Abstract
The intestinal immune system can respond to invading pathogens yet maintain immune tolerance to self-antigens and microbiota. Myeloid cells are central to these processes, but the signaling pathways that underlie tolerance versus inflammation are unclear. Here we show that mice lacking Calcineurin B in CD11chighMHCII+ cells (Cnb1CD11c mice) spontaneously develop intestinal inflammation and are susceptible to induced colitis. In these mice, colitis is associated with expansion of T helper type 1 (Th1) and Th17 cell populations and a decrease in the number of FoxP3+ regulatory T (Treg) cells, and the pathology is linked to the inability of intestinal Cnb1-deficient CD11chighMHCII+ cells to express IL-2. Deleting IL-2 in CD11chighMHCII+ cells induces spontaneous colitis resembling human inflammatory bowel disease. Our findings identify that the calcineurin–NFAT–IL-2 pathway in myeloid cells is a critical regulator of intestinal homeostasis by influencing the balance of inflammatory and regulatory responses in the mouse intestine.
Similar content being viewed by others
Introduction
The intestine is a unique and challenging immune microenvironment in which homeostasis requires active tolerance towards self-antigens, dietary-antigens, and commensal microorganisms, balanced with the requirement to detect and respond rapidly to pathogen invasion. Dysregulation of the intestinal immune compartment can result in inflammatory bowel disease (IBD), which affects ~3.5 million people in the US and Europe1.
The mouse intestinal lamina propria (LP) contains distinct subsets of mononuclear phagocytes that maintain tolerance and immune reactivity towards pathogens2,3. These subsets include conventional dendritic cells (DC), monocytes, and tissue-resident macrophages. LP DCs express high levels of CD11c, MHCII, and FMS-like tyrosine kinase 3 (FLT3, also known as CD135) and FLT3 ligand (FLT3L), and are negative for the lineage markers CD3, CD19, B220, NK1.1, and CD64, whereas the monocytes and macrophages predominantly express CD64 and CD11b3. Intestinal DCs comprise three major subsets divided by CD103 and CD11b expression: CD103+CD11b−, CD103+CD11b+, and CD103−CD11b+. Similar to LP populations, these DCs are present in the mesenteric lymph nodes (MLN), suggesting that they contribute to initiation of immune responses. Despite the importance of these myeloid cells in supporting intestinal immune homeostasis, we know little about the cellular signaling networks that modulate tolerance-versus-inflammation in the intestine.
The nuclear factor of activated T-cells (NFAT) family of transcription factors is an important mediator of DC function4,5. Initially, NFAT members were thought to be involved solely in the transcriptional activation of T cells6, but different NFAT proteins are now known to have a complex influence on host tolerance to self and foreign antigens7,8,9. Upon calcium influx induced by ligation of pattern recognition receptors, activated NFAT triggers calmodulin-mediated activation of the protein phosphatase calcineurin, which in turn induces NFAT nuclear translocation4,10.
NFAT is a master transcription factor that regulates expression of IL-2, an immuno-regulatory cytokine that controls T-cell expansion and differentiation, Treg-cell maintenance, and NK-cell activation6. DCs are a source of IL-211,12 and IL-2 can regulate immune responses in the mouse lung during fungal infection5,13, and promote adaptive immune responses to alum immunization14. Whether the calcineurin–NFAT–IL-2 axis in antigen-presenting cells (APC) can contribute to immune homeostasis under basal and inflammatory conditions in other organs, such as the intestine, is unknown.
Here we use mice lacking calcineurin B (encoded by Cnb1) or IL-2 (encoded by Il2) in CD11chighMHCII+ cells, and show that calcineurin–NFAT–IL-2 signaling in intestinal CD11chighMHCII+ APCs prevents spontaneous chronic intestinal inflammation in acute and chronic models of induced colitis. Calcineurin–NFAT signaling in intestinal DCs mediates IL-2 production, which supports local Treg cell expansion in the intestinal LP and restrains inflammatory T-cell expansion and effector function. Our data shed light on the processes underpinning tolerance versus inflammation in the mouse intestine, and identify the calcineurin–NFAT–IL-2 pathway in intestinal APCs as a potential therapeutic target for IBD.
Results
Calcineurin–NFAT is active in colonic CD11chighMHCII+ cells
mRNA expression analysis of Cnb1 and the major Nfat isoforms in leukocytes Nfat1 and Nfat2 revealed that T cells from mesenteric lymph nodes (MLN) and CD11b– and CD11b+ CD11chighMHCII+ cells from colonic lamina propria (LP-colon) expressed Cnb1 and Nfat1 at comparable levels, but Nfat2 was less abundant in both populations compared to T cells (Fig. 1a). At the protein level, NFAT-1 was expressed by ~80% of CD11chighCD11b– and CD11chighCD11b+ myeloid cells of the spleen, MLN, Peyer’s patches (PP) and LP-colon and spleen CD3+CD4+ T cells (Fig. 1b). Fewer LP-colon CD11chighCD11b+ cells expressed NFAT-1 (~70%). Despite comparable frequencies of NFAT-1+ cells, the mean fluorescence intensity of NFAT-1 in both myeloid cell populations was significantly lower than in colonic CD3+CD4+ T cells (Fig. 1c). Cytoplasmic NFAT-1 in LP-colon CD11chighCD11b– and CD11chighCD11b+ cells was confirmed by confocal microscopy (Fig. 1d) and its translocation to the nucleus was observed in response to the calcium mobilizer thapsigargin (Fig. 1e). Using a mouse DC line (D1) stably expressing an NFAT-luciferase reporter, we found that whole-glucan particles (WGP; particulated dectin-1 agonist) and thapsigargin could robustly activate NFAT; lipopolysaccharide (LPS) and soluble β-(1,3)-glucan PGG activated NFAT to a lesser extent (Fig. 1f). Finally, inhibition of calcineurin signaling with cyclosporin A or tacrolimus (FK506) effectively suppressed WGP-induced NFAT-luciferase activity (Fig. 1g). These data indicate that the calcineurin–NFAT pathway is active under steady-state conditions in colonic CD11chighMHCII+ cells, and can respond to calcium flux and TLR/dectin-1 ligands.
Calcineurin B and NFAT expression in mouse intestinal myeloid cells. a Relative expression levels of Cnb1, Nfat1, and Nfat2 mRNAs in intestinal CD11chighMHCII+ cells (CD11b+ and CD11b−) and MLN CD3+ T cells, assessed by qRT-PCR. Data represent the means ± standard error of three experiments (n ≥ 7 mice/exp). b, c Percentage of NFAT-1+ cells (b) and NFAT-1 protein levels (c) in CD11chighMHCII+ cells (CD11b+ and CD11b−) obtained from spleen, MLN, PP, and colonic LP (LP-colon), assessed by flow cytometry. Splenic CD3+CD4+ T cells are included for comparison. Data represent the means ± standard error of three experiments (n = 4–5 mice/experiment). **P < 0.01 versus CD4+ T cells (ANOVA followed by Dunnett’s multiple comparisons test). d Representative images of NFAT-1 labeling in sorted colonic CD11chighMHCII+ cells (CD11b+ and CD11b−). Scale bar 5 μm. e NFAT-1 nuclear translocation in CD11chighMHCII+ cells (CD11b+ and CD11b−) after 30 min stimulation with thapsigargin (Thap; 200 nm), as assessed by confocal analysis. Data represent the means ± standard error of two experiments (n = 6–10 mice/experiment). Scale bar 5 μm. f NFAT-dependent luciferase activity measured in a DC cell line in response to TLRs (Poly I:C, LPS), dectin-1 ligands (PGG and WGP), and the calcium mobilizer thapsigargin. g Dose-dependent inhibition of NFAT nuclear translocation by cyclosporin A (CsA) and tacrolimus (FK506) in WGP-stimulated D1 cells, as assessed by NFAT-luciferase activity. AU arbitrary units, Ctrl control, Luc luciferase, LP lamina propria, LPS Lipopolysaccharide, MLN mesenteric lymph node, PGG soluble β-(1,3)-glucan, PP Peyer’s Patches, WGP whole glucan particles, Thap thapsigargin
Calcineurin B in myeloid cells maintains intestinal homeostasis
We next asked whether the calcineurin–NFAT pathway has a functional role in intestinal immune homeostasis. We generated Cnb1CD11c mice in which calcineurin B expression is lost in cells expressing CD11c at high level, by crossing Cnb1fl/fl mice with CD11c-specific Cre-mice15. Cnb1 mRNA was significantly diminished in LP-colon of CD11chighMHCII+CD11b− and CD11chighMHCII+CD11b+ myeloid cells (mostly belonging to the DC pool) of Cnb1CD11c mice, compared to CD11clowCD11b+CD64+ cells (mostly macrophages) (Fig. 2a). Cnb1 deletion prevented thapsigargin-driven NFAT-1 nuclear translocation in CD11b+ and CD11b− CD11chighMHCII+ cell populations in the MLN (Fig. 2b). However, calcineurin–NFAT-1 deficiency in CD11chighMHCII+ cells did not affect the relative abundance of these myeloid populations in LP-colon of Cnb1CD11c mice (Fig. 2c), or the expression of maturation markers of CD11chighMHCII+ myeloid cells in LP-colon and LP-small intestine (LP-SI) (Supplementary Fig. 1). Calcineurin B expression remained intact in B, NK, and mast cells, and naïve, memory CD4+ T cells and Treg cells from MLN of Cnb1CD11c mice, which also released normal cytokine levels in vitro (Supplementary Fig. 2a–c).
