcite precipitated from the decarboxylation of organic acids concentrates from −3 to −25‰, rarely yielding values below −25‰35,37. Authigenic calcite in the study area shows δ13C values falling mainly into the range of −50.0 to −36.0‰, with a minimum value of −69.8‰, consistent with the oxidation model for gaseous hydrocarbons11,12,36.
During the oxidation of gaseous hydrocarbons, 12C is preferentially oxidized to CO2, leaving the remaining hydrocarbon pool to become increasingly enriched in 13C as the reaction proceeds24,38,39. Figure 6 shows that the δ13C value of methane in T1b natural gas samples increases from around −45‰ to values above −40‰ as the CO2 contents of the gas increases; however, the isotopic composition of heavier hydrocarbon gases (C2–C4) remains comparatively stable. This result demonstrates that, within the T1b natural gas pool, abundant CH4 was oxidized during diagenesis. The δ13C values of the authigenic calcite in T1b are generally lower than those of the associated methane (−46.8 to 39.3‰; Fig. 7), further supporting our interpretation that the calcite is mainly derived from CO2 generated by methane oxidation11,12,36.
Carbon isotopes of natural gases from T1b sandy conglomerate reservoirs plotted against [CO2]/[CO2 + ∑n=1nCnH2n+2]. The latter is a parameter estimating the extent of thermochemical sulfate reduction39. The error bar (±SEM) is smaller than the symbol size
Carbon isotopes of natural gas, calcites from reservoir strata, and calcite veins from source rocks. Region I represents carbonates associated with decarboxylation of organic acids, while Region III denotes carbonates derived from the oxidation of methane. Region II indicates carbonates associated with a combination of the oxidation of methane and small quantities of C2+, as well as decarboxylation. The error bar (±SEM) is smaller than the symbol size
There are two mechanisms for oxidization of methane in sedimentary systems, i.e., AOM and TOM10,11,13,14,15. AOM has been observed mainly in modern marine sediments, especially near cold seeps on the seafloor, where the δ13C values of the associated authigenic carbonate can be as low as −125‰10,11,36. TOM can occur with or without TSR reactions. Methane is usually not the most active reductant in systems where TSR occurs compared with C2+ gaseous alkanes, so the minimum δ13C value of the associated calcite is not usually lower than −31‰13,15,39,40. TOM reactions induced by high-valence metal oxides instead of sulfate have never been reported in natural systems, though they have been simulated in the laboratory24,25,26,27.
All the three processes described above may have contributed to the precipitation of calcites in T1b. The range of measured δ13C values (−69.8 to −22.5‰) is compatible with calcite formed via AOM, but is also lower than the typical value of calcite formed via methane-dominated TSR (δ13C = −31‰)13. While there are no carbon isotope data reported for the carbonate associated with TOM induced by high-valence metal oxides in geological systems, the fractionation factors may be assumed to be similar to those in laboratory-simulated experiments. Thus, we cannot rule out the TOM process based on our isotopic measurements.
Anaerobic oxidation of methane by bacteria or archaea can be ruled out. Firstly, although archaeal groups such as ANME-1 and ANME-2 can anaerobically metabolize methane in the presence of oxidants such as MnO2, SO42−, and Fe3+ at temperatures < 80 °C10,18,19,41, the high formation temperature (> 80 °C) does not favor the metabolism of these archaeal groups19. The T1b reservoir strata were buried at depths of 2250–3200 m when they were firstly charged with methane in the Early Jurassic32. Taking the geothermal gradient as 28–32 °C km−1 and the surface temperature as 20 °C during the Early Jurassic42, this corresponds to a formation temperature of 83–122 °C. The δ18O values of authigenic calcites also indicate precipitation of the cements at relatively high temperatures. A similar diagenetic condition was reported for the Brent Group of the North Sea with a burial depth of 2500–3500 m43,44. The δ18O value of T1b pore water is likely to have increased by the same value (about 4‰) as the Brent Group from that of the initial meteoric water (−10 to −8‰) to −6 to −4‰44,45,46. Using this δ18O value, we calculated that the temperature was greater than 90 °C during formation of more than 85% of the total calcite, with a background temperature range of 90–135 °C and an average of 109.6 °C (see Supplementary Data 2), consistent with the temperatures estimated from the geothermal gradient.
Secondly, T1b hydrocarbons show no evidence of biological degradation, indicating that archaea had negligible influence on the composition of oil and gas. The first manifestation of hydrocarbon biodegradation is typically the selective removal of C8–C12 normal alkanes19. Light C2–C6 hydrocarbons exhibit a consistent degradation sequence: propane is the first compound to be altered, followed by butane, pentane, and C6+ hydrocarbons47. The net effect of biodegradation is an overall decrease in wet gas components (C2+) and enrichment in methane19. The crude oil hosted in the Baikouquan Formation shows a complete alkane series, with no obvious loss of C8–C12 alkanes, and no hump of unresolved complex mixtures (Fig. 5). Natural gas samples contain 5.14–22.68% wet gas (C2+) and 0.79–5.23% propane. Because AOM depends on the presence of biological mediators, and there is no evidence for biological modification, we can rule out this process of methane oxidation19,47.
