Abstract
The genetic code used in nuclear genes is almost universal, but here we report that it changed three times in parallel during the evolution of budding yeasts. All three changes were reassignments of the codon CUG, which is translated as serine (in 2 yeast clades), alanine (1 clade), or the ‘universal’ leucine (2 clades). The newly discovered Ser2 clade is in the final stages of a genetic code transition. Most species in this clade have genes for both a novel tRNASer(CAG) and an ancestral tRNALeu(CAG) to read CUG, but only tRNASer(CAG) is used in standard growth conditions. The coexistence of these alloacceptor tRNA genes indicates that the genetic code transition occurred via an ambiguous translation phase. We propose that the three parallel reassignments of CUG were not driven by natural selection in favor of their effects on the proteome, but by selection to eliminate the ancestral tRNALeu(CAG).
Similar content being viewed by others
Introduction
In the vast majority of organisms, translation of mRNAs into proteins is carried out according to the standard (‘universal’) genetic code, which assigns each of the 64 possible codons to one of the 20 canonical amino acids or as a stop codon. The genetic code is implemented by tRNAs—the adapter molecules that physically connect amino acids to anticodons—and the aminoacyl tRNA synthetases that charge them. The code was initially suggested to be an immutable ‘frozen accident’, because any change to it would alter the sequences of most proteins and would be lethal or highly disadvantageous1. Later discoveries showed that the code is not completely frozen because some codon reassignments have occurred, albeit infrequently, during evolution2,3,4. Deviations from the standard code are most commonly seen in mitochondria, which are susceptible to drift because they have their own ribosomes and tRNAs, and only a few genes5. Genetic code changes in nuclear genomes are much rarer, and most of the known examples are reassignments where a former stop codon becomes a sense codon3,4,5,6.
Genetic code changes in which the meaning of a sense codon is switched from one amino acid to another are particularly rare in nuclear genomes. From 1989 until 2016, the only known example in all eukaryotes was the reassignment of CUG from leucine (its standard meaning) to serine in a clade of budding yeasts that includes Candida albicans7,8,9,10. In 2016, a second reassignment was discovered11,12, and surprisingly it also involved reassignment of CUG in a yeast species (Pachysolen tannophilus), this time to alanine (Fig. 1).
Theoretical models of how a sense codon could be reassigned from one amino acid to another during evolution can be grouped into two broad categories: ambiguous intermediate models, and unassigned codon models3. In the ambiguous intermediate models, a codon becomes reassigned by going through a transition phase during which it can be translated as both the old amino acid and the new one. In their original proposal of this model, Schultz and Yarus13 envisaged a cell containing two tRNAs that were charged with different amino acids (alloacceptor tRNAs) but which could both read the same codon, so that the cell produced mixtures of proteins with different translations at each ambiguous site. When it was later discovered that the single tRNASer that reads CUG codons in C. albicans is mischarged with leucine instead of serine approximately 3% of the time, the ambiguous intermediate model was extended to include ambiguous charging of a single tRNA species6,14,15. In the unassigned codon models, one of the 64 codons becomes untranslatable because its tRNA has been lost from the genome, or at least lost its function3. The codon can later be captured by another amino acid if a tRNA for that amino acid mutates so that it can read the unassigned codon.
Recently, a ‘tRNA loss-driven’ model of genetic code change was proposed6,11,16. In this model, a codon becomes ‘free’ because the tRNA that previously read it has been lost. Translation of the free codon is therefore disturbed or abolished6, but the loss of the original tRNA may be partially compensated by wobble decoding of the free codon by other tRNAs, including ones for other amino acids16. Depending on the extent to which the free codon is translatable by alloacceptor tRNAs, the tRNA loss-driven model can be regarded as a variant of the unassigned codon model (if there is no translation) or of the ambiguous intermediate model (if alloacceptor tRNAs could read the codon by wobble, even before the original tRNA was lost). Previous studies, both by experimentation and by comparative genomics, have shown that mutations in the anticodon of tRNA genes occur frequently17,18. These anticodon shifts can be synonymous, altering the balance between isoacceptor tRNAs for the same amino acid, or nonsynonymous, redeploying the tRNA to a codon for a different amino acid and so causing mistranslation18. Many tRNAs are encoded by multigene families, so a mutation in the anticodon of one tRNA gene in a family will often not abolish the organism’s ability to translate the original codon.
The discovery of the CUG-Ala genetic code in P. tannophilus11,12, and its phylogenetic closeness to the well-known CUG-Ser code in Candida, motivated us to investigate the phylogenetic relationship among yeasts with standard and non-standard genetic codes8,19. Using whole-genome data to establish phylogeny, and mass spectrometry to determine genetic codes, we show that the CUG codon was reassigned on three separate occasions during the evolution of budding yeasts. We identify a new clade, CUG-Ser2, that transitioned from CUG-Leu to CUG-Ser translation, independently of the similar transition that occurred in the Candida clade. We discuss the mechanism of genetic code change, and the cause of the evolutionary instability of CUG-Leu translation in budding yeasts.
