Abstract
All-inorganic perovskites have a special place in halide perovskite family because of their potential for better stability. However, the representative cesium lead iodide (CsPbI3) is metastable and spontaneously converts to the non-perovskite structure at room temperature. Here, we demonstrate that what appears to be all-inorganic CsPbI3 stabilized in its perovskite form using the purported intermediate known as hydrogen lead iodide (HPbI3) is, in fact, the hybrid perovskite cesium dimethylammonium lead iodide (Cs1−xDMAxPbI3, x = 0.2 to 0.5). Thus, many of the reported all-inorganic perovskites are actually still hybrid organic-inorganic perovskites, as strongly evidenced by a wide battery of experimental techniques presented here. Solar cells based on the representative composition Cs0.7DMA0.3PbI3 can achieve an average power conversion efficiency of 9.27 ± 1.28% (max 12.62%). These results provide an alternative angle to look at previous results pertaining all-inorganic CsPbI3 while the DMA cation is now revealed as an alternative A site cation.
Similar content being viewed by others
Introduction
Halide perovskites have unique properties such as tunable band gaps, long diffusion lengths, high optical absorption coefficients, and low exciton binding energies1,2,3,4,5,6,7,8. Halide perovskite solar cells have achieved a record power conversion efficiency (PCE) of 22.7%9, which are a great candidate for the next generation of photovoltaics. High-performance perovskite solar cells typically employ organic-inorganic hybrid Pb-based perovskites as light absorbers such as methylammonium lead iodide (MAPbI3), formamidinium lead iodide (FAPbI3), and their mixtures, due to their excellent optical and electrical properties10,11,12,13,14,15,16. However, the intrinsic thermal and light instabilities of organic cations of MA and FA present serious hurdles in the further development and commercialization of long-term operating devices17. The all-inorganic perovskite analog cesium lead iodide (CsPbI3) could present an important way forward, since it possesses a relatively wider band gap with large band widths and especially a significantly better thermal stability compared with the organic cations-based perovskites17,18,19,20. However, the black CsPbI3 perovskite is metastable and it spontaneously converts to the undesired yellow polytype (δ-phase, NH4CdCl3 structure-type) at room temperature5,18,21,22. Yellow δ-CsPbI3 has a wide band gap of Eg = 2.82 eV and has poor transport properties18. By contrast, black CsPbI3 has a narrow band gap of around 1.7 eV and it is suitable for solar cell applications21,23. Therefore, much effort has been devoted to improving the phase-stability of the black-phase CsPbI3 perovskites so that highly efficient all-inorganic solar cells can be made18,24,25,26,27,28,29,30,31,32,33,34,35. In one approach, researchers used pre-synthesized CsPbI3 nanocrystals to make solar cells. For example, Protesescu et al. demonstrated that the black CsPbI3 perovskite (γ-phase) could be stabilized as nanocrystals36, due to the large contribution of surface energy. Subsequent work by Luther et al. demonstrated that colloidal CsPbI3 quantum dots synthesized by a hot-injection method can be phase-stable for several months in ambient air and the solar cells achieved a PCE as high as 13.43%27,28. In another approach, You et al. used dimethylformamide (DMF)/dimethyl sulfoxide (DMSO) solvent followed by high-temperature annealing process to obtain the black α-phase CsPbI333. These solar cells yielded a record PCE of 15.7% and showed a good light-soaking stability under the protection of N2-filled glovebox. Much more commonly, the use of hydriodic acid (HI)18,26 or the so-called hydrogen lead iodide (HPbI3) precursor31,37, produced by HI treatment, are the most used approaches to stabilize the black α-CsPbI3. The use of HPbI3 was first reported as an intermediate in the synthesis of FAPbI3 solar cells38, and was prepared by mixing PbI2 and HI in DMF. Eperon et al. subsequently reported that HI can be used directly as an additive in DMF solutions of CsPbI3 to stabilize the black α-CsPbI3 films at room temperature18. The planar device made from this black CsPbI3 film achieved a PCE of 2.9%. Recently, Zhang et al. reported the formation of black CsPbI3 by a low-temperature deposition using the pre-synthesized PbI2·xHI, which was also made from DMF, PbI2, and HI31. Combined with the use of larger ethylenediammonium (en) cation, the champion α-CsPbI3 solar cell yielded a PCE of 11.8% with enhanced stability and good reproducibility31. More recently, Jiang et al. also used HPbX3 (X = I, Br) as the precursor together with phenylethylammonium (PEA) cation to fabricate low-dimensional α-CsPbI3 and the solar cells achieved a high PCE of 12.4%37. Despite the apparent success of the HPbI3 methodology, the mechanism of the stabilization of the black CsPbI3 by HI and HPbI3 remains unclear. The claim alone that HPbI3 even exists is extraordinary and there is no structural or spectroscopic validation for the existence of this compound. From the chemistry point of view, HPbI3 would be a solid acid with HI fragment in it and intuitively is unlikely to exist. This is because the HI molecule itself is not known to engage in binding to metals and it is likely that cannot form a stable structure39 and will readily dissociate to PbI2 and HI. Thus, the claim that HPbI3 is a real compound deserves closer attention and we set out to investigate it in the context of the present manuscript.
Here, we report that HPbI3 does not exist and instead what was believed to be it is in fact a compound of DMAPbI3 (DMA = dimethylammonium, (CH3)2NH2+), where DMA is a decomposition product of the acidic hydrolysis of DMF catalyzed by HI. We demonstrate that the black CsPbI3 films deposited from DMF containing HI/HPbI3 and reported to be all-inorganic black phase of CsPbI3 are not. Instead, they are the mixed-cation perovskite phase of cesium dimethylammonium lead iodide (Cs1-xDMAxPbI3, x = 0.2 to 0.5). The α-Cs1-xDMAxPbI3 perovskite films have similar characteristics with the black γ-CsPbI3 films arising from HI addition, but they exhibit better charge transport due to the superior band structure characteristics (larger bandwidth, smaller band gap) of the cubic phase. Our best-performing solar cell based on α-Cs0.7DMA0.3PbI3 perovskite absorber achieves a remarkable PCE of 12.62%. Our results reveal the existence of α-Cs0.7DMA0.3PbI3 a black 3D perovskite which is a great absorber for the fabrication of high efficiency solar cells.