Cnb1 deletion in CD11chighMHCII+ cells abrogates NFAT-1 nuclear translocation. a Cnb1 mRNA levels in DCs (CD11chighMHCII+CD11b+, CD11chighMHCII+CD11b−) and macrophages (CD11clowMHCII+CD11b+CD64+) isolated from the LP-colon of Cnb1CD11c and Cnb1fl/fl mice, measured by qRT-PCR. Data represent the means ± standard error of three experiments (n = 3 mice/experiment). **P < 0.01. b Confocal microscopy analysis of NFAT-1 nuclear translocation in CD11chighMHCII+ cells (CD11b+ and CD11b−) from the MLN of Cnb1CD11c and Cnb1fl/fl mice after thapsigargin stimulation for 30 min. Data represent the means ± standard error of two experiments (n = 3 mice/experiment). **P < 0.01. Scale bar 10 μm. c Representative flow cytometric analysis of intestinal LP mononuclear myeloid CD11c+ cells evaluated by CD11c, CD103, CD11b, and CD64 expression. Four myeloid cell subsets were identified: three DC populations, CD11chighCD103+, CD11chighCD103+CD11b+, CD11chighCD103−CD11b+ and one CD11clowCD103−CD11b+ macrophage population. The macrophage population was identified based on the expression of CD64 within the CD11clowCD103−CD11b+ population (CD11clow CD64+). The frequency of the four different myeloid subsets in LP-colon (left) and LP-SI (right) of Cnb1CD11c and Cnb1fl/fl mice is shown. Data represent the means ± standard error of three experiments (n = 2–3 mice/group per experiment, aged 8–12 weeks). LP lamina propria, SI small intestine
To confirm that Cnb1 deletion in CD11chighMHCII+ cells did not impact T-cell function in vivo, we monitored the development of colitis following adoptive transfer of naïve CD4+CD45RBhighCD25− T cells from Cnb1CD11c and Cnb1fl/fl mice into immune-deficient Rag2KO mice16. A normal course of colitis development was observed in Rag2KO mice receiving naïve CD4+ T cells from Cnb1CD11c mice (Supplementary Fig. 2d, e). These data indicate that CD11chighMHCII+ cells are the predominant cell population targeted by the Cnb1 deletion and the abundance and activation status of myeloid cells and effector function of CD4+ T cells in the intestine of Cnb1CD11c mice remain unaffected.
Having confirmed the specificity of our Cnb1 deletion to CD11chighMHCII+ cells, we asked whether disruption of calcineurin–NFAT signaling in these cells affected intestinal homeostasis in vivo. Macroscopic examination of Cnb1CD11c mice (aged 10–14 weeks) revealed moderate enlargement of the MLN compared to Cnb1fl/fl control mice (Fig. 3a), and spontaneous inflammation of the SI and colon, characterized by an inflammatory infiltrate in the submucosa with marked erosion of the mucosal lining (Fig. 3b). Although Cnb1CD11c mice did not develop evident symptoms of colitis (such as weight loss, diarrhea or rectal bleeding), they exhibited significantly higher intestinal permeability, titers of fecal IgA (Fig. 3c), and myeloperoxidase activity in homogenates of the terminal ileum (Fig. 3d) than Cnb1fl/fl controls. Increased levels of mucosal TNF and IFNγ were evident in the SI and colon of Cnb1CD11c mice, compared to Cnb1fl/fl controls, while IL-17 levels were comparable (Fig. 3d).
Cnb1CD11c mice exhibit high intestinal permeability and inflammation. a MLN from Cnb1CD11c and Cnb1fl/fl mice. b Histological inflammation index of proximal (prox; n = 15–20 mice), medial (med; n = 22–27 mice), and terminal (term; n = 20–25 mice) small intestine (SI) and colon (n = 24–29 mice) in Cnb1CD11c and Cnb1fl/fl mice. *P < 0.05, ***P < 0.0001. Representative images of H&E-stained terminal ileum (SI) and colon sections from Cnb1CD11c and Cnb1fl/fl mice (×10 magnification). Scale bar 0.1 mm. c Intestinal permeability of Cnb1CD11c and Cnb1fl/fl mice was assessed by measuring the FITC-dextran concentration in the plasma after FITC-dextran administration by oral gavage (upper panel). *P < 0.05. IgA levels in the fecal content of colons from Cnb1CD11c and Cnb1fl/fl mice (lower panel). **P < 0.01. Data shown are from three independent experiments (n = 5–8 mice/group per experiment). d Myeloperoxidase, TNF, IFNγ, and IL-17 levels in terminal SI and colon tissue homogenates from Cnb1CD11c and Cnb1fl/fl mice, assessed by ELISA. Data shown are from four experiments (n = 5–6 mice/group per experiment). *P < 0.05, **P < 0.01. e Leukocyte abundance and population composition in the SI and colonic LP of Cnb1CD11c and Cnb1fl/fl mice. Data represent the mean number of CD45+ LP mononuclear cells obtained from each colon or SI, the total percentage of CD4+ T cells, the percentage of antigen-experienced CD4+ T cells (CD44highCD62Lneg), and of CD4+ T cells producing IL-17 or IFNγ. Data represent the means ± standard error of 7–9 experiments (n = 5–7 mice/group per experiment). *P < 0.05, **P < 0.01. f Percentage of FoxP3+ CD4+ T cells in the thymus, spleen, MLN, LP-colon, and LP-SI of Cnb1CD11c and Cnb1fl/fl mice. Data represent the means ± standard error of 3–6 experiments (n = 3–6 mice/group). *P < 0.05. DX4000 4 kDa dextran, FITC fluorescein isothiocyanate, LP lamina propria, MLN mesenteric lymph node, MPO myeloperoxidase
Analysis of the immune-cell composition and cytokine profile in the Cnb1CD11c LP revealed a higher frequency of immune cells (CD45+ and CD4+ T cells), and a higher percentage of antigen-experienced CD44+CD62L− CD4+ T cells in the LP-SI and LP-colon compared to Cnb1fl/fl controls (Fig. 3e). Cnb1 deficiency in CD11chighMHCII+ myeloid cells also significantly increased the frequency of IL-17-producing and IFNγ-producing CD4+ T cells infiltrating the LP-SI, and IFNγ-producing CD4+ T cells in the LP-colon (Fig. 3e). No difference in IL-4-producing CD4+ T cells or the CD8+ T-cell phenotype was observed in the LP-colon of Cnb1CD11c mice compared to Cnb1fl/fl controls (Supplementary Fig. 3a). Importantly, Cnb1CD11c mice exhibited significantly reduced frequencies of FoxP3+ Treg cells in the LP-colon, LP-SI, and MLN, but not in the spleen or thymus (Fig. 3f). Spontaneous chronic inflammation occurred exclusively in the intestine and systemic inflammation, immune phenotype and cytokine release from splenic CD4+ T cells were comparable between Cnb1CD11c and Cnb1fl/fl mice (Supplementary Fig. 3b, c).
These data support that the calcineurin–NFAT axis in CD11chighMHCII+ cells is critical for CD4+ T-cell homeostasis in the intestine under steady-state conditions.
Cnb1 CD11c mice have unrestrained induced gut inflammation
We next asked how dysregulated T-cell responses would affect the ability of Cnb1CD11c mice to restrain pathologic mucosal inflammation during acute and chronic models of IBD. Intra-rectal instillation of trinitrobenzene sulfonate (TNBS) rapidly induces intestinal inflammation in mice that recapitulates key features of Crohn’s disease17,18. Compared to TNBS-treated Cnb1fl/fl controls, TNBS-treated Cnb1CD11c mice exhibited a significant loss in body weight by 6 days post treatment, worse stool consistency (Fig. 4a), and severe ulceration and thickening of the colon wall (Fig. 4b). Using a histopathological scoring approach, we found that Cnb1CD11c mice exhibited severe leukocyte infiltration that penetrated all colon layers (Fig. 4c) and a significant increase in myeloperoxidase activity, TNF, and IL-17 in colon homogenates (Fig. 4d). Treatment of Cnb1CD11c mice with naproxen, a non-steroidal anti-inflammatory drug that can exacerbate colitis in susceptible IBD patients, also induced notable colonic mucosal inflammation compared to untreated Cnb1CD11c mice or naproxen-treated Cnb1fl/fl control mice (Supplementary Fig. 4). These results show that calcineurin B expression in CD11chighMHCII+ cells not only prevents spontaneous colitis under steady-state conditions, but also helps suppress severe intestinal pathology in response to acute and chronic inflammation.