Thermochemical sulfate reduction can also be ruled out. There is very little SO42− in T1b formation waters48, and no H2S has been detected in T1b natural gas samples (Supplementary Table 2) or the associated crude oil in adjacent formations28,29. Pyrite is also absent in T1b reservoir strata. The lack of both key reactants (i.e., sulfate) and characteristic products (sulfide in gaseous or mineral forms) strongly suggests that the TSR reaction did not occur in our study area. Thus, the highly negative δ13C values seen in the authigenic calcites cannot be attributed to methane oxidation by sulfate.
Therefore, we infer that the extremely negative δ13C observed in the study area results from methane oxidation by non-sulfate oxidants at high temperatures. It is shown that methane can serve as an electron acceptor for high-valence Mn and Fe oxides, leading to the formation of CO2 and the precipitation of calcite10. This process can be expressed with the following chemical formulas:
The Gibbs free energy increment (ΔG) differs substantially between the first two reactions and the third one under the same conditions, with oxidation by Mn2O3 or MnO2 being approximately twice as energetically favorable as oxidation by Fe(OH)310. In geological systems where high-valence Mn and Fe coexist, CH4 can be preferentially oxidized by Mn rather than Fe. During the interval of T1b deposition, the northwestern margin of the Junggar Basin experienced an arid or semiarid climate31,33. Mafic-ultramafic rocks were weathered extensively from the provenance during the period of deposition and were ultimately deposited in the organic matter-depleted red layers of T1b (Supplementary Fig. 2). These red beds provided the material basis for subsequent methane oxidation by the high-valence Mn and Fe oxides49. XRF analysis shows that the Mn content of sandy conglomerates in T1b ranges from 0.07 to 0.48 wt.% (as Mn2O3) in bulk rock, with an average of 0.17 wt.%; the Fe content ranges from 2.88 to 12.71 wt.% (as Fe2O3), with an average of 6.25 wt.% (Supplementary Table 4). Further XRD, FE-SEM, and EPMA analyses indicate that high-valence Mn(Fe) exists as hematite in the clastic rocks in T1b. Most of the hematite is disseminated in the clay-rich matrix, while minor occurrences as isolated amorphous aggregates are observed. The manganese generally occurs as the isomorphous Mn3+/4+ substitution in the hematite lattice structure50,51 at levels of 0.95–1.47 wt.% in the isolated hematite and 0.61–0.74 wt.% in the disseminated hematite in the form of Mn2O3 (see Supplementary Note 3 and Supplementary Fig. 3 for detailed descriptions).
The authigenic calcites in T1b are generally enriched in manganese relative to whole rock samples, with MnO content ranging from 0.79 to 14.67 wt.% (average = 5.05 wt.%). However, the FeO content of the calcite cements is uniformly less than 1 wt.%, averaging at 0.12 wt.%. This reflects the energetic favorability of Mn as an oxidant relative to Fe. Thus, the precipitation of calcite was closely associated with methane oxidation by high-valence manganese.
It should be noted that C2+ gaseous alkanes (especially ethane and propane) are usually preferentially oxidized during TSR reactions, whereas methane is the last to react14,15,21,40. The oxidation of abundant methane in T1b rocks has been influenced by several factors. One is the sufficient supply of Mn3+/4+, which is present as hematite within brown clastic rocks and guarantees that almost all of the gaseous alkanes, including methane, will be consumed in the bleaching pathways49. The other is that since methane is the main component of the T1b natural gas, ultimately it will have dominated the thermochemical oxidation process. Moreover, the T1b clastic strata are low-permeability due to their high clay content and complex grain composition32. Consequently, gaseous alkanes mainly migrated by diffusion. Due to its higher initial concentration and higher diffusion coefficient, the quantity of methane reaching the matrix of reservoir rocks or mudstones at a distance from the oil-gas interface is much higher than that of C2+ gaseous alkanes52. Ultimately, as shown in Fig. 6, abundant methane was oxidized in the T1b clastic rocks.
Microbial metabolism and high temperature can both serve to facilitate reactions (1) and (2). The former may reduce the energy threshold, while the latter can provide the activation energy necessary for the reaction to occur10,24. Since microbial activity in the T1b reservoir strata appears to have been minimal, heating would have been the only way to promote oxidation reactions. Considering the formation temperature (90–122 °C) during infiltration of natural gas and the main precipitation temperature (90–135 °C) of authigenic calcites, we can infer that methane oxidation in the study area mainly took place at 90–135 °C. Laboratory experiments have shown that reactions (1) and (2) do not occur at temperatures below 300 °C and 250 °C, respectively24,27. However, during extended geological processes, long intervals of time can compensate for the slow rate of reactions at low temperatures. In the case of thermochemical sulfate reduction, experiments show that the reaction occurs at temperatures greater than 250 °C53, though evidence for TSR has been observed in sedimentary basins at temperatures of ~100 °C40. As another example, while the standard Rock Eval pyrolysis procedure shows the peak of hydrocarbon generation at 420–480 °C, in natural geological systems the peak of hydrocarbon generation typically occurs at 120–150 °C54. This also appears to be the case for TOM. The long duration of geological time can compensate for a low reaction rate at relatively low temperatures. With the increasing burial depth of T1b, the formation temperature generally increased and reached 90 °C in the Middle Jurassic (~170 Ma, see Supplementary Note 4 and Supplementary Fig. 4 for thermal evolution). Since then, methane has been oxidized more efficiently.