Results
Phylogeny and genetic code determination
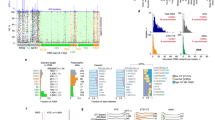
To identify species with modified genetic codes, we systematically examined the genomes of 52 yeast species (including 7 newly sequenced) and two outgroups. The species phylogeny was inferred by maximum likelihood from whole-genome amino acid data under a site-homogeneous model (Fig. 2). An almost identical tree containing the same set of five clades was obtained using a site-heterogeneous model (Supplementary Fig. 1).
Phylogenomic tree and CUG decoding in 52 yeast species. Blue, pink and yellow indicate CUG translation as Leu, Ser, and Ala, respectively. Point X indicates the last common ancestor of the clades with altered genetic codes. Circles indicate the presence of tRNA genes with the indicated anticodons. The VLE content column shows species where a characterized Virus-Like Element with killer activity is present (Killer), a VLE-like plasmid is present but killer activity has not been demonstrated (Plasmid), or VLE-like pseudogenes are present in the nuclear genome (ψ). Asterisks beside species names indicate genomes sequenced in this study. The tree was constructed from 1237 proteins by maximum likelihood
We initially used a BLAST-based method to make a bioinformatic prediction of the genetic code in each species, and then empirically determined the codes of 18 species at key phylogenetic positions by liquid chromatography-tandem mass spectrometry (LC–MS/MS). The data analysis pipeline is summarized in Supplementary Fig. 2. It should be noted that although the LC–MS/MS experiments enable us to identify with confidence the major translation product of CUG codons in each species analyzed (Supplementary Table 1), they do not rule out the possibility of low-level incorporation of other amino acids. Detailed LC–MS/MS and BLAST results for each species are shown in Supplementary Data 1. Examples of LC–MS/MS spectra for species with non-standard codes are shown in Supplementary Fig. 3. The mass error values of CUG-translated residues were similar to those of other residues (Supplementary Fig. 4).
Extents of the clades with non-standard genetic codes
Examining the BLAST and mass spectrometry results in the context of the inferred species phylogeny, we conclude that there are five monophyletic groups (clades) that differ in their translations of CUG, which we refer to as the Ala, Ser1, Ser2, Leu1, and Leu2 clades (Fig. 2), as well as paraphyletic outgroup taxa (Leu0) with the standard code. The Ala clade contains the genera Nakazawaea and Peterozyma, as well as Pachysolen11,12. The split between the Ala and Leu2 clades forms a deep division within the yeast family Pichiaceae20. The Leu2 clade includes Citeromyces and Kuraishia, as well as the industrial yeasts Komagataella, Ogataea, and Pichia. The Ser1 clade contains many pathogenic Candida species and extends as far as Babjeviella12, whose code we confirmed by LC–MS/MS. The newly identified Ser2 clade contains only the genera Ascoidea and Saccharomycopsis. Ser2 clade species have few CUG codons in conserved genes and gave conflicting results in the BLAST analysis, but the mass spectrometry data showed that CUG is translated as serine in the two species analyzed, Saccharomycopsis capsularis and Ascoidea rubescens (Fig. 2; Supplementary Table 1). The Ser2 clade is sister to the Leu1 clade, which contains Saccharomyces cerevisiae and extends as deep as Cyberlindnera and Wickerhamomyces (families Saccharomycetaceae, Saccharomycodaceae, and Phaffomycetaceae20).
The branches separating the clades with different codes are short, so we evaluated the support for alternative topologies using the Shimodaira-Hasegawa and Approximately Unbiased bootstrapping tests (Supplementary Table 2). These analyses rejected the possibility that the Ser1 and Ser2 clades shared a common ancestor after they diverged from the Ala, Leu1, and Leu2 clades. They also rejected the hypothesis that the Leu1 and Leu2 clades are sisters. Therefore, the most parsimonious explanation of the data is that the CUG codon has been reassigned three times, on three separate branches of the Saccharomycotina tree: once from Leu to Ala, and twice from Leu to Ser (Fig. 2).