Exacerbation of acute and chronic colitis in Cnb1CD11c mice. a Severity of TNBS-induced colitis evaluated according to weight loss (left panel) and stool consistency (right panel) in Cnb1CD11c and Cnb1fl/fl mice. b Macroscopic index (left) and overt anatomy of the colon (right) and c histological inflammation score for colons of Cnb1CD11c and Cnb1fl/fl mice after TNBS administration. Representative H&E-stained colon sections showing infiltrating leukocytes (×10 magnification, scale bar 0.1 mm). d Levels of inflammatory markers (MPO, TNF, IL-17, and IFNγ) in total colon homogenates from Cnb1CD11c and Cnb1fl/fl mice. All data represent the means ± standard error of two experiments (n = 6–7 mice/group per experiment). *P < 0.05, ***P < 0.001. TNBS trinitrobenzenesulfonic acid, MPO myeloperoxidase
Cnb1 deletion in non-DC phagocytes reduces colitis severity
To examine the effect of Cnb1 depletion in monocytes, macrophages, and granulocytes on intestinal inflammation, we generated a Cnb1LysM mouse model, in which the Cnb1 floxed allele was deleted by Cre-recombinase in cells expressing LysM19. The intestinal phenotype of LP mononuclear cells from Cnb1LysM mice under steady-state conditions was indistinguishable from Cnb1fl/fl controls with respect to: maintenance of body weight, colon histology, LP-colon immune-cell phenotypes, and levels of pro-inflammatory cytokines (Supplementary Fig. 5a–c). Moreover, upon TNBS treatment, symptoms of acute colitis were significantly less severe in Cnb1LysM mice than in Cnb1fl/fl controls (Supplementary Fig. 5d–f).
These data highlight that calcineurin B expression in CD11chighMHCII+LysM− cells, which mainly represent the conventional DCs in the intestine, participates in intestinal homeostasis by restraining pathological inflammation during colitis while calcineurin B expressed in LysM+ monocytes/macrophages and granulocytes has little effect on intestinal homeostasis during steady-state conditions, but exacerbates intestinal immunopathology during TNBS-induced colitis.
Calcineurin B partially regulates DC-derived IL-2 production
To delineate the mechanistic basis for calcineurin-mediated protection from colitis elicited by CD11chighMHCII+ DCs, we examined a role for calcineurin B in the production of DC cytokines regulating T-helper differentiation. Colonic CD11chighMHCII+CD11b− cells from Cnb1CD11c mice produced significantly less IL-2 following thapsigargin exposure compared to cells from Cnb1fl/fl mice, while the levels of IL-6, IL-12, IL-23, IL-10, and TGFβ were comparable (Fig. 5a). Similarly, we found that bone marrow (BM)-derived DCs released IL-2 in response to thapsigargin, LPS, and zymosan, as assessed by ELISA and intracellular IL-2 labeling (Fig. 5b, c and Supplementary Fig. 6a), whereas IL-2 production from Cnb1CD11c DCs, but not IL-12p40 or IL-6, was significantly reduced. Moreover, exposure to NFκB and p38 MAPK inhibitors fully abrogated IL-2 release from Cnb1CD11c DCs (Supplementary Fig. 6b). These data indicate that IL-2 synthesis in DCs depends on calcineurin–NFAT, NFκB and AP1 signaling.
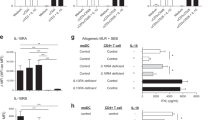
Calcineurin-NFAT signaling in CD11c+MHCIIhigh myeloid cells regulates IL-2 synthesis. a CD11chighMHCIIhighCD11b− cells sorted from MLN of Cnb1CD11c and Cnb1fl/fl mice were stimulated with thapsigargin for 16–20 h. Cytokine levels were measured in the culture supernatants using Luminex technology. Data represent the means ± standard error of three experiments (n = 10 mice/group per experiment). *P < 0.05. b, c IL-2 production from bone marrow-derived DCs of Il2KO, Cnb1CD11c, and Cnb1fl/fl mice in response to thapsigargin stimulation for 16 h, as assessed by ELISA (b) and by intracellular labeling (c). Data represent the means ± standard error of three experiments (n = 1–2 mice/group; mice aged 6–7 weeks old). **P < 0.01, ***P < 0.001. d GFP (corresponding to IL-2) expression in CD11chighMHCII+ cells, CD3+CD4+ T cells, and total myeloid CD11b+ (CD4−CD11c−) cells isolated from the spleen and MLN of IL-2-GFP reporter mice. Data represent the means ± standard error of four experiments (n ≥ 3 mice/group). *P < 0.05. e Intracellular IL-2 labeling of colonic CD3−CD11chighMHCII+ cells from Il2KO, Cnb1fl/fl and Cnb1CD11c mice. Data are presented as the relative percentage of IL-2+ cells and represent the means ± standard error of five experiments (n = 3–4 mice/group per experiment). **P < 0.01, ***P < 0.001. f Percentage of IL-2+ cells in intestinal DC (CD45+Lin−CD3−CD11c+MHCII+CD24+CD64−F4/80−) and macrophage (CD45+Lin−CD3−CD11c+MHCII+CD24−CD64+F4/80+/−) populations in LP-colon and LP-small intestine of Cnb1fl/fl mice at steady state, or during colitis induced in immunocompromised Rag1KO mice by adoptive transfer of naïve CD4+ T cells (CD45RBhighCD62L+CD44−CD25−) isolated from the spleens of C57BL/6 mice. Data represent the means ± standard error of three experiments (5–6 mice/group). g IL-2 expression in CD103− and CD103+ CD11chighMHCII+ cells from colons of Cnb1fl/fl and Cnb1CD11c mice. Representative dot plots of IL-2 labeling in colonic CD3−CD11chighMHCII+ cells are shown. Data represent the means ± standard error of three experiments (n = 2–5 mice/group per experiment). *P < 0.05. KO knockout, LP lamina propria, MLN mesenteric lymph node, SI small intestine, Thap thapsigargin, UT untreated, ND not detected
We also evaluated the ability of CD11chighMHCII+ cells to produce IL-2 in vivo using an IL-2–GFP reporter mouse20. GFP+ (IL-2+) cells among the CD11chighMHCII+ compartment were present in the spleen (~2–4%), MLN (~8–10%), and were absent in CD11b+ (CD4−CD11c−) cells belonging to the monocyte/macrophage/granulocyte lineage (Fig. 5d and Supplementary Fig. 7). Intracellular IL-2 labeling of CD11chighMHCII+ cells isolated from spleen and LP-colon showed that up to ~13% of cells produced IL-2 under steady-state conditions (Fig. 5e and Supplementary Fig. 8). During acute colitis, the proportion of IL-2-producing CD11c+MHCII+ (CD64−F4/80−) DCs in LP-colon further increased (Fig. 5f and Supplementary Fig. 9a, b). Moreover, intestinal macrophage population (CD11c+MHCII+CD64+F4/80+/−) was negative for IL-2 labeling both during steady-state and colitic conditions (Fig. 5f and Supplementary Fig. 9a, b). IL-2 production by CD11c+MHCII+ DCs, but not macrophages, was also confirmed by qRT-PCR (Supplementary Fig. 9c). These data indicate that IL-2 expression in vivo is high in the intestine and restricted to the CD11c+MHCII+ DC population (predominantly CD103+ cells) (Fig. 5g and Supplementary Fig. 9).
To examine whether deficient IL-2 expression in CD11chighMHCII+ DCs of Cnb1CD11c mice was the key factor involved in dysregulation of intestinal immune responses, we administered IL-2:anti-IL-2 complexes (IL-2C), which increase IL-2 bioactivity in vivo, and assessed whether normal intestinal homeostasis could be restored in these mice. While the proportions of Foxp3+ Treg cells were increased to a similar extent in the spleens of both Cnb1CD11c and Cnb1fl/fl mice after IL-2C treatment, these cells expanded only in the intestinal mucosa of Cnb1CD11c mice compared to Cnb1fl/fl controls, suggesting that lack of Cnb1 in CD11c+ cells is partly responsible for altered numbers of Treg cells in these mice (Supplementary Fig. 10a). To confirm that the impairment in DC-derived IL-2 signaling induces intestinal CD4+ T-cell dysregulation observed in Cnb1CD11c mice, we adoptively transferred wild-type or Il2-null DCs into the Cnb1CD11c mice. Wild-type DCs selectively restored the levels of activated CD4+ T cells producing IFNγ in the LP-colon of Cnb1CD11c mice compared to untreated or Cnb1CD11c mice receiving Il2-null DCs (Supplementary Fig. 10b). Moreover, wild-type DC transfer into Cnb1CD11c mice normalized the percentage of Treg cells in MLN, whereas the injection of Il2-null DCs induced an additional reduction of colonic FoxP3+ Treg cells. (Supplementary Fig. 10c).