Results
Phase diversity of CsPbI3
The structure of halide perovskites which have a general formula of AMX3 can be only stabilized by limited cations, according to the rule of tolerance factor, t39,40. Where A, M, and X represent a nonbonding univalent cation, an octahedrally coordinated bivalent metal ion, and a monoanionic halide ion, respectively5,41. A cubic structure of perovskite materials typically has a suitable t value ranged from 0.9 to 1.0, which is defined by the equation of t = (rA + rX)/√2(rM + rX). rA, rM, and rX represent the ionic sizes of A, B, and X, respectively42. Different A cations have different ionic sizes, only cubic MAPbI3 could be stable at room temperature. Both FAPbI3 and CsPbI3 normally adopt the yellow phase at room temperature, due to the too large and too small size of A cations, respectively5,42. Specifically, CsPbI3 can adopt two structures:21 the NH4CdCl3-type yellow phase (δ-phase) stable at room temperature (Fig. 1a) and the black cubic CaTiO3-type (α-phase) structure stable above 300 °C (Fig. 1b). The perovskite structure can be kinetically stabilized at room temperature, where it adopts the black orthorhombic γ-phase (bandgap of around 1.73 eV) but it converts within hours to the δ-phase5,42,43. Thus, CsPbI3 is typically stabilized by adjusting the t via formation of solid solutions such as partial replacement of I with smaller Br anions to form CsPbI3−xBrx compositions, which exhibit a wider band gap44,45,46,47,48,49,50,51,52,53,54, or by substituting the A-site with larger organic cations such as FA to form Cs1−xFAxPbI3 solid solutions55. DMA (2.72 Å) is very similar in shape to FA (2.53 Å) but has a slightly larger effective radius owing to the conjugated bonding in the latter (Fig. 1c)56,57. Because of this, pure DMAPbI3, which has a large t = 1.026 and is not able to form the perovskite structure, unlike FAPbI3. Pure DMAPbI3, which is identical to what previously reported as HPbI3, by inspection of the reported and simulated powder diffraction patterns (PXRD), has a 1D structure assembled by face-sharing [PbI6]4− octahedra58, and it is isostructural with yellow FAPbI35. Unlike FAPbI3, DMAPbI3 cannot be isolated as a black perovskite due to the too large size of DMA which leads to a prohibitively large t, even at elevated temperatures. On the other hand, the effective radius of Cs cation is only 1.88 Å and pure CsPbI3 has a low tolerance factor of t = 0.851 which is below the ideal range of cubic structure (Fig. 1d)17,19. However, mixing Cs with DMA, i.e., Cs1-xDMAxPbI3, can on average bring the effective tolerance factor within the desirable region, thus enabling the stabilization of a black perovskite phase, namely Cs1−xDMAxPbI3 (x = 0.2 to 0.5), having a t = 0.904 (x = 0.3) that allows it to adopt the cubic α-phase (Fig. 1d), similarly to Cs1−xFAxPbI3.
Crystal and molecular structures. a Crystal structure of CsPbI3 with non-perovskite yellow orthorhombic (δ) phase. b Crystal structure of CsPbI3 with perovskite black cubic (α) phase. c Molecular structures of FA (top) and DMA (bottom) cations. d Tolerance factors of CsPbI3, DMAPbI3, and Cs0.7DMA0.3PbI3
Cs1-xDMAxPbI3 film properties
This has indeed turned out to be the case in our thin films, where the nominal Cs0.7DMA0.3PbI3 composition is able to adopt the perovskite phase. As shown in Fig. 2a, the Cs0.7DMA0.3PbI3 film without HI addition shows a black color, indicting the successful fabrication of black-phase perovskite structure with lattice constant a = 6.272 Å and Pm-3m space group. On the contrary, the pristine CsPbI3 and DMAPbI3 films deposited on FTO/PEDOT substrates are yellow and pale-yellow color (Fig. 2a), respectively, because they are non-perovskite NH4CdCl3 structure-type56. For comparison, we also fabricated CsPbI3 film with HI addition (see details in Methods). Similar to the previously reported results18,25,26,59, the CsPbI3 film prepared with HI addition also shows a black color and a shiny surface (Fig. 2a), indicative of the formation of the similar black-phase perovskite structure. The color of the films is in good agreement with the band gaps of the respective compounds as determined by optical absorption measurements. Fig. 2b shows that the Cs0.7DMA0.3PbI3 film has an absorbance onset at around 730 nm, indicating a band gap of 1.7 eV. The CsPbI3 film with HI addition shows a similar absorption but slightly blue-shifted from 730 nm to 710 nm. As expected, the pure CsPbI3 and DMAPbI3 films only show an absorption at around 440 nm and 390 nm, respectively, due to the wide band gaps of these yellow phases18. Photoluminescence (PL) measurements reveal the same trend. As shown in Fig. 2c, the Cs0.7DMA0.3PbI3 film has a PL emission at around 724 nm. The black CsPbI3 film with HI addition shows a blue-shift PL peak at around 704 nm, consistent with the trend of UV-vis absorption spectra. Both CsPbI3 and DMAPbI3 films only show negligible PL emission, due to the indirect nature of the band gaps.
Film properties and component studies. a Photographs, b UV-vis absorption spectra, c PL spectra, and d XRD patterns of a pure CsPbI3 film, a pure DMAPbI3 film, a HI-treated CsPbI3 film, and a Cs0.7DMA0.3PbI3 film. e NMR spectra of the powder obtained from scratching away Cs0.7DMA0.3PbI3 films and DMAI polycrystalline powder synthesized from DMF and HI, which were dissolved in DMSO-d6
X-ray diffraction (XRD) patterns on the thin-films confirm the structure description discussed above. As shown in Fig. 2d, the main peaks of the neat yellow CsPbI3 film locates at 9.9°, 13.2°, 22.8° etc. are assigned to the orthorhombic (Pnma) yellow δ-phase (Supplementary Fig. 1)18. The XRD pattern of the DMAPbI3 film only shows two main reflections at 2θ = 11.8° and 20.4° (Fig. 2d), due to the preferred orientation, corresponding to the hexagonal (P63/mmc) yellow phase (Supplementary Fig. 2). The Cs0.7DMA0.3PbI3 film shows peaks at 2θ = 14.4°, 20.2°, 28.9°, and 40.9° etc., which can be indexed to the (100), (110), (200), and (220) reflections of cubic CsPbI3, showing some orientation of the perovskite in both (100) and (110) directions of the cubic (Pm-3m) α-phase (Supplementary Fig. 3)18,30. While the CsPbI3 film with HI additive exhibits similar peaks, it has no preferred orientation showing reflections that correspond to the black orthorhombic (Pbnm) perovskite γ-phase (Supplementary Figs. 4–5). In addition, the CsPbI3 film with HI also reveals a weak reflection at 2θ = 9.9°, which corresponds to the yellow δ-phase, indicating that the conversion to the black phase is incomplete.