Carbon isotope fractionation is expected to occur during thermal oxidation of methane, with the fractionation factor controlled by a number of factors, including the type of oxidant and the temperature24,27,53. In general, the residual methane is enriched in 13C as the reaction proceeds, while the generated CO2 is depleted, resulting in CO2 with a lower δ13C value than the initial methane pool (δ13Ci-CH4). Taking the carbon isotope kinetic fractionation factor (α) to be 103/(103−∆) in reactions (1) and (2), where ∆ = δ13Ci-CH4−δ13CCO2, we calculate an α value of 1.0175–1.0193 for Reaction (2) at 90–135 °C24. This value is close to that reported for the methane-dominated TSR in the East Sichuan Basin of China (1.0166) and the ethane-dominated TSR in the Jurassic Norphlet Formation of Mobile Bay (1.109)55. Then, a ∆ value of 17–19‰ for a closed system at 90–135 °C can be obtained. Carbon isotope fractionation also occurs during the calcite precipitation. At 90 to 135 °C, the δ13C of precipitated CaCO3 is expected to be 2 to 5‰ lower than that of the original gaseous CO256. Integrating the methane oxidation and calcite precipitation processes, the δ13C of authigenic calcite is expected to be 19 to 24‰ lower than that of the residual methane. Thus, the expected δ13C of authigenic calcite is −70 to −59‰. The extremely 13C-depleted calcites may also precipitate during the oxidation of primary microbial or kerogen-cracking-derived low δ13C methane, although their relative quantities involved in the reaction remain ambiguous. The former is formed via bacterial methanogenesis at temperatures generally below 50–60 °C in the P1f source rock57, whereas the latter is generated as a concomitant of liquid oil expelled from low-maturity source rock19. In addition, it should be noted that the contributions of oxidation of minor C2+ and decarboxylation of organic acids from the source rock cannot be ruled out (Fig. 8). As shown in Fig. 7, the decarboxylation reaction can produce CO2 with a δ13C value of around −25‰. The addition of small amounts of CO2 from C2+ oxidation and decarboxylation yields a range of potential δ13C values in authigenic calcite from −70 to −22.5‰, consistent with our observations (Fig. 8).
Schematic model of thermochemical methane oxidation in the clastic strata of petroliferous basins. R-COOH represents carboxylic acids. High-valence Mn (Fe) exists in hematite, which is generally found in the matrix of sandstone and conglomerate. Due to the high diffusivity of methane, minor high-valence Mn (Fe) in the brown-red mudstone adjacent to the reservoir rocks may also be reduced
Methane is generally believed to be stable in the crust, especially in sedimentary basins; oxidation is thought to occur mainly after the methane has reached the surface and been exposed to atmospheric oxygen8,9. The potentially important role of TOM as a methane sink in the deep strata of sedimentary basins has largely been neglected so far; methane oxidization in the deep crust has not been documented, and only a few relevant simulation experiments have been conducted to date24,27,53. In this study, we provide the first evidence for the occurrence of TOM in a clastic reservoir, and propose that TOM is an important CH4 sink in the deep crustal carbon cycle.
In order to evaluate the extent to which TOM serves as a CH4 sink, we made a series of semi-quantitative estimates based on the M18-Aihu2 oil producing area (highlighted in yellow in Fig. 1a), which contains a relatively dense concentration of exploration wells. Details of the calculations are described in the Methods section. Assuming an average authigenic calcite content of 2.5% in the relevant reservoir beds, we calculated a total authigenic calcite mass of 420 Tg. The calcite would require 57 Tg of CH4 to have been oxidized, assuming only 85% of the carbonate carbon is derived from methane oxidation. If only taking account of the total oil-charging area in the sag, a fairly conservative CH4 consumption is 155 Tg. However, authigenic calcites are not restricted in the conglomerates and sandstones charged by liquid oil, they also occur in the wells far away from the oil-charging area. Extending these calculations to encompass the entire Mahu Sag (~5000 km2), we estimate that as much as 1224 Tg of CH4 might be oxidized in the reservoir strata. The global submarine CH4 flux to the atmosphere is only 8 to 65 Tg per year58, suggesting that TOM in sedimentary basins may play a geologically significant role in reducing CH4 seepage to the surface. In other words, extensive TOM may be an important methane sink in sedimentary basins, while it is unfavorable for the accumulation of recoverable natural gas reserves.