DC IL-2 production prevents severe spontaneous chronic colitis
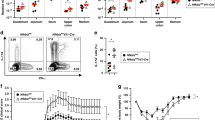
A role for T-cell-derived IL-2 in regulating T-cell responses and maintaining Treg-cell-mediated self-tolerance is well-established, but a distinct contribution of myeloid IL-2 to intestinal homeostasis in vivo is unknown. To address this issue, we used an Il2CD11c knock-out mouse model5 to monitor the effects of myeloid Il2 deficiency. Il2CD11c mice were initially indistinguishable from Il2fl/fl mice, but from ~5 weeks of age they exhibited significant weight loss, resulting in ~40–45% lower body mass compared to Il2fl/fl mice by 22 weeks (Fig. 6a). Enlargement of the MLN and macroscopic thickening and shortening of the colon without splenomegaly was observed in 7–10-week-old Il2CD11c mice compared to Il2fl/fl mice (Fig. 6b). Histopathological analysis of colons from 8-week-old Il2CD11c mice revealed moderate leukocyte infiltration of the mucosa and sub-mucosa, which affected 50–100% of the colon surface (Fig. 6c); this pathology increased in severity with age, until systemic inflammation (high serum levels of CRP and IgG1) had developed by 14–16 weeks (Fig. 6d). Consistently, Il2CD11c mice also exhibited substantially higher numbers of CD45+ leukocytes and total, antigen-experienced, and IFNγ-producing or IL-17-producing CD4+ T cells (Fig. 6e), and a markedly smaller population of FoxP3+ Treg cells in colons and the MLN (but not thymus and spleen), compared to Il2fl/fl mice (Fig. 6f). MLN CD4+ T cells isolated from Il2CD11c mice robustly expanded with age (Supplementary Fig. 11a) and produced more IFNγ and IL-17, while IL-2 release was normal when cells were re-stimulated ex vivo with antiCD3/CD28 antibodies (Supplementary Fig. 11b).
Th1/Th17-cell expansion and Treg-cell contraction in the gut of Il2CD11c mice. a Change in body weight (expressed as Δ percentage relative to the mean weight of the control group) in Il2CD11c mice compared to Il2fl/fl mice. Data represent the means ± standard error (n = 20 mice/group). *P < 0.05, **P < 0.01. b Representative images of the MLN (left) and colons (right) from Il2CD11c and Il2fl/fl mice aged 10 weeks old. c H&E staining of colon sections showing massive leukocyte infiltration in Il2CD11c compared to Il2fl/fl mice (×10 magnification, scale bar 0.1 mm). Histological inflammation index of the colon was also evaluated over time. Data represent the means ± standard error (14 mice/group aged 6–10 weeks old, and 6 mice/group aged 14–17 weeks old). ***P < 0.001. d Plasma levels of C-reactive protein (CRP) and IgG1 in the sera of Il2CD11c and Il2fl/fl mice. Data represent the means ± standard error (5–7 mice/group). **P < 0.01. e Immune phenotype of LP-colon mononuclear cells from Il2CD11c and Il2fl/fl mice. Data represent the means ± standard error of the total number of mononuclear cells obtained from the LP, the percentage of total and antigen-experienced CD44highCD62L- CD4+ T cells, and the proportion of CD4+ T cells producing IL-17, IFNγ or IL-4. f Frequency of FoxP3+ Treg cells in thymus, spleen, MLN, and LP-colon isolated from Il2CD11c and Il2fl/fl mice. Data represent the means ± standard error of 3–4 experiments (n = 2–4 mice/group per experiment, aged 6–10 weeks). **P < 0.01, ***P < 0.001. LP lamina propria, MLN mesenteric lymph node
To address the contribution of myeloid IL-2 in LP during differentiation of naïve CD4+ T cells in vivo, syngeneic naïve T helper cells from wild-type donor mice were adoptively transferred into Il2CD11c and Il2fl/fl, Rag-deficient mice (Rag2KOIl2CD11c and Rag2KOIl2fl/fl, respectively), alone or in combination with wild-type Treg cells16. Loss of myeloid IL-2 in the absence of lymphocytes did not affect the LP composition of colonic immune cells (Supplementary Fig. 12). By contrast, Rag2KOIl2CD11c mice receiving naïve CD4+ T cells showed an early and severe onset of wasting disease characterized by substantial weight loss (Fig. 7a), shortening of colon (Fig. 7b), mucosal inflammation (Fig. 7c), and an increase in activated CD4+ T cells (Fig. 7d) secreting higher levels of IL-17 and IFNγ (Fig. 7e) compared to Rag2KOIl2fl/fl control mice. Co-transfer of Treg cells prevented colitis by reducing the expansion of activated IFNγ-producing and IL-17-producing CD4+ T cells (Fig. 7a–e). After the induction of T-cell-mediated experimental colitis, IL-2 loss in CD11chighMHCII+ cells was also associated with increased signs of skin and joint inflammation, which are the most common extra-intestinal manifestations of IBD in humans (Supplementary Fig. 13)21. These results indicate that a deficiency in myeloid IL-2 is necessary and sufficient to trigger spontaneous autoimmune colitis characterized by spontaneous and chronic mucosal damage reminiscent of Crohn’s disease22,23.
DC-derived IL-2 suppresses pathogenic CD4+ T-cell expansion in the intestine. a Change in body weight and b severity of colitis based on colon length of Il2CD11c and Il2fl/fl, Rag2 knockout (KO) mice (Rag2KOIL-2fl/fl and Rag2KOIL-2CD11c mice) adoptively transferred with naïve CD4+ T cells either alone or in combination with Treg cells isolated from spleens of C57BL/6 mice. Representative pictures of colons after adoptive T-cell transfer are shown. Data represent the means ± standard error of two experiments (n = 2–5 mice/group per experiment). **P < 0.01. c Histological inflammatory score based on H&E staining of colon sections (×10 magnification, scale bar 0.5 mm) obtained from Rag2KOIl2fl/fl and Rag2KOIl2CD11c mice after 110 days of naïve CD4+ T-cell transfer. Data represent the means ± standard error. ***P < 0.001. d, e Phenotypic analysis of colonic lamina propria CD4+ T cells evaluated 110 days after adoptive T-cell transfer. The total number and the relative percentage of total and activated CD4+ T cells (d), as well as the proportions of CD4+ T cells producing IL-17, IFNγ or both (e) are shown. *P < 0.05, **P < 0.01, ***P < 0.001. All data represent the means ± standard error of two experiments (n = 3–4 mice/group per experiment)
Treg cells are essential to intestinal homeostasis as they monitor effector T helper-cell activation17. As we observed a consistent reduction in Treg cells in both Cnb1CD11c and Il2CD11c mice in the MLN and LP, but not in the thymus or spleen (Figs. 3f and 6f), we examined whether myeloid IL-2 is important for the induction of antigen-specific Treg cells in the LP. Naïve CD4+ T cells from OTII mice were adoptively transferred into Rag2KOIl2CD11c and Rag2KOIl2fl/fl mice, and the mice were then fed with ovalbumin (OVA) for 5 days. Conversion of naïve OTII CD4+ T cells into FoxP3+ Treg cells was significantly impaired, while Th1 differentiation (and to a lesser extent Th17) was significantly enhanced in the MLN of Rag2KOIl2CD11c mice compared to controls (Fig. 8a); similar results were obtained in Cnb1CD11c mice (Fig. 8b). These data indicate that myeloid IL-2 regulates Treg cell induction, which can directly suppress effector T helper cell expansion and activity.
Il2 or Cnb1 deficiency in DCs causes a dysregulated T-cell response. a, b Naïve congenic OTII cells were sorted from spleens of donor mice and transferred intravenously into Rag2KOIl2fl/fl and Rag2KOIl2CD11c (a) or Cnb1CD11c and Cnb1fl/fl mice (b). After 24 h, mice were administered OVA protein antigen by daily oral gavage for 5 days. The frequencies of CD4+ T cells expressing FoxP3, IL-17, and IFNγ were examined in the mesenteric lymph node 7 days after adoptive cell transfer. Representative FACS plots of OTII donor CD4+ T cells are shown (left panels). Data represent the means ± standard deviation of two experiments (n = 1–2 mice/group). *P < 0.05 (two-tailed, unpaired Student’s t test)
DC and T cell IL-2 have distinct functions in gut homeostasis
To dissect the contributions of myeloid cell-derived IL-2 and T helper cell-derived IL-2 to intestinal immune homeostasis, we compared the systemic and intestinal phenotypes of Il2CD11c, Il2CD4 (generated by crossing Il2fl/fl and CD4-cre mice), and Il2KO (carrying IL-2 deletion in myeloid and CD4+ T cells) mice. Il2 deletion in the CD4+ T-cell compartment resulted in characteristic IL-2 Deficiency Syndrome24,25,26 originally described in Il2KO mice and characterized by severe anemia (Fig. 9a), splenomegaly (Fig. 9b), uncontrolled expansion of splenic T cells associated with reduction of Treg cells and loss of B cells. None of these features were observed in Il2CD11c mice (Fig. 9b), indicating that IL-2 Deficiency Syndrome is mostly driven by the loss of “adaptive” IL-2. The intestinal phenotypes of Il2CD11c mice were even more divergent from Il2CD4 mice: although both Il2CD11c and Il2CD4 mice developed colitis at 10–13 weeks, the intestinal pathology was exacerbated in Il2CD11c mice compared to Il2CD4 mice, and more similar to that of Il2KO mice (Fig. 9c). A higher increase in total leukocytes, total and activated CD4+ T cells, and IL-17-producing CD4+ T cells was observed in the LP-colon of Il2CD11c mice compared to Il2CD4 mice, and was similar to that seen in Il2KO mice, whereas accumulation of CD8+ T cells was specific to Il2KO mice (Fig. 9d, e). Similar to the LP-colon of Il2KO mice, frequencies of Treg cells were reduced in the LP-colon of Il2CD11c and to a lesser extent in Il2CD4 mice compared to Il2fl/fl mice (Fig. 9f). These results indicate that innate (myeloid cells) and adaptive (CD4+ T cells) IL-2 exert both distinct and overlapping functions at the systemic level and in the intestine.