To further prove that DMA cation is actually present in the black CsPbI3 films, we measured the proton nuclear magnetic resonance (1H NMR) spectrum of the powder obtained from scratching away the Cs0.7DMA0.3PbI3 films and dissolving it in dimethyl sulfoxide-d6 (DMSO-d6) (Fig. 2e). The signals of –NH2+– (singlet) and –CH3 (doublet) protons are located at δ = 8.17 ppm and δ = 2.55 ppm, respectively. The ratio integrated from the signals of –NH2+– and –CH3 is 1:3, indicating that DMA is protonated. The NMR spectrum of the pure DMAI powder confirms the peak position (Supplementary Fig. 6), thus demonstrating DMA is incorporated in the CsPbI3 structure as would be expected by the Cs0.7DMA0.3PbI3 composition. Remarkably, the NMR spectrum of the DMSO-d6 solution of the powder obtained from scratching away the CsPbI3 film with HI addition also shows the same two peaks in the 1:3 for –NH2+– and –CH3 ratio (Supplementary Fig. 7).
The chemical mechanism and reality of so-called HPbI3
Thus, it transpires from the NMR results that the DMA cation is produced via a side reaction (of DMF with HI) and then gets incorporated in the CsPbI3 films. Since only CsI and PbI2 were dissolved in the DMF solvent, the only possible source of DMA in the precursor solution is DMF itself, which is known to slowly hydrolyze to DMA and formic acid (HCOOH)60,61,62,63,64,65. Addition of HI significantly accelerates the hydrolysis according to Eq. 1, heavily shifting the equilibrium towards the dissociation products, thus generating significant amounts of DMA in the mixture66.
In order to further demonstrate this reaction, we synthesized the DMAI from the reaction of DMF and HI directly (see Methods). As Fig. 2e illustrate, DMAI can indeed be readily formed by the reaction between DMF and HI, with 1H-NMR spectra and XRD patterns confirming its identity in comparison with the commercial DMAI compound (Supplementary Figs. 6 and 8). Note that the two peaks at δ = 7.22 ppm and δ = 6.18 ppm shown in Fig. 2e are assigned to the –PH2– protons of hypophosphorous acid (H3PO2) which presents as a stabilizer in concentrated HI aqueous solutions. Therefore, this is the reason why 1H-NMR spectra of Cs0.7DMA0.3PbI3, CsPbI3 with HI, DMAI, and DMF + HI samples are identical. Most importantly, it explains why the UV-vis absorption, PL, XRD, and 1H-NMR results between the intentionally made Cs0.7DMA0.3PbI3 film and the DMF derived CsPbI3 with HI addition films are so similar. Note that the red shift (around 20 nm) of PL and UV-vis spectra of Cs0.7DMA0.3PbI3 with respect to the CsPbI3 sample with HI is because the former (which contains significant amounts of DMA) adopts the cubic α-CsPbI3 phase, whereas the latter (containing only small amounts of DMA) stabilizes the orthorhombic γ-phase (as would be expected by the effective tolerance factor discussed above).
The hydrolysis of DMF in the presence of acid such as HI as a film fabrication tool has great implications in the fabrication of perovskite films and solar cells. Since HI addition has been reported to alter the phase transition temperature and improve the film morphology of MAPbI367, and also was identified as the critical reagent for stabilizing the perovskite phase of CsPbI3, the nature and exact composition of these crystalline films will need to be re-examined18,25,26,59. In addition, HPbI3, obtained from the reaction of HI, DMF, and PbI2, has been widely used for making stable FAPbI338, CsPbI331,37,53, and MAPbI3 solar cells68, which actually does not exist and should be reformulated to DMAPbI3. It is confirmed by the XRD patterns of our DMAPbI3 film (Fig. 2d and Supplementary Fig. 2) and the reported HPbI338, showing the exact same reflections. Given that the use of HPbI3 in a serendipitous manner, these systems also need to be revisited and studied carefully under the understanding of the DMF hydrolysis mechanism. It is important to re-evaluate these systems counting the time as a synthetic parameter since the preparation will significantly vary over time as different amounts of DMA are produced at different times of the reactions and therefore the composition of the films will vary accordingly.
In a previous report, we demonstrated that HI addition can alter the MAPbI3 phase transition temperature67. A room temperature phase transition from tetragonal to cubic was observed for MAPbI3 films treated with high HX (X = Cl, Br, I) concentrations. In our original hypothesis, we and others speculated that the reduction of the crystallite size induced by the acid were responsible for this phase transition18,67. HI can improve the perovskite film morphology and eliminate the hysteresis of MAPbI3 solar cells69,70. However, in light of the results reported here, we can now revise our original concept and ascribe the compositional phase transition to the presence of DMA, induced by the presence of acid. Note that the concept of the role of acid expands beyond the HX acids, as other acids such as H3PO271 appear to have a similar effect. Therefore, based on the above results, all those compositionally-induced phase transitions may be attributed to the introduction of the in-situ forming DMA cation in the perovskite lattice and it applies equally well in the MAPbI3 and CsPbI3 systems. Note that FAPbI3 is excluded since FA is already too large to stabilize the perovskite structure.