Separate sources of the two tS CAG genes
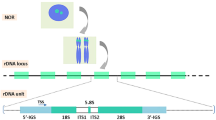
Analysis of the tRNA gene sets of each genome shows that the Ser1, Ser2, and Ala clade species each use a different novel tRNA with anticodon CAG to translate CUG codons11,21 (Fig. 3; Supplementary Figs. 5–7). The genes for these novel tRNAs were formed by mutating the anticodons of pre-existing tRNASer or tRNAAla genes. Because these pre-existing ‘source’ genes were members of multigene families, the ability of the organism to translate the original codon that they recognized was not lost. The tSCAG gene (i.e., the gene for tRNASer with anticodon CAG) of the Ser2 clade was derived from a different source gene than the tSCAG gene of the Ser1 clade, which supports the phylogenomic evidence (Fig. 2) that the Leu → Ser reassignments in the Ser1 and Ser2 clades were separate events. Specifically, the novel tSCAG genes in the two clades are derived from source genes that read the two different serine codon boxes (Fig. 1). In the Ser2 clade, the novel tSCAG was formed by mutating one of the tSGCU genes for the tRNA that reads the two AGY serine codons, whereas in the Ser1 clade the novel tSCAG was formed by mutating one of the tSAGA or tSUGA genes for a tRNA that reads some of the four UCN serine codons. The AGY- and UCN-decoding tRNASer molecules are distinct and form separate clades in phylogenetic analysis (Supplementary Figs. 6–8; Supplementary Note 1). Notably, the tRNASer(CAG) molecules of Ser2 clade species contain an A at position 37 which is immediately 3′ of the anticodon (Fig. 3). In C. albicans in the Ser1 clade, a G at this position is responsible for the observed 3% mischarging of its tRNASer(CAG) with leucine14, and G37 is conserved among all the Ser1 clade species, we analyzed except B. inositovora (Fig. 3; Supplementary Fig. 6).
Retention of tL CAG as well as tS CAG in the Ser2 clade
Surprisingly, the genome sequences of four of the five Ser2 clade species indicate that they have a tLCAG gene as well as a tSCAG gene (Fig. 3; Supplementary Fig. 9; Supplementary Note 2). In other words, they have two different tRNAs capable of reading CUG: a tRNALeu and a tRNASer. To our knowledge, there is no precedent for a genome that naturally produces competing tRNAs that read the same codon but insert different amino acids. This suggests that Ser2 clade species might produce ‘statistical proteins’ with a mix of serine and leucine incorporation at CUG sites22,23, but in our mass spectrometry analysis (of Saccharomycopsis capsularis) we found only robust evidence for translation of CUG as serine (Supplementary Table 1). We also found that tSCAG but not tLCAG is transcribed in two Saccharomycopsis species when grown in the same conditions (YPD media) as were used for mass spectrometry (Supplementary Fig. 10). However, the tLCAG gene is conserved among the three Saccharomycopsis species sequenced, whereas its flanking DNA has diverged, so it is unlikely to be a pseudogene (Supplementary Fig. 11). It is also syntenic with the functional tLCAG gene in the Leu1 clade (Supplementary Fig. 9).
Selection against tRNALeu(CAG) in the Leu1 and Leu2 clades
What made the CUG codon so unstable in yeasts? The meaning of sense codons in nuclear genes has been stable throughout all of eukaryotic evolution2,3,4,5,6, except in the three yeast clades where CUG became reassigned. The observations that three reassignments occurred independently in three closely related eukaryotic lineages, and that they all involved the same codon, strongly suggest that the reassignments shared a common evolutionary cause. As described below, we hypothesize that the shared cause was natural selection acting against the ancestral tRNALeu(CAG), and that the selective pressure was caused by a killer toxin that attacked this specific tRNA molecule (see Discussion).
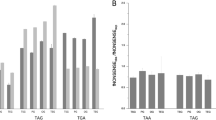
The primary evidence for selection against tRNALeu(CAG) is that it has been lost at least seven times during budding yeast evolution (Fig. 4), in stark contrast to the general stability of tRNA anticodon repertoires in yeasts16. Three losses of tRNALeu(CAG) occurred in the clades that changed their genetic codes, and were facilitated by the prior emergence of tRNASer(CAG) or tRNAAla(CAG) tRNAs to decode CUG. Four additional losses of ancestral tLCAG genes occurred in the Leu1 and Leu2 clades, without changing their genetic codes (Fig. 4, ‘–Z’ and ‘–P’ symbols). The sets of tRNAs that species in these clades now use to translate CUN as leucine show three unusual features that are consistent with an hypothesis of selective pressure against the ancestral tRNALeu(CAG). These features are summarized here and described in more detail in Supplementary Note 3. First, the tLCAG gene was modified in some Leu1 clade species by unprecedented expansion of its intron, making it the largest canonical tRNA intron known (134–318 nt; Supplementary Fig. 12). The intron contains extensive secondary structure, which is likely to slow the rate of formation of the mature spliced and base-modified tRNALeu(CAG). Second, the gene was replaced in other Leu1 and Leu2 species (represented by Pichia and Lachancea thermotolerans in Fig. 4) that eliminated the ancestral gene and later regained paralogous tLCAG genes, probably by horizontal gene transfer. Third, the gene was eliminated, without replacement, in Saccharomyces by losing the standard eukaryotic modification of the wobble base U34 in tRNALeu(UAG), allowing this tRNA to read CUG as well as CUA codons. Leu1 clade species also show other unusual deviations from the normal wobble rules used by eukaryotes, in the way they read the CUN codon box (Supplementary Note 3).