Systemic and intestinal immune homeostasis in Il2CD11c and Il2CD4 mice. a RBC count and b total number of splenocytes and proportion of CD4+ T cells, FoxP3+ Treg cells, and B220+ B cells in spleens of Il2fl/fl, Il2CD11c, and Il2CD4 mice. Representative images of spleens are shown. Data represent the means ± standard error of 2–6 experiments (n = 2–3 mice/group per experiment, 6–13 weeks old). **P < 0.01, ***P < 0.001. c Representative images of H&E-stained colon sections (×4 magnification, scale bar 0.5 mm; ×10 magnification, scale bar 0.1 mm) from Il2CD11c, Il2CD4, and Il2KO mice. d Total LP-colon leukocyte number and percentage of total and activated (CD44highCD62L−) CD4+ T cells, and B cells. e, f Percentage of IFNγ- and IL-17-producing CD4+ T cells, and FoxP3+ Treg cells obtained from the LP-colon of Il2fl/fl, Il2CD11c, Il2CD4, and Il2KO mice. Data represent the means ± standard error of 2–4 experiments (n = 2–3 mice/group per experiment, 9–13 weeks old). **P < 0.01, ***P < 0.001. LP lamina propria, RBC red blood cells
Discussion
This study shows that activation of the calcineurin–NFAT pathway in CD11chighMHCII+ cells (representing the intestinal DC pool) prevents spontaneous intestinal inflammation in vivo by orchestrating a balance between inflammatory and regulatory responses in the colonic mucosa. By characterizing mice in which Cnb1 gene was deleted either in CD11chighMHCII+ APCs (mostly representing the DC pool) but not CD64+ macrophages, or LysM-expressing cells (mainly macrophages, granulocytes, and monocytes), we identified two distinct roles for the calcineurin–NFAT pathway: calcineurin activation has an anti-inflammatory function in intestinal CD11c+MHCIIhigh DCs, but a pro-inflammatory role in macrophages, granulocytes, and monocytes. Accordingly, Cnb1CD11c mice developed spontaneous intestinal inflammation, whereas Cnb1LyzM mice were unaffected by targeted Cnb1 deletion, and were protected from induced colitis.
We verified that spontaneous chronic colitis in Cnb1CD11c mice was not due to anomalous clonal composition of the myeloid compartment, as the DC and macrophage populations were normally represented in the LP-colon and no overt differences in co-stimulatory markers expressed by these cells were found in our model. The CD4+ T-cell compartment of Cnb1CD11c mice was also normal: T helper cells (both naïve and memory) and Treg cells expressed normal calcineurin B levels, were able to produce effector cytokines (including IL-2, IFNγ, and IL-10) at normal levels, and naïve T helper cells isolated from these mice were able to induce severe colitis when adoptively transferred into lymphopenic mice.
We also identified the importance of calcineurin–NFAT-mediated production of IL-2 by the intestinal DC pool in regulating intestinal homeostasis. The colonic CD103+ DC subset is the main producer of IL-2, whereas CD64+F4/80+/− macrophages do not produce this cytokine. Colonic DCs from Cnb1CD11c mice still retain the ability to release IL-6, IL-12, IL-23. IL-2 deletion in CD11chighMHCII+ DCs caused more severe, spontaneous intestinal inflammation in Il2CD11c mice compared to Cnb1CD11c mice, which was characterized by expansion of activated CD4+ effector T cells and substantial reduction of Treg cells. IL-2 complex treatment and DCs transfer results support that reduced levels of DC-derived IL-2 could be the primary cause triggering the dysregulation of intestinal CD4+ T cells observed in Cnb1CD11c mice.
Spontaneous colitis in Cnb1CD11c mice was milder compared to Il2CD11c mice, suggesting that other signaling pathways that trigger DC-IL-2 production may exist. Using pharmacological inhibitors, we found that IL-2 release also depends on NFκB in response to LPS, zymosan, and thapsigargin stimulation, and on p38 MAPK in response to zymosan and thapsigargin stimulation. A recent study reported that deletion of TRAF6, a component of the TLR–NFκB pathway, is associated with ~50% reduction of DC-derived IL-227. Our study identifies the calcineurin–NFAT pathway as an additional axis that operates in parallel with the TRAF6–NFκB pathway in myeloid cells to produce IL-2, suggesting that both NFκB and NFAT cooperate to activate IL-2 transcription. Although these pathways contribute to the overall production of IL-2, the nature of the intestinal immune-pathologies observed in the Traf6-DC and Cnb1-DC-deficient mice diverge: loss of Traf6 in DCs causes reduced inflammatory cytokines secretion, including IL-12, whereas Cnb1-deficient DCs release normal levels of inflammatory cytokines. Moreover, partial loss of IL-2 in Traf6-deficient DCs causes spontaneous Th2-mediated enteritis characterized by eosinophilic infiltration and decreased Treg-cell numbers specifically in the small intestine27. This phenotype markedly differs from Cnb1CD11c mice, as these mice develop a spontaneous form of enteritis in both the small and large intestine resembling Crohn’s disease, and is associated with Th1 and Th17 colitogenic T-cell expansion, mucosal accumulation of mononuclear cells and progressive colon thickening28. Through a series of in vivo experiments, our data suggest that the calcineurin–NFAT–IL-2 pathway in myeloid cells is crucial for the induction of Treg cells, which contributes to maintaining immune tolerance in the gut by preventing autoimmunity. Indeed, Il2CD11c and lymphopenic Rag2KOIl2CD11c mice receiving normal naïve T cells developed an exacerbated form of spontaneous colitis associated with extra-intestinal manifestations, including cutaneous and articular inflammatory reactions, which are the foremost comorbidities of IBD21.
To date, the regulatory and effector functions of the calcineurin–NFAT–IL-2 axis in orchestrating intestinal immune homeostasis has been largely studied in terms of T helper-cell and Treg-cell biology6. The current literature supports that IL-2 is pivotal for maintaining tolerance to self and microbial antigens8,9,18,22, which is achieved via various mechanisms. First, CD4+ naïve T cells constitutively produce IL-2 in the intestinal mucosa, which induces and maintains the intestinal Treg-cell pool, as these cells are uniquely dependent upon IL-229. Second, IL-2 can directly suppress the differentiation of pathogenic Th17 cells30. Although T-cell-derived IL-2 likely has an important role in promoting intestinal tolerance, our data also highlight the requirement for DC-derived IL-2 in this process. Of note, in the absence of ex vivo restimulation, the detected IL-2 signals in intestinal DCs are relatively weak. However, it is important to bear in mind that levels of IL-2 and intensity of the signal cannot be compared to the IL-2 levels in T cells. Like DCs, unstimulated T cells do have weak IL-2 transcription/production levels, which is dramatically boosted upon stimulation. In this scenario, low level IL-2 production by DCs seems to have an important biological function and can be directly used by naïve CD4+ T cells to differentiate into Treg cells thereby suppressing the differentiation of pathogenic Th17 cells. It is also possible that some regulatory feedback mechanisms might be in place in DCs to limit and fine tune the level of IL-2 expression, as very high levels could disrupt optimal function of these cells, as well as affecting their interaction with other cells such as CD4+ T cells. Using various in vivo models, we have shown that DC-derived IL-2 in the gastrointestinal tract plays a crucial biological role in maintaining the immune homeostasis. Thus, we are confident that the immune phenotype observed in our DC-IL-2-deficient mouse model was mostly due to reduction of IL-2 production by DCs.