Now that we have established that the so-called all-inorganic CsPbI3 films processed with HI and HPbI3 are in fact Cs1-xDMAxPbI3 films, we return to the study of the latter and their behavior and performance in complete solar cells. The CsPbI3 film with HI addition was prepared by a conventional one-step method and using DMF as solvent18, actually is unintentionally-made Cs1−xDMAxPbI3 but we still referred it as HI-treated CsPbI3 in the following discussion. A small amount of HI aqueous solution was added into the CsPbI3 precursor after all materials were dissolved in DMF solvent. All other intentionally-made Cs1−xDMAxPbI3 (x = 0 to 0.5) films were fabricated by CsI, PbI2, and pre-synthesized DMAI using a solvent-engineering method14,72 with a mixture of DMF and DMSO as the solvent. Scanning electron microscopy (SEM) images show that the yellow-phase CsPbI3 film has a rough surface with big grains and pin-holes, Fig. 3a. The morphology of the neat yellow DMAPbI3 film is even worse, showing some long and big cracks (Fig. 3b). Fig. 3c shows the one-step method prepared CsPbI3 film treated with HI which clearly has much smoother surface, still containing some pin holes. The best film quality was obtained from the CsPbI3 film with DMA cation i.e., Cs0.7DMA0.3PbI3. As shown in Fig. 3d, the Cs0.7DMA0.3PbI3 film has a smooth surface, big grains and few pinholes. The average grain size of the Cs0.7DMA0.3PbI3 film is around 500 nm, which is comparable with the film thickness. This is desirable since big grains facilitate charge transfer and reduce the recombination at grain boundaries14. Fig. 3e shows a cross-sectional SEM of a complete device based on the Cs0.7DMA0.3PbI3 absorber. The 380 nm thick Cs0.7DMA0.3PbI3 film is sandwiched between a thin poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) hole transporting layer and a thin fullerene (C60) electron transporting layer. The ultra-smooth Cs0.7DMA0.3PbI3 film enables the fabrication of solar cells with an inverted planar structure.
Film and device morphology. Top view SEM images of a a CsPbI3, b a DMAPbI3, c a HI-treated CsPbI3, and d a Cs0.7DMA0.3PbI3 film deposited on FTO/PEDOT substrates. e Cross-sectional SEM image of a completed device using a PEDOT:PSS hole transporting layer, a Cs0.7DMA0.3PbI3 absorber, a C60/BCP electron transporting layer, and a Ag metal electrode
Effects of DMA cation on solar cell performance
We then investigated the effects of various absorbers on device performance. Fig. 4a shows the photocurrent density-voltage (J–V) curves of the representative solar cells based on the yellow CsPbI3, the pale-yellow DMAPbI3, the black HI-treated CsPbI3, and the black Cs0.7DMA0.3PbI3 absorbers, measured under a reverse voltage scan. The performance of the solar cells based on different absorbers shows huge difference. As expected, the solar cells with yellow CsPbI3 and DMAPbI3 absorbers show very poor efficiencies. As shown in Fig. 4a, the solar cell employed the yellow CsPbI3 film achieved a very low PCE of 0.002% with an open-circuit voltage (Voc) of 0.05 V, a short-circuit current density (Jsc) of 0.21 mA cm−2, a fill factor (FF) of 22.63%. A similarly low PCE of 0.001% with a Voc of 0.03 V, a Jsc of 0.30 mA cm−2, and an FF of 14.23% was obtained for the solar cell employing the yellow DMAPbI3 film. Both CsPbI3 and DMAPbI3 devices were almost short-circuited, due to the poor film quality, non-perovskite structure, and limited light absorption due to their wide band gaps. The device performance of the so-called black CsPbI3 was significantly enhanced after adding HI into the perovskite precursor. The HI-treated CsPbI3 solar cell achieved a Voc of 0.82 V, a Jsc of 9.56 mA cm−2, an FF of 61.56%, and a PCE of 4.84%, which is comparable with other reports18,31. The enhanced performance can be mainly attributed to the better morphology and the unintended incorporation of DMA cation which can stabilize the Cs1−xDMAxPbI3 perovskite as a black phase. Nevertheless, adding HI into the perovskite precursor makes it difficult to control the amount of DMA in the final film in a consistent manner, so after realizing the role of HI in the film formation the main focus was given in the easily reproducible fabrication of devices containing controlled amounts of DMA. As expected, the solar cells based on the designed Cs0.7DMA0.3PbI3 absorber yielded much better performance. A significantly enhanced PCE of 10.39% with a high Voc of 1.03 V, a Jsc of 15.43 mA cm−2, an FF of 65.61% was achieved for an optimized device, with both the high-quality of the films and narrow band gap contributing to the decent performance of the Cs0.7DMA0.3PbI3 solar cells.
Solar cell performance. a J–V curves of devices using various absorbers measured under reverse voltage scans (from Voc to 0 V). b J–V curves of the champion Cs0.7DMA0.3PbI3 solar cell measured under different voltage scan directions. c EQE spectrum and integrated Jsc of a Cs0.7DMA0.3PbI3 solar cell. d PCE statistics for 40 Cs0.7DMA0.3PbI3 solar cells
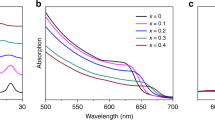
We find that the amount of DMA cation incorporated in the perovskite structure of CsPbI3 has a huge effect on the material properties. Unlike the HI addition to DMF which results in the uncontrollable generation of DMA, it is easy to fabricate Cs1-xDMAxPbI3 films using precise DMA ratios. First, the film morphology was affected by different amounts of DMA. As shown in Supplementary Fig. 9, Cs0.8DMA0.2PbI3, Cs0.7DMA0.3PbI3, Cs0.6DMA0.4PbI3, and Cs0.5DMA0.5PbI3 films show different morphology. All films are compact and have smooth surface with Cs0.7DMA0.3PbI3 film having the biggest grains. The Cs0.6DMA0.4PbI3, and Cs0.5DMA0.5PbI3 films are compact and have smaller grains. In addition, these films show similar band baps and PL peaks but different absorption intensities (Supplementary Fig. 10). In particular, the Cs0.7DMA0.3PbI3 film shows the strongest absorption at long wavelength range. Supplementary Fig. 11 shows that the different films also have different XRD results. The diffraction intensities of the cubic Cs0.6DMA0.4PbI3 and Cs0.5DMA0.5PbI3 films are weaker while the extra peak at 2θ = 11.8° indicates that DMAPbI3 is present as a second phase, suggesting that the substitution of DMA for Cs is limited. Cs0.7DMA0.3PbI3 and Cs0.8DMA0.2PbI3 show the exclusive formation of the black α-CsPbI3 phase along with the former orienting preferentially along both (100) and (110) planes and the latter exclusively along the (110) planes. From the combined data, around 30% of DMA seems to be the optimum amount to make high-quality perovskite film. This is also reflected in the device performance of the corresponding solar cells based on the CsPbI3 absorbers with various amounts of DMA. Supplementary Fig. 12 shows the J–V curves of the solar cells using the Cs0.8DMA0.2PbI3, Cs0.7DMA0.3PbI3, Cs0.6DMA0.4PbI3, and Cs0.5DMA0.5PbI3 absorbers. The device performance first increases and then decreases as the DMA amount increases. As expected, the solar cell derived from the best-quality Cs0.7DMA0.3PbI3 film achieved the highest performance (Supplementary Table 1).