Summary of evolutionary losses and gains of tRNAs with CAG anticodons. Colored branches on the tree indicate presence of tRNALeu (blue), tRNAAla (orange), and tRNASer (pink) molecules, with shades of blue indicating different tRNALeu orthogroups (designated P, Z, L, Q, R, and T; Supplementary Fig. 9; Supplementary Note 3). Plus and minus symbols indicate inferred gains and losses of tRNA types. For each branch, only some representative genera or species are named. Asterisks indicate taxa with large introns in tLCAG-Z. Species with no tRNACAG gene (thin lines) translate CUG as Leu by wobble using tRNALeu(UAG)
Discussion
The coexistence of ancestral tLCAG and novel tSCAG genes in Ser2 clade species provides direct support for the ambiguous intermediate model of genetic code evolution3,13. Because the tLCAG gene in the Ser2 clade is orthologous to the functional tLCAG gene in the Leu1 clade (Supplementary Fig. 9), the genomic data indicate that the Ser2 clade underwent a transition from CUG-Leu to CUG-Ser translation via an intermediate phase in which both types of tRNA gene were present and functional in the same species. It is evident that tLCAG was not lost before tSCAG was gained, because tLCAG is still present. The Ser2 clade species appear to be in the final stages of the evolutionary transition between genetic codes. Only Ascoidea rubescens has completed the transition and lost the tLCAG gene. In the other four species, the novel tRNASer(CAG) is now the major tRNA species decoding CUG, but the gene for tRNALeu(CAG) has been conserved even though it was not transcribed or used for translation in our experiments. The evolutionary conservation of tLCAG in four Ser2 clade species is puzzling, but a possible explanation is that tRNALeu(CAG) may be required for translation of some genes specifically expressed in conditions that we did not examine, for example meiosis. It suggests that some CUG sites in these four species still code for essential leucine residues.
We propose that the genetic code changes were driven by natural selection against the existence of a particular tRNA species (the ancestral form of tRNALeu(CAG)), and not by selection in favor of the proteomic consequences of the code changes. Selection in favor of the proteomic changes seems implausible in view of the phylogenomic evidence that three independent reassignments occurred (Fig. 2), because it is difficult to envisage how the replacement of thousands of leucine residues with both alanine (in one lineage) and serine (in two other lineages) could have had beneficial effects on protein sequences and been advantageous in both situations. In contrast, AlaRS and SerRS are the only two aminoacyl tRNA synthetases that do not require particular bases to be present in the anticodon of the tRNAs they charge24, so these are the only two amino acids to which CUG could have readily been reassigned from leucine simply by mutating the anticodon of an existing tRNAAla or tRNASer16, although multiple mutations are required in the anticodon. Our hypothesis of selection against tRNALeu(CAG) is also consistent with the tendency for CUG codons to be located at non-essential sites in species that have changed their genetic code9. CUG codons are relatively rare in the clades that changed codes. They tend to occur in orphan genes, or in regions of genes that do not align well with other species, as opposed to sites that are well conserved (Supplementary Figs. 13, 14). This pattern would not be expected if the genetic code changes were favored because of their effect on protein sequences.
We hypothesize that the agent of selection against tRNALeu(CAG) may have been a Virus-Like Element (VLE). VLEs are cytoplasmic linear DNA plasmids (also called killer plasmids) that code for a toxin and an antitoxin25,26. VLEs or VLE-like plasmids are present in 1–2% of budding yeast strains27. Cells carrying a VLE secrete a toxin that kills cells, from the same or other species, that lack the VLE. The toxins are ribonucleases that cleave the anticodon loops of specific tRNAs. The two known targets of this class of killer element are tRNAGlu(UUC), which is cleaved by a toxin found in some strains of Kluyveromyces lactis (Leu1 clade), and tRNAGln(UUG), which is cleaved by toxins from strains of Millerozyma acaciae and Debaryomyces robertsiae (both Ser1 clade)25. Similar, but uncharacterized, VLE-like plasmids have been described in Leu2 and Ser2 clade species28,29. BLAST searches show that the nuclear genomes of many species in the Ser1, Ser2, Leu1, and Leu2 clades contain pseudogenes of VLE-like plasmids30,31 (Fig. 2; Supplementary Table 3), which indicates that these species have been infected by VLEs in the past. In contrast, VLE-like sequences are absent from the genomes of the Leu0 species, an outcome whose probability is 0.01 under the assumption of uniform distribution of the 18 found VLE-like sequences across the 54 analyzed species.