We further analyzed the distinct contributions of innate and adaptive IL-2 to intestinal immune homeostasis. Here, we compared the systemic and intestinal phenotype of Il2CD11c mice (lacking Il2 in the myeloid compartment), Il2CD4 mice (lacking Il2 in the CD4+ T cells), and Il2KO mice (in which both myeloid and lymphoid Il2 is absent). Il2CD4 mice exhibited IL-2 Deficiency Syndrome24,25,26 characterized by severe anemia, splenomegaly, uncontrolled expansion of T cells associated with a reduction of Treg cells, and loss of B cells. The same phenotypes noted in the Il2KO model. Mice deficient in myeloid IL-2 (Il2CD11c mice) did not develop the same pathology, but instead showed a transmural infiltration of mononuclear cells, without affecting CD8+ T cells compartment involved in erosive mucosal lesion observed in Il2KO mice, indicating that lymphoid IL-2 is crucial for immune homeostasis in the peripheral immune tissues. The difference between Il2CD4 and Il2CD11c is even more striking in the intestine, where loss of myeloid IL-2 resulted in a higher exacerbation of spontaneous colitis and a higher increase in the total number of leukocytes and activated CD4+ T effector cells producing IFNγ and IL-17 compared to mice lacking T-cell-produced IL-2. A significant reduction in the frequencies of Treg cells in the LP-colon of both Il2CD11c and Il2CD4 mice was observed, indicating that both myeloid and adaptive IL-2 are equally important for the induction and/or maintenance of Treg cells in the mouse intestine20,31. Of note, the aberrant Treg:T effector-cell ratio that accompanied pathologic inflammation in Cnb1CD11c and Il2CD11c mice seems to be restricted to the intestine of these mice, as we did not detect any changes in Treg-cell frequency in the lung or axillary and popliteal lymph nodes, suggesting a fundamental role for the gut microbiota in the activation of this pathway and thus IL-2 production by intestinal myeloid cells.
The results presented here pose interesting questions for future research. It would now be interesting to investigate how DC-derived IL-2 mechanistically regulates the induction of intestinal Treg cells, as well as how it modulates effector T-cell functions in the mouse intestine. Moreover, the requirement of DC-derived IL-2 for the development and maintenance of innate cells, such as innate lymphoid cells, should be examined. Considering the differential effects of calcineurin signaling in various myeloid cell subsets, it will be important to investigate the effects of calcineurin inhibitors on different cell subsets in vivo. Calcineurin inhibitors are already in clinical use and elicit some benefits in patients with selected forms of IBD, including acute severe ulcerative colitis and steroid-refractory IBD. Data remain insufficient to recommend the use of calcineurin inhibitors (cyclosporin A or FK506) in Crohn’s disease32.
In summary, our data support that the calcineurin–NFAT–IL-2 pathway in intestinal DCs has a central role in eliciting proper T-cell responses in the mouse intestine under both steady-state and inflammatory conditions, by supporting Treg-cell induction and by suppressing excessive expansion of Th1/Th17 cells (directly or indirectly). We propose that therapeutic strategies that selectively boost DC-derived IL-2 production may have a beneficial effect in patients with IBD.
Methods
Mice
C57BL/6-Ppp3r1tm1Stl/J (Cnb1fl/fl, Stock No. #6581), B6.Cg-Tg(Itgax-cre)1-1Reiz/J (CD11c-cre, Stock No. #8068), B6.129P2-Lyz2tm1(cre)Ifo/J (LyzM-cre, Stock No. #4781), Tg(Cd4-cre)1Cwi/BfluJ (Stock No. #017336), and B6.129P2-Il2tm1Hor/J (Stock No. #2252) mice were obtained from The Jackson Laboratory, USA. C57BL/6 and Rag2KO mice were obtained from the Biological Resource Center, Agency for Science, Technology and Research (A*STAR), Singapore. Il2fl/fl mice were generated by Ozgene Pte5. All mice were maintained at the Biological Resource Center (A*STAR), under specific pathogen-free conditions. Cnb1fl/fl mice were crossed with B6.Cg-Tg(Itgax-cre)1-1Reiz/J (CD11c-cre) and B6.129P2-Lyz2tm1(cre)Ifo/J (LysM-cre) mice. Mice lacking Il2 specifically in CD11chighMHCII+ myeloid cells or CD4+ T cells were obtained by crossing Il2fl/fl mice with B6.Cg-Tg(Itgax-cre)1-1Reiz/J (CD11c-cre) or Tg(Cd4-cre)1Cwi/BfluJ (CD4-cre) mice, respectively. Il2 deletion was quantified in splenic CD11chighMHCII+ DCs and CD4+ T cells of Cnb1CD11c mice, as previously described5. Il2 deletion in Rag2KOIl2CD11c mice was evident in the majority of CD11chighMHCII+ cells, but not in the CD11clow compartment. Male and female mice, and congenic gender-matched littermate controls were used in all experiments.
All the experiments described in this study were allowed and carried out in accordance with the guidelines of the Institutional Animal Care and Use Committee of the Biological Resource Centre (A*STAR, Singapore). B6.TgIL-2-GFP mice20 were maintained in the animal facility at the Institut Pasteur (Paris, France) in accordance with European animal welfare regulations. These animal studies were approved by the Institut Pasteur Safety Committee and by the Paris Ethics Committee 1.
Mouse models of colitis
Mice were exposed to 150 µl TNBS (Sigma-Aldrich) pre-sensitization solution (1% TNBS dissolved in 4:1 acetone:olive oil solution) that was applied to a shaved area of abdominal skin. Control mice were subjected to the same protocol with pre-sensitization solution lacking TNBS. After 7 days, mice were anesthetized by intraperitoneal ketamine/xylazine injection (100 µl/10 g body weight of 100 mg/ml ketamine and 20 mg/ml xylazine in saline) and then administered 1 mg TNBS (dissolved in 50% ethanol) intra-rectally using a 3.5 French catheter equipped with a 1 ml syringe. To ensure even distribution of TNBS throughout the entire colon and cecum, mice were held in an inverted vertical position for 30 s after instillation. In some experiments to induce colitis, naproxen (40 mg/kg/day) was administrated for 4 weeks via drinking water. The mice were then monitored daily for weight loss and fecal score. The criteria and numerical scores for macroscopic damage were as follows: 0, no ulcer, no inflammation; 1, no ulcer, local hyperemia; 2, colon wall thickening/edema; 3, ulceration and inflammation at one site only; 4, two or more sites of ulceration and inflammation. Additional points were scored for the presence of indurations, intestinal adhesions, and each additional site of inflammation or ulceration >1 cm2 in surface area.
T-cell transfer model of chronic colitis
Splenocytes were collected from C57BL/6 mice aged 6–10 weeks, and naïve CD4+CD45RBhigh T cells and CD4+CD45RBlowCD25+ Treg cells were sorted by fluorescence-activated cell sorting (FACS). Naïve T cells (3×105/mouse) were injected intravenously alone or in combination with Treg cells (1×105/mouse) into Rag2KOIl2CD11c and Rag2KOIl2fl/fl recipient mice. Mouse weight was recorded to evaluate irritable bowel disease. Mice were then killed by CO2 asphyxiation at 110 or 140 days after adoptive cell transfer for macroscopic analysis, histopathological scoring and flow cytometric analyses of leukocytes.
Intestinal permeability assay
Mice were fasted for 6 h and then administered 500 mg/kg FITC-dextran (4 kDa; Sigma-Aldrich) by oral gavage. Blood samples were collected 1 and 3 h later, centrifuged at 12,000 x g at 4 °C for 3 min, and the plasma was collected for analysis. The plasma was diluted in an equal volume of PBS and then analyzed for FITC-dextran concentration using a fluorescence spectrophotometer (485 nm excitation, 535 nm emission). A standard curve was obtained by diluting FITC-dextran in untreated plasma diluted with PBS (1:3 v/v).
Inflammatory histological score
Sections of medial colon and small intestine were fixed in buffered formalin, cut (5 µm sections, ~150 µm between each section, 4–8 per fragment), and then stained with hematoxylin and eosin (H&E). Stained sections were examined and scored in a blinded fashion. The “degree of inflammation” was graded semi-quantitatively from 0 to 4: 0, no signs of inflammation; 1, very low level; 2, low level of leukocyte infiltration; 3, high level of leukocyte infiltration, high vascular density, and thickening of the colon wall; and 4, transmural infiltration, loss of goblet cells, high vascular density, and thickening of the colon wall. The “extent of inflammation” was graded from 0 to 4: 0, none; 1, mucosal; 2, submucosal; 3, mucosal and submucosal; 4, full thickness. Finally, the “involved surface area” was graded from 0 to 4: 0, none; 1, 0–25% of the mucosal surface; 2, 25–50%; 3, 50–75%; 4, 75–100%. The overall inflammatory score was obtained by summing the “degree” and “extent” scores, and multiplying by the ‘involved surface area’ (minimum score 0, maximum 32).
Measurement of tissue inflammatory markers
Frozen fragments of small intestine and colon were homogenized using Tissue Protein Extraction Reagent (Thermo Scientific) and the homogenates were used to assess protein concentrations and cytokine levels. Neutrophil infiltration into the colon was monitored by measuring MPO activity using a spectrophotometric assay with tri-methylbenzidine (TMB) as the substrate and myeloperoxidase activity expressed as mU/mg of protein.
IL-2-mAb complex treatment
IL-2-mAb complex was prepared by mixing 1 µg of recombinant IL-2 (Miltenyi Biotec) with 5 µg of anti-IL-2 mAb (clone JES6-1A12, eBioscience) in PBS per mouse for i.p. administration 3 per week, every 2 days for 4–5 weeks.