Figure 4b shows the J–V curves of the best-performing Cs0.7DMA0.3PbI3 solar cell measured under different voltage scan directions. This solar cell achieved a Voc of 0.99 V, a Jsc of 16.65 mA cm−2, an FF of 76.49%, and therefore a high PCE of 12.62% when measured under the reverse voltage scan. A similar PCE of 12.31% with a Voc of 0.99 V, a Jsc of 16.44 mA cm−2, and an FF of 75.71% was achieved for the solar cell measured under the forward voltage scan, suggesting small hysteresis behavior of our devices. External quantum efficiency (EQE) measurement was taken to confirm the high Jsc obtained from the J–V curves. The EQE spectrum of the solar cell based on the Cs0.7DMA0.3PbI3 absorber is shown in Fig. 4c. The cell shows high average value in the whole visible wavelength range. The Jsc integrated from the EQE curve is about 15.95 mA cm−2, which is very close to the Jsc measured from the J–V curves. To check the reproducibility of the device performance, we then made 40 solar cells based on the Cs0.7DMA0.3PbI3 absorbers. The histograms of statistics PCEs for these cells are shown in Fig. 4d, ranged from 6.73 to 12.62% due to the inhomogeneous film morphology. The 40 solar cells achieved an average PCE of 9.27 ± 1.28% with a Voc of 1.01 ± 0.03 V, a Jsc of 15.45 ± 1.80 mA cm−2, and an FF of 59.40 ± 4.01%.
Furthermore, we also added the DMA cation into solutions of CsPbI2Br and CsPbIBr2 perovskites. Supplementary Fig. 13a-b shows UV-vis absorption and XRD pattern of a Cs0.7DMA0.3I2Br film coated on a FTO/PEDOT:PSS substrate. The Cs0.7DMA0.3I2Br film shows an absorption onset at around 685 nm, according to a band gap of around 1.8 eV (Supplementary Fig. 13a). The Cs0.7DMA0.3I2Br film has good crystalline quality and shows peaks at 14.7°, 20.5°, 29.5°, and 41.6° (Supplementary Fig. 13b), which can be indexed to the (100), (110), (200), and (220) planes. Supplementary Fig. 13c shows the J–V curve of a solar cell based on the CsPbI2Br absorber. The Cs0.7DMA0.3I2Br solar cell achieved a PCE of 5.24%, a Voc of 1.02 V, a Jsc of 12.39 mA cm−2, and an FF of 41.67%. The lower Jsc is consistent with the wider band gap compared to Cs0.7DMA0.3PbI3. The Cs0.7DMA0.3PbIBr2 film has a more blue-shifted absorption onset at around 620 nm, according to a wider band gap of around 2.0 eV, Supplementary Fig. 14a. The Cs0.7DMA0.3PbIBr2 solar cell achieved a PCE of 2.80%, a Voc of 1.11 V, a Jsc of 8.55 mA cm−2, and an FF of 29.57% (Supplementary Fig. 14c). A much lower Jsc is obtained from the Cs0.7DMA0.3PbIBr2 cell because of the much wider band gap. Higher performance for the Cs1-xDMAxPbI2Br and Cs1−xDMAxPbIBr2 cells is anticipated after further device optimization.
Discussion
We have demonstrated that small cation DMA can be formed in suite by the HI induced decomposition of DMF and can subsequently stabilize the black perovskite phase of CsPbI3. However, the films are not really the all-inorganic CsPbI3 phase but the Cs1-xDMAxPbI3 (x = 0.2 to 0.5) cubic perovskite solid solution. Cs0.7DMA0.3PbI3 has a band gap of around 1.7 eV and can enable high efficiency solar cells. In the context of this work, we realize that DMA is likely present in many other devices reported in the past, since it can be inadvertently created from the degradation of the commonly used DMF solvent and then incorporated to some extend in the perovskite structures as an A cation. This DMF decomposition process is accelerated by acidic hydrolysis, and must be taken into account when considering the role of the solvent degradation in device performance. DMF hydrolysis therefore explains the effects of the widely used HI addition and HPbI3 methods to stabilize the purported all-inorganic black CsPbI3 perovskite. Based on above results, we show that HPbI3 does not exist while at the same time it is actual in situ formation of the DMA cation, which should widely exist in the perovskites processed with DMF, HI, and HPbI3. We anticipate that the DMA cation, which now has been revealed as a viable A-site cation, may be unsuspectingly occurring not only in "CsPbI3 films" but also in films of other perovskite families, for example, pure MA, MA/FA, Cs/FA, and Cs/MA/FA mixed-cation perovskites. We suggest that NMR spectroscopy be used for such films by dissolving them in pure DMF or DMSO to check for the presence of the DMA cation.