In our hypothesis, a VLE with a toxin specific for tRNALeu(CAG) infected the common ancestor of five clades (point X in Fig. 2). The infection reduced the availability of tRNALeu(CAG) in the pool of leucine tRNAs, causing selection in favor of alternative ways to read CUG codons without using the susceptible tRNA. Some yeast lineages responded by changing their genetic codes, whereas others altered the sets of tRNALeu genes they contain and managed to retain the standard code, either by changing their wobble rules or by acquiring versions of tRNALeu(CAG) that were resistant to the toxin. Incompatibilities between different genetic codes may have contributed to reproductive isolation among the clades that emerged shortly after point X. The outcome of the infection resembles the predictions of the ‘tRNA loss-driven’ model6,11,16, but the initial event was destruction of tRNALeu(CAG) molecules by the postulated toxin rather than loss of the tLCAG gene. We cannot tell if the hypothesized VLE still exists or was transient. Stochastic losses of infection could explain how ancestral tLCAG genes survive in a few species. If our hypothesis is correct, reorganization of the genetic code can be regarded as a radical mechanism of host defense against an infectious agent32,33.
Methods
Genome sequences
We sequenced and assembled the genomes of seven species using the iWGS pipeline34, selecting the assembly with the highest N50: the Leu2 clade species Saturnispora dispora (strain NRRL Y-1447, SPAdes assembly), Ambrosiozyma philentoma (NRRL Y-7523, SPAdes), Candida boidinii (NRRL Y-2332, DISCOVAR), and Citeromyces matritensis (NRRL Y-2407, MASURCA); the Ala clade species Nakazawaea wickerhamii (NRRL Y-2563, DISCOVAR) and Peterozyma xylosa (NRRL Y-12939, DISCOVAR); and the Ser2 clade species Saccharomycopsis capsularis (NRRL Y-17639, DISCOVAR).
Phylogenetic tree construction
The phylogenetic tree in Fig. 2 was constructed20 from genomic data for 52 yeast taxa and 2 fungal outgroups, using a set of 1237 genes from BUSCO35. Each locus sampled had minimum sequence occupancy ≥27 taxa and sequence length ≥167 amino acid residues. To avoid any influence of mistranslated CUG codons on the phylogenetic tree, amino acids encoded by CUG codons were substituted by “X”. We used RAxML36 version 8.2.0 to perform maximum likelihood analyses of the concatenation data matrix (607,754 sites) under an unpartitioned scheme (a LG+GAMMA model) and a gene-based partition scheme (1237 partitions; each has its own model). The two ML trees produced by RAxML were topologically identical and were also found by the program IQ-TREE37 version 1.5.1. Branch support for each internode was evaluated with 100 rapid bootstrapping replicates using RAxML38. Because running an analysis using the site-heterogeneous CAT model in PhyloBayes is computationally intractable for our concatenated dataset, the C60 model (a maximum likelihood variant of Bayesian CAT model) implemented in IQ-TREE was used to infer ML phylogeny with passing ‘-m C60+LG+G4 -bb’ to specify the site-heterogeneous model and to conduct 1000 ultrafast bootstrap replicates (Supplementary Fig. 1). Running time was ~25 days with 32 CPUs. Phylogenetic trees for tRNA genes were constructed using PhyML after intron removal and MUSCLE alignment39. To calculate the probability of all VLEs appearing outside the Leu0 clade under the hypothesis that they are uniformly distributed across the tree, we calculated the number (44 choose 18)/(54 choose 18) which is approximately equal to 1.01%.
Bioinformatics methods
For bioinformatic inference of genetic codes, to maintain consistency of annotation all genomes were annotated using a simple method that identified all open reading frames (ORFs, the region between one stop codon and the next) as potential genes, provided that they were ≥180 bp long and did not overlap by >50 bp with a longer ORF. ORFs were translated using the standard genetic code and searched by BLASTP against the Yeast Gene Order Browser database of proteins40 with a cutoff of E ≤ 1e−10. The set of BLAST high-scoring pairs (HSPs) from a genome was then processed to populate a matrix of 64 codons × 20 amino acids (Supplementary Data 2) for the query species, as follows. For every codon site inside a predicted ORF, if there were ≥5 YGOB database proteins aligned against the site, and > 80% of these had the same amino acid at the site, then for every HSP at this site we assigned to the amino acid aligned against that site a score of 1/n, where n is the total number of proteins aligned against the site (Supplementary Fig. 2). Counts of CUG codon occurrence in ORFs, in genes with HSPs, and in HSP regions (Supplementary Figs. 13, 14) were calculated from the results of BLAST searches against the BUSCO Ascomycota ‘ancestral’ database35.
tRNA genes were predicted using tRNAscan-SE41, with introns removed by our own Python code. In some Kluyveromyces species, the tLCAG gene was not predicted by tRNAscan-SE due to its unusually long intron but was found by BLASTN. In our phylogenetic trees, tRNAs are identified with names such as S_CAG1_Ser1_Babino_r2_i_25. The fields (separated by underscores) in these names are: inferred amino acid; anticodon; clade; 3-letter genus and species abbreviations; “r” indicates the repeat count of genes coding for identical tRNAs (ignoring introns) in this species; “i” or “n” indicates presence or absence of an intron in the gene; intron length.