DC transfer in vivo
Bone marrow cells from C57BL/6 CD45.1 or Il2KO mice were cultured in conditioned media containing recombinant mouse granulocytes macrophages colony-stimulating factor (GM-CSF) or recombinant mouse Flt3-L for 11 days. CD103+ and CD11b+ DCs (CD11c+MHCIIhigh) were FACS-sorted and adoptively transferred intravenously into 12-week-old Cnb1fl/fl or Cnb1CD11c mice at a ratio of 1:6 CD103:CD11b (total number of transferred cells: 5×105). Two weeks post-transfer, mice were killed and the immune phenotype of mononuclear cells from spleen, SI, and colon was investigated by flow cytometry.
Isolation of LP leukocytes from the colon
Isolated colons were cut longitudinally and divided into segments of 0.5–1 cm in length. The tissue segments pieces were then added to RPMI medium supplemented with 2% FBS, and incubated for 20 min at 37 oC with constant stirring. After incubation, the resultant suspension was passed through a 70 µm sterile strainer and pieces of colon tissue were placed into a 50 ml tube containing 15 ml serum-free RPMI, shaken vigorously for 30 s, and then filtered twice through a 70 µm cell strainer. The remaining fragments of colon were washed in calcium-free and magnesium-free HBSS and treated with 1 mm EDTA in PBS (twice for 20 min) to remove the epithelium. The tissue was then digested with type-IV collagenase (0.8 mg/ml; Sigma-Aldrich) containing DNase and the leukocytes were enriched on a 40:75 Percoll gradient (Pharmacia) and the interface collected after centrifugation at 700 × g for 20 min.
Isolation of LP leukocytes from the small intestine
Isolated SI was flushed with HBSS, the PP was removed, and then were cut longitudinally and divided into pieces of 1 cm in length. Epithelial cells were removed by incubation with 2 mm EDTA in HBSS supplemented with 10% FCS for 40 min at 37 °C, followed by vigorous shaking for 20 s. The samples were then collected and incubated with type-VIII collagenase (0.1 mg/ml, Sigma-Aldrich) and DNase I (50 µg/ml) in HBSS containing 10% FCS and 10 mm HEPES for 45 min at 37 °C. After digestion, the samples were shaken vigorously for 10 s, the supernatants were collected by filtration through a nylon mesh, and the tissue underwent a second round of enzymatic digestion. Leukocytes were enriched on a 40:75 Percoll gradient and the interface was collected after centrifugation at 700 × g for 20 min.
Cell culture and stimulation
Splenic D1 DCs were cultured as described previously33. NFAT-luciferase reporter D1 cells (D1-NFAT-Luc) were generated using the Cignal Lenti NFAT Reporter (luc) kit (Qiagen) as previously described5. Briefly, D1 cells were grown to 80% confluence before transduction with Cignal Lenti NFAT reporter (MOI 5) using SureENTRYTM Transduction reagent (2 μg/ml; Qiagen). Transduced D1 cells were selected in the presence of 150 μg/ml puromycin dihydrochloride (Sigma-Aldrich). Single D1 cell sub-cloning was carried out to generate a stable NFAT-luciferase reporter D1 cell line. D1-NFAT-Luc cells were seeded into a 96-well plate (5×104 per well) and rested overnight prior to 30 min pre-treatment with cyclosporin A or tacrolimus (FK506) at the indicated concentrations. Cells were then stimulated for 4 h with CpG (10 μg/ml), polyI:C (10 μg/ml), LPS (10 ng/ml), soluble β-(1,3)-glucan (PGG; 1 μg/ml), whole glucan particles (WGP; 10 μg/ml) or thapsigargin (60 nm). NFAT nuclear translocation was detected using the ONE-Glo Luciferase Assay System (Promega) and the luminescence signal was quantified with the GloMax-Multi Detection System Luminometer module (Promega).
BM-derived DCs (5×105 cells/ml) were cultured in Iscove’s modified Dulbecco’s medium containing GM-CSF (10 ng/ml), 10% heat-inactivated FCS, streptomycin (100 mg/ml) and penicillin (100 U/ml). The cells were harvested on day 6 and contaminating Gr-1+ cells were depleted using an AutoMACS apparatus (Miltenyi Biotec) prior to DC stimulation with LPS (10 μg/ml), zymosan (10 μg/ml) or thapsigargin (60 nm) for 18 h alone or in combination with pharmacological inhibitors of NFκB (BAY 11-7821, 1 µm) or p38 MAPK (SB 202190, 1 µm) added 30 min before stimulation.
Sorted CD11c+MHCIIhigh (CD11b+ and CD11b−) cells were seeded into 96-well plates (150,000 cells/100 µl/well) in complete RPMI medium supplemented with glutamine (2 mm), HEPES (10 mm) and β-mercaptoethanol (50 µm; all from GIBCO) and stimulated with thapsigargin (60 nm) for 16–20 h at 37 °C. Culture supernatants were then used for IL-2, IL-6, IL-10, IL-12p70, and IL-23p19 (eBioscience), and TGFβ1 (R&D System) ELISA.
OTII cell conversion
Naïve congenic OTII cells (CD45.1+CD4+CD45RBhighCD62L+CD44low) were sorted from spleens of donor mice and then transferred by intravenous injection into gender and age-matched Rag2KOIl2CD11c, Rag2KOIl2fl/fl, Cnb1CD11c, and Cnb1fl/fl mice (1×106 cells/mouse). After 24 h, mice were administered OVA protein (5 mg, Hyglos GmbH) dissolved in PBS daily for 7 days by oral gavage. The MLN were harvested on day 7 and OTII cells were analyzed for FoxP3, IFNγ, and IL-17 expression by flow-cytometry.
Flow cytometry and sorting of cell populations
Cell suspensions prepared from spleen, LP, and MLN were subjected to flow cytometry or cell sorting. The anti-mouse antibodies were used at 1:100 dilution unless specified otherwise. CD3ε (clone 500A2), CD16/CD32 (clone 2.4G2), CD62L (clone MEL-14) were obtained from BD Pharmingen; CD4 (clone RM4–5) and CD11b (clone M1/70) were from eBioscience; CD11c (clone N418), CD44 (clone IM7), CD45 (clone 30-F11 used at 1:200 dilution), CD45R B220 (clone RA3-6B2), CD45RB (clone C36-16A), CD69 (clone H1.2F3), CD80 (clone 16-10A1), CD86 (clone GL1), CD103 (clone 2E7), I-A/I-E (MHCII, clone M5/114.15.2) were obtained from Biolegend. For Treg-cell quantification, cells were permeabilized using the Mouse FoxP3 Buffer Set (BD Bioscience) reagents, and labeled with a FoxP3 antibody (clone MF23). For NFAT-1 flow analysis staining, cells were fixed and permeabilized with BD Cytofix/Cytoperm™ Fixation/Permeabilization Solution Kit (BD Bioscience) and labeled with an anti-mouse-NFAT-1 (1:100, Rabbit anti-mouse IgG) for 1 h followed by labeling with an anti-rabbit IgG-FITC secondary antibody (1:500). For CD4+ T-cell intracellular cytokine labeling, cells were stimulated with PMA (1 µg/ml) and ionomycin (1 µg/ml) for 1.5 h before addition of Brefeldin A (10 µg/ml) and incubation for a further 5 h. The cells were then harvested and surface-labeled before permeabilization using BD Cytofix/Cytoperm™ reagents (BD Bioscience) and intracellular staining for 30 min at 4 °C with a 50 μl mix of anti-IL17 (clone TCII-18H101 used at 1:50 dilution) and anti-IFNγ (clone XMG1.2 used at 1:50 dilution). For cytokine release quantification, CD4+ T cells were sorted and then seeded into 96-well plates for stimulation with plate-bound anti-CD3 (5 µg/ml) and soluble anti-CD28 (2 µg/ml) in complete RPMI medium for 36 h at 37 °C. After incubation, the culture supernatant was analyzed for cytokine content by ELISA. For intracellular IL-2 labeling, DCs were incubated for 30 min at 4 °C with anti-CD45, anti-CD3, anti-CD11c, anti-MHC-II, and anti-CD103 antibodies. For intracellular labeling, cells were permeabilized using the BD Cytofix/Cytoperm™ Fixation/Permeabilization Solution Kit (BD Bioscience) and then incubated with anti-mouse-IL-2 antibody (clone JES6-1A12; eBioscience) (1:16, 20 µl/tube for 30 min at 4 °C) or the same amount of fluorescently conjugated Rat-IgG2a, as an isotype control.
Immunofluorescent NFAT-1 staining
Cells were fixed in 2% paraformaldehyde for 10 min, washed with PBS and permeabilized with PBS containing 0.1% saponin, 0.2% gelatin, and 5 mg/ml BSA. The cells were then incubated with antibodies diluted in PBS containing 0.01% saponin and 0.2% gelatin. The following antibodies were used at a dilution of 1:500: NFAT-1 (cat. 4389S from Cell Signaling Technology) and CD11c PE (clone N418 from BioLegend). Cells were cytospun and mounted with Vectashield (Vector Laboratories). Images were acquired using a camera mounted on an Olympus FV1000 confocal microscope.