This study reports geological evidence for widespread thermochemical oxidation of methane (TOM) induced by high-valence metal oxides in a natural environment, from the Junggar Basin in northwestern China. Methane was oxidized at high temperatures (90–135 °C) in the T1b stratum, with high-valence metal oxides acting as the electron acceptor. This reaction resulted in the precipitation of authigenic calcite cements, characterized by extremely negative δ13C values (−70 to −22.5‰) and high manganese contents (average MnO = 5 wt.%). While both Mn and Fe oxides served as methane oxidants, less Fe oxide was consumed due to the relatively higher Gibbs free energy increment of the reaction. Based on the abundance and distribution of authigenic calcite, we estimate that thousands of Tg of CH4 were consumed by TOM in the Lower Triassic reservoir strata of the Mahu Sag. We further propose that the geologic importance of TOM has been underestimated to date, and that this critical methane sink should be a focus of future studies of the deep carbon cycle.
Methods
Sampling
A total of 103 rock samples were collected from T1b reservoir beds in 11 wells from the western slope area of the Mahu Sag (Supplementary Table 5). In addition to rock samples, 9 crude oil and 12 natural gas samples were collected from the wells (Supplementary Table 6).
Elemental analyses
Backscattering electron (BSE) microprobe analyses were conducted on 38 sandy conglomerate samples, using a Japan Electron Optics Laboratory (JEOL) x-ray analyzer (JXA)-8800 electron microprobe operated at 15 kV, with a 10 nA beam current. The calcite cements in 14 sandy conglomerate samples were analyzed for their major element composition, using a 2 μm beam diameter and 10 s counting time. The major element compositions of whole-rock samples were measured on a Thermo Scientific ARL 9900 × -ray fluorescence (XRF) spectrometer. The relative standard deviation of measured major element concentrations is around ± 1% for elements with concentrations > 1.0 wt.%, and about ± 10% for the elements with concentrations < 1.0 wt.%, based on repeated measurements of Chinese national standards GBW-07103 (granite) and GBW-07105 (basalt). Both EPMA and XRF analyses were carried out at the State Key Laboratory for Mineral Deposits Research, Nanjing University.
Carbonate isotope analyses of bulk rock samples
The calcite cements in 50 T1b sandy conglomerate (i.e., reservoir rock) samples and 5 P2w mudstone (i.e., source rock) samples were analyzed for stable carbon and oxygen isotopes. Prior to analysis, each sample was broken into small pieces, and ~ 1 g of calcite cement was manually selected under a binocular microscope. All samples were then crushed to a fine powder. Twenty milligrams of each powdered sample was reacted with 100% H3PO4 at 25 °C for more than 12 h, in a Kiel IV autosampler (ThermoFinnigan). The produced CO2 was then introduced into a Finnegan MAT 253 mass spectrometer for isotopic analysis. Carbon and oxygen isotope measurements were corrected using Chinese national standard GBW 04405 (δ13C = 0.57 ± 0.03‰ VPDB; δ18O = −8.49 ± 0.13‰ VPDB), an Ordovician carbonate from a site near Beijing. All analyses were performed at the Nanjing Institute of Geology and Paleontology, Chinese Academy of Sciences. The external precisions (1σ) of oxygen and carbon isotope analyses were ± 0.08 and ± 0.03‰, respectively.
SIMS δ13Ccalcite analyses
Small pieces of polished thin sections of carbonate samples were mounted in Buehler EpoFix epoxy resin, together with carbonate reference materials: NBS-19 calcite, UWC-3 calcite (δ13CVPDB = –0.91 ± 0.08‰, 2σ, n = 9; cf. ref. 59.), UW6220 dolomite (δ13CVPDB = 0.84 ± 0.02‰, 2σ, n = 5; cf. ref. 60.) and Hammerfall dolomite (δ13CVPDB = –0.28 ± 0.07‰, 2σ, n = 3; Ian S. Williams, personal communication). Backscattered electron images were taken using a JEOL JSM-6610A Analytical Scanning Electron Microscope in Research School of Earth Science (RSES), Australian National University (ANU), Canberra.
Secondary ion mass spectroscopy (SIMS) analyses of carbon isotopes were performed in situ using the Sensitive High Resolution Ion Microprobe for Stable Isotopes (SHRIMP SI) located in RSES, ANU. The analytical procedure was similar to ref. 61 for oxygen isotopes, though without electron-induced secondary ionization emission (EISIE) correction at the presence of the electron gun. Carbon isotopes (12C and 13C) were measured simultaneously by two Faraday cups, using an ~15 kV Cs + primary ion beam focused over an area of ~30 μm in diameter and source slit of 60 µm. 12CH was completely resolved with collector slits of 200 µm for 13C and 300 µm for 12C.
Duplicate δ13C analyses of the calcite standard UWC-3 yield an external (spot to spot) precision (1σ) of 0.40–0.50‰. Mn content (shown as Mn/(Ca + Mg + Fe + Mn); Supplementary Table 1) in the calcite samples is as high as 4.6–12.7 mol. %. Such small amount of Mn has less measurable matrix effects on SIMS δ13C bias than Fe2+ 60. Contamination of measurements by organic carbon was avoided by carefully choosing spots free of micro fractures or inclusions. Thus the potential error associated with matrix effect and organic inclusions is considered to be insignificant relative to isotopic variation.