To identify pseudogenes of genes from VLEs or VLE-like plasmids (cytosolic linear DNA plasmids without demonstrated killer activity) located in yeast nuclear genomes, we first constructed a database of known yeast VLEs25,27 (Supplementary Table 3). Proteins encoded by these elements were used as queries in TBLASTN searches against a database of all 54 fungal genomes, and potential pseudogenes were then tested for reciprocal BLASTX hits to VLE proteins.
LC–MS/MS
Total protein was extracted from cultures grown in YPD, and analyzed by LC–MS/MS. Triplicate samples were run on a Thermo Scientific Q Exactive mass spectrometer connected to a Dionex Ultimate 3000 (RSLCnano) chromatography system. Tryptic peptides were resuspended in 0.1% formic acid. Each sample was loaded onto a fused silica emitter (75 μm i.d., pulled using a Sutter Instruments P2000 laser puller), packed with 1.8 μm 120 Å UChrom C18 packing material (NanoLCMS Solutions) and was separated by an increasing acetonitrile gradient over 60 min at a flow rate of 250 nL/min. The mass spectrometer was operated in positive ion mode with a capillary temperature of 320 °C, and with a potential of 2300 V applied to the frit. All data were acquired with the mass spectrometer operating in automatic data-dependent switching mode. A high resolution MS scan (300–1600 m/z; Supplementary Table 4) was performed using the Q Exactive to select the eight most intense ions prior to MS/MS analysis using HCD.
In a first approach to empirical genetic code determination (Supplementary Fig. 2), de novo peptide sequences were extracted from the LC–MS/MS data using PEAKS42 Studio 7 software. Settings were Parent Mass Error Tolerance 10.0 ppm, Fragment Mass Error Tolerance 0.03 Da, fixed modifications: carbamidomethylation, variable modifications: oxidation. Peptides that mapped to a unique site in the genome with ≤1 mismatch to the standard-code translation were identified12. If a genomic site mapped to multiple peptides, all peptides were required to agree. This method deduced the complete genetic code table of each species (Supplementary Data 3), except for ambiguity of Leu and Ile, which cannot be differentiated by mass, and showed that no species had reassigned any codon other than CUG.
In a second approach (Supplementary Fig. 2), which used peptide mass fingerprinting rather than complete de novo peptide sequences, we generated 19 hypothetical proteome databases from each genome, corresponding to every possible sense translation of CUG11. We then used MaxQuant43,44 version 1.5.5.1 to identify peptides that had a unique match to only one of these databases, filtered the matches to include only CUG-encoded residues that were individually supported by b- and/or y-ion data, and tabulated the translations of CUG seen at each genomic site (Supplementary Table 1). The accepted mass ranges for individual amino acids in b/y-ion fragment determination are listed in Supplementary Table 5. MaxQuant parameters were set to a false discovery rate of 1% (other parameters are given in Supplementary Data 4).
The nature of the LC–MS/MS experiment does not allow us to directly quantify the levels of (mis)incorporation of different amino acids at any particular CUG site of interest. Because LC–MS/MS involves sampling a limited number of tryptic peptides for fragmentation (an average of 32,051 unique peptides per species in our data; Supplementary Table 4), and these peptides were chosen randomly by the mass spectrometer (data-dependent acquisition mode), in general each genomic CUG site whose translation was detected was spanned by only one peptide. Therefore, we tabulated for each species, for all the CUG sites in its genome that were spanned by a peptide, the proportion of those sites that matched Ser, Ala, Leu, etc., in the peptide. This proportion exceeds 90% for the major translation product of CUG in all species except C. boidinii (86%) and A. rubescens (80%) (Supplementary Table 1).
RT-PCR of Ser2 clade tL CAG and tS CAG expression
Genomic DNA of S. capsularis and S. malanga was extracted by homogenization of stationary phase cultures with glass beads followed by phenol-chloroform extraction and ethanol precipitation. RNA was extracted from log-phase cultures by hot acid phenol-chloroform extraction. Following DNase I (Invitrogen) treatment, cDNA was synthesized using random primers (High-Capacity cDNA Reverse Transcription Kit with RNase Inhibitor; Applied Biosystems). Primer sequences are listed in Supplementary Table 6. RT-PCR amplification was performed using GoTaq (Promega) polymerase or Q5 high fidelity polymerase (NEB) for 30 cycles with an annealing temperature of 55 °C.
Data availability
Genome sequences and raw reads have been deposited in GenBank as BioProject PRJNA386659, under DDBJ/ENA/GenBank accessions NHAL00000000–NHAR00000000. The versions described in this paper are versions NHAL01000000–NHAR01000000. The genome sequences of the other 47 species analyzed are from public sources (Supplementary Table 7). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository45 with the dataset identifier PXD008827.
References
Crick, F. H. The origin of the genetic code. J. Mol. Biol. 38, 367–379 (1968).