Methods
Device fabrication
For the DMAI synthesis, DMF was slowly added into the HI solution with continue stirring at 0 °C for 30 min. Water and excess DMF in solution was removed by low-vacuum rotary evaporation and then a white polycrystalline powder (DMAI powder) was obtained. Pre-cleaned FTO substrates were treated with UV-Ozone for 30 min. PEDOT:PSS films were coated on the FTO substrates with a spin-rate of 4000 rpm for 30 s and then annealing for 30 min at 150 °C. Perovskite films were deposited on FTO/PEDOT:PSS substrates in a N2-filled glovebox. Neat CsPbI3, neat DMAPbI3, and Cs1−xDMAxPbX3 (X = I, Br) films were prepared by a solvent-engineering method14 at a spin rate of 4000 rpm for 60 s. 0.7 ml of diethyl ether was quickly dropped onto the rotating substrates during the spin-coating process. CsPbI3 films with HI addition were prepared by a one-step method at a spin rate of 2000 rpm for 60 s18. The neat CsPbI3 precursor was prepared by dissolving 260 mg of CsI (99.999%, Sigma-Aldrich) and 461 mg of PbI2 (beads, 99.999%, Sigma-Aldrich) in 0.8 ml of DMF (anhydrous, 99.8%, Sigma-Aldrich) and 0.2 ml of DMSO (anhydrous,≥ 99.9%, Sigma-Aldrich). The neat DMAPbI3 precursor was prepared by dissolving 173 mg of DMAI and 461 mg of PbI2 in 0.8 ml of DMF and 0.2 ml of DMSO. The Cs1−xDMAxPbI3 precursor was prepared by dissolving (1−x) × 260 mg of CsI, x × 173 mg of DMAI, and 461 mg of PbI2 in 0.8 ml of DMF and 0.2 ml of DMSO. The CsPbI3 precursor with HI addition was prepared by dissolving 260 mg of CsI and 461 mg of PbI2 in 2 ml of DMF and then adding 3.3 vol% HI (57 wt.% in H2O, 99.95%, Sigma-Aldrich) into the dissolved solution. All the films were annealed for 2 min at 60 °C and then 5 min at 100 °C on a hot plate. To complete the devices, a thin C60 (20 nm), a thin 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline (BCP) (5 nm), and a Ag (80 nm) films were sequentially thermal evaporated on top of the absorber layers using a metal mask. The active area of the solar cells was 0.09 cm2.
Film and device characterization
1H-NMR spectra were measured with Bruker Avance III 600 MHz system with BBI probe. The morphology of the films and devices was examined on a high-resolution field emission SEM (Hitachi SU8030). XRD patterns of the films were characterized by a Rigaku Miniflex600 pXRD (Cu Kα graphite, λ = 1.5406 Å) operating at 40 kV/15 mA with a Kβ foil filter. PL spectra were taken on a Horiba LabRAM HR Evolution confocal Raman microscope spectrometer (600 g mm−1 diffraction grating) equipped with a diode continuouswave laser (473 nm, 25 mW) and a Synapse charge-coupled device camera. UV-vis absorption spectra of the films were measured with a Shimadzu UV-3600 UV-vis NIR spectrometer operating in the 200–2000 nm region at room temperature. EQE spectrum was obtained on an Oriel model QE-PV-SI instrument equipped with a NIST-certified Si diode. J–V curves were recorded using a Keithley model 2400 instrument under AM1.5 G simulated irradiation with a standard solar simulator (Abet Technologies). J–V curves were measured from 1.5 V to −0.2 V (reverse) or from −0.2 V to 1.5 V (forward) with an integration time of 16.67 ms and a voltage step of 11.4 mV. The light intensity of the solar simulator was calibrated by a NREL-certified monocrystalline silicon solar cell.
Data availability
The authors declare that the main data supporting the findings of this study are available within the article and its Supplementary Information. Extra data are available from the authors upon request.
References
Mitzi, D. B. Synthesis, structure, and properties of organic-inorganic perovskites and related materials. Prog. Inorg. Chem. 48, 1–121 (1999).
Yin, W. J., Shi, T. & Yan, Y. Unique properties of halide perovskites as possible origins of the superior solar cell performance. Adv. Mater. 26, 4653–4658 (2014).
Dong, Q. et al. Electron-hole diffusion lengths>175 μm in solution-grown CH3NH3PbI3 single crystals. Science 347, 967–970 (2015).
Stranks, S. D. et al. Electron-hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science 342, 341–344 (2013).
Stoumpos, C. C., Malliakas, C. D. & Kanatzidis, M. G. Semiconducting tin and lead iodide perovskites with organic cations: phase transitions, high mobilities, and near-infrared photoluminescent properties. Inorg. Chem. 52, 9019–9038 (2013).
Nagabhushana, G. P., Shivaramaiah, R. & Navrotsky, A. Thermochemistry of multiferroic organic-inorganic hybrid perovskites [(CH3)2NH2][M(HCOO)3] (M = Mn, Co, Ni, and Zn). J. Am. Chem. Soc. 137, 10351–10356 (2015).
Ke, W., Stoumpos, C. C. & Kanatzidis, M. G. “Unleaded” perovskites: status quo and future prospects of tin-based perovskite solar cells. Adv. Mater. 1803230. https://doi.org/10.1002/adma.201803230 (2018).
Ke, W. et al. Enhanced photovoltaic performance and stability with a new type of hollow 3D perovskite {en}FASnI3. Sci. Adv. 3, e1701293 (2017).
Li, X. et al. A vacuum flash–assisted solution process for high-efficiency large-area perovskite solar cells. Science 353, 58–62 (2016).
Kim, H. S. et al. Lead iodide perovskite sensitized all-solid-state submicron thin film mesoscopic solar cell with efficiency exceeding 9%. Sci. Rep. 2, 591 (2012).
Lee, M. M., Teuscher, J., Miyasaka, T., Murakami, T. N. & Snaith, H. J. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012).
Yang, W. S. et al. Iodide management in formamidinium-lead-halide–based perovskite layers for efficient solar cells. Science 356, 1376–1379 (2017).
Saliba, M. et al. Incorporation of rubidium cations into perovskite solar cells improves photovoltaic performance. Science 354, 206–209 (2016).
Ke, W. et al. Employing lead thiocyanate additive to reduce the hysteresis and boost the fill factor of planar perovskite solar cells. Adv. Mater. 28, 5214–5221 (2016).
Ke, W. et al. Low-temperature solution processed tin oxide as an alternative electron transporting layer for efficient perovskite solar cells. J. Am. Chem. Soc. 137, 6730–6733 (2015).
Gao, L.-L., Li, C.-X., Li, C.-J. & Yang, G.-J. Large-area high-efficiency perovskite solar cells based on perovskite films dried by the multi-flow air knife method in air. J. Mater. Chem. A 5, 1548–1557 (2017).
Chen, H. et al. Inorganic perovskite solar cells: a rapidly growing field. Sol. RRL 2, 1700188 (2018).