SIMS δ18Ocalcite analyses
The oxygen isotope composition of calcite was measured using a SHRIMP II ion microprobe at the RSES facility of ANU. Analytical methods were similar to those described by ref. 61. Sequences of sample analyses (typically three measurements) were bracketed by 1–2 analyses of standards. Corrected 18O/16O ratios are reported in standard δ18O notation, relative to Vienna Pee Dee Belemnite (VPDB). All δ18O values were calibrated against the UWC-3 calcite standard (δ18O = −17.87‰, 1σ = 0.03‰; cf. ref. 62). The spot-to-spot reproducibility (i.e., external precision) was typically better than ± 0.30‰ (1σ).
Composition and δ13C of natural gas
The composition of natural gas was determined using a Hewlett Packard 6890 II gas chromatograph (GC). Stable carbon isotope values were measured on an Optima isotope ratio mass spectrometer (IR-MS) coupled to a Hewlett Packard 6890 II GC. Measurements followed the procedure described by ref. 29. An internal CO2 reference gas was introduced into the spectrometer prior to and after the peaks of interest. Measured δ13C values were calibrated using the national ‘charcoal black’ standard, with a value of −22.43‰ relative to VPDB. All gas samples were analyzed in triplicate.
Biomarkers and δ13C in oil samples
The biomarker compositions of oil samples were analyzed on a Hewlett Packard 6890 II GC, using a Hewlett Packard PONA capillary column with dimensions of 50 m × 0.25 mm × 25 μm. Stable carbon isotope analyses of alkanes and arenes were performed on the same instrument, following the procedure described by ref. 63.
Estimating the methane sink capacity of the Mahu Sag
We first take the Mahu18-Aihu2 district, highlighted in blue dashed line in Fig. 1a, to be representative of the broader Mahu Sag. At least eight cores have been drilled in the 234 km2 area, providing a good overview of the T1b reservoir strata. The combined thickness of the horizons with secondary calcite cements ranges from 19.6 m to 48.3 m, with an average value of 28.7 m. Thus, the volume of rock containing secondary calcite can be estimated as: 234 × 106 m2 (area of the region of interest) × 28.7 m (average thickness of relevant horizons) = 6.7 × 109 m3 of calcite cement-bearing strata. Multiplying by a typical sedimentary rock density of 2.5 × 103 kg m-3 converts this volume to a total mass of 16.8 × 1012 kg.
Based on the microscopic observations, the content of calcite cement in the relevant strata ranges from 1% to 5%, with the average value of 2.5%. Thus, we can calculate the total mass of authigenic calcite to be: 16.8 × 1012 kg × 2.5% = 420.0 × 109 kg of calcite cement.
With the above estimate of the total mass of calcite cement in the Mahu18-Aihu2 district, we can calculate the mass of methane that would have been necessary to generate this cement. Due to its diffusion superiority of volatile natural gas relative to liquid oil, it can be approximately assumed that all carbon in the cement is derived from natural gas. As the average relative content of CH4 is 85% in the T1b natural gas (Supplementary Table 2), the methane consumption can be estimated as: 420.0 × 109 kg of CaCO3 ÷ 100.09 g mol−1 CaCO3 × 16.06 g mol−1 × 0.85 CH4 = 57.3 × 109 kg (57.3 Tg) of CH4. Converting this mass to volume yields: 57.3 × 109 kg CH4 ÷ 0.72 kg m−3 ≈ 79.6 × 1010 m3 of CH4.
If only taking account of the oil-charging area, a fairly conservative methane consumption is 155 Tg, as the total discovered oil-charging area has reached 633 km2 to the end of 2017 in the Mahu Sag (Fig. 1a). However, authigenic calcites are not restricted in the conglomerates and sandstones charged by oil, they also occur in the wells far away from the oil-charging area. The entire sag has an area of approximately 5000 km2, which is roughly 20 times greater than the area of the Mahu18-Aihu2 district. Thus, we can estimate that the total mass of CH4 abiotically oxidized by Fe-Mn oxides in the sag reaches 1,224 Tg.
The parameters used in the mass balance calculations are as follows: MassCH4 = 16.05 g mol−1; MassCaCO3 = 100.09 g mol−1; Density of CH4 = 0.72 kg m−3 (defined at 273.15 K and 101.325 kPa); Density of rock = 2.50 × 103 kg m−3.
Data availability
All necessary data generated or analyzed during this study are included in this published article, and other auxiliary data are available from the corresponding authors on reasonable request.
References
U.S. Energy Information Administration. Annual Energy Outlook 2015 https://www.eia.gov/outlooks/aeo/pdf/0383(2015).pdf (EIA, 2015).
International Energy Agency. Natural Gas Information 2016 https://doi.org/10.1787/nat_gas-2016-en (IEA, 2016).
Wuebbles, D. J. & Hayhoe, K. Atmospheric methane and global change. Earth Sci. Rev. 57, 177–210 (2002).