Koonin, E. V. & Novozhilov, A. S. Origin and evolution of the universal genetic code. Annu. Rev. Genet. 51, 45–62 (2017).
Sengupta, S. & Higgs, P. G. Pathways of genetic code evolution in ancient and modern organisms. J. Mol. Evol. 80, 229–243 (2015).
Keeling, P. J. Genomics: evolution of the genetic code. Curr. Biol. 26, R851–R853 (2016).
Ling, J., O’Donoghue, P. & Soll, D. Genetic code flexibility in microorganisms: novel mechanisms and impact on physiology. Nat. Rev. Microbiol. 13, 707–721 (2015).
Kollmar, M. & Mühlhausen, S. Nuclear codon reassignments in the genomics era and mechanisms behind their evolution. Bioessays 39, 1600221 (2017).
Kawaguchi, Y., Honda, H., Taniguchi-Morimura, J. & Iwasaki, S. The codon CUG is read as serine in an asporogenic yeast Candida cylindracea. Nature 341, 164–166 (1989).
Ohama, T. et al. Non-universal decoding of the leucine codon CUG in several Candida species. Nucleic Acids Res. 21, 4039–4045 (1993).
Butler, G. et al. Evolution of pathogenicity and sexual reproduction in eight Candida genomes. Nature 459, 657–662 (2009).
Moura, G. R., Paredes, J. A. & Santos, M. A. Development of the genetic code: insights from a fungal codon reassignment. FEBS Lett. 584, 334–341 (2010).
Mühlhausen, S., Findeisen, P., Plessmann, U., Urlaub, H. & Kollmar, M. A novel nuclear genetic code alteration in yeasts and the evolution of codon reassignment in eukaryotes. Genome Res. 26, 945–955 (2016).
Riley, R. et al. Comparative genomics of biotechnologically important yeasts. Proc. Natl Acad. Sci. USA 113, 9882–9887 (2016).
Schultz, D. W. & Yarus, M. On malleability in the genetic code. J. Mol. Evol. 42, 597–601 (1996).
Suzuki, T., Ueda, T. & Watanabe, K. The ‘polysemous’ codon—a codon with multiple amino acid assignment caused by dual specificity of tRNA identity. EMBO J. 16, 1122–1134 (1997).
Gomes, A. C. et al. A genetic code alteration generates a proteome of high diversity in the human pathogen Candida albicans. Genome Biol. 8, R206 (2007).
Kollmar, M. & Mühlhausen, S. How tRNAs dictate nuclear codon reassignments: only a few can capture non-cognate codons. RNA Biol. 14, 293–299 (2017).
Yona, A. H. et al. tRNA genes rapidly change in evolution to meet novel translational demands. Elife 2, e01339 (2013).
Rogers, H. H. & Griffiths-Jones, S. tRNA anticodon shifts in eukaryotic genomes. RNA 20, 269–281 (2014).
Sugita, T. & Nakase, T. Non-universal usage of the leucine CUG codon and the molecular phylogeny of the genus Candida. Syst. Appl. Microbiol 22, 79–86 (1999).
Shen, X. X. et al. Reconstructing the backbone of the Saccharomycotina yeast phylogeny using genome-scale data. G3 6, 3927–3939 (2016).
Yokogawa, T. et al. Serine tRNA complementary to the nonuniversal serine codon CUG in Candida cylindracea: evolutionary implications. Proc. Natl Acad. Sci. USA 89, 7408–7411 (1992).
Bezerra, A. R. et al. Reversion of a fungal genetic code alteration links proteome instability with genomic and phenotypic diversification. Proc. Natl Acad. Sci. USA 110, 11079–11084 (2013).
Weil, T. et al. Adaptive mistranslation accelerates the evolution of fluconazole resistance and induces major genomic and gene expression alterations in Candida albicans. mSphere 2, e00167–17 (2017).
Giegé, R., Sissler, M. & Florentz, C. Universal rules and idiosyncratic features in tRNA identity. Nucleic Acids Res 26, 5017–5035 (1998).
Satwika, D., Klassen, R. & Meinhardt, F. Anticodon nuclease encoding virus-like elements in yeast. Appl. Microbiol. Biotechnol. 96, 345–356 (2012).
Wickner, R. B. & Edskes, H. K. Yeast killer elements hold their hosts hostage. PLoS Genet. 11, e1005139 (2015).
Fukuhara, H. Linear DNA plasmids of yeasts. FEMS Microbiol. Lett. 131, 1–9 (1995).
Sturmberger, L. et al. Refined Pichia pastoris reference genome sequence. J. Biotechnol. 235, 121–131 (2016).
Shepherd, H. S., Ligon, J. M., Bolen, P. L. & Kurtzman, C. P. Cryptic DNA plasmids of the heterothallic yeast Saccharomycopsis crataegensis. Curr. Genet. 12, 297–304 (1987).