Eperon, G. E. et al. Inorganic caesium lead iodide perovskite solar cells. J. Mater. Chem. A 3, 19688–19695 (2015).
Liang, J., Liu, J. & Jin, Z. All-inorganic halide perovskites for optoelectronics: progress and prospects. Solar RRL 1, 1700086 (2017).
Nagabhushana, G. P., Shivaramaiah, R. & Navrotsky, A. Direct calorimetric verification of thermodynamic instability of lead halide hybrid perovskites. Proc. Natl Acad. Sci. USA 113, 7717–7721 (2016).
Marronnier, A. et al. Anharmonicity and disorder in the black phases of cesium lead iodide used for stable inorganic perovskite solar cells. ACS Nano 12, 3477–3486 (2018).
Sutton, R. J. et al. Cubic or orthorhombic? Revealing the crystal structure of metastable black-phase CsPbI3 by theory and experiment. ACS Energy Lett. 3, 1787–1794 (2018).
Chung, I. et al. CsSnI3: Semiconductor or metal? High electrical conductivity and strong near-infrared photoluminescence from a single material. High hole mobility and phase-transitions. J. Am. Chem. Soc. 134, 8579–8587 (2012).
Lau, C. F. J. et al. Strontium-doped low-temperature-processed CsPbI2Br perovskite solar cells. ACS Energy Lett. 2, 2319–2325 (2017).
Hu, Y. et al. Bismuth incorporation stabilized α-CsPbI3 for fully inorganic perovskite solar cells. ACS Energy Lett. 2, 2219–2227 (2017).
Luo, P. et al. Solvent engineering for ambient-air-processed, phase-stable CsPbI3 in perovskite solar cells. J. Phys. Chem. Lett. 7, 3603–3608 (2016).
Sanehira, E. M. et al. Enhanced mobility CsPbI3 quantum dot arrays for record-efficiency, high-voltage photovoltaic cells. Sci. Adv. 3, eaao4204 (2017).
Swarnkar, A. et al. Quantum dot–induced phase stabilization of α-CsPbI3 perovskite for high-efficiency photovoltaics. Science 354, 92–95 (2016).
Frolova, L. A. et al. Highly efficient all-inorganic planar heterojunction perovskite solar cells produced by thermal coevaporation of CsI and PbI2. J. Phys. Chem. Lett. 8, 67–72 (2017).
Wang, Q. et al. Stabilizing the α-phase of CsPbI3 perovskite by sulfobetaine zwitterions in one-step spin-coating films. Joule 1, 371–382 (2017).
Zhang, T. et al. Bication lead iodide 2D perovskite component to stabilize inorganic α-CsPbI3 perovskite phase for high-efficiency solar cells. Sci. Adv 3, e1700841 (2017).
Wang, Q. et al. µ-Graphene crosslinked CsPbI3 quantum dots for high efficiency solar cells with much improved stability. Adv. Energy Mater. 8, 1800007 (2018).
Wang, P. et al. Solvent-controlled growth of inorganic perovskite films in dry environment for efficient and stable solar cells. Nat. Commun. 9, 2225 (2018).
Zhao, B. et al. Thermodynamically stable orthorhombic γ-CsPbI3 thin films for high-performance photovoltaics. J. Am. Chem. Soc. 140, 11716–11725 (2018).
Wang, Y., Zhang, T., Kan, M. & Zhao, Y. Bifunctional stabilization of all-inorganic α-CsPbI3 perovskite for 17% efficiency photovoltaics. J. Am. Chem. Soc. 140, 12345–12348 (2018).
Protesescu, L. et al. Nanocrystals of cesium lead halide perovskites (CsPbX3, X = Cl, Br, and I): novel optoelectronic materials showing bright emission with wide color gamut. Nano Lett. 15, 3692–3696 (2015).
Jiang, Y. et al. Reduced-dimensional α-CsPbX3 perovskites for efficient and stable photovoltaics. Joule 2, 1356–1368 (2018).
Wang, F., Yu, H., Xu, H. & Zhao, N. HPbI3: a new precursor compound for highly efficient solution-processed perovskite solar cells. Adv. Funct. Mater. 25, 1120–1126 (2015).
Kieslich, G., Sun, S. & Cheetham, A. K. Solid-state principles applied to organic-inorganic perovskites: new tricks for an old dog. Chem. Sci. 5, 4712–4715 (2014).
Goldschmidt, V. M. Die gesetze der krystallochemie. Naturwissenschaften 14, 477–485 (1926).
Stoumpos, C. C. & Kanatzidis, M. G. Halide perovskites: poor man’s high-performance semiconductors. Adv. Mater. 28, 5778–5793 (2016).
Stoumpos, C. C. & Kanatzidis, M. G. The renaissance of halide perovskites and their evolution as emerging semiconductors. Acc. Chem. Res. 48, 2791–2802 (2015).
Møller, C. K. [The structure of perovskite-like caesium plumbo trihalides]. Mat. Fys. Medd. Dan. Vid. Sels. 32, 1–18 (1959).
Nam, J. K. et al. Potassium incorporation for enhanced performance and stability of fully inorganic cesium lead halide perovskite solar cells. Nano. Lett. 17, 2028–2033 (2017).
Kulbak, M. et al. Cesium enhances long-term stability of lead bromide perovskite-based solar cells. J. Phys. Chem. Lett. 7, 167–172 (2016).
Sutton, R. J. et al. Bandgap-tunable cesium lead halide perovskites with high thermal stability for efficient solar cells. Adv. Energy Mater. 6, 1502458 (2016).
Ma, Q., Huang, S., Wen, X., Green, M. A. & Ho-Baillie, A. W. Y. Hole transport layer free inorganic CsPbIBr2 perovskite solar cell by dual source thermal evaporation. Adv. Energy Mater. 6, 1502202 (2016).
Lau, C. F. J. et al. CsPbIBr2 perovskite solar cell by spray-assisted deposition. ACS Energy Lett. 1, 573–577 (2016).
Chang, X. et al. Carbon-based CsPbBr3 perovskite solar cells: all-ambient processes and high thermal stability. ACS Appl. Mater. Interfaces 8, 33649–33655 (2016).