Hopcroft, P. O., Valdes, P. J., O’Connor, F. M., Kaplan, J. O. & Beerling, D. J. Understanding the glacial methane cycle. Nat. Commun. 8, 14383 (2017).
Pavlov, A. A., Kasting, J. F., Brown, L. L., Rages, K. A. & Freedman, R. Greenhouse warming by CH4 in the atmosphere of early Earth. J. Geophys. Res. Atm. 105, 11981–11990 (2000).
Jiang, G., Kennedy, M. J. & Christie-Blick, N. Stable isotopic evidence for methane seeps in Neoproterozoic postglacial cap carbonates. Nature 426, 822–826 (2003).
Kirschke, S. et al. Three decades of global methane sources and sinks. Nat. Geosci. 6, 813–823 (2013).
Etiope, G., Milkov, A. V. & Derbyshire, E. Did geologic emissions of methane play any role in Quaternary climate change? Glob. Planet. Chang 61, 79–88 (2008).
Berbesi, L. A., di Primio, R., Anka, Z., Horsfield, B. & Wilkes, H. Methane leakage from evolving petroleum systems: Masses, rates and inferences for climate feedback. Earth Planet. Sci. Lett. 387, 219–228 (2014).
Beal, E. J., House, C. H. & Orphan, V. J. Manganese- and iron-dependent marine methane oxidation. Science 325, 184–187 (2009).
Michaelis, W. et al. Microbial reefs in the black sea fueled by anaerobic oxidation of methane. Science 297, 1013–1015 (2002).
Drake, H. et al. Extreme 13C depletion of carbonates formed during oxidation of biogenic methane in fractured granite. Nat. Commun. 6, 1–9 (2015).
Worden, R. H. & Smalley, P. C. H2S-producing reactions in deep carbonate gas reservoirs: Khuff Formation, Abu Dhabi. Chem. Geol. 133, 157–171 (1996).
Cai, C., Worden, R. H., Bottrell, S. H., Wang, L. & Yang, C. Thermochemical sulphate reduction and the generation of hydrogen sulphide and thiols (mercaptans) in Triassic carbonate reservoirs from the Sichuan Basin, China. Chem. Geol. 202, 39–57 (2003).
Cai, C. et al. Methane-dominated thermochemical sulphate reduction in the Triassic Feixianguan Formation East Sichuan Basin, China: towards prediction of fatal H2S concentrations. Mar. Petrol. Geol. 21, 1265–1279 (2004).
Mullis, J., Dubessy, J., Poty, B. & O’Neil, J. Fluid regimes during late stages of a continental collision: physical, chemical, and stable isotope measurements of fluid inclusions in fissure quartz from a geotraverse through the Central Alps, Switzerland. Geochim. Cosmochim. Acta 58, 2239–2267 (1994).
Allen, P. A. & Allen, J. R. Basin Analysis: Principles and Application to Petroleum Play Assessment (John Wiley & Sons, New York, 2013).
Kallmeyer, J. & Boetius, A. Effects of temperature and pressure on sulfate reduction and anaerobic oxidation of methane in hydrothermal sediments of Guaymas Basin. Appl. Environ. Microbiol. 70, 1231–1233 (2004).
Peters, K. E., Walters, C. C. & Moldowan, J. M. The Biomarker Guide Ch. 16 (Cambridge University Press, Cambridge, 2005).
Jenden, P. D., Titley, P. A. & Worden, R. H. Enrichment of nitrogen and 13C of methane in natural gases from the Khuff Formation, Saudi Arabia, caused by thermochemical sulfate reduction. Org. Geochem. 82, 54–68 (2015).
Liu, Q. Y. et al. TSR versus non-TSR processes and their impact on gas geochemistry and carbon stable isotopes in Carboniferous, Permian and Lower Triassic marine carbonate gas reservoirs in the Eastern Sichuan Basin, China. Geochim. Cosmochim. Acta 100, 96–115 (2013).
Tarantola, A., Mullis, J., Vennemann, T., Dubessy, J. & de Capitani, C. Oxidation of methane at the CH4/H2O-(CO2) transition zone in the external part of the Central Alps, Switzerland: Evidence from stable isotope investigations. Chem. Geol. 237, 329–357 (2007).
Tarantola, A. et al. Oxidation of CH4 to CO2 and H2O by chloritization of detrital biotite at 270 ± 5 °C in the external part of the Central Alps, Switzerland. Lithos 112, 497–510 (2009).
Kiyosu, Y. & Imaizumi, S. Carbon and hydrogen isotope fractionation during oxidation of methane by metal oxides at temperatures from 400 to 530 C. Chem. Geol. 133, 279–287 (1996).
Stobbe, E. R., De Boer, B. A. & Geus, J. W. The reduction and oxidation behaviour of manganese oxides. Catal. Today 47, 161–167 (1999).
Seewald, J. S. Aqueous geochemistry of low molecular weight hydrocarbons at elevated temperatures and pressures: constraints from mineral buffered laboratory experiments. Geochim. Cosmochim. Acta 65, 1641–1664 (2001).