Frank, A. C. & Wolfe, K. H. Evolutionary capture of viral and plasmid DNA by yeast nuclear chromosomes. Eukaryot. Cell 8, 1521–1531 (2009).
Satwika, D., Klassen, R. & Meinhardt, F. Repeated capture of a cytoplasmic linear plasmid by the host nucleus in Debaryomyces hansenii. Yeast 29, 145–154 (2012).
Shackelton, L. A. & Holmes, E. C. The role of alternative genetic codes in viral evolution and emergence. J. Theor. Biol. 254, 128–134 (2008).
Taylor, D. J., Ballinger, M. J., Bowman, S. M. & Bruenn, J. A. Virus-host co-evolution under a modified nuclear genetic code. PeerJ 1, e50 (2013).
Zhou, X. et al. In silico whole genome sequencer & analyzer (iWGS): a computational pipeline to guide the design and analysis of de novo genome sequencing studies. G3 6, 3655–3662 (2016).
Simao, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Nguyen, L. T., Schmidt, H. A., von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Stamatakis, A., Hoover, P. & Rougemont, J. A rapid bootstrap algorithm for the RAxML Web servers. Syst. Biol. 57, 758–771 (2008).
Gouy, M., Guindon, S. & Gascuel, O. SeaView version 4: a multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol. Biol. Evol. 27, 221–224 (2010).
Byrne, K. P. & Wolfe, K. H. The yeast gene order browser: combining curated homology and syntenic context reveals gene fate in polyploid species. Genome Res. 15, 1456–1461 (2005).
Lowe, T. M. & Eddy, S. R. tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25, 955–964 (1997).
Zhang, J. et al. PEAKS DB: de novo sequencing assisted database search for sensitive and accurate peptide identification. Mol. Cell Proteomics 11, M111.010587 (2012).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Tyanova, S. et al. Visualization of LC–MS/MS proteomics data in MaxQuant. Proteomics 15, 1453–1456 (2015).
Vizcaino, J. A. et al. 2016 update of the PRIDE database and its related tools. Nucleic Acids Res 44, D447–D456 (2016).
Acknowledgements
We thank K. Wynne, E. Dillon, A. Salamov, J. DeVirgilio, and A.B. Hulfachor for assistance, M. Takashima for permission to use JCM strains data, and G. Butler and P. Baranov for discussion. We thank the UW Biotechnology Center DNA Sequencing Facility, UW Center for High Throughput Computing, Vanderbilt Advanced Computing Center for Research and Education, and Lucigen Corporation for access to infrastructure. This study was supported by Science Foundation Ireland (13/IA/1910 to K.H.W.), the Wellcome Trust (109165/Z/15/Z to T.K., 102414/Z/13/Z to N.M.), the National Science Foundation (DEB-1442148 to C.T.H. and C.P.K., DEB-1442113 to A.R.), the National Institutes of Health NIAID (AI105619 to A.R.), the USDA National Institute of Food and Agriculture (Hatch Project 1003258 to C.T.H.), and in part by the DOE Great Lakes Bioenergy Research Center (DOE Office of Science BER DE-FC02-07ER64494 to T.J. Donohue). The work conducted by the U.S. Department of Energy Joint Genome Institute, a DOE Office of Science User Facility, is supported by the DOE Office of Science under Contract No. DE-AC02-05CH11231. C.T.H. is a Pew Scholar in the Biomedical Sciences, supported by the Pew Charitable Trusts. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
A.Y.C. conducted LC–MS/MS experiments and RT-PCR; T.K. and K.H.W. conducted most bioinformatics analyses; A.Y.C., N.M., and D.C.S. analyzed LC–MS/MS data; C.P.K., C.T.H., A.R., and K.H.W. selected taxa for genome sequencing; J.K., D.A.O., and C.T.H. generated and performed preliminary analyses of new genome sequences; X.-X.S., X.Z., and A.R. performed phylogenomic analyses; T.K., A.Y.C., R.R., I.V.G., C.P.K., C.T.H., A.R., and K.H.W. designed research; T.K., A.Y.C., and K.H.W. drafted the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Krassowski, T., Coughlan, A.Y., Shen, XX. et al. Evolutionary instability of CUG-Leu in the genetic code of budding yeasts. Nat Commun 9, 1887 (2018). https://doi.org/10.1038/s41467-018-04374-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-04374-7
This article is cited by
-
Candida boidinii isolates from olive curation water: a promising platform for methanol-based biomanufacturing
AMB Express (2024)
-
Invasive candidiasis
Nature Reviews Disease Primers (2024)
-
Candida parapsilosis complex in the clinical setting
Nature Reviews Microbiology (2024)
-
Use of the Saccharomycopsis schoenii MET17 promoter for regulated heterologous gene expression
Current Genetics (2024)
-
Immune cells alter genetic decoding in cancer
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.