Niezgoda, J. S., Foley, B. J., Chen, A. Z. & Choi, J. J. Improved charge collection in highly efficient CsPbBrI2 solar cells with light-induced dealloying. ACS Energy Lett. 2, 1043–1049 (2017).
Zeng, Q. et al. Polymer-passivated inorganic cesium lead mixed-halide perovskites for stable and efficient solar cells with high open-circuit voltage over 1.3 V. Adv. Mater. 30, 1705393 (2018).
Ma, Q. et al. The effect of stoichiometry on the stability of inorganic cesium lead mixed-halide perovskites solar cells. J. Phys. Chem. C. 121, 19642–19649 (2017).
Wang, Y., Zhang, T., Xu, F., Li, Y. & Zhao, Y. A facile low temperature fabrication of high performance CsPbI2Br all-inorganic perovskite solar cells. Sol. RRL 2, 1700180 (2018).
Bian, H. et al. Graded bandgap CsPbI2+xBr1−x perovskite solar cells with a stabilized efficiency of 14.4%. Joule 2, 1500–1510 (2018).
Li, Z. et al. Stabilizing perovskite structures by tuning tolerance factor: formation of formamidinium and cesium lead iodide solid-state alloys. Chem. Mater. 28, 284–292 (2015).
Stoumpos, C. C., Mao, L., Malliakas, C. D. & Kanatzidis, M. G. Structure-band gap relationships in hexagonal polytypes and low-dimensional structures of hybrid tin iodide perovskites. Inorg. Chem. 56, 56–73 (2017).
Kieslich, G., Sun, S. & Cheetham, A. K. Solid-state principles applied to organic–inorganic perovskites: new tricks for an old dog. Chem. Sci. 5, 4712–4715 (2014).
Mancini, A. et al. Synthesis, structural and optical characterization of APbX3 (A = methylammonium, dimethylammonium, trimethylammonium; X = I, Br, Cl) hybrid organic-inorganic materials. J. Solid State Chem. 240, 55–60 (2016).
Kim, Y. G. et al. Cesium lead iodide solar cells controlled by annealing temperature. Phys. Chem. Chem. Phys. 19, 6257–6263 (2017).
Muzart, J. N,N-Dimethylformamide: much more than a solvent. Tetrahedron 65, 8313–8323 (2009).
Juillard, J. Dimethylformamide: purification, tests for purity and physical properties. Pure Appl. Chem. 49, 885–892 (1977).
Noel, N. K. et al. Unveiling the influence of pH on the crystallization of hybrid perovskites, delivering low voltage loss photovoltaics. Joule 1, 328–343 (2017).
Dou, B. et al. Degradation of highly alloyed metal halide perovskite precursor inks: mechanism and storage solutions. ACS Energy Lett. 3, 979–985 (2018).
Nayak, P. K. et al. Mechanism for rapid growth of organic-inorganic halide perovskite crystals. Nat. Commun. 7, 13303 (2016).
Spanopoulos, I. et al. Enhanced gas-sorption properties of a high surface area, ultramicroporous magnesium formate. CrystEngComm 17, 532–539 (2015).
Jain, P., Dalal, N. S., Toby, B. H., Kroto, H. W. & Cheetham, A. K. Order−disorder antiferroelectric phase transition in a hybrid inorganic−organic framework with the perovskite architecture. J. Am. Chem. Soc. 130, 10450–10451 (2008).
Soe, C. M. et al. Room temperature phase transition in methylammonium lead iodide perovskite thin films induced by hydrohalic acid additives. ChemSusChem 9, 2656–2665 (2016).
Pang, S. et al. Transformative evolution of organolead triiodide perovskite thin films from strong room-temperature solid-gas interaction between HPbI3-CH3NH2 precursor pair. J. Am. Chem. Soc. 138, 750–753 (2016).
Heo, J. H., Han, H. J., Kim, D., Ahn, T. K. & Im, S. H. Hysteresis-less inverted CH3NH3PbI3 planar perovskite hybrid solar cells with 18.1% power conversion efficiency. Energy Environ. Sci. 8, 1602–1608 (2015).
Heo, J. H. et al. Planar CH3NH3PbI3 perovskite solar cells with constant 17.2% average power conversion efficiency irrespective of the scan rate. Adv. Mater. 27, 3424–3430 (2015).
Xiao, Z. et al. Unraveling the hidden function of a stabilizer in a precursor in improving hybrid perovskite film morphology for high efficiency solar cells. Energy Environ. Sci. 9, 867–872 (2016).
Jeon, N. J. et al. Solvent engineering for high-performance inorganic–organic hybrid perovskite solar cells. Nat. Mater. 13, 897–903 (2014).
Acknowledgements
This work was supported in part by the LEAP Center, an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, and Office of Basic Energy Sciences under Award DE-SC0001059 (solar cell fabrication and characterization). Work in sample synthesis, processing and structural characterization was supported by grant Department of Energy, Office of Science grant SC0012541. This work made use of the EPIC facility (NUANCE Center-Northwestern University), which has received support from the MRSEC program (NSF DMR-1720139) at the Materials Research Center, and the Nanoscale Science and Engineering Center (EEC-0118025/003), both programs of the National Science Foundation; the State of Illinois; and Northwestern University.
Author information
Authors and Affiliations
Contributions
W.K., C.C.S., and M.G.K. conceived the idea, designed the experiments, and wrote the manuscript. I.S. performed the NMR measurements and analyzed the data. W.K. carried out film and device fabrication and characterization. All authors discussed the results and commented on the manuscript. M.G.K. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ke, W., Spanopoulos, I., Stoumpos, C.C. et al. Myths and reality of HPbI3 in halide perovskite solar cells. Nat Commun 9, 4785 (2018). https://doi.org/10.1038/s41467-018-07204-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-07204-y
This article is cited by
-
rGO aerogel embedded with organic–inorganic hybrid perovskite for lightweight broadband electromagnetic wave absorption
Nano Research (2024)
-
Intermediate-phase engineering via dimethylammonium cation additive for stable perovskite solar cells
Nature Materials (2023)
-
Methylammonium-free wide-bandgap metal halide perovskites for tandem photovoltaics
Nature Reviews Materials (2023)
-
Recent progress in perovskite solar cells: material science
Science China Chemistry (2023)
-
Near-room-temperature martensitic actuation profited from one-dimensional hybrid perovskite structure
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.