Pan, C., Yu, L., Liu, J. & Fu, J. Chemical and carbon isotopic fractionations of gaseous hydrocarbons during abiogenic oxidation. Earth Planet. Sci. Lett. 246, 70–89 (2006).
Cao, J. et al. The Permian hybrid petroleum system in the northwest margin of the Junggar Basin, northwest China. Mar. Petrol. Geol. 22, 331–349 (2005).
Tao, K. et al. Geochemistry and origin of natural gas in the petroliferous Mahu sag, northwestern Junggar Basin, NW China: Carboniferous marine and Permian lacustrine gas systems. Org. Geochem. 100, 62–79 (2016).
Qi, W. et al. Fluid inclusion and hydrocarbon charge history for reservoir of Baikouquan Formation in the Mahu Sag, Junggar Basin. Nat. Gas. Geosci. 26, 64–71 (2015).
Jia, H. et al. A retreating fan-delta system in the northwestern Junggar Basin, northwestern China—Characteristics, evolution and controlling factors. J. As. Earth Sci. 123, 162–177 (2016).
Kang, X. et al. Selective dissolution of alkali feldspars and its effect on Lower Triassic sandy conglomerate reservoirs in the Junggar Basin, northwestern China. Geol. J. 53, 475–499 (2018).
Retallack, G. J. et al. Multiple Early Triassic greenhouse crises impeded recovery from Late Permian mass extinction. Palaeogeogr. Palaeoclim. Palaeoecol. 308, 233–251 (2011).
Wronkiewicz, D. J. & Condie, K. C. Geochemistry of Archean shales from the Witwatersrand Supergroup, South Africa: source-area weathering and provenance. Geochim. Cosmochim. Acta 51, 2401–2416 (1987).
Irwin, H., Curtis, C. & Coleman, M. Isotopic evidence for source of diagenetic carbonates formed during burial of organic-rich sediments. Nature 269, 209–213 (1977).
Campbell, K. A., Farmer, J. D. & Des Marais, D. Ancient hydrocarbon seeps from the Mesozoic convergent margin of California: carbonate geochemistry, fluids and palaeoenvironments. Geofluids 2, 63–94 (2002).
Macaulay, C. I., Fallick, A. E., McLaughlin, O. M., Haszeldine, R. S. & Pearson, M. J. The significance of δ13C of carbonate cements in reservoir sandstones: a regional perspective from the Jurassic of the northern North Sea. Intern. Assoc. Sediment. Spec. Publ. 26, 395–408 (1998).
Coleman, D. D., Risatti, J. B. & Schoell, M. Fractionation of carbon and hydrogen isotopes by methane-oxidizing bacteria. Geochim. Cosmochim. Acta 45, 1033–1037 (1981).
Krouse, H. R., Viau, C. A., Eliuk, L. S., Ueda, A. & Halas, S. Chemical and isotopic evidence of thermochemical sulphate reduction by light hydrocarbon gases in deep carbonate reservoirs. Nature 333, 415–419 (1988).
Machel, H. G. Bacterial and thermochemical sulfate reduction in diagenetic settings—old and new insights. Sediment. Geol. 140, 143–175 (2001).
Orphan, V. J., House, C. H., Hinrichs, K. U., McKeegan, K. D. & DeLong, E. F. Methane-consuming archaea revealed by directly coupled isotopic and phylogenetic analysis. Science 293, 484–487 (2001).
Qiu, N. S., Yang, H. B. & Wang, X. L. Tectono-thermal evolution in the Junggar Basin. Chn J. Geol. 37, 423–429 (2002).
Bjørlykke, K., Nedkvitne, T., Ramm, M. & Saigal, G. C. Diagenetic processes in the Brent Group (Middle Jurassic) reservoirs of the North Sea: an overview. Geol. Soc. Lond. Spec. Publ. 61, 263–287 (1992).
Haszeldine, R. S., Brint, J. F., Fallick, A. E., Hamilton, P. J. & Brown, S. Open and restricted hydrologies in Brent Group diagenesis: North Sea. Geol. Soc. Lond. Spec. Publ. 61, 401–419 (1992).
Sun, Y. et al. Lethally hot temperatures during the Early Triassic greenhouse. Science 338, 366–370 (2012).
Roberts, J., Turchyn, A. V., Wignall, P. B., Newton, R. J. & Vane, C. H. Disentangling diagenesis from the rock record: an example from the Permo-Triassic Wordie Creek Formation, East Greenland. Geochem. Geophys. Geosyst. 19, 99–113 (2018).
James, A. T. & Burns, B. J. Microbial alteration of subsurface natural gas accumulations. AAPG Bull. 68, 957–960 (1984).
Chen, J. P., Cha, M. & Zhou, Y. Q. Chemical characteristics of formation water in relation with oil/gas in the northwesterh parts of Junggar Basin. Geol. Geochem 28, 54–58 (2000).
Chan, M. A., Parry, W. T. & Bowman, J. R. Diagenetic hematite and manganese oxides and fault-related fluid flow in Jurassic sandstones, southeastern Utah. AAPG Bull. 84, 1281–1310 (2000).