Quantitative real‐time PCR
Total cellular RNA was extracted using the Arcturus® PicoPure® RNA Isolation Kit. High‐capacity cDNA Reverse Transcription Kits with RNase Inhibitor (Applied Biosystems) or the Super‐Script III First Strand Synthesis System for RT‐PCR (Invitrogen) were used for reverse transcription. Real‐time PCR was carried out with the following validated SYBR Green primer pairs: Cnb1 forward 5′-TGTTCCGTGCCTTGAGGTTG-3′, reverse 5′-TCTGTTCCTTATCGCCTTTGAC-3′; Nfat1 forward 5′-CTGGTCTACGGGGGCCAGCA-3′, reverse 5′-GGCAGGGACTGG GTGGTAGG-3′; Nfat2 forward 5′-TGCAAGCCAAATTCCCTGGTGG-3′, reverse 5′-GGGGTCGGGAGGCATGGTGA-3′; Il2 forward 5′-CCCAGGATGCTCACCTTC-3′, reverse 5′-CAACAGTTACTCTGATATTGCTGATG-3′; Gapdh forward 5′-TCGTCCCGTAGACAAAATGG-3′, reverse 5′-TTGAGGTCAATGAAGGGGTC-3′. Amplification was performed on a 7500 Real-Time QPCR system (Applied Biosystems) and the relative gene expression was calculated using the comparative Ct method (2−ΔΔCt).
Statistical analyses
Data are expressed as the means ± standard error. One-way ANOVA was used for comparisons of more than two groups, followed by Tukey’s test for the analysis of naproxen-induced colitis, or by Dunnett’s multiple comparisons test to assess the levels of Cnb1, Nfat1, and Nfat2 expression in CD11c-expressing cells compared with controls. A two-tailed, unpaired Student’s t test was used to compare two groups of data. P values < 0.05 were considered statistically significant. GraphPad Prism version 6 (GraphPad Software) was used to prepare graphics and perform the statistical analyses.
Data availability
The authors declare that all data supporting the findings of this study are available within the article and its supplementary information files or from the corresponding authors on reasonable request.
References
Kaplan, G. G. The global burden of IBD: from 2015 to 2025. Nat. Rev. Gastroenterol. Hepatol. 12, 720–727 (2015).
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Joeris, T., Muller-Luda, K., Agace, W. W. & Mowat, A. M. Diversity and functions of intestinal mononuclear phagocytes. Mucosal Immunol. 10, 845–864 (2017).
Fric, J. et al. NFAT control of innate immunity. Blood 120, 1380–1389 (2012).
Zelante, T. et al. CD103(+) dendritic cells control Th17 cell function in the lung. Cell Rep. 12, 1789–1801 (2015).
Macian, F. NFAT proteins: key regulators of T-cell development and function. Nat. Rev. Immunol. 5, 472–484 (2005).
Fehr, T. et al. A CD8 T cell-intrinsic role for the calcineurin-NFAT pathway for tolerance induction in vivo. Blood 115, 1280–1287 (2010).
Baine, I., Abe, B. T. & Macian, F. Regulation of T-cell tolerance by calcium/NFAT signaling. Immunol. Rev. 231, 225–240 (2009).
Shin, D. S. et al. Regulatory T cells suppress CD4+ T cells through NFAT-dependent transcriptional mechanisms. EMBO Rep. 15, 991–999 (2014).
Liu, J. O. Calmodulin-dependent phosphatase, kinases, and transcriptional corepressors involved in T-cell activation. Immunol. Rev. 228, 184–198 (2009).
Granucci, F. et al. Inducible IL-2 production by dendritic cells revealed by global gene expression analysis. Nat. Immunol. 2, 882–888 (2001).
Slack, E. C. et al. Syk-dependent ERK activation regulates IL-2 and IL-10 production by DC stimulated with zymosan. Eur. J. Immunol. 37, 1600–1612 (2007).
Zelante, T. et al. Impaired calcineurin signaling in myeloid cells results in downregulation of pentraxin-3 and increased susceptibility to aspergillosis. Mucosal Immunol. 10, 470–480 (2017).
Khameneh, H. J. et al. The Syk-NFAT-IL-2 pathway in dendritic cells is required for optimal sterile immunity elicited by alum adjuvants. J. Immunol. 198, 196–204 (2017).
Caton, M. L., Smith-Raska, M. R. & Reizis, B. Notch-RBP-J signaling controls the homeostasis of CD8- dendritic cells in the spleen. J. Exp. Med. 204, 1653–1664 (2007).
Mottet, C., Uhlig, H. H. & Powrie, F. Cutting edge: cure of colitis by CD4+ CD25+ regulatory T cells. J. Immunol. 170, 3939–3943 (2003).
Scheiffele, F. & Fuss, I. J. Induction of TNBS colitis in mice. Curr. Protoc. Immunol. 49, 15.19.1–15.19.14 (2002).
Strober, W., Fuss, I. J. & Blumberg, R. S. The immunology of mucosal models of inflammation. Annu. Rev. Immunol. 20, 495–549 (2002).
Clausen, B. E., Burkhardt, C., Reith, W., Renkawitz, R. & Forster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 8, 265–277 (1999).
Amado, I. F. et al. IL-2 coordinates IL-2-producing and regulatory T cell interplay. J. Exp. Med. 210, 2707–2720 (2013).
Levine, J. S. & Burakoff, R. Extraintestinal manifestations of inflammatory bowel disease. Gastroenterol. Hepatol. 7, 235–241 (2011).
Manicassamy, S. & Pulendran, B. Dendritic cell control of tolerogenic responses. Immunol. Rev. 241, 206–227 (2011).
Maloy, K. J. & Powrie, F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature 474, 298–306 (2011).
Schimpl A. et al. IL-2 and autoimmune disease. Cytokine Growth Factor Rev. 13, 369–378 (2002).
Sadlack, B. et al. Ulcerative colitis-like disease in mice with a disrupted interleukin-2 gene. Cell 75, 253–261 (1993).
Contractor, N. V. et al. Lymphoid hyperplasia, autoimmunity, and compromised intestinal intraepithelial lymphocyte development in colitis-free gnotobiotic IL-2-deficient mice. J. Immunol. 160, 385–394 (1998).
Han, D. et al. Dendritic cell expression of the signaling molecule TRAF6 is critical for gut microbiota-dependent immune tolerance. Immunity 38, 1211–1222 (2013).
Neurath, M. F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 14, 329–342 (2014).
Tanoue, T., Atarashi, K. & Honda, K. Development and maintenance of intestinal regulatory T cells. Nat. Rev. Immunol. 16, 295–309 (2016).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Liao, W., Lin, J. X. & Leonard, W. J. Interleukin-2 at the crossroads of effector responses, tolerance, and immunotherapy. Immunity 38, 13–25 (2013).
Bryant, R. V., Brain, O. & Travis, S. P. Conventional drug therapy for inflammatory bowel disease. Scand. J. Gastroenterol. 50, 90–112 (2015).
Winzler, C. et al. Maturation stages of mouse dendritic cells in growth factor-dependent long-term cultures. J. Exp. Med. 185, 317–328 (1997).
Acknowledgements
The authors would like to thank I. Low, T. Seng, S. Mustafah, and N. Shadan from the SIgN flow-cytometry facility for their help with cell sorting, the SIgN Immunomonitoring Platform for assisting with luminex processing, and the SIgN Mutant Mouse Collection Core Facility for providing some mice. We wish to thank H.S. Tay for providing technical assistance, K. Neo for performing mouse genotyping and Insight Editing London for critically reviewing the manuscript. This work was supported by a SIgN core grant (A*STAR, Singapore). J.F. was supported by European Social Fund and European Regional Development Fund—Project MAGNET (No. CZ.02.1.01/0.0/0.0/15_003/0000492).
Author information
Authors and Affiliations
Contributions
A.M. conceived, designed, and performed the experiments, analyzed primary and aggregated data, and wrote the manuscript; H.J.K., J.F., M.V., and T.S.L. performed some experiments; S.E.D. analyzed data from the IL-2-GFP reporter mice experiments; H.J.K. critically revised the manuscript; J.P.T. performed qRT-PCR experiments and genotyped mice; S.N. performed animal screening; A.B. performed confocal microscopy; B.J. contributed with DC transfer experiments; F.G. provided methodological guidance for identifying myeloid cell subsets; P.R.-C. conceived the research, supervised the project and secured funds; A.Mo. conceived and supervised the project, designed experiments, analyzed aggregated data, secured funds, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mencarelli, A., Khameneh, H.J., Fric, J. et al. Calcineurin-mediated IL-2 production by CD11chighMHCII+ myeloid cells is crucial for intestinal immune homeostasis. Nat Commun 9, 1102 (2018). https://doi.org/10.1038/s41467-018-03495-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-03495-3
This article is cited by
-
Duan-Nai-An, A Yeast Probiotic, Improves Intestinal Mucosa Integrity and Immune Function in Weaned Piglets
Scientific Reports (2020)
-
Sensing the gut microbiota
Nature Immunology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.