Manceau, A. et al. Crystal chemistry of trace elements in natural and synthetic goethite. Geochim. Cosmochim. Acta 64, 3643–3661 (2000).
Liu, H. et al. Geochemical fates and unusual distribution of arsenic in natural ferromanganese duricrust. Appl. Geochem. 76, 74–87 (2017).
Crank, J. The Mathematics of Diffusion Ch. 7 (Clarendon Press, Oxford, 1975).
Kiyosu, Y. & Krouse, H. R. Carbon isotope effect during abiogenic oxidation of methane. Earth Planet. Sci. Lett. 95, 302–306 (1989).
Peters, K. E. Guidelines for evaluating petroleum source rock using programmed pyrolysis. AAPG Bull. 70, 318–329 (1986).
Cai, C., Zhang, C., He, H. & Tang, Y. Carbon isotope fractionation during methane-dominated TSR in East Sichuan Basin gasfields, China: A review. Mar. Petrol. Geol. 48, 100–110 (2013).
Halas, S., Szaran, J. & Niezgoda, H. Experimental determination of carbon isotope equilibrium fractionation between dissolved carbonate and carbon dioxide. Geochim. Cosmochim. Acta 61, 2691–2695 (1997).
Whiticar, M. J. Carbon and hydrogen isotope systematics of bacterial formation and oxidation of methane. Chem. Geol. 161, 291–314 (1999).
Hovland, M., Judd, A. G. & Burke, R. A. The global flux of methane from shallow submarine sediments. Chemosphere 26, 559–578 (1993).
Kozdon, R., Ushikubo, T., Kita, N. T., Spicuzza, M. & Valley, J. W. Intratest oxygen isotope variability in the planktonic foraminifer N. pachyderma: Real vs. apparent vital effects by ion microprobe. Chem. Geol. 258, 327–337 (2009).
Śliwiński, M. G. et al. Secondary ion mass spectrometry bias on isotope ratios in dolomite-ankerite, part II: δ13C matrix effects. Geostand. Geoanal. Res. 40, 173–184 (2016).
Ickert, R. B. et al. Determining high precision, in situ, oxygen isotope ratios with a SHRIMP II: analyses of MPI-DING silicate-glass reference materials and zircon from contrasting granites. Chem. Geol. 257, 114–128 (2008).
Śliwiński, M. G. et al. Secondary ion mass spectrometry bias on isotope ratios in dolomite-ankerite, part I: δ18O matrix effects. Geostand. Geoanal. Res. 40, 157–172 (2016).
Cao, J. et al. Geochemistry and origins of natural gases in the central Junggar Basin, northwest China. Org. Geochem. 53, 166–176 (2012).
Acknowledgements
This study was supported by funds from the Natural Science Foundation of China to W.-X.H. (41830425, 41230312) and X.-L.W. (41573054), the Fundamental Research Funds for the Central Universities to X.-L.W. (020614380056), and the Outstanding PhD Candidate Program of Nanjing University to X.K. Thanks are extended to Jun Jin, Bao-li Xiang, and Zhao Yang for their assistances with core, oil and gas sampling; Ian S. Williams, Peter Holden, and Peter Lanc for their assistance with in situ stable carbon and oxygen isotope analyses; Wen-lan Zhang for her assistance with major element analyses; Huan Liu for his assistance with the analyses of Fe/Mn valence in hematite; Shane Schoepfer for improving the manuscript language.
Author information
Authors and Affiliations
Contributions
W.-X.H. and X.K. designed this study; X.K. wrote the manuscript with contributions from W.-X.H. and X.-L.W.; X.K. collected the samples and performed the experiments; W.-X.H. developed the evaluation model for CH4 sink; J.C. provided the oil and gas samples and participated in the organic geochemical experiments; X.-L.W. participated in the interpretation of the data; B.F. contributed a lot in the in situ stable isotope analyses; H.-G.W. participated in the core sampling and EPMA analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hu, WX., Kang, X., Cao, J. et al. Thermochemical oxidation of methane induced by high-valence metal oxides in a sedimentary basin. Nat Commun 9, 5131 (2018). https://doi.org/10.1038/s41467-018-07267-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-07267-x
This article is cited by
-
Sulfur isotopes from the Paleoproterozoic Francevillian Basin record multigenerational pyrite formation, not depositional conditions
Communications Earth & Environment (2024)
-
Unrecognized extensive charge of microbial gas in the Junggar basin
Scientific Reports (2024)
-
Thermochemical oxidation of methane by manganese oxides in hydrothermal sediments
Communications Earth & Environment (2023)
-
Influence of hydrogen fugacity on thermal transformation of sedimentary organic matter: Implications for hydrocarbon generation in the ultra-depth
Science China Earth Sciences (2022)
-
Multisourced CO2 Injection in Fan Delta Conglomerates and Its Influence on Reservoir Quality: Evidence from Carbonate Cements of the Baikouquan Formation of Mahu Sag, Junggar Basin, Northwestern China
Journal of Earth Science